Atomic Force Microscopy Vs Total Internal Reflection Microscopy: Efficiency
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AFM and TIRM Technology Background and Objectives
Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) represent two distinct yet complementary approaches in the field of microscopy that have revolutionized our ability to visualize and characterize surfaces at the nanoscale. Since its invention in 1986 by Binnig, Quate, and Gerber, AFM has evolved from a simple imaging tool to a versatile platform capable of measuring various surface properties including topography, mechanical characteristics, and even chemical compositions with nanometer resolution.
TIRM, developed in the 1980s based on the physical principle of total internal reflection, has followed a parallel but distinct evolutionary path. This technique exploits the evanescent wave generated when light undergoes total internal reflection at an interface, allowing for the visualization of objects within approximately 100-200 nm of a surface without direct contact.
The technological progression of both methods has been driven by advances in electronics, computing power, and materials science. AFM has seen significant improvements in probe design, feedback mechanisms, and operational modes, transitioning from contact mode to dynamic modes that reduce sample damage. Similarly, TIRM has benefited from developments in light sources, detectors, and image processing algorithms, enhancing its sensitivity and resolution.
Current technological trends indicate a convergence of these methodologies with other techniques to create hybrid systems capable of providing complementary information. The integration of AFM with optical microscopy, including TIRM, represents a frontier in correlative microscopy, allowing researchers to simultaneously obtain topographical, mechanical, and optical data from the same sample region.
The primary objective of this technical research is to comprehensively evaluate the efficiency of AFM versus TIRM across various applications, considering factors such as resolution capabilities, sample preparation requirements, data acquisition speed, and operational complexity. This assessment aims to identify the optimal conditions and scenarios for employing each technique, recognizing that efficiency encompasses not only the quality of results but also practical considerations like cost, time, and expertise requirements.
Furthermore, this research seeks to explore potential synergies between AFM and TIRM, investigating how their complementary strengths might be leveraged in integrated systems to overcome the limitations inherent to each individual technique. By understanding the fundamental principles, current capabilities, and future directions of these technologies, we aim to provide strategic insights that will inform research priorities and investment decisions in microscopy infrastructure.
The ultimate goal is to establish a framework for selecting the most efficient microscopy approach based on specific research or industrial requirements, thereby optimizing resource allocation and enhancing productivity in fields ranging from materials science to biological research.
TIRM, developed in the 1980s based on the physical principle of total internal reflection, has followed a parallel but distinct evolutionary path. This technique exploits the evanescent wave generated when light undergoes total internal reflection at an interface, allowing for the visualization of objects within approximately 100-200 nm of a surface without direct contact.
The technological progression of both methods has been driven by advances in electronics, computing power, and materials science. AFM has seen significant improvements in probe design, feedback mechanisms, and operational modes, transitioning from contact mode to dynamic modes that reduce sample damage. Similarly, TIRM has benefited from developments in light sources, detectors, and image processing algorithms, enhancing its sensitivity and resolution.
Current technological trends indicate a convergence of these methodologies with other techniques to create hybrid systems capable of providing complementary information. The integration of AFM with optical microscopy, including TIRM, represents a frontier in correlative microscopy, allowing researchers to simultaneously obtain topographical, mechanical, and optical data from the same sample region.
The primary objective of this technical research is to comprehensively evaluate the efficiency of AFM versus TIRM across various applications, considering factors such as resolution capabilities, sample preparation requirements, data acquisition speed, and operational complexity. This assessment aims to identify the optimal conditions and scenarios for employing each technique, recognizing that efficiency encompasses not only the quality of results but also practical considerations like cost, time, and expertise requirements.
Furthermore, this research seeks to explore potential synergies between AFM and TIRM, investigating how their complementary strengths might be leveraged in integrated systems to overcome the limitations inherent to each individual technique. By understanding the fundamental principles, current capabilities, and future directions of these technologies, we aim to provide strategic insights that will inform research priorities and investment decisions in microscopy infrastructure.
The ultimate goal is to establish a framework for selecting the most efficient microscopy approach based on specific research or industrial requirements, thereby optimizing resource allocation and enhancing productivity in fields ranging from materials science to biological research.
Market Applications and Demand Analysis
The market for advanced microscopy technologies has witnessed substantial growth in recent years, driven by increasing demand for high-resolution imaging across multiple sectors. Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) represent two distinct approaches that serve different market segments with varying efficiency considerations.
In the life sciences and biomedical research sector, there is growing demand for non-invasive imaging techniques that can visualize cellular processes at nanoscale resolution. TIRM has gained significant traction in this space due to its ability to visualize membrane-proximal events with minimal photodamage to living specimens. Market research indicates that approximately 65% of academic life science research facilities now incorporate some form of TIRM in their imaging suites, particularly for studies involving membrane trafficking and cell adhesion dynamics.
