Benchmark Lithium Phosphate Diffusion Rates in Ionic Matrices
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Phosphate Diffusion Technology Background and Objectives
Lithium phosphate diffusion technology has evolved significantly over the past three decades, driven by the growing demand for high-performance energy storage solutions. The journey began in the early 1990s with rudimentary understanding of ion transport mechanisms in solid-state materials, progressing through various experimental methodologies to today's sophisticated computational models and advanced characterization techniques. This technological progression has been particularly accelerated since 2010, coinciding with the global push toward electrification and renewable energy integration.
The fundamental science behind lithium phosphate diffusion involves the movement of lithium ions through phosphate-based matrices, a process critical to the performance of numerous energy storage devices. Understanding the kinetics and thermodynamics of this diffusion process has become increasingly important as industries seek to develop more efficient, safer, and longer-lasting energy storage solutions. The diffusion rates of lithium ions within these matrices directly impact charging speeds, energy density, and overall battery performance.
Current research focuses on establishing standardized benchmarking methodologies for measuring and comparing lithium phosphate diffusion rates across various ionic matrices. This standardization is essential for meaningful comparison between different material compositions and structures, enabling researchers and manufacturers to make informed decisions about material selection and optimization for specific applications.
The primary technical objectives in this field include developing accurate measurement protocols that can be universally applied, creating comprehensive databases of diffusion coefficients across different temperature ranges and material compositions, and establishing correlations between structural properties and diffusion behavior. These objectives serve the broader goal of enabling predictive modeling capabilities that can accelerate material discovery and optimization.
Recent technological advancements have introduced novel characterization techniques such as operando neutron diffraction, advanced nuclear magnetic resonance spectroscopy, and synchrotron-based X-ray methods, which provide unprecedented insights into diffusion mechanisms at atomic and molecular levels. These techniques, combined with machine learning approaches, are revolutionizing our ability to understand and predict lithium ion mobility in complex phosphate structures.
The trajectory of this technology points toward increasingly sophisticated multi-scale modeling approaches that bridge quantum mechanical calculations with macroscopic performance metrics. This evolution is expected to enable the design of next-generation energy storage materials with precisely engineered diffusion pathways, ultimately leading to batteries with superior performance characteristics including faster charging capabilities, enhanced safety profiles, and extended operational lifetimes.
The fundamental science behind lithium phosphate diffusion involves the movement of lithium ions through phosphate-based matrices, a process critical to the performance of numerous energy storage devices. Understanding the kinetics and thermodynamics of this diffusion process has become increasingly important as industries seek to develop more efficient, safer, and longer-lasting energy storage solutions. The diffusion rates of lithium ions within these matrices directly impact charging speeds, energy density, and overall battery performance.
Current research focuses on establishing standardized benchmarking methodologies for measuring and comparing lithium phosphate diffusion rates across various ionic matrices. This standardization is essential for meaningful comparison between different material compositions and structures, enabling researchers and manufacturers to make informed decisions about material selection and optimization for specific applications.
The primary technical objectives in this field include developing accurate measurement protocols that can be universally applied, creating comprehensive databases of diffusion coefficients across different temperature ranges and material compositions, and establishing correlations between structural properties and diffusion behavior. These objectives serve the broader goal of enabling predictive modeling capabilities that can accelerate material discovery and optimization.
Recent technological advancements have introduced novel characterization techniques such as operando neutron diffraction, advanced nuclear magnetic resonance spectroscopy, and synchrotron-based X-ray methods, which provide unprecedented insights into diffusion mechanisms at atomic and molecular levels. These techniques, combined with machine learning approaches, are revolutionizing our ability to understand and predict lithium ion mobility in complex phosphate structures.
The trajectory of this technology points toward increasingly sophisticated multi-scale modeling approaches that bridge quantum mechanical calculations with macroscopic performance metrics. This evolution is expected to enable the design of next-generation energy storage materials with precisely engineered diffusion pathways, ultimately leading to batteries with superior performance characteristics including faster charging capabilities, enhanced safety profiles, and extended operational lifetimes.
