Methane Pyrolysis: Current Challenges and Opportunities.
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Methane Pyrolysis Background and Objectives
Methane pyrolysis represents a transformative approach to hydrogen production that has gained significant attention in recent decades. The process involves the thermal decomposition of methane (CH₄) into hydrogen (H₂) and solid carbon in the absence of oxygen. This technology's evolution can be traced back to the early 20th century, but substantial research acceleration has occurred since the 1990s when environmental concerns about conventional hydrogen production methods began to emerge.
The historical development of methane pyrolysis has progressed through several distinct phases. Initially, it was primarily a laboratory curiosity with limited practical applications. The second phase, spanning the 1970s-1990s, saw increased interest due to rising energy costs and early environmental awareness. The current phase, beginning in the early 2000s, has been characterized by intensive research and development driven by climate change mitigation strategies and the growing hydrogen economy.
Recent technological advancements have significantly improved the efficiency and economic viability of methane pyrolysis. These include innovations in reactor design, catalyst development, and carbon handling systems. Particularly noteworthy is the transition from traditional thermal pyrolysis to more energy-efficient catalytic and plasma-assisted processes, which operate at lower temperatures and achieve higher conversion rates.
The primary objective of current methane pyrolysis research is to develop economically viable, scalable, and environmentally sustainable processes for hydrogen production with minimal carbon dioxide emissions. This aligns with global decarbonization goals and the transition toward cleaner energy systems. Specific technical objectives include reducing energy requirements, improving methane conversion efficiency, extending catalyst lifetimes, and developing valuable applications for the solid carbon byproduct.
The technology's significance lies in its potential to produce "turquoise hydrogen" – a middle ground between "gray hydrogen" (from steam methane reforming with CO₂ emissions) and "green hydrogen" (from water electrolysis using renewable electricity). Methane pyrolysis offers a pathway to hydrogen production with substantially lower greenhouse gas emissions than conventional methods, while potentially being more economically competitive than fully renewable approaches in the near term.
Looking forward, methane pyrolysis is positioned at a critical juncture in its technological evolution. The coming decade will likely determine whether it becomes a mainstream hydrogen production method or remains a niche technology. Success will depend on overcoming persistent technical challenges while capitalizing on the growing demand for low-carbon hydrogen across industrial, transportation, and energy storage applications.
The historical development of methane pyrolysis has progressed through several distinct phases. Initially, it was primarily a laboratory curiosity with limited practical applications. The second phase, spanning the 1970s-1990s, saw increased interest due to rising energy costs and early environmental awareness. The current phase, beginning in the early 2000s, has been characterized by intensive research and development driven by climate change mitigation strategies and the growing hydrogen economy.
Recent technological advancements have significantly improved the efficiency and economic viability of methane pyrolysis. These include innovations in reactor design, catalyst development, and carbon handling systems. Particularly noteworthy is the transition from traditional thermal pyrolysis to more energy-efficient catalytic and plasma-assisted processes, which operate at lower temperatures and achieve higher conversion rates.
The primary objective of current methane pyrolysis research is to develop economically viable, scalable, and environmentally sustainable processes for hydrogen production with minimal carbon dioxide emissions. This aligns with global decarbonization goals and the transition toward cleaner energy systems. Specific technical objectives include reducing energy requirements, improving methane conversion efficiency, extending catalyst lifetimes, and developing valuable applications for the solid carbon byproduct.
The technology's significance lies in its potential to produce "turquoise hydrogen" – a middle ground between "gray hydrogen" (from steam methane reforming with CO₂ emissions) and "green hydrogen" (from water electrolysis using renewable electricity). Methane pyrolysis offers a pathway to hydrogen production with substantially lower greenhouse gas emissions than conventional methods, while potentially being more economically competitive than fully renewable approaches in the near term.