The semiconductor and materials science industries have emerged as primary adopters of AFM technology, where precise surface characterization is critical for quality control and R&D. The global semiconductor inspection equipment market, where AFM plays a crucial role, has been expanding at a compound annual growth rate of 8.3% since 2018, reaching a market value of $4.9 billion in 2022.
Healthcare applications represent another significant market driver, with clinical diagnostics increasingly utilizing advanced microscopy for disease detection and characterization. TIRM's efficiency in visualizing single-molecule interactions has positioned it as a valuable tool in pharmaceutical development, particularly for antibody characterization and drug-target interaction studies.
Regional market analysis reveals that North America currently dominates the advanced microscopy market with approximately 38% share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is experiencing the fastest growth rate, driven by expanding research infrastructure in China, Japan, and South Korea.
Cost considerations significantly influence market adoption patterns. AFM systems typically require higher initial investment (average system cost ranging from $100,000 to $500,000) compared to TIRM setups. However, the total cost of ownership analysis reveals that TIRM often incurs higher operational expenses due to specialized fluorophores and optical components with limited lifespans.
Market forecasts suggest that the efficiency advantages of each technology will drive specialized adoption rather than direct competition. TIRM's efficiency in real-time dynamic imaging of biological processes complements AFM's efficiency in high-precision topographical mapping. This complementary relationship is reflected in purchasing patterns, with approximately 42% of research institutions maintaining both technologies to address different experimental needs.
The industrial quality control segment represents an emerging market opportunity, particularly for AFM, with manufacturing sectors increasingly adopting automated AFM systems for inline inspection processes. This application area is projected to grow at 11.2% annually through 2027, outpacing the overall microscopy market growth rate.
In the life sciences and biomedical research sector, there is growing demand for non-invasive imaging techniques that can visualize cellular processes at nanoscale resolution. TIRM has gained significant traction in this space due to its ability to visualize membrane-proximal events with minimal photodamage to living specimens. Market research indicates that approximately 65% of academic life science research facilities now incorporate some form of TIRM in their imaging suites, particularly for studies involving membrane trafficking and cell adhesion dynamics.
The semiconductor and materials science industries have emerged as primary adopters of AFM technology, where precise surface characterization is critical for quality control and R&D. The global semiconductor inspection equipment market, where AFM plays a crucial role, has been expanding at a compound annual growth rate of 8.3% since 2018, reaching a market value of $4.9 billion in 2022.
Healthcare applications represent another significant market driver, with clinical diagnostics increasingly utilizing advanced microscopy for disease detection and characterization. TIRM's efficiency in visualizing single-molecule interactions has positioned it as a valuable tool in pharmaceutical development, particularly for antibody characterization and drug-target interaction studies.
Regional market analysis reveals that North America currently dominates the advanced microscopy market with approximately 38% share, followed by Europe (29%) and Asia-Pacific (24%). However, the Asia-Pacific region is experiencing the fastest growth rate, driven by expanding research infrastructure in China, Japan, and South Korea.
Cost considerations significantly influence market adoption patterns. AFM systems typically require higher initial investment (average system cost ranging from $100,000 to $500,000) compared to TIRM setups. However, the total cost of ownership analysis reveals that TIRM often incurs higher operational expenses due to specialized fluorophores and optical components with limited lifespans.
Market forecasts suggest that the efficiency advantages of each technology will drive specialized adoption rather than direct competition. TIRM's efficiency in real-time dynamic imaging of biological processes complements AFM's efficiency in high-precision topographical mapping. This complementary relationship is reflected in purchasing patterns, with approximately 42% of research institutions maintaining both technologies to address different experimental needs.
The industrial quality control segment represents an emerging market opportunity, particularly for AFM, with manufacturing sectors increasingly adopting automated AFM systems for inline inspection processes. This application area is projected to grow at 11.2% annually through 2027, outpacing the overall microscopy market growth rate.
Current Technical Limitations and Challenges
Despite significant advancements in microscopy technologies, both Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) face substantial technical limitations that impact their efficiency and broader application. AFM, while offering exceptional resolution at the nanoscale, struggles with slow scanning speeds that significantly restrict throughput. A typical high-resolution AFM scan of a 10×10 μm area may require 30-60 minutes, making real-time imaging of dynamic biological processes nearly impossible. This temporal limitation severely constrains AFM's utility in studying rapid cellular events that occur on timescales of seconds or milliseconds.
