Atomic Force Microscopy Vs Confocal Laser Scanning Microscopy: Clarity
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
AFM and CLSM Technology Background and Objectives
Microscopy techniques have evolved significantly over the past century, with Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) emerging as two powerful tools for nanoscale and microscale imaging. Developed in the 1980s, AFM revolutionized surface science by enabling three-dimensional topographical imaging with nanometer resolution without requiring sample preparation procedures that might alter the specimen's natural state.
CLSM, introduced in the 1950s but significantly refined in the 1980s, transformed optical microscopy by eliminating out-of-focus light through a confocal pinhole, allowing for high-resolution optical sectioning of samples. This innovation enabled researchers to construct three-dimensional representations of specimens with unprecedented clarity in the optical domain.
The technological evolution of both techniques has been driven by the increasing demand for higher resolution, better contrast, and more detailed structural information across various scientific disciplines. AFM has progressed from basic contact mode operations to advanced multimodal capabilities including force spectroscopy and nanomechanical mapping, while CLSM has benefited from improvements in laser technology, detector sensitivity, and computational processing.
The primary objective in comparing these technologies is to establish their respective strengths and limitations regarding image clarity, which encompasses resolution, contrast, depth of field, and signal-to-noise ratio. AFM excels in providing topographical details with nanometer precision but is generally limited to surface features, whereas CLSM offers optical sectioning capabilities throughout transparent samples with diffraction-limited resolution.
Understanding the complementary nature of these techniques is crucial for researchers seeking to obtain comprehensive structural and functional information. AFM provides unparalleled surface detail and mechanical property measurements, while CLSM excels in visualizing specific labeled components within complex biological systems through fluorescence detection.
The technological trajectory suggests continued refinement in both fields, with AFM moving toward faster scanning rates, less invasive tip-sample interactions, and integration with other analytical techniques. CLSM development focuses on breaking the diffraction barrier through super-resolution techniques, enhancing sensitivity for detecting weak signals, and improving three-dimensional reconstruction algorithms.
This comparative analysis aims to provide clarity on when each technology offers optimal results, guiding researchers and industry professionals in selecting the appropriate microscopy technique based on specific application requirements, sample characteristics, and desired information output. The ultimate goal is to leverage the strengths of both technologies, potentially through correlative approaches, to address complex scientific and industrial challenges.
CLSM, introduced in the 1950s but significantly refined in the 1980s, transformed optical microscopy by eliminating out-of-focus light through a confocal pinhole, allowing for high-resolution optical sectioning of samples. This innovation enabled researchers to construct three-dimensional representations of specimens with unprecedented clarity in the optical domain.
The technological evolution of both techniques has been driven by the increasing demand for higher resolution, better contrast, and more detailed structural information across various scientific disciplines. AFM has progressed from basic contact mode operations to advanced multimodal capabilities including force spectroscopy and nanomechanical mapping, while CLSM has benefited from improvements in laser technology, detector sensitivity, and computational processing.
The primary objective in comparing these technologies is to establish their respective strengths and limitations regarding image clarity, which encompasses resolution, contrast, depth of field, and signal-to-noise ratio. AFM excels in providing topographical details with nanometer precision but is generally limited to surface features, whereas CLSM offers optical sectioning capabilities throughout transparent samples with diffraction-limited resolution.
Understanding the complementary nature of these techniques is crucial for researchers seeking to obtain comprehensive structural and functional information. AFM provides unparalleled surface detail and mechanical property measurements, while CLSM excels in visualizing specific labeled components within complex biological systems through fluorescence detection.
The technological trajectory suggests continued refinement in both fields, with AFM moving toward faster scanning rates, less invasive tip-sample interactions, and integration with other analytical techniques. CLSM development focuses on breaking the diffraction barrier through super-resolution techniques, enhancing sensitivity for detecting weak signals, and improving three-dimensional reconstruction algorithms.
This comparative analysis aims to provide clarity on when each technology offers optimal results, guiding researchers and industry professionals in selecting the appropriate microscopy technique based on specific application requirements, sample characteristics, and desired information output. The ultimate goal is to leverage the strengths of both technologies, potentially through correlative approaches, to address complex scientific and industrial challenges.
Market Applications and Demand Analysis
The market for advanced microscopy technologies continues to expand rapidly, driven by increasing demands across multiple sectors including life sciences, materials science, and semiconductor industries. Both Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) serve critical roles in these markets, though with distinct application profiles and demand patterns.
In the life sciences sector, CLSM dominates with an estimated market share exceeding 65% of advanced microscopy applications. This prevalence stems from its superior capabilities in visualizing biological specimens, particularly in cellular and subcellular imaging where three-dimensional reconstruction is essential. The pharmaceutical and biotechnology industries have become major consumers of CLSM technology, utilizing it extensively in drug discovery processes and fundamental research.