Looking forward, methane pyrolysis is positioned at a critical juncture in its technological evolution. The coming decade will likely determine whether it becomes a mainstream hydrogen production method or remains a niche technology. Success will depend on overcoming persistent technical challenges while capitalizing on the growing demand for low-carbon hydrogen across industrial, transportation, and energy storage applications.
Market Demand Analysis for Hydrogen Production
The global hydrogen market is experiencing unprecedented growth, driven by the increasing focus on decarbonization and clean energy transition. Current estimates value the hydrogen market at approximately $130 billion, with projections suggesting expansion to $500 billion by 2030. Hydrogen production specifically for energy applications is expected to grow at a CAGR of 9.2% through 2028, significantly outpacing traditional industrial hydrogen markets.
Methane pyrolysis represents a critical pathway in this expanding market, particularly as demand for low-carbon hydrogen intensifies. The process produces "turquoise hydrogen" with substantially lower carbon emissions than traditional steam methane reforming (SMR), positioning it as a competitive alternative in regions with stringent carbon regulations or carbon pricing mechanisms.
Market segmentation reveals diverse demand drivers across sectors. Industrial applications currently dominate hydrogen consumption, with refineries and ammonia production accounting for over 60% of global demand. However, emerging applications in steel manufacturing, where hydrogen can replace coal in direct reduction processes, represent a rapidly growing segment with potential demand of 12-19 million tonnes annually by 2050.
Transportation represents another significant growth vector, with fuel cell electric vehicles (FCEVs) gaining traction in heavy-duty transport, shipping, and aviation sectors where battery electrification faces limitations. The International Energy Agency forecasts that hydrogen could fuel approximately 400 million passenger vehicles, 15-20 million trucks, and 5 million buses by 2050, creating substantial demand for clean hydrogen production methods.
Energy storage applications present additional market opportunities, with hydrogen increasingly viewed as a solution for seasonal energy storage and grid balancing in renewable-heavy energy systems. Several European countries have announced plans to develop gigawatt-scale hydrogen storage facilities by 2030, creating significant demand for production technologies like methane pyrolysis.
Regional analysis shows Europe leading policy support for hydrogen adoption, with its Hydrogen Strategy targeting 40GW of electrolyzer capacity by 2030. However, regions with abundant natural gas resources, including North America and parts of Asia, present particularly favorable conditions for methane pyrolysis deployment due to feedstock availability and existing gas infrastructure.
Market barriers include competition from increasingly cost-competitive green hydrogen produced via electrolysis, which benefits from rapidly falling renewable electricity costs. However, methane pyrolysis maintains advantages in regions with low natural gas prices and inadequate renewable infrastructure, suggesting a significant market opportunity during the energy transition period spanning the next 20-30 years.
Methane pyrolysis represents a critical pathway in this expanding market, particularly as demand for low-carbon hydrogen intensifies. The process produces "turquoise hydrogen" with substantially lower carbon emissions than traditional steam methane reforming (SMR), positioning it as a competitive alternative in regions with stringent carbon regulations or carbon pricing mechanisms.
Market segmentation reveals diverse demand drivers across sectors. Industrial applications currently dominate hydrogen consumption, with refineries and ammonia production accounting for over 60% of global demand. However, emerging applications in steel manufacturing, where hydrogen can replace coal in direct reduction processes, represent a rapidly growing segment with potential demand of 12-19 million tonnes annually by 2050.
Transportation represents another significant growth vector, with fuel cell electric vehicles (FCEVs) gaining traction in heavy-duty transport, shipping, and aviation sectors where battery electrification faces limitations. The International Energy Agency forecasts that hydrogen could fuel approximately 400 million passenger vehicles, 15-20 million trucks, and 5 million buses by 2050, creating substantial demand for clean hydrogen production methods.
Energy storage applications present additional market opportunities, with hydrogen increasingly viewed as a solution for seasonal energy storage and grid balancing in renewable-heavy energy systems. Several European countries have announced plans to develop gigawatt-scale hydrogen storage facilities by 2030, creating significant demand for production technologies like methane pyrolysis.