Sample preparation for AFM presents another major challenge, requiring specimens to be immobilized on substrates—a process that can potentially alter native biological structures or functions. The physical interaction between the AFM tip and sample also introduces concerns about sample damage, particularly with delicate biological specimens, limiting repeated measurements on the same sample area.
TIRM, while excelling at visualizing membrane-proximal events with minimal phototoxicity, suffers from diffraction-limited resolution (approximately 200-300 nm laterally), significantly inferior to AFM's capabilities. This resolution barrier prevents detailed visualization of molecular structures and nanoscale cellular components that are increasingly important in modern biological research.
The depth of field in TIRM is inherently restricted to approximately 100-200 nm from the coverslip surface, severely limiting its application to membrane-proximal events and preventing whole-cell or tissue-level imaging. This constraint makes TIRM unsuitable for studying intracellular structures beyond the basal membrane region.
Both technologies face substantial integration challenges with complementary techniques. AFM systems struggle with simultaneous correlation with fluorescence microscopy due to mechanical constraints and optical access limitations. TIRM, while more compatible with other optical methods, faces challenges in quantitative analysis and interpretation of signal intensity in relation to actual molecular distances from the interface.
Cost and accessibility remain significant barriers for both technologies. High-end AFM systems can exceed $500,000, while TIRM requires specialized optical components and expertise. The complexity of operation demands extensive training, limiting widespread adoption in standard research laboratories and clinical settings.
Environmental sensitivity presents another shared challenge, with both techniques requiring vibration isolation and temperature stability. AFM is particularly vulnerable to acoustic noise and thermal drift, while TIRM performance can be compromised by refractive index variations in the imaging medium or optical aberrations in the light path.
Sample preparation for AFM presents another major challenge, requiring specimens to be immobilized on substrates—a process that can potentially alter native biological structures or functions. The physical interaction between the AFM tip and sample also introduces concerns about sample damage, particularly with delicate biological specimens, limiting repeated measurements on the same sample area.
TIRM, while excelling at visualizing membrane-proximal events with minimal phototoxicity, suffers from diffraction-limited resolution (approximately 200-300 nm laterally), significantly inferior to AFM's capabilities. This resolution barrier prevents detailed visualization of molecular structures and nanoscale cellular components that are increasingly important in modern biological research.
The depth of field in TIRM is inherently restricted to approximately 100-200 nm from the coverslip surface, severely limiting its application to membrane-proximal events and preventing whole-cell or tissue-level imaging. This constraint makes TIRM unsuitable for studying intracellular structures beyond the basal membrane region.
Both technologies face substantial integration challenges with complementary techniques. AFM systems struggle with simultaneous correlation with fluorescence microscopy due to mechanical constraints and optical access limitations. TIRM, while more compatible with other optical methods, faces challenges in quantitative analysis and interpretation of signal intensity in relation to actual molecular distances from the interface.
Cost and accessibility remain significant barriers for both technologies. High-end AFM systems can exceed $500,000, while TIRM requires specialized optical components and expertise. The complexity of operation demands extensive training, limiting widespread adoption in standard research laboratories and clinical settings.
Environmental sensitivity presents another shared challenge, with both techniques requiring vibration isolation and temperature stability. AFM is particularly vulnerable to acoustic noise and thermal drift, while TIRM performance can be compromised by refractive index variations in the imaging medium or optical aberrations in the light path.