AFM, conversely, has established a stronger foothold in materials science and semiconductor industries, where surface characterization at the nanoscale is paramount. The semiconductor industry alone accounts for approximately 40% of AFM market applications, with demand growing steadily as chip manufacturers pursue ever-smaller feature sizes requiring atomic-level inspection capabilities.
Market analysis reveals a significant price differential between these technologies, with high-end CLSM systems typically commanding 30-50% higher prices than comparable AFM systems. Despite this premium, CLSM adoption continues to accelerate in clinical and research settings due to its versatility and compatibility with fluorescence techniques that enable specific molecular targeting.
Regional market distribution shows North America leading in both technologies, accounting for roughly 40% of global installations, followed by Europe and Asia-Pacific. However, the Asia-Pacific region demonstrates the highest growth rate, particularly in China and South Korea where government initiatives are heavily funding advanced microscopy capabilities in both academic and industrial settings.
The clarity advantage of CLSM in biological applications has created a distinct market segmentation, with research institutions often maintaining both technologies to address different imaging needs. This complementary relationship has resulted in a combined annual growth rate of approximately 8% for advanced microscopy technologies overall, outpacing many other scientific instrumentation categories.
Customer demand increasingly focuses on integrated solutions that combine multiple imaging modalities, with approximately 25% of new installations featuring some form of correlative microscopy capability. This trend reflects the growing recognition that complex research questions often require complementary data from different microscopy approaches, driving manufacturers to develop more flexible and interoperable systems.
In the life sciences sector, CLSM dominates with an estimated market share exceeding 65% of advanced microscopy applications. This prevalence stems from its superior capabilities in visualizing biological specimens, particularly in cellular and subcellular imaging where three-dimensional reconstruction is essential. The pharmaceutical and biotechnology industries have become major consumers of CLSM technology, utilizing it extensively in drug discovery processes and fundamental research.
AFM, conversely, has established a stronger foothold in materials science and semiconductor industries, where surface characterization at the nanoscale is paramount. The semiconductor industry alone accounts for approximately 40% of AFM market applications, with demand growing steadily as chip manufacturers pursue ever-smaller feature sizes requiring atomic-level inspection capabilities.
Market analysis reveals a significant price differential between these technologies, with high-end CLSM systems typically commanding 30-50% higher prices than comparable AFM systems. Despite this premium, CLSM adoption continues to accelerate in clinical and research settings due to its versatility and compatibility with fluorescence techniques that enable specific molecular targeting.
Regional market distribution shows North America leading in both technologies, accounting for roughly 40% of global installations, followed by Europe and Asia-Pacific. However, the Asia-Pacific region demonstrates the highest growth rate, particularly in China and South Korea where government initiatives are heavily funding advanced microscopy capabilities in both academic and industrial settings.
The clarity advantage of CLSM in biological applications has created a distinct market segmentation, with research institutions often maintaining both technologies to address different imaging needs. This complementary relationship has resulted in a combined annual growth rate of approximately 8% for advanced microscopy technologies overall, outpacing many other scientific instrumentation categories.
Customer demand increasingly focuses on integrated solutions that combine multiple imaging modalities, with approximately 25% of new installations featuring some form of correlative microscopy capability. This trend reflects the growing recognition that complex research questions often require complementary data from different microscopy approaches, driving manufacturers to develop more flexible and interoperable systems.
Current Technical Limitations and Challenges
Despite significant advancements in microscopy technologies, both Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) face distinct technical limitations that impact their clarity and overall performance. These challenges represent critical barriers to further development and application expansion in various scientific and industrial domains.
AFM encounters resolution limitations due to tip-sample interactions, where the physical dimensions of the probe tip (typically 5-10 nm radius) fundamentally restrict lateral resolution. This creates artifacts in imaging, particularly when examining samples with steep topographical features or deep trenches. The mechanical nature of AFM scanning also introduces vibration sensitivity issues that can significantly degrade image quality in non-isolated environments.
Speed constraints represent another significant challenge for AFM technology. The serial nature of data acquisition means that high-resolution imaging can be extremely time-consuming, with some detailed scans requiring hours to complete. This limitation makes AFM impractical for dynamic processes or high-throughput applications where rapid imaging is essential.
CLSM faces different but equally significant challenges. The diffraction limit of light (approximately 200-300 nm laterally and 500-700 nm axially) fundamentally restricts optical resolution, preventing visualization of nanoscale structures that fall below this threshold. This limitation is particularly problematic when examining subcellular structures or nanomaterials.
Photobleaching and phototoxicity present additional challenges for CLSM, especially in biological applications. Prolonged or intense laser exposure can damage fluorophores and living specimens, limiting observation time and potentially introducing artifacts in dynamic studies. This restricts the applicability of CLSM in long-term live-cell imaging experiments.
Both technologies struggle with specific sample preparation requirements. AFM typically requires relatively flat samples with moderate hardness, limiting its application to certain material types. CLSM demands fluorescent labeling, which can alter native biological structures and functions, potentially leading to misinterpretation of results.