Regional analysis shows Europe leading policy support for hydrogen adoption, with its Hydrogen Strategy targeting 40GW of electrolyzer capacity by 2030. However, regions with abundant natural gas resources, including North America and parts of Asia, present particularly favorable conditions for methane pyrolysis deployment due to feedstock availability and existing gas infrastructure.
Market barriers include competition from increasingly cost-competitive green hydrogen produced via electrolysis, which benefits from rapidly falling renewable electricity costs. However, methane pyrolysis maintains advantages in regions with low natural gas prices and inadequate renewable infrastructure, suggesting a significant market opportunity during the energy transition period spanning the next 20-30 years.
Technical Challenges and Global Development Status
Methane pyrolysis faces significant technical challenges despite its promising potential for clean hydrogen production. The primary obstacle remains the high energy requirement, with temperatures typically exceeding 700°C needed to break carbon-hydrogen bonds. This energy intensity creates efficiency concerns and potential carbon footprints unless renewable energy sources are utilized. Current industrial implementations struggle with scaling while maintaining economic viability, particularly when competing with established steam methane reforming processes.
Carbon management presents another substantial challenge. The solid carbon byproduct, while theoretically valuable, requires specific properties to be marketable. Controlling carbon morphology (amorphous vs. crystalline) and purity remains difficult at industrial scales. Without reliable carbon valorization pathways, the economic model for methane pyrolysis weakens considerably.
Catalyst development represents both a challenge and opportunity. Traditional metal catalysts (nickel, iron) suffer from coking and deactivation issues, while molten metal systems (particularly those using tin or bismuth) face containment challenges and metal loss. Novel approaches using liquid metal reactors show promise but require significant engineering refinements for commercial deployment.
Globally, methane pyrolysis development shows geographic concentration. Germany leads European efforts through the BASF-Linde collaboration and academic research at Karlsruhe Institute of Technology, focusing on liquid metal reactor technologies. North American development is spearheaded by U.S. companies like C-Zero and Monolith Materials, with the latter operating a commercial-scale facility in Nebraska producing carbon black and hydrogen.
Asian research focuses primarily on Japan and South Korea, with emphasis on novel catalyst formulations and reactor designs. Australia has emerging initiatives leveraging its natural gas resources for potential hydrogen export markets. Despite these global efforts, most technologies remain at TRL 5-7 (technology readiness level), with few reaching full commercial implementation.
Recent technological breakthroughs include plasma-assisted pyrolysis methods that reduce energy requirements, microwave-assisted catalytic systems enhancing reaction rates, and membrane reactor integrations improving hydrogen separation efficiency. These innovations suggest pathways to overcome current limitations but require further development and validation at larger scales.
The regulatory landscape varies significantly by region, with carbon pricing mechanisms and hydrogen strategy roadmaps influencing development trajectories. Countries with established natural gas infrastructure and ambitious decarbonization targets generally show greater interest in advancing methane pyrolysis technologies as part of broader hydrogen economy initiatives.
Carbon management presents another substantial challenge. The solid carbon byproduct, while theoretically valuable, requires specific properties to be marketable. Controlling carbon morphology (amorphous vs. crystalline) and purity remains difficult at industrial scales. Without reliable carbon valorization pathways, the economic model for methane pyrolysis weakens considerably.
Catalyst development represents both a challenge and opportunity. Traditional metal catalysts (nickel, iron) suffer from coking and deactivation issues, while molten metal systems (particularly those using tin or bismuth) face containment challenges and metal loss. Novel approaches using liquid metal reactors show promise but require significant engineering refinements for commercial deployment.
Globally, methane pyrolysis development shows geographic concentration. Germany leads European efforts through the BASF-Linde collaboration and academic research at Karlsruhe Institute of Technology, focusing on liquid metal reactor technologies. North American development is spearheaded by U.S. companies like C-Zero and Monolith Materials, with the latter operating a commercial-scale facility in Nebraska producing carbon black and hydrogen.