Comparative Analysis of AFM and TIRM Solutions
01 Combined AFM and TIRM systems for enhanced imaging
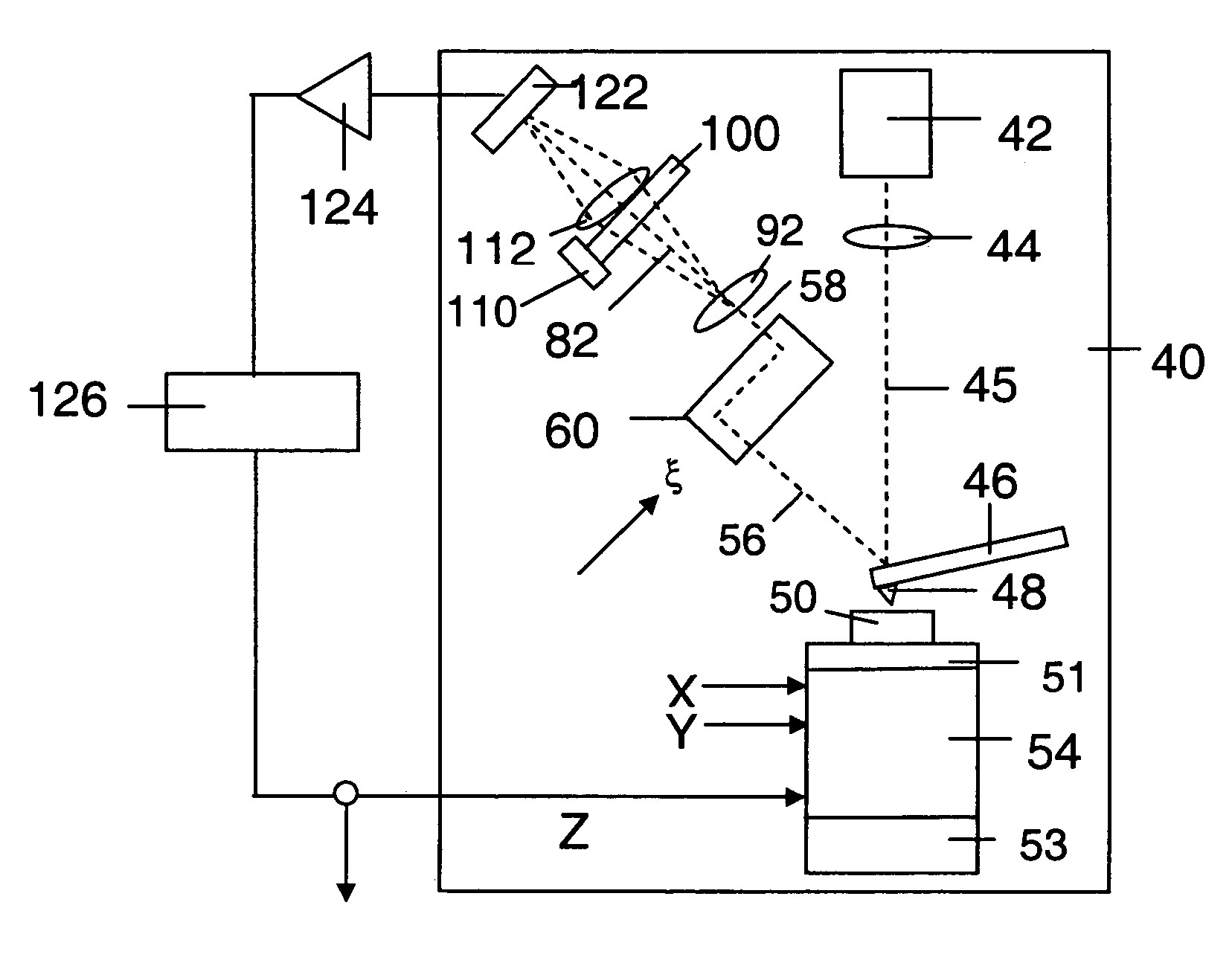
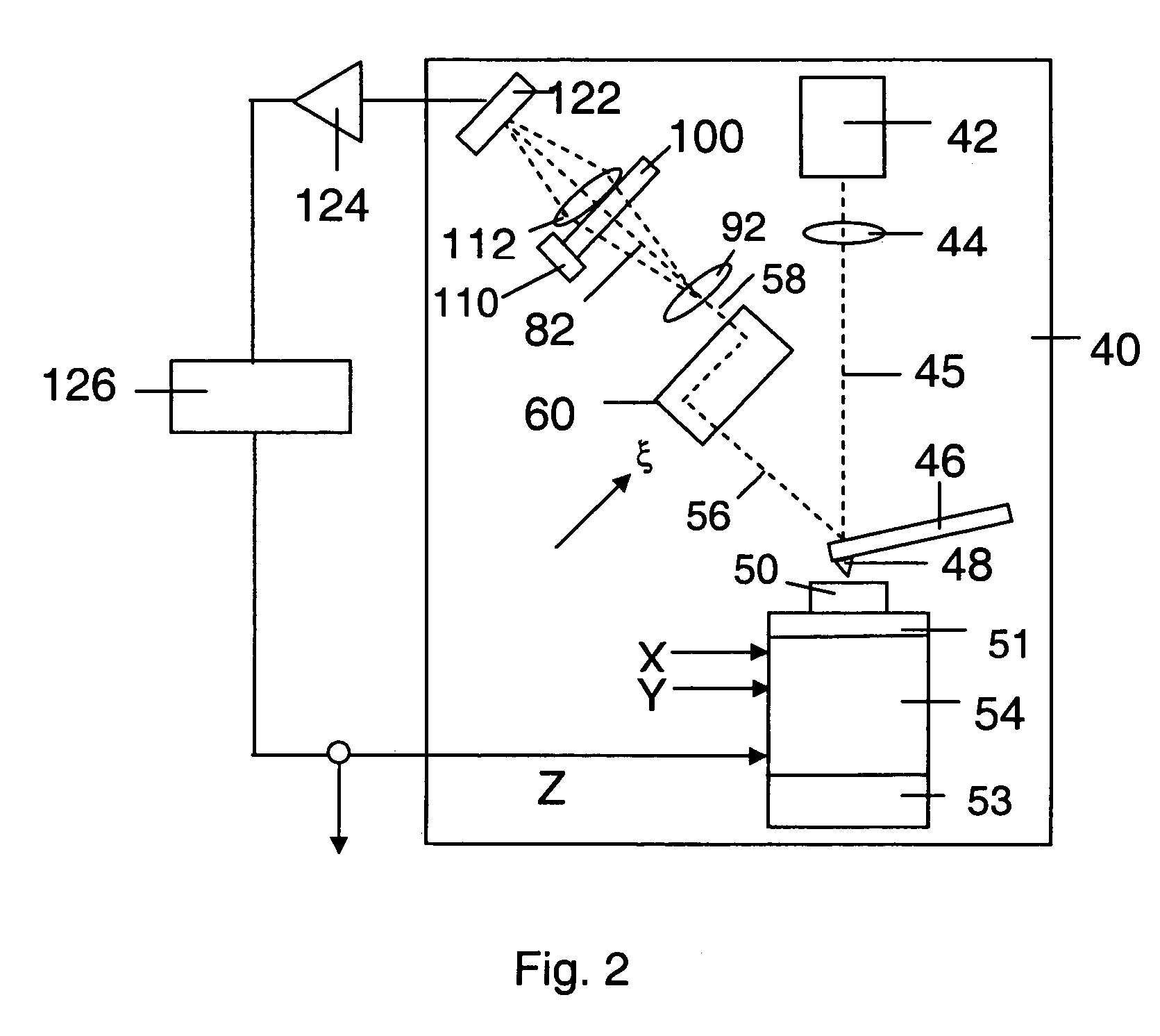
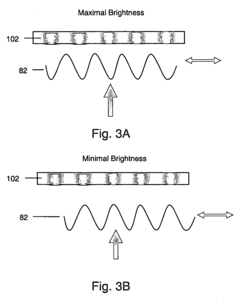
Integration of Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) into combined systems allows for simultaneous topographical and optical measurements. This combination enhances imaging capabilities by providing complementary data about sample surfaces and subsurface features. The integrated approach improves efficiency by allowing correlation between mechanical properties measured by AFM and optical properties detected by TIRM in a single measurement session.- Combined AFM and TIRM systems for enhanced imaging: Systems that integrate Atomic Force Microscopy (AFM) with Total Internal Reflection Microscopy (TIRM) provide complementary imaging capabilities. This combination allows for simultaneous topographical measurements from AFM and optical information from TIRM, enhancing the efficiency of sample analysis. These integrated systems enable researchers to correlate surface structures with optical properties at the nanoscale level.
- Efficiency improvements in AFM probe designs: Advanced probe designs for Atomic Force Microscopy significantly improve imaging efficiency and resolution. These innovations include specialized tip geometries, materials, and coatings that enhance sensitivity and reduce artifacts. Some designs incorporate optical elements that work in conjunction with TIRM techniques, allowing for more efficient data collection and reduced sample damage during extended imaging sessions.
- TIRM illumination and detection optimization: Optimized illumination and detection systems for Total Internal Reflection Microscopy improve efficiency by maximizing signal-to-noise ratios. These systems employ specialized optical components, precise angle control mechanisms, and advanced light sources to enhance evanescent field generation. Improved detection schemes, including high-sensitivity cameras and spectral filtering, allow for better visualization of surface phenomena with reduced background interference.
- Software and algorithms for microscopy data processing: Advanced software solutions and algorithms significantly enhance the efficiency of both AFM and TIRM data processing. These computational tools enable real-time image correction, noise reduction, and feature extraction. Machine learning approaches help automate analysis tasks, while specialized algorithms facilitate the correlation of data from multiple microscopy techniques, allowing researchers to extract more meaningful information from combined AFM-TIRM datasets.
- Sample preparation techniques for dual-mode microscopy: Specialized sample preparation methods optimize specimens for simultaneous analysis by both AFM and TIRM. These techniques include surface treatments that enhance optical contrast while maintaining topographical features, substrate modifications that improve evanescent field generation, and environmental control systems that stabilize samples during extended imaging sessions. Proper sample preparation significantly increases the efficiency and reliability of measurements across both microscopy techniques.