Data interpretation challenges persist across both platforms. AFM data can be affected by tip-sample convolution effects that distort true surface topography, while CLSM images may contain optical aberrations and noise that complicate accurate analysis. These interpretation difficulties necessitate sophisticated post-processing algorithms and expert knowledge.
Cost and accessibility remain significant barriers to widespread adoption. High-end AFM and CLSM systems require substantial capital investment (often exceeding $100,000) and specialized training for operation and maintenance. This restricts their availability to well-funded research institutions and industrial R&D departments, limiting broader scientific advancement.
AFM encounters resolution limitations due to tip-sample interactions, where the physical dimensions of the probe tip (typically 5-10 nm radius) fundamentally restrict lateral resolution. This creates artifacts in imaging, particularly when examining samples with steep topographical features or deep trenches. The mechanical nature of AFM scanning also introduces vibration sensitivity issues that can significantly degrade image quality in non-isolated environments.
Speed constraints represent another significant challenge for AFM technology. The serial nature of data acquisition means that high-resolution imaging can be extremely time-consuming, with some detailed scans requiring hours to complete. This limitation makes AFM impractical for dynamic processes or high-throughput applications where rapid imaging is essential.
CLSM faces different but equally significant challenges. The diffraction limit of light (approximately 200-300 nm laterally and 500-700 nm axially) fundamentally restricts optical resolution, preventing visualization of nanoscale structures that fall below this threshold. This limitation is particularly problematic when examining subcellular structures or nanomaterials.
Photobleaching and phototoxicity present additional challenges for CLSM, especially in biological applications. Prolonged or intense laser exposure can damage fluorophores and living specimens, limiting observation time and potentially introducing artifacts in dynamic studies. This restricts the applicability of CLSM in long-term live-cell imaging experiments.
Both technologies struggle with specific sample preparation requirements. AFM typically requires relatively flat samples with moderate hardness, limiting its application to certain material types. CLSM demands fluorescent labeling, which can alter native biological structures and functions, potentially leading to misinterpretation of results.
Data interpretation challenges persist across both platforms. AFM data can be affected by tip-sample convolution effects that distort true surface topography, while CLSM images may contain optical aberrations and noise that complicate accurate analysis. These interpretation difficulties necessitate sophisticated post-processing algorithms and expert knowledge.
Cost and accessibility remain significant barriers to widespread adoption. High-end AFM and CLSM systems require substantial capital investment (often exceeding $100,000) and specialized training for operation and maintenance. This restricts their availability to well-funded research institutions and industrial R&D departments, limiting broader scientific advancement.
Comparative Analysis of AFM and CLSM Solutions
01 Combined AFM and CLSM systems for enhanced imaging
Integration of Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) into combined systems allows for simultaneous topographical and optical imaging. This combination provides enhanced clarity by correlating surface structures with fluorescence or optical data, enabling researchers to obtain comprehensive sample characterization with nanometer resolution alongside optical sectioning capabilities.- Combined AFM and CLSM systems for enhanced imaging: Integration of Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) into combined systems allows for simultaneous or correlated imaging of samples. This combination provides both high-resolution topographical information from AFM and optical/fluorescence data from CLSM, enabling comprehensive sample characterization. The integrated systems often feature specialized sample stages, alignment mechanisms, and software for correlative analysis.
- Sample preparation techniques for dual microscopy: Specific sample preparation methods are essential for achieving optimal clarity in both AFM and CLSM imaging. These techniques include specialized fixation protocols, surface treatments, and mounting procedures that maintain sample integrity while allowing access by both microscopy methods. Proper preparation ensures minimal artifacts, reduced background noise, and enhanced signal clarity for correlative microscopy studies.
- Resolution enhancement and artifact reduction: Advanced techniques for improving resolution and reducing artifacts in AFM and CLSM imaging include specialized probe designs, signal processing algorithms, and optical configurations. These innovations help overcome limitations such as diffraction limits in CLSM and tip-sample interactions in AFM. Methods for noise reduction, contrast enhancement, and deconvolution significantly improve image clarity and data reliability in both microscopy techniques.
- Environmental control for improved imaging clarity: Environmental factors significantly impact the clarity of both AFM and CLSM imaging. Systems incorporating temperature control, vibration isolation, humidity regulation, and atmospheric composition management help maintain stable imaging conditions. These environmental control mechanisms minimize thermal drift, reduce mechanical noise, and prevent sample degradation, resulting in sharper images and more reliable data acquisition.
- Data processing and analysis for correlative microscopy: Specialized software and algorithms for processing and analyzing data from AFM and CLSM enable meaningful correlation between different imaging modalities. These tools facilitate image registration, feature extraction, 3D reconstruction, and quantitative analysis of multimodal datasets. Advanced data processing techniques help overcome differences in scale, resolution, and contrast mechanisms between the two microscopy methods, providing comprehensive sample characterization with enhanced clarity.