Asian research focuses primarily on Japan and South Korea, with emphasis on novel catalyst formulations and reactor designs. Australia has emerging initiatives leveraging its natural gas resources for potential hydrogen export markets. Despite these global efforts, most technologies remain at TRL 5-7 (technology readiness level), with few reaching full commercial implementation.
Recent technological breakthroughs include plasma-assisted pyrolysis methods that reduce energy requirements, microwave-assisted catalytic systems enhancing reaction rates, and membrane reactor integrations improving hydrogen separation efficiency. These innovations suggest pathways to overcome current limitations but require further development and validation at larger scales.
The regulatory landscape varies significantly by region, with carbon pricing mechanisms and hydrogen strategy roadmaps influencing development trajectories. Countries with established natural gas infrastructure and ambitious decarbonization targets generally show greater interest in advancing methane pyrolysis technologies as part of broader hydrogen economy initiatives.
Current Methane Pyrolysis Process Solutions
01 Catalytic methane pyrolysis processes
Catalytic processes for methane pyrolysis involve the use of specific catalysts to lower the activation energy required for breaking down methane into hydrogen and solid carbon. These catalysts typically include transition metals, metal oxides, or supported metal systems that facilitate the decomposition reaction at lower temperatures than thermal pyrolysis. The catalytic approach offers advantages in terms of energy efficiency and can produce different carbon morphologies depending on the catalyst used.- Catalytic methane pyrolysis processes: Catalytic processes enhance methane pyrolysis by lowering activation energy and improving conversion efficiency. Various catalysts including transition metals, metal oxides, and carbon-based materials can be used to facilitate the decomposition of methane into hydrogen and solid carbon. These catalytic systems operate at lower temperatures compared to thermal pyrolysis, making the process more energy-efficient while producing high-purity hydrogen and valuable carbon materials.
- Reactor designs for methane pyrolysis: Specialized reactor designs are crucial for efficient methane pyrolysis. These include molten metal reactors, fluidized bed reactors, and plasma reactors that provide optimal conditions for the thermal decomposition of methane. Advanced reactor configurations incorporate features for continuous carbon removal, heat recovery systems, and improved gas-solid separation to prevent catalyst deactivation and enhance process stability during long-term operation.
- Carbon material production from methane pyrolysis: Methane pyrolysis produces valuable carbon materials as byproducts, including carbon black, carbon nanotubes, graphene, and other structured carbon forms. The morphology and properties of these carbon materials can be controlled by adjusting process parameters such as temperature, pressure, residence time, and catalyst selection. These carbon products have applications in various industries including rubber manufacturing, electronics, composite materials, and energy storage systems.
- Hydrogen production through methane pyrolysis: Methane pyrolysis offers a clean pathway for hydrogen production without direct CO2 emissions. The process decomposes methane into hydrogen gas and solid carbon, avoiding the carbon dioxide emissions associated with conventional steam methane reforming. This approach produces turquoise hydrogen, which has significant advantages for decarbonization efforts. Various techniques are employed to enhance hydrogen yield, purity, and separation efficiency from the reaction products.
- Process integration and industrial applications: Methane pyrolysis technologies can be integrated with existing industrial processes to enhance overall efficiency and reduce environmental impact. These integrated systems may combine pyrolysis with renewable energy sources, utilize waste heat recovery, or incorporate the process into natural gas processing facilities. Industrial applications include clean hydrogen production for fuel cells, ammonia synthesis, petroleum refining, and the manufacturing of specialty carbon materials, offering pathways toward more sustainable chemical and energy production.