02 Efficiency improvements in AFM probe technology
Advanced probe designs for Atomic Force Microscopy significantly improve measurement efficiency and accuracy. These innovations include specialized tip geometries, cantilever materials, and coating technologies that enhance sensitivity and durability. Some designs incorporate feedback mechanisms that optimize tip-sample interactions, reducing artifacts and improving resolution. These technological advancements allow for faster scanning speeds while maintaining high-quality imaging results.Expand Specific Solutions03 Optical configurations for enhanced TIRM performance
Specialized optical configurations in Total Internal Reflection Microscopy improve light coupling efficiency and detection sensitivity. These designs optimize the evanescent field generation at the interface, enhancing signal-to-noise ratio and imaging depth. Advanced optical components such as specialized objectives, beam shapers, and custom prism designs maximize the total internal reflection effect. These configurations enable more efficient illumination and collection of the reflected light, resulting in higher quality images with improved contrast.Expand Specific Solutions04 Signal processing and data analysis methods
Advanced signal processing and data analysis methods enhance the efficiency of both AFM and TIRM systems. These techniques include noise reduction algorithms, image enhancement procedures, and automated feature recognition. Machine learning approaches are increasingly applied to interpret complex microscopy data, enabling faster and more accurate analysis. Real-time processing capabilities allow for immediate feedback during measurements, optimizing experimental parameters and reducing the need for repeated scans.Expand Specific Solutions05 Sample preparation techniques for improved microscopy efficiency
Specialized sample preparation methods significantly impact the efficiency and quality of both AFM and TIRM measurements. These techniques include surface functionalization, substrate selection, and environmental control during sample mounting. Proper preparation minimizes contamination and reduces artifacts that can interfere with imaging. Advanced sample holders and positioning systems enable precise alignment between the sample and the microscope components, facilitating more efficient data acquisition and reproducible results.Expand Specific Solutions
Leading Manufacturers and Research Institutions
Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) compete in a maturing market with growing applications in nanoscale imaging. The industry is experiencing steady growth, with an estimated market size of $1.5-2 billion annually. AFM technology, championed by companies like Bruker Nano, JEOL, and Olympus, offers superior resolution for surface topography but at higher costs and slower scanning speeds. TIRM, advanced by Leica Microsystems, Canon, and Agilent Technologies, provides better real-time imaging of biological samples with less sample preparation. Academic institutions (including Beihang University, National University of Singapore, and McGill University) are driving innovation through collaborative research with industry leaders, pushing both technologies toward greater efficiency and specialized applications in semiconductor, biological, and materials science fields.
Bruker Nano, Inc.
Technical Solution: Bruker Nano has developed advanced Atomic Force Microscopy (AFM) systems with proprietary PeakForce Tapping technology that allows simultaneous acquisition of multiple sample properties while protecting both tip and sample. Their BioScope Resolve AFM system integrates with optical microscopes to provide correlative microscopy capabilities, enabling researchers to combine AFM's nanoscale resolution with fluorescence imaging. Bruker's FastScan technology allows imaging speeds up to 20 times faster than conventional AFMs, with scan rates reaching 125 Hz. Their systems feature automated probe exchange and alignment, reducing setup time from hours to minutes. Bruker has also developed specialized AFM probes with tailored properties for specific applications, including high-resolution imaging in liquid environments, which is particularly valuable for biological samples[1][2]. Their systems offer force measurement capabilities down to piconewton levels, enabling detailed studies of molecular interactions.
Strengths: Industry-leading resolution (sub-nanometer) and force sensitivity (piconewton range); extensive automation reducing operator expertise requirements; versatile platform supporting multiple measurement modes. Weaknesses: Higher cost compared to academic-focused systems; complex operation requiring specialized training despite automation; larger footprint than some competing systems.
Leica Microsystems CMS GmbH
Technical Solution: Leica Microsystems has pioneered advanced Total Internal Reflection Fluorescence (TIRF) microscopy systems that utilize the evanescent wave generated when light is totally internally reflected at the glass-water interface. Their DMi8 S platform features a proprietary infinity port design that enables simultaneous multi-channel TIRF imaging with precise laser angle control, allowing researchers to adjust penetration depth from 70-300nm with nanometer precision. Leica's Infinity TIRF module provides automated laser alignment and calibration, ensuring reproducible illumination across experiments. Their systems incorporate adaptive focus control technology that maintains focus stability over extended time-lapse experiments, critical for live-cell imaging. Leica has also developed specialized objectives with high numerical apertures (>1.45) specifically optimized for TIRF applications, maximizing light collection efficiency. Their THUNDER technology employs computational clearing to remove out-of-focus blur in real-time, enhancing contrast in TIRF images[3][4]. Recent innovations include multi-angle TIRF capabilities that enable 3D reconstruction of membrane-proximal structures.