02 Sample preparation techniques for improved microscopy clarity
Specialized sample preparation methods significantly improve imaging clarity in both AFM and CLSM. These techniques include surface treatment protocols, specialized mounting procedures, and environmental control systems that minimize artifacts and noise. Proper sample preparation ensures optimal interaction between the probe/laser and the specimen, resulting in higher resolution images and more accurate data interpretation.Expand Specific Solutions03 Advanced data processing algorithms for microscopy image enhancement
Sophisticated data processing algorithms are employed to enhance image clarity in both AFM and CLSM. These computational methods include noise reduction filters, deconvolution techniques, and 3D reconstruction algorithms that significantly improve image quality. Machine learning approaches can also be applied to extract meaningful features from raw microscopy data, resulting in clearer visualization and more accurate quantitative analysis.Expand Specific Solutions04 Probe and scanner design innovations for higher resolution
Innovations in probe tip design for AFM and scanner technology for CLSM have led to substantial improvements in imaging clarity. Advanced cantilever materials, optimized tip geometries, and improved laser focusing systems enable higher resolution and reduced artifacts. These design innovations allow for more precise measurements of surface topography and optical properties, particularly important when examining nanoscale structures.Expand Specific Solutions05 Multi-modal microscopy approaches for comprehensive sample analysis
Multi-modal microscopy approaches combine AFM and CLSM with additional imaging techniques to provide comprehensive sample characterization. These hybrid systems may incorporate spectroscopic methods, fluorescence lifetime imaging, or other complementary techniques. By collecting multiple data types simultaneously, researchers can correlate mechanical, optical, and chemical properties of samples, leading to deeper insights and clearer understanding of complex specimens.Expand Specific Solutions
Leading Manufacturers and Research Institutions
Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM) represent complementary imaging technologies in a mature market dominated by established players. The industry is in a consolidation phase with key manufacturers like Carl Zeiss, Olympus, Leica Microsystems, and Bruker Nano leading technological innovation. The global microscopy market exceeds $8 billion, with advanced microscopy segments growing at 6-8% annually. Technology maturity varies: AFM offers superior resolution for surface topography analysis but with complex operation, while CLSM provides excellent optical sectioning with simpler workflow. Academic institutions (Zhejiang University, Heidelberg University) collaborate with industry leaders to advance capabilities, particularly in biological applications where Evident Corp. and Caliber Imaging are developing specialized clinical solutions.
Leica Microsystems CMS GmbH
Technical Solution: Leica Microsystems has developed the TCS SP8 STED 3X super-resolution platform that bridges the gap between confocal and atomic force microscopy resolutions. Their proprietary STED (Stimulated Emission Depletion) technology achieves optical resolution down to 30nm, approaching the mechanical sensing capabilities of AFM systems[1]. For comparative studies between AFM and CLSM, Leica offers the DMi8 S platform with modular design allowing integration of AFM components from partner manufacturers. Their Matrix Screener technology enables automated multi-position imaging across both modalities, facilitating direct comparison of the same regions using both techniques[2]. Leica's adaptive confocal technology (LIGHTNING) uses computational approaches to enhance resolution while maintaining the non-destructive nature of optical imaging, providing an alternative to AFM for samples sensitive to mechanical probing[3]. Their LAS X software includes modules specifically designed for correlative microscopy workflows.
Strengths: Industry-leading super-resolution capabilities that narrow the resolution gap with AFM; excellent software integration for correlative workflows; strong focus on live-cell imaging applications. Weaknesses: Requires partnerships for full AFM integration; higher maintenance costs; complex system operation requiring specialized training.
Olympus Corp.
Technical Solution: Olympus has developed the FLUOVIEW FV3000 confocal laser scanning microscope system that incorporates their TruSpectral detection technology, allowing precise wavelength selection for enhanced multicolor imaging capabilities. Their approach to the AFM vs CLSM comparison focuses on complementary integration rather than competition between technologies. The Olympus LEXT OLS5000 confocal laser scanning microscope specifically addresses industrial measurement applications with 4K scanning technology and a resolution of 0.2μm, providing non-contact surface measurements comparable to certain AFM applications[1]. For correlative microscopy, Olympus has partnered with leading AFM manufacturers to create integrated workflows where their high-speed resonant scanner technology in CLSM systems (achieving up to 438 fps) complements the high-resolution but slower scanning capabilities of AFM[2]. Their cellSens software platform includes specific modules for correlative microscopy that align and overlay data from both imaging modalities.
Strengths: Exceptional speed in confocal scanning; robust industrial measurement capabilities; user-friendly software interface; strong focus on practical applications. Weaknesses: Less specialized in high-end research applications; requires third-party partnerships for complete AFM solutions; more limited resolution compared to leading super-resolution systems.