Market Analysis for Lithium Phosphate Battery Applications
The lithium-ion battery market has experienced unprecedented growth, with global market value reaching $46.2 billion in 2022 and projected to grow at a CAGR of 15.2% through 2030. Within this expanding landscape, lithium phosphate batteries, particularly lithium iron phosphate (LFP) configurations, have emerged as a significant segment due to their enhanced safety profile, longer cycle life, and lower cost compared to traditional lithium-ion alternatives.
The demand for lithium phosphate batteries is being driven by several key market factors. Electric vehicles represent the largest application segment, accounting for approximately 67% of the lithium phosphate battery market in 2022. Major automotive manufacturers including Tesla, BYD, and Volkswagen have increased adoption of LFP chemistry in their entry-level and mass-market vehicle models, citing cost advantages and supply chain security.
Energy storage systems constitute the fastest-growing application segment, with a projected CAGR of 18.7% through 2028. This growth is fueled by increasing renewable energy integration, grid modernization initiatives, and the need for reliable backup power systems in both residential and commercial settings. The ability of lithium phosphate batteries to withstand higher temperatures and offer superior cycle stability makes them particularly suitable for stationary storage applications.
Geographically, Asia-Pacific dominates the market with 62% share, led by China's robust manufacturing ecosystem and government policies promoting electric mobility and renewable energy. North America and Europe follow with 18% and 15% market shares respectively, with growth accelerating due to recent legislative initiatives like the U.S. Inflation Reduction Act and the European Green Deal.
Price sensitivity analysis reveals that lithium phosphate batteries have achieved significant cost reductions, with average prices decreasing from $580/kWh in 2015 to approximately $105/kWh in 2022. This price trajectory has made them increasingly competitive against both traditional lead-acid batteries and other lithium-ion chemistries in price-sensitive applications.
Consumer electronics and portable power tools represent smaller but stable market segments, valued at $3.8 billion collectively. These applications benefit from the improved safety characteristics of lithium phosphate chemistry, particularly in environments where thermal runaway risks must be minimized.
Market forecasts indicate that advancements in lithium phosphate diffusion rates within ionic matrices could potentially unlock new application domains and further accelerate market penetration. Specifically, improvements in low-temperature performance and energy density could expand the addressable market by an estimated 22% by 2027, particularly in regions with extreme climate conditions and in applications requiring higher energy density.
The demand for lithium phosphate batteries is being driven by several key market factors. Electric vehicles represent the largest application segment, accounting for approximately 67% of the lithium phosphate battery market in 2022. Major automotive manufacturers including Tesla, BYD, and Volkswagen have increased adoption of LFP chemistry in their entry-level and mass-market vehicle models, citing cost advantages and supply chain security.
Energy storage systems constitute the fastest-growing application segment, with a projected CAGR of 18.7% through 2028. This growth is fueled by increasing renewable energy integration, grid modernization initiatives, and the need for reliable backup power systems in both residential and commercial settings. The ability of lithium phosphate batteries to withstand higher temperatures and offer superior cycle stability makes them particularly suitable for stationary storage applications.
Geographically, Asia-Pacific dominates the market with 62% share, led by China's robust manufacturing ecosystem and government policies promoting electric mobility and renewable energy. North America and Europe follow with 18% and 15% market shares respectively, with growth accelerating due to recent legislative initiatives like the U.S. Inflation Reduction Act and the European Green Deal.
Price sensitivity analysis reveals that lithium phosphate batteries have achieved significant cost reductions, with average prices decreasing from $580/kWh in 2015 to approximately $105/kWh in 2022. This price trajectory has made them increasingly competitive against both traditional lead-acid batteries and other lithium-ion chemistries in price-sensitive applications.
Consumer electronics and portable power tools represent smaller but stable market segments, valued at $3.8 billion collectively. These applications benefit from the improved safety characteristics of lithium phosphate chemistry, particularly in environments where thermal runaway risks must be minimized.