Strengths: Exceptional signal-to-noise ratio for membrane studies; non-invasive live cell imaging capabilities; compatibility with various fluorophores and simultaneous multi-color imaging; relatively simple sample preparation. Weaknesses: Limited to studying structures within 300nm of the coverslip; requires specialized high-NA objectives; more expensive than conventional fluorescence microscopy; potential photobleaching during extended imaging sessions.
Key Patents and Technical Innovations
High sensitivity scanning probe system
PatentInactiveUS7230719B2
Innovation
- The system employs a scanning probe microscope with an interferometer that splits and recombines light beams to form an interference pattern, using a transmission grating and actuators to modulate the fringe pattern and cancel noise, allowing for enhanced sensitivity and independent measurement of bending and torsion modes without the need for complex alignment or expensive manufacturing.
Multi-head AFM for defect inspection and review
PatentWO2024215203A1
Innovation
- A multi-head scanning probe microscope system with multiple scanning heads, each capable of operating in different modes: a high-speed, low-resolution first scanning mode and a high-resolution second scanning mode, allowing for simultaneous scanning and verification of defects, with the substrate carrier facilitating precise movement and tip types optimized for each mode.
Cost-Benefit Analysis and ROI Considerations
When evaluating the financial implications of implementing either Atomic Force Microscopy (AFM) or Total Internal Reflection Microscopy (TIRM) technologies, organizations must conduct thorough cost-benefit analyses to determine the most efficient allocation of resources. The initial capital expenditure for AFM systems typically ranges from $100,000 to $500,000, significantly higher than TIRM systems which generally cost between $50,000 and $200,000. This substantial difference in upfront investment necessitates careful consideration of long-term returns.
Operational expenses present another critical dimension for comparison. AFM systems require specialized probe tips that need regular replacement (approximately $100-300 per tip), with each tip having a limited lifespan depending on usage intensity. Additionally, AFM systems consume more electricity and demand more stringent environmental controls. TIRM systems, while having lower consumable costs, often require specialized optical components and precise alignment maintenance, resulting in different but still significant operational expenditures.
The return on investment timeline differs markedly between these technologies. AFM typically demonstrates a longer ROI period (3-5 years) but offers versatility across multiple research domains and applications. TIRM may show faster ROI (2-3 years) in specialized applications, particularly in biological imaging where its non-invasive nature provides distinct advantages. Organizations must align these ROI considerations with their specific research objectives and funding cycles.
Productivity metrics reveal that while AFM offers higher resolution capabilities, the sample preparation time and imaging process can be significantly longer than TIRM. In high-throughput environments, TIRM's faster imaging capabilities may translate to greater sample processing efficiency, potentially offsetting its lower resolution limitations in certain applications. This throughput advantage can substantially impact the cost-per-result calculation in production environments.
Maintenance costs over a five-year operational period typically amount to 15-20% of the initial investment for AFM systems, compared to 10-15% for TIRM systems. However, the availability of service contracts and the organization's internal technical expertise can significantly influence these figures. Organizations with established microscopy facilities may leverage existing expertise to reduce maintenance costs for either technology.
Grant funding opportunities and collaborative research potential should also factor into the financial analysis. AFM technologies often attract broader funding opportunities due to their wider application range, while TIRM may access specialized funding in fields like cell biology and membrane research. The potential for generating intellectual property and commercialization opportunities differs between these technologies and should be quantified in comprehensive ROI calculations.
Operational expenses present another critical dimension for comparison. AFM systems require specialized probe tips that need regular replacement (approximately $100-300 per tip), with each tip having a limited lifespan depending on usage intensity. Additionally, AFM systems consume more electricity and demand more stringent environmental controls. TIRM systems, while having lower consumable costs, often require specialized optical components and precise alignment maintenance, resulting in different but still significant operational expenditures.
The return on investment timeline differs markedly between these technologies. AFM typically demonstrates a longer ROI period (3-5 years) but offers versatility across multiple research domains and applications. TIRM may show faster ROI (2-3 years) in specialized applications, particularly in biological imaging where its non-invasive nature provides distinct advantages. Organizations must align these ROI considerations with their specific research objectives and funding cycles.
Productivity metrics reveal that while AFM offers higher resolution capabilities, the sample preparation time and imaging process can be significantly longer than TIRM. In high-throughput environments, TIRM's faster imaging capabilities may translate to greater sample processing efficiency, potentially offsetting its lower resolution limitations in certain applications. This throughput advantage can substantially impact the cost-per-result calculation in production environments.