Key Patents and Technical Innovations
High sensitivity scanning probe system
PatentInactiveUS20050117163A1
Innovation
- The proposed scanning probe system utilizes an interferometer to split and recombine light beams, forming an interference pattern that is processed to enhance sensitivity and noise rejection, with a transmission grating modulating the fringe pattern to distinguish between bending and torsion modes, and actuators to adjust the fringe pattern orientation and period, allowing for improved surface characterization without the need for expensive photodetector adjustments.
Confocal laser scanning microscopy apparatus
PatentInactiveUS20050258375A1
Innovation
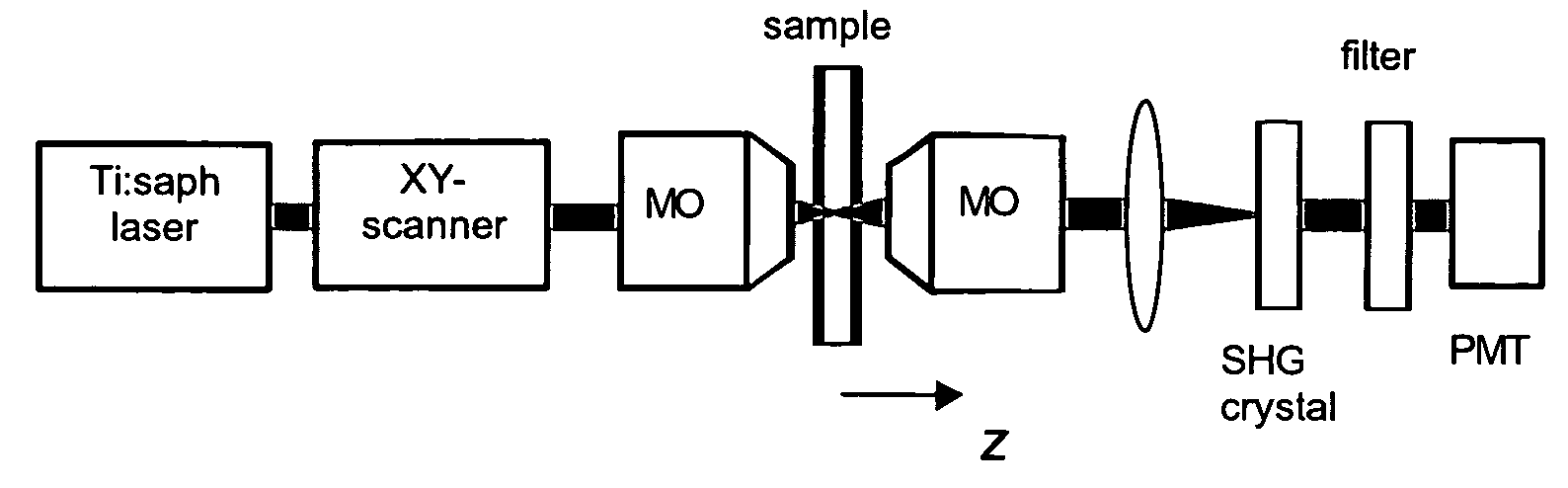
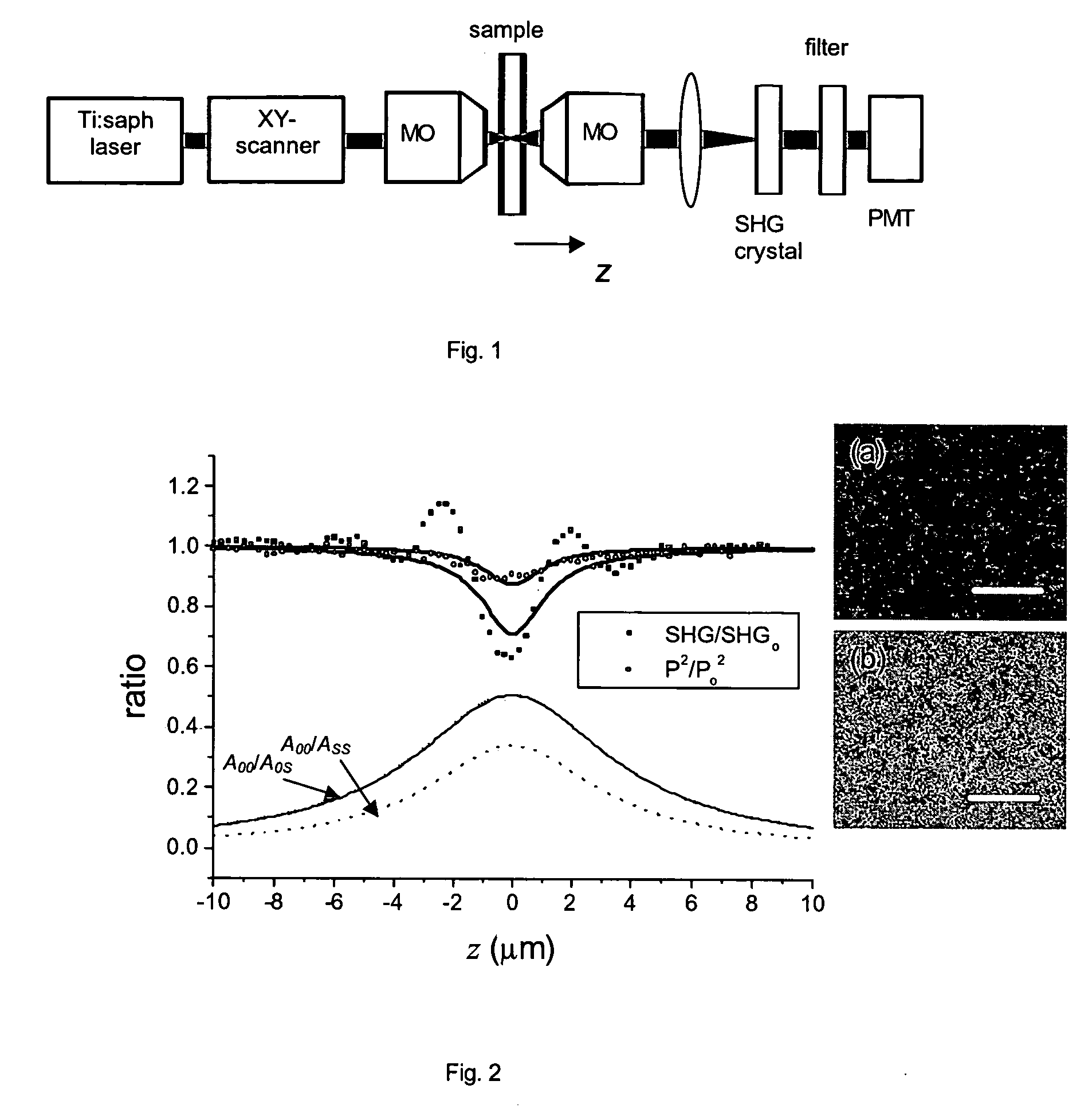
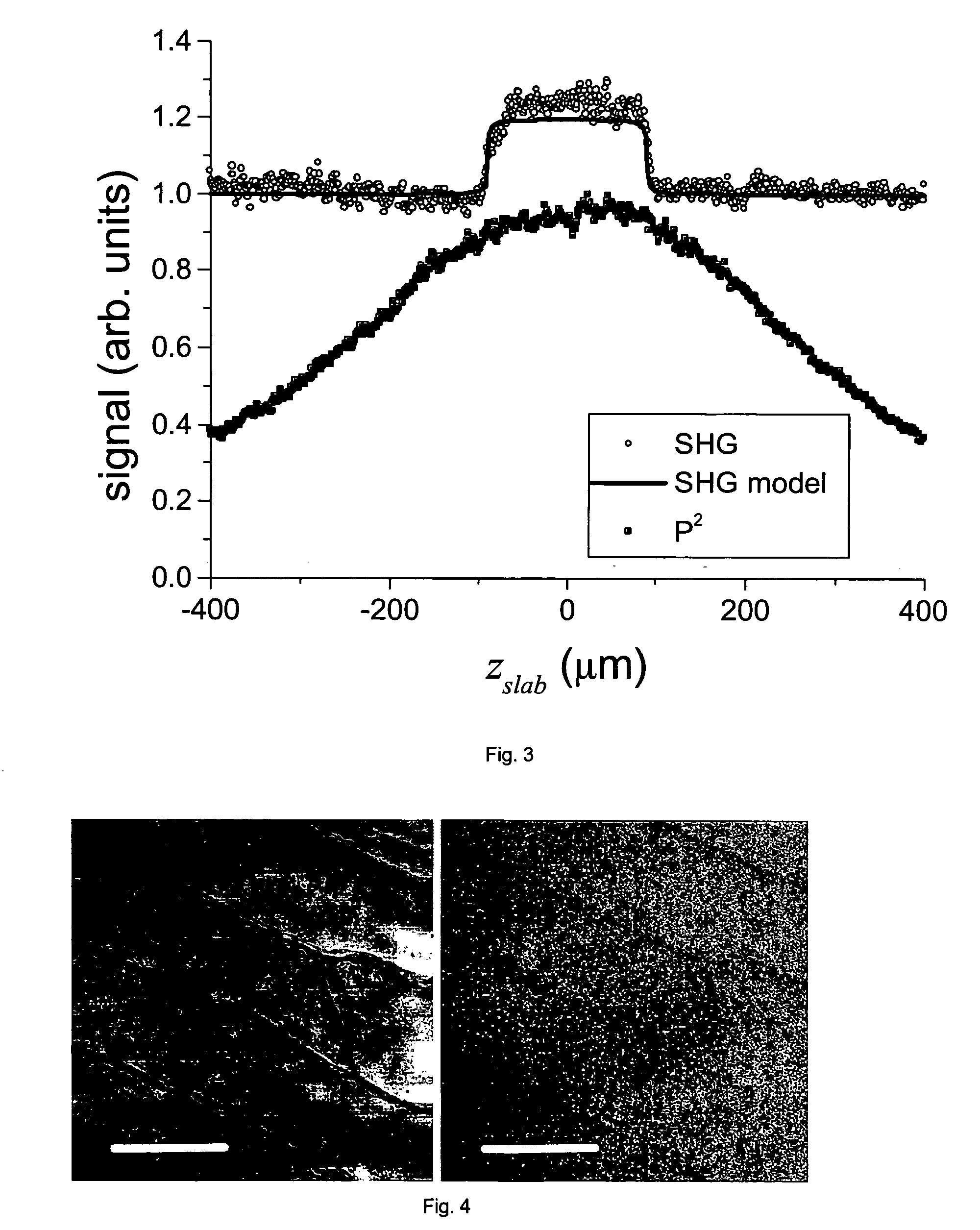
- The use of a second harmonic generation (SHG) crystal as a virtual pinhole, which scales quadratically with incident intensity, allowing for self-aligned confocality without the need for de-scanning, and preferentially rejects out-of-focus scattered light by detecting only SHG signals.