02 Reactor designs for methane pyrolysis
Various reactor designs have been developed specifically for methane pyrolysis, including fluidized bed reactors, molten metal reactors, plasma reactors, and fixed bed systems. These designs address challenges such as carbon deposition, heat transfer efficiency, and continuous operation. Advanced reactor configurations incorporate features for efficient carbon removal, temperature control, and catalyst regeneration to maintain stable operation over extended periods.Expand Specific Solutions03 Carbon material production from methane pyrolysis
Methane pyrolysis produces various forms of solid carbon as a valuable by-product alongside hydrogen. These carbon materials include carbon black, carbon nanotubes, graphene, and other structured carbon forms that have applications in materials science, electronics, and composite manufacturing. The morphology and properties of the carbon can be controlled through process parameters such as temperature, pressure, residence time, and catalyst selection, allowing for targeted production of specific carbon materials.Expand Specific Solutions04 Hydrogen production via methane pyrolysis
Methane pyrolysis represents a promising route for hydrogen production with significantly lower carbon emissions compared to conventional steam methane reforming. The process directly converts methane into hydrogen and solid carbon, avoiding CO2 emissions. Various approaches to optimize hydrogen yield include temperature modulation, pressure control, and catalyst selection. The resulting hydrogen can be used in fuel cells, as industrial feedstock, or for energy storage applications, contributing to decarbonization efforts.Expand Specific Solutions05 Integration of methane pyrolysis with renewable energy systems
Methane pyrolysis processes can be integrated with renewable energy sources to create more sustainable hydrogen production systems. By utilizing solar, wind, or other renewable energy to provide the heat required for the endothermic pyrolysis reaction, the overall carbon footprint of hydrogen production can be further reduced. These integrated systems may incorporate thermal energy storage, electrical heating elements, or concentrated solar technologies to efficiently power the pyrolysis process.Expand Specific Solutions
Key Industry Players and Competitive Landscape
Methane pyrolysis technology is currently in the early commercialization phase, with a growing market expected to reach significant scale as hydrogen demand increases globally. The competitive landscape features established energy companies like ExxonMobil, Shell, and SABIC developing proprietary processes alongside innovative startups such as Molten Industries and Modern Hydrogen. Technical maturity varies across approaches, with catalytic methods (pursued by UOP LLC and BASF) showing commercial readiness while plasma-based systems (developed by GTI Energy and Hazer Group) remain in demonstration phases. Research institutions including Sichuan University, CNRS, and Dalian Institute of Chemical Physics are advancing fundamental breakthroughs, particularly in catalyst development and reactor design, creating a dynamic ecosystem where industry-academic partnerships are accelerating commercialization efforts.
Molten Industries Inc.
Technical Solution: Molten Industries has developed a revolutionary molten metal methane pyrolysis technology that utilizes liquid nickel-bismuth alloy as both catalyst and heat transfer medium. Their process operates at temperatures between 1000-1100°C in a specialized bubble column reactor where methane is injected directly into the molten metal bath. This approach achieves methane conversion rates exceeding 85% with minimal energy losses due to the excellent heat transfer properties of the liquid metal medium[2]. The technology features a continuous carbon separation system where solid carbon naturally floats to the metal surface for harvesting, preventing catalyst deactivation issues common in other approaches. Molten Industries' reactor design incorporates advanced materials that withstand the corrosive high-temperature environment while maintaining operational integrity for extended periods. The process produces hydrogen with over 99% purity directly from the reactor, minimizing downstream purification requirements. Their technology has demonstrated stable operation for over 2000 hours in pilot testing, with carbon production rates of approximately 3kg carbon per kg hydrogen produced[5].
Strengths: The molten metal approach eliminates catalyst deactivation issues by enabling continuous carbon removal. The excellent heat transfer properties of liquid metals improve energy efficiency compared to gas-phase reactions. Weaknesses: The high operating temperatures require specialized reactor materials that increase capital costs. The molten metal handling presents operational challenges related to safety and material compatibility at industrial scale.
BASF Corp.