Market forecasts indicate that advancements in lithium phosphate diffusion rates within ionic matrices could potentially unlock new application domains and further accelerate market penetration. Specifically, improvements in low-temperature performance and energy density could expand the addressable market by an estimated 22% by 2027, particularly in regions with extreme climate conditions and in applications requiring higher energy density.
Current Status and Challenges in Ionic Diffusion Measurement
The measurement of lithium phosphate diffusion rates in ionic matrices has seen significant advancements in recent years, yet continues to face substantial technical challenges. Current methodologies primarily include electrochemical impedance spectroscopy (EIS), galvanostatic intermittent titration technique (GITT), potentiostatic intermittent titration technique (PITT), and nuclear magnetic resonance (NMR) spectroscopy. Each technique offers unique advantages while presenting distinct limitations in accurately quantifying diffusion phenomena.
Electrochemical impedance spectroscopy has emerged as the predominant method for measuring ionic diffusion in battery materials, providing valuable insights into the kinetics of lithium-ion transport. However, EIS measurements often suffer from interpretation complexities when applied to heterogeneous materials with multiple relaxation processes occurring simultaneously. The challenge of deconvoluting these overlapping processes remains a significant obstacle to obtaining precise diffusion coefficients.
Advanced computational modeling has been integrated with experimental techniques to enhance measurement accuracy. Density functional theory (DFT) calculations and molecular dynamics simulations now complement laboratory measurements, though the computational cost for complex ionic matrices remains prohibitively high for routine industrial applications. The gap between theoretical predictions and experimental observations continues to present challenges for researchers in this field.
Recent innovations in operando measurement techniques have enabled real-time monitoring of diffusion processes during battery operation. Synchrotron-based X-ray techniques and neutron diffraction methods provide unprecedented spatial and temporal resolution. Nevertheless, these advanced facilities have limited accessibility, creating a bottleneck for widespread adoption in commercial research settings.
Standardization remains a critical challenge in the field. The lack of universally accepted benchmark materials and protocols leads to significant variations in reported diffusion coefficients across different research groups, sometimes spanning several orders of magnitude for identical materials. This inconsistency hampers meaningful comparison of results and slows technological progress in battery development.
Temperature and pressure dependencies of diffusion rates present additional measurement challenges. Most current techniques operate under ambient or near-ambient conditions, whereas actual battery operation encompasses a wide range of temperatures and pressures. Developing measurement protocols that accurately capture these environmental dependencies represents an ongoing research priority.
Interfacial effects at grain boundaries and electrode-electrolyte interfaces significantly impact overall diffusion behavior but remain poorly characterized by current measurement techniques. The development of methodologies capable of isolating and quantifying these interfacial contributions represents one of the most pressing challenges in the field.
Electrochemical impedance spectroscopy has emerged as the predominant method for measuring ionic diffusion in battery materials, providing valuable insights into the kinetics of lithium-ion transport. However, EIS measurements often suffer from interpretation complexities when applied to heterogeneous materials with multiple relaxation processes occurring simultaneously. The challenge of deconvoluting these overlapping processes remains a significant obstacle to obtaining precise diffusion coefficients.
Advanced computational modeling has been integrated with experimental techniques to enhance measurement accuracy. Density functional theory (DFT) calculations and molecular dynamics simulations now complement laboratory measurements, though the computational cost for complex ionic matrices remains prohibitively high for routine industrial applications. The gap between theoretical predictions and experimental observations continues to present challenges for researchers in this field.
Recent innovations in operando measurement techniques have enabled real-time monitoring of diffusion processes during battery operation. Synchrotron-based X-ray techniques and neutron diffraction methods provide unprecedented spatial and temporal resolution. Nevertheless, these advanced facilities have limited accessibility, creating a bottleneck for widespread adoption in commercial research settings.