Sample Preparation Techniques and Optimization
Sample preparation represents a critical determinant in the quality and reliability of microscopic imaging results, particularly when comparing Atomic Force Microscopy (AFM) and Confocal Laser Scanning Microscopy (CLSM). The fundamental differences in these technologies necessitate distinct approaches to specimen preparation, each with specific requirements and optimization strategies.
For AFM analysis, sample preparation typically requires specimens to be immobilized on atomically flat substrates such as mica, silicon wafers, or glass slides. Surface cleanliness is paramount, often requiring rigorous cleaning protocols involving sonication in organic solvents followed by plasma treatment to remove contaminants that could interfere with the nanoscale resolution. Biological samples frequently require chemical fixation with glutaraldehyde or formaldehyde, followed by careful dehydration to preserve structural integrity while minimizing artifacts.
CLSM sample preparation, conversely, focuses on fluorescent labeling efficiency and specificity. Samples generally require fixation, permeabilization, and incubation with fluorophore-conjugated antibodies or fluorescent proteins. The optimization of fluorophore concentration is essential to balance signal intensity against background fluorescence. Additionally, mounting media selection significantly impacts image clarity, with antifade reagents being crucial for preventing photobleaching during extended imaging sessions.
Environmental conditions during preparation substantially influence imaging outcomes for both techniques. AFM samples are highly sensitive to humidity and temperature fluctuations, which can induce conformational changes in biomolecules or alter surface properties. CLSM specimens require controlled pH environments to maintain fluorophore quantum yield and prevent quenching effects that diminish signal quality.
Cross-platform sample preparation protocols have emerged as valuable approaches for comparative studies. These methods involve preparing identical specimens suitable for sequential imaging with both AFM and CLSM, often utilizing specialized substrates with fiducial markers for precise correlation of images. Such protocols typically incorporate compromises in fixation methods and buffer compositions to accommodate the requirements of both techniques.
Recent advances in cryogenic preparation techniques have significantly enhanced preservation of native structures for both microscopy methods. Flash-freezing samples in vitreous ice maintains near-native conformations while minimizing preparation artifacts. Similarly, developments in correlative microscopy workflows have streamlined the transition between imaging modalities, allowing researchers to leverage the complementary strengths of AFM's topographical precision and CLSM's molecular specificity.
For AFM analysis, sample preparation typically requires specimens to be immobilized on atomically flat substrates such as mica, silicon wafers, or glass slides. Surface cleanliness is paramount, often requiring rigorous cleaning protocols involving sonication in organic solvents followed by plasma treatment to remove contaminants that could interfere with the nanoscale resolution. Biological samples frequently require chemical fixation with glutaraldehyde or formaldehyde, followed by careful dehydration to preserve structural integrity while minimizing artifacts.
CLSM sample preparation, conversely, focuses on fluorescent labeling efficiency and specificity. Samples generally require fixation, permeabilization, and incubation with fluorophore-conjugated antibodies or fluorescent proteins. The optimization of fluorophore concentration is essential to balance signal intensity against background fluorescence. Additionally, mounting media selection significantly impacts image clarity, with antifade reagents being crucial for preventing photobleaching during extended imaging sessions.
Environmental conditions during preparation substantially influence imaging outcomes for both techniques. AFM samples are highly sensitive to humidity and temperature fluctuations, which can induce conformational changes in biomolecules or alter surface properties. CLSM specimens require controlled pH environments to maintain fluorophore quantum yield and prevent quenching effects that diminish signal quality.