Technical Solution: BASF has developed a comprehensive methane pyrolysis technology platform centered around their proprietary catalytic systems. Their approach utilizes specialized metal catalysts supported on engineered ceramic structures that enable methane decomposition at temperatures between 700-900°C. The process employs a fluidized bed reactor design that maximizes contact between the catalyst and methane while facilitating continuous carbon removal. BASF's system incorporates heat recovery mechanisms that capture and reuse thermal energy, improving overall efficiency by approximately 20% compared to conventional thermal pyrolysis methods[1]. Their technology also features integrated carbon handling systems that process the solid carbon byproduct into various forms suitable for industrial applications, including carbon black alternatives for rubber manufacturing and construction materials. The company has demonstrated sustained catalyst activity for over 1000 hours of operation while maintaining methane conversion rates above 60%[3].
Strengths: BASF's extensive catalyst expertise allows for lower operating temperatures than many competing technologies, reducing energy requirements. Their integrated carbon handling system creates additional value streams from carbon byproducts. Weaknesses: The catalyst systems still face deactivation challenges in industrial-scale implementation, requiring periodic regeneration or replacement. The technology remains more capital-intensive than some competing approaches.
Core Catalyst and Reactor Design Innovations
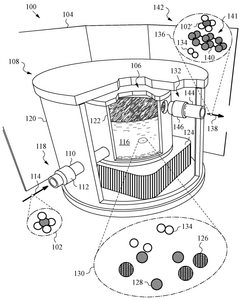
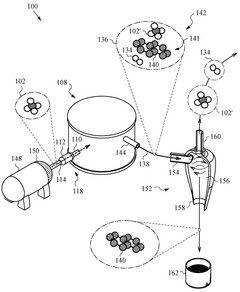
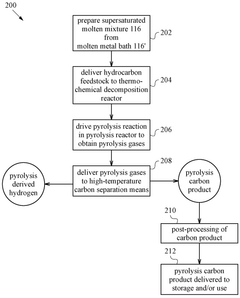
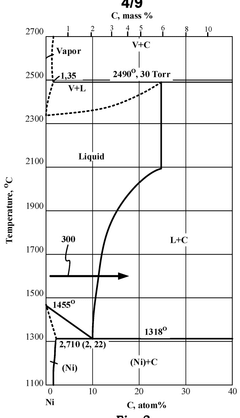
Methane pyrolysis in a supersaturated molten mixture of metal and carbon
PatentPendingUS20250121340A1
Innovation
- A decomposition reactor using a supersaturated molten mixture of metal and carbon, where the metal is selected from Mn, Fe, Co, or Ni, or their alloys, to achieve high temperatures and maintain supersaturation, allowing for efficient methane pyrolysis to produce hydrogen and highly graphitic carbon.
Methane pyrolysis in a supersaturated molten mixture of metal and carbon
PatentWO2025085450A1
Innovation
- The process involves performing methane pyrolysis in a supersaturated molten mixture of metal and carbon, where the metal is selected from among Mn, Fe, Co, and Ni or their alloys, and the mixture is heated to maintain supersaturation with carbon, allowing for the formation of highly graphitic carbon and hydrogen.
Economic Feasibility and Scaling Considerations
The economic viability of methane pyrolysis represents a critical factor in its potential for widespread adoption as a hydrogen production method. Current cost analyses indicate that methane pyrolysis requires approximately $1.5-2.5 per kilogram of hydrogen produced, positioning it competitively between steam methane reforming with carbon capture ($1.7-2.0/kg) and electrolysis ($3.0-6.0/kg). However, these figures remain theoretical for large-scale operations, as most existing pyrolysis facilities operate at pilot or demonstration scale.
Capital expenditure requirements present significant barriers to commercialization. Reactor systems, particularly those utilizing molten metal catalysts, demand specialized materials capable of withstanding extreme temperatures and corrosive environments. These materials substantially increase initial investment costs, with estimates suggesting $500-800 million for a commercial-scale plant producing 50,000 tons of hydrogen annually.