Maintenance costs over a five-year operational period typically amount to 15-20% of the initial investment for AFM systems, compared to 10-15% for TIRM systems. However, the availability of service contracts and the organization's internal technical expertise can significantly influence these figures. Organizations with established microscopy facilities may leverage existing expertise to reduce maintenance costs for either technology.
Grant funding opportunities and collaborative research potential should also factor into the financial analysis. AFM technologies often attract broader funding opportunities due to their wider application range, while TIRM may access specialized funding in fields like cell biology and membrane research. The potential for generating intellectual property and commercialization opportunities differs between these technologies and should be quantified in comprehensive ROI calculations.
Integration Possibilities with Emerging Technologies
The integration of Atomic Force Microscopy (AFM) and Total Internal Reflection Microscopy (TIRM) with emerging technologies presents significant opportunities for advancing microscopy capabilities. Machine learning algorithms are increasingly being applied to both AFM and TIRM data processing, enabling automated feature recognition, noise reduction, and enhanced image resolution. These computational approaches can compensate for efficiency limitations in both techniques, particularly addressing AFM's slower scanning speeds and TIRM's limited resolution in certain applications.
Quantum sensing technologies represent another promising integration pathway. Quantum-enhanced AFM probes utilizing nitrogen-vacancy centers in diamond can achieve unprecedented sensitivity levels while maintaining the spatial resolution advantages of traditional AFM. Similarly, quantum light sources could enhance TIRM's detection capabilities beyond classical limits, potentially overcoming its efficiency constraints in low-light conditions.
Microfluidic platforms are revolutionizing sample preparation and handling for both microscopy techniques. Integration of AFM or TIRM with lab-on-a-chip devices enables real-time imaging of biological processes under precisely controlled conditions. This combination particularly benefits TIRM, which excels at interface studies, by providing controlled environmental parameters that maximize its efficiency advantages.
Correlative microscopy approaches that combine AFM or TIRM with complementary techniques are gaining traction. For instance, integrating AFM with super-resolution optical microscopy creates powerful hybrid systems that leverage the mechanical sensitivity of AFM with the molecular specificity of fluorescence techniques. Similarly, TIRM combined with Raman spectroscopy provides both interfacial dynamics visualization and chemical composition analysis simultaneously.
Automation and robotics integration represents a significant efficiency enhancement for both techniques. Automated sample handling, probe positioning, and measurement protocols can dramatically increase throughput for AFM, addressing one of its primary efficiency limitations. For TIRM, automated focus maintenance and illumination angle optimization can ensure consistent performance across extended imaging sessions.
Cloud computing and edge processing architectures are enabling remote operation and collaborative analysis of microscopy data. These developments particularly benefit AFM, which generates complex multidimensional datasets that require substantial computational resources. Real-time data processing at the edge can accelerate AFM image formation, while cloud-based platforms facilitate advanced analysis without local hardware constraints.
Quantum sensing technologies represent another promising integration pathway. Quantum-enhanced AFM probes utilizing nitrogen-vacancy centers in diamond can achieve unprecedented sensitivity levels while maintaining the spatial resolution advantages of traditional AFM. Similarly, quantum light sources could enhance TIRM's detection capabilities beyond classical limits, potentially overcoming its efficiency constraints in low-light conditions.
Microfluidic platforms are revolutionizing sample preparation and handling for both microscopy techniques. Integration of AFM or TIRM with lab-on-a-chip devices enables real-time imaging of biological processes under precisely controlled conditions. This combination particularly benefits TIRM, which excels at interface studies, by providing controlled environmental parameters that maximize its efficiency advantages.
Correlative microscopy approaches that combine AFM or TIRM with complementary techniques are gaining traction. For instance, integrating AFM with super-resolution optical microscopy creates powerful hybrid systems that leverage the mechanical sensitivity of AFM with the molecular specificity of fluorescence techniques. Similarly, TIRM combined with Raman spectroscopy provides both interfacial dynamics visualization and chemical composition analysis simultaneously.
Automation and robotics integration represents a significant efficiency enhancement for both techniques. Automated sample handling, probe positioning, and measurement protocols can dramatically increase throughput for AFM, addressing one of its primary efficiency limitations. For TIRM, automated focus maintenance and illumination angle optimization can ensure consistent performance across extended imaging sessions.
Cloud computing and edge processing architectures are enabling remote operation and collaborative analysis of microscopy data. These developments particularly benefit AFM, which generates complex multidimensional datasets that require substantial computational resources. Real-time data processing at the edge can accelerate AFM image formation, while cloud-based platforms facilitate advanced analysis without local hardware constraints.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!