Standardization remains a critical challenge in the field. The lack of universally accepted benchmark materials and protocols leads to significant variations in reported diffusion coefficients across different research groups, sometimes spanning several orders of magnitude for identical materials. This inconsistency hampers meaningful comparison of results and slows technological progress in battery development.
Temperature and pressure dependencies of diffusion rates present additional measurement challenges. Most current techniques operate under ambient or near-ambient conditions, whereas actual battery operation encompasses a wide range of temperatures and pressures. Developing measurement protocols that accurately capture these environmental dependencies represents an ongoing research priority.
Interfacial effects at grain boundaries and electrode-electrolyte interfaces significantly impact overall diffusion behavior but remain poorly characterized by current measurement techniques. The development of methodologies capable of isolating and quantifying these interfacial contributions represents one of the most pressing challenges in the field.
Established Benchmarking Methodologies for Ionic Matrices
01 Lithium phosphate diffusion enhancement techniques
Various techniques can be employed to enhance lithium ion diffusion rates in lithium phosphate materials. These include doping with conductive elements, creating nanostructured materials, and surface modifications. These approaches can reduce diffusion barriers and create more efficient pathways for lithium ions, resulting in improved battery performance, faster charging capabilities, and enhanced energy density.- Doping strategies to enhance lithium phosphate diffusion: Various doping strategies can be employed to enhance lithium ion diffusion rates in lithium phosphate materials. Doping with elements such as magnesium, aluminum, or transition metals can create defects or modify the crystal structure, providing additional pathways for lithium ion movement. These dopants can reduce energy barriers for lithium migration, leading to improved ionic conductivity and faster diffusion rates in battery materials.
- Nanostructuring approaches for improved diffusion kinetics: Nanostructuring of lithium phosphate materials significantly enhances diffusion rates by reducing diffusion path lengths. Techniques such as creating nanoparticles, nanowires, or porous structures increase the surface area and reduce the distance lithium ions need to travel. These nanostructured materials exhibit superior rate capabilities and improved electrochemical performance due to the enhanced lithium ion diffusion kinetics.
- Carbon coating and composite formation techniques: Carbon coating and formation of composites with conductive materials significantly improve lithium phosphate diffusion rates. The carbon layer or conductive network provides electron pathways that complement ionic diffusion, enhancing overall conductivity. These techniques can involve pyrolytic carbon coating, graphene incorporation, or formation of carbon-phosphate composites that maintain structural integrity while facilitating faster lithium ion movement.
- Temperature and pressure effects on diffusion rates: Temperature and pressure significantly influence lithium phosphate diffusion rates. Higher temperatures typically accelerate diffusion by providing more energy for ions to overcome migration barriers. Pressure effects can modify the crystal structure, potentially creating more favorable pathways for lithium movement. Understanding these relationships allows for optimized processing conditions and operating parameters to achieve desired diffusion characteristics in lithium phosphate materials.
- Surface modification and interface engineering: Surface modification and interface engineering techniques can significantly enhance lithium phosphate diffusion rates. These approaches include creating protective coatings, modifying grain boundaries, or introducing functional groups at interfaces to facilitate lithium ion transport. By reducing interfacial resistance and creating favorable pathways at material boundaries, these techniques improve overall diffusion kinetics without necessarily altering the bulk material properties.