Operational economics demonstrate promising aspects through the production of solid carbon as a valuable by-product. When marketed as carbon black or graphite, this carbon can generate additional revenue streams of $500-1,500 per ton, potentially offsetting 15-30% of production costs. However, market saturation concerns arise if methane pyrolysis scales significantly, potentially depressing carbon prices and undermining this economic advantage.
Energy efficiency considerations reveal that methane pyrolysis consumes approximately 15-18 kWh per kilogram of hydrogen produced, comparing favorably to electrolysis (50-55 kWh/kg) but less favorably to steam methane reforming (10-12 kWh/kg). This energy requirement translates directly to operational costs, particularly in regions with high energy prices.
Scaling challenges manifest in several dimensions. Reactor design optimization remains problematic at larger scales, with heat transfer inefficiencies and carbon handling complications increasing disproportionately with reactor size. Current technology demonstrates diminishing efficiency returns beyond certain capacity thresholds, necessitating modular approaches rather than single large-scale facilities.
Infrastructure integration presents additional scaling hurdles. Methane pyrolysis facilities require robust natural gas supply infrastructure and hydrogen storage/transport capabilities. The solid carbon handling logistics also demand specialized equipment and transportation networks that do not currently exist at the required scale.
Economic viability ultimately depends on policy frameworks and carbon pricing mechanisms. Without carbon taxes or emissions trading systems valuing the carbon-neutral nature of pyrolysis, the technology struggles to compete with conventional hydrogen production methods purely on cost basis. Analysis suggests carbon prices of $30-50 per ton would create sufficient economic incentives for widespread methane pyrolysis adoption.
Capital expenditure requirements present significant barriers to commercialization. Reactor systems, particularly those utilizing molten metal catalysts, demand specialized materials capable of withstanding extreme temperatures and corrosive environments. These materials substantially increase initial investment costs, with estimates suggesting $500-800 million for a commercial-scale plant producing 50,000 tons of hydrogen annually.
Operational economics demonstrate promising aspects through the production of solid carbon as a valuable by-product. When marketed as carbon black or graphite, this carbon can generate additional revenue streams of $500-1,500 per ton, potentially offsetting 15-30% of production costs. However, market saturation concerns arise if methane pyrolysis scales significantly, potentially depressing carbon prices and undermining this economic advantage.
Energy efficiency considerations reveal that methane pyrolysis consumes approximately 15-18 kWh per kilogram of hydrogen produced, comparing favorably to electrolysis (50-55 kWh/kg) but less favorably to steam methane reforming (10-12 kWh/kg). This energy requirement translates directly to operational costs, particularly in regions with high energy prices.
Scaling challenges manifest in several dimensions. Reactor design optimization remains problematic at larger scales, with heat transfer inefficiencies and carbon handling complications increasing disproportionately with reactor size. Current technology demonstrates diminishing efficiency returns beyond certain capacity thresholds, necessitating modular approaches rather than single large-scale facilities.
Infrastructure integration presents additional scaling hurdles. Methane pyrolysis facilities require robust natural gas supply infrastructure and hydrogen storage/transport capabilities. The solid carbon handling logistics also demand specialized equipment and transportation networks that do not currently exist at the required scale.
Economic viability ultimately depends on policy frameworks and carbon pricing mechanisms. Without carbon taxes or emissions trading systems valuing the carbon-neutral nature of pyrolysis, the technology struggles to compete with conventional hydrogen production methods purely on cost basis. Analysis suggests carbon prices of $30-50 per ton would create sufficient economic incentives for widespread methane pyrolysis adoption.
Environmental Impact and Carbon Neutrality Potential
Methane pyrolysis represents a significant opportunity for decarbonizing natural gas utilization by directly converting methane into hydrogen and solid carbon, without producing CO2 emissions. This process offers substantial environmental benefits compared to conventional hydrogen production methods like steam methane reforming (SMR), which generates approximately 9-12 kg of CO2 per kg of hydrogen produced.