Cross-platform sample preparation protocols have emerged as valuable approaches for comparative studies. These methods involve preparing identical specimens suitable for sequential imaging with both AFM and CLSM, often utilizing specialized substrates with fiducial markers for precise correlation of images. Such protocols typically incorporate compromises in fixation methods and buffer compositions to accommodate the requirements of both techniques.
Recent advances in cryogenic preparation techniques have significantly enhanced preservation of native structures for both microscopy methods. Flash-freezing samples in vitreous ice maintains near-native conformations while minimizing preparation artifacts. Similarly, developments in correlative microscopy workflows have streamlined the transition between imaging modalities, allowing researchers to leverage the complementary strengths of AFM's topographical precision and CLSM's molecular specificity.
Cost-Benefit Analysis and Implementation Considerations
When evaluating the implementation of Atomic Force Microscopy (AFM) versus Confocal Laser Scanning Microscopy (CLSM) for clarity-focused applications, organizations must conduct a thorough cost-benefit analysis to make informed decisions. The initial investment for AFM systems typically ranges from $100,000 to $500,000, while CLSM systems generally cost between $200,000 and $700,000, depending on specifications and capabilities.
Beyond acquisition costs, operational expenses differ significantly. AFM requires specialized probes that need regular replacement ($100-300 per probe), with each having a limited lifespan depending on usage intensity and sample characteristics. CLSM operational costs center around laser maintenance, fluorescent dyes, and specialized sample preparation materials, averaging $15,000-25,000 annually for moderate usage facilities.
Maintenance requirements present another critical consideration. AFM systems demand regular calibration and probe replacement but generally have fewer complex optical components that might fail. CLSM systems require periodic laser alignment, detector calibration, and more complex maintenance protocols, typically resulting in 15-20% higher annual maintenance costs compared to AFM.
Space and facility requirements also impact implementation decisions. AFM systems are more compact, requiring approximately 2-3 square meters of vibration-isolated space. CLSM installations need 4-6 square meters with specific environmental controls for temperature stability and dust management. Both technologies require trained operators, though the learning curve for basic AFM operation is generally steeper than for CLSM.
Return on investment calculations must consider application-specific benefits. For nanoscale topographical analysis, AFM delivers superior value despite higher per-sample processing times (30-60 minutes versus 10-20 minutes for CLSM). Conversely, for fluorescence-based cellular studies requiring rapid throughput, CLSM provides better economic efficiency despite higher initial costs.
Hybrid implementation strategies are increasingly common in research facilities. Organizations often begin with either AFM or CLSM based on immediate research priorities, then expand capabilities through service contracts or collaborative arrangements before committing to purchasing both technologies. This phased approach optimizes capital expenditure while maintaining research flexibility.
Ultimately, the clarity advantage of either technology must be weighed against specific research objectives, sample throughput requirements, and available technical expertise. Organizations should develop a 3-5 year technology roadmap that accounts for evolving research needs, potential technology advancements, and maintenance cost projections to maximize the return on microscopy investments.
Beyond acquisition costs, operational expenses differ significantly. AFM requires specialized probes that need regular replacement ($100-300 per probe), with each having a limited lifespan depending on usage intensity and sample characteristics. CLSM operational costs center around laser maintenance, fluorescent dyes, and specialized sample preparation materials, averaging $15,000-25,000 annually for moderate usage facilities.
Maintenance requirements present another critical consideration. AFM systems demand regular calibration and probe replacement but generally have fewer complex optical components that might fail. CLSM systems require periodic laser alignment, detector calibration, and more complex maintenance protocols, typically resulting in 15-20% higher annual maintenance costs compared to AFM.
Space and facility requirements also impact implementation decisions. AFM systems are more compact, requiring approximately 2-3 square meters of vibration-isolated space. CLSM installations need 4-6 square meters with specific environmental controls for temperature stability and dust management. Both technologies require trained operators, though the learning curve for basic AFM operation is generally steeper than for CLSM.
Return on investment calculations must consider application-specific benefits. For nanoscale topographical analysis, AFM delivers superior value despite higher per-sample processing times (30-60 minutes versus 10-20 minutes for CLSM). Conversely, for fluorescence-based cellular studies requiring rapid throughput, CLSM provides better economic efficiency despite higher initial costs.
Hybrid implementation strategies are increasingly common in research facilities. Organizations often begin with either AFM or CLSM based on immediate research priorities, then expand capabilities through service contracts or collaborative arrangements before committing to purchasing both technologies. This phased approach optimizes capital expenditure while maintaining research flexibility.
Ultimately, the clarity advantage of either technology must be weighed against specific research objectives, sample throughput requirements, and available technical expertise. Organizations should develop a 3-5 year technology roadmap that accounts for evolving research needs, potential technology advancements, and maintenance cost projections to maximize the return on microscopy investments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!