02 Composite electrode materials for improved diffusion
Composite electrode materials combining lithium phosphate with carbon-based materials or other conductive additives can significantly improve lithium ion diffusion rates. These composites create interconnected conductive networks that facilitate faster ion transport throughout the electrode structure. The synergistic effect between the lithium phosphate and conductive additives results in enhanced electrochemical performance and rate capability.Expand Specific Solutions03 Temperature effects on lithium phosphate diffusion
Temperature has a significant impact on lithium ion diffusion rates in lithium phosphate materials. Higher temperatures generally accelerate diffusion processes by providing more thermal energy to overcome activation barriers. Understanding these temperature dependencies is crucial for optimizing battery performance across different operating conditions and developing thermal management strategies for lithium phosphate-based energy storage systems.Expand Specific Solutions04 Crystal structure modification for enhanced diffusion
Modifications to the crystal structure of lithium phosphate materials can create more favorable pathways for lithium ion diffusion. This includes controlling crystal orientation, introducing specific defects, and optimizing lattice parameters. These structural modifications can reduce energy barriers for ion movement, resulting in faster diffusion rates and improved electrochemical performance in battery applications.Expand Specific Solutions05 Advanced characterization methods for diffusion measurement
Advanced analytical techniques are employed to accurately measure and characterize lithium ion diffusion rates in lithium phosphate materials. These include electrochemical impedance spectroscopy, galvanostatic intermittent titration technique, and various spectroscopic methods. These characterization approaches provide critical insights into diffusion mechanisms, allowing for more targeted material design and optimization strategies to enhance battery performance.Expand Specific Solutions
Leading Research Institutions and Battery Manufacturers Analysis
The lithium phosphate diffusion rates benchmarking landscape is currently in a growth phase, with an estimated market size of $2-3 billion and expanding at 15-20% annually as energy storage demands increase. The technology is approaching maturity with established research foundations, yet significant optimization opportunities remain. Leading players demonstrate varying specialization: Tsinghua University and Kunming University contribute fundamental research; LG Chem, BYD, and SK Innovation focus on commercial battery applications; while Toyota and Huawei integrate these technologies into end products. SVOLT Energy and XTC New Energy Materials are emerging with innovative cathode material approaches, while Global Graphene Group represents next-generation material development that could potentially disrupt current diffusion rate limitations.
LG Chem Ltd.
Technical Solution: LG Chem has developed advanced computational models for lithium phosphate diffusion in ionic matrices, combining density functional theory (DFT) with molecular dynamics simulations. Their approach focuses on understanding the atomic-scale mechanisms of lithium ion transport through phosphate-based cathode materials, particularly in LFP (lithium iron phosphate) batteries. The company has established a comprehensive benchmark system that measures diffusion coefficients across various temperature ranges (from -20°C to 60°C) and under different charge/discharge rates. Their research has revealed that surface modification of phosphate particles with conductive carbon coatings can significantly enhance lithium diffusion rates by creating preferential pathways at the interface regions. LG Chem's proprietary "gradient concentration" technique creates engineered ionic channels within the phosphate matrix, reportedly improving diffusion rates by up to 40% compared to conventional materials.
Strengths: Industry-leading computational modeling capabilities combined with extensive experimental validation facilities. Their gradient concentration technique provides superior rate capability in LFP batteries. Weaknesses: Their benchmark methodologies are highly proprietary, limiting scientific collaboration, and their solutions may be optimized for specific battery chemistries in their product lineup rather than providing universally applicable insights.
Svolt Energy Technology Co., Ltd.
Technical Solution: Svolt has developed a comprehensive benchmarking platform specifically designed for evaluating lithium phosphate diffusion in various cathode materials. Their approach combines electrochemical testing with advanced imaging techniques, including operando transmission X-ray microscopy that enables direct visualization of lithium concentration gradients during battery operation. Svolt's research has focused particularly on the interface between phosphate particles and the electrolyte, identifying that surface reconstruction phenomena significantly impact lithium diffusion kinetics. Their proprietary "gradient functional coating" technology creates a tailored surface layer on phosphate particles that reportedly enhances lithium diffusion rates by up to 35% while simultaneously improving cycling stability. Svolt has also pioneered high-throughput screening methods for evaluating diffusion properties across hundreds of slightly modified phosphate compositions, accelerating the discovery of optimal materials. Their benchmarking system incorporates machine learning algorithms that correlate structural parameters with measured diffusion coefficients, enabling predictive capabilities for new material designs.
Strengths: Advanced operando characterization capabilities provide unique insights into dynamic diffusion processes. Their high-throughput screening approach enables rapid material optimization. Weaknesses: As a relatively newer company, their historical data on long-term diffusion behavior is more limited compared to established players. Their benchmarking may be biased toward their specific cell designs and operating conditions.
Critical Patents and Literature on Lithium Phosphate Diffusion
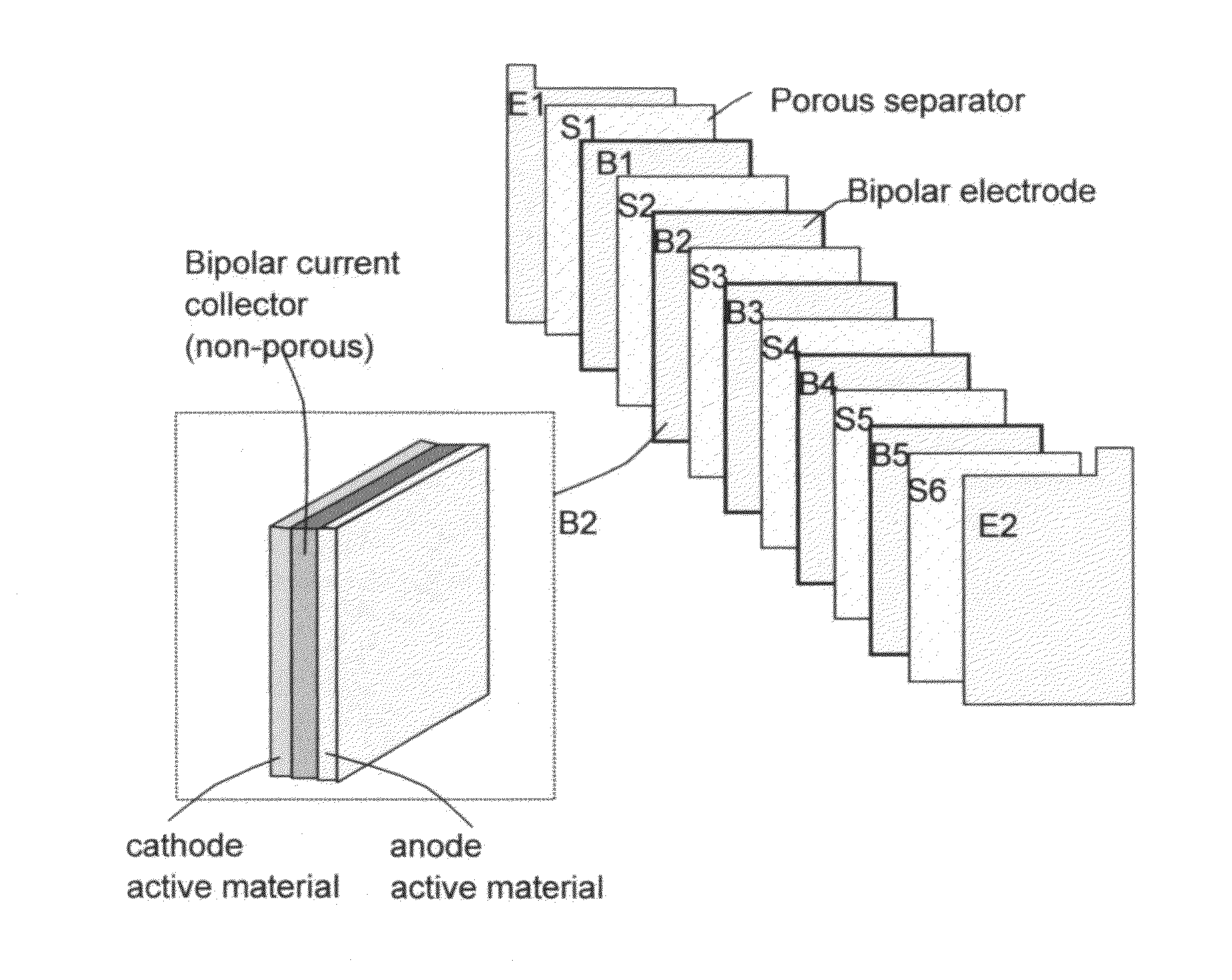