The carbon neutrality potential of methane pyrolysis is particularly promising. When powered by renewable electricity, the process can achieve near-zero greenhouse gas emissions across its lifecycle. Studies indicate that methane pyrolysis could reduce carbon emissions by up to 85-90% compared to traditional hydrogen production methods, positioning it as a key technology for meeting climate targets while maintaining energy security.
Beyond direct emission reductions, methane pyrolysis creates valuable solid carbon byproducts that can permanently sequester carbon. These carbon materials, ranging from carbon black to graphite and carbon nanotubes, represent a form of carbon capture that avoids the uncertainties and monitoring requirements associated with geological carbon storage. The market value of these carbon products further enhances the economic viability of the process.
Methane pyrolysis also offers significant advantages in addressing fugitive methane emissions. By creating economic incentives for methane capture and utilization, particularly from sources like landfills, wastewater treatment facilities, and agricultural operations, the technology could help reduce emissions of methane, which has 28-36 times the global warming potential of CO2 over a 100-year period.
Water conservation represents another environmental benefit of methane pyrolysis. Unlike SMR, which consumes approximately 9 kg of water per kg of hydrogen produced, methane pyrolysis requires minimal water input. This characteristic makes it particularly suitable for water-stressed regions where conventional hydrogen production would face resource constraints.
Land use impacts of methane pyrolysis facilities are also favorable compared to other clean energy alternatives. The compact nature of pyrolysis plants means they require significantly less land area than equivalent renewable energy installations. Analysis suggests that hydrogen production via methane pyrolysis could be 50-100 times more land-efficient than hydrogen production using solar-powered electrolysis.
However, comprehensive lifecycle assessments are still needed to fully quantify the environmental benefits. These assessments must account for upstream methane leakage, energy inputs for the pyrolysis process, and the ultimate fate of carbon products to accurately determine the net climate impact of widespread methane pyrolysis deployment.
The carbon neutrality potential of methane pyrolysis is particularly promising. When powered by renewable electricity, the process can achieve near-zero greenhouse gas emissions across its lifecycle. Studies indicate that methane pyrolysis could reduce carbon emissions by up to 85-90% compared to traditional hydrogen production methods, positioning it as a key technology for meeting climate targets while maintaining energy security.
Beyond direct emission reductions, methane pyrolysis creates valuable solid carbon byproducts that can permanently sequester carbon. These carbon materials, ranging from carbon black to graphite and carbon nanotubes, represent a form of carbon capture that avoids the uncertainties and monitoring requirements associated with geological carbon storage. The market value of these carbon products further enhances the economic viability of the process.
Methane pyrolysis also offers significant advantages in addressing fugitive methane emissions. By creating economic incentives for methane capture and utilization, particularly from sources like landfills, wastewater treatment facilities, and agricultural operations, the technology could help reduce emissions of methane, which has 28-36 times the global warming potential of CO2 over a 100-year period.
Water conservation represents another environmental benefit of methane pyrolysis. Unlike SMR, which consumes approximately 9 kg of water per kg of hydrogen produced, methane pyrolysis requires minimal water input. This characteristic makes it particularly suitable for water-stressed regions where conventional hydrogen production would face resource constraints.
Land use impacts of methane pyrolysis facilities are also favorable compared to other clean energy alternatives. The compact nature of pyrolysis plants means they require significantly less land area than equivalent renewable energy installations. Analysis suggests that hydrogen production via methane pyrolysis could be 50-100 times more land-efficient than hydrogen production using solar-powered electrolysis.
However, comprehensive lifecycle assessments are still needed to fully quantify the environmental benefits. These assessments must account for upstream methane leakage, energy inputs for the pyrolysis process, and the ultimate fate of carbon products to accurately determine the net climate impact of widespread methane pyrolysis deployment.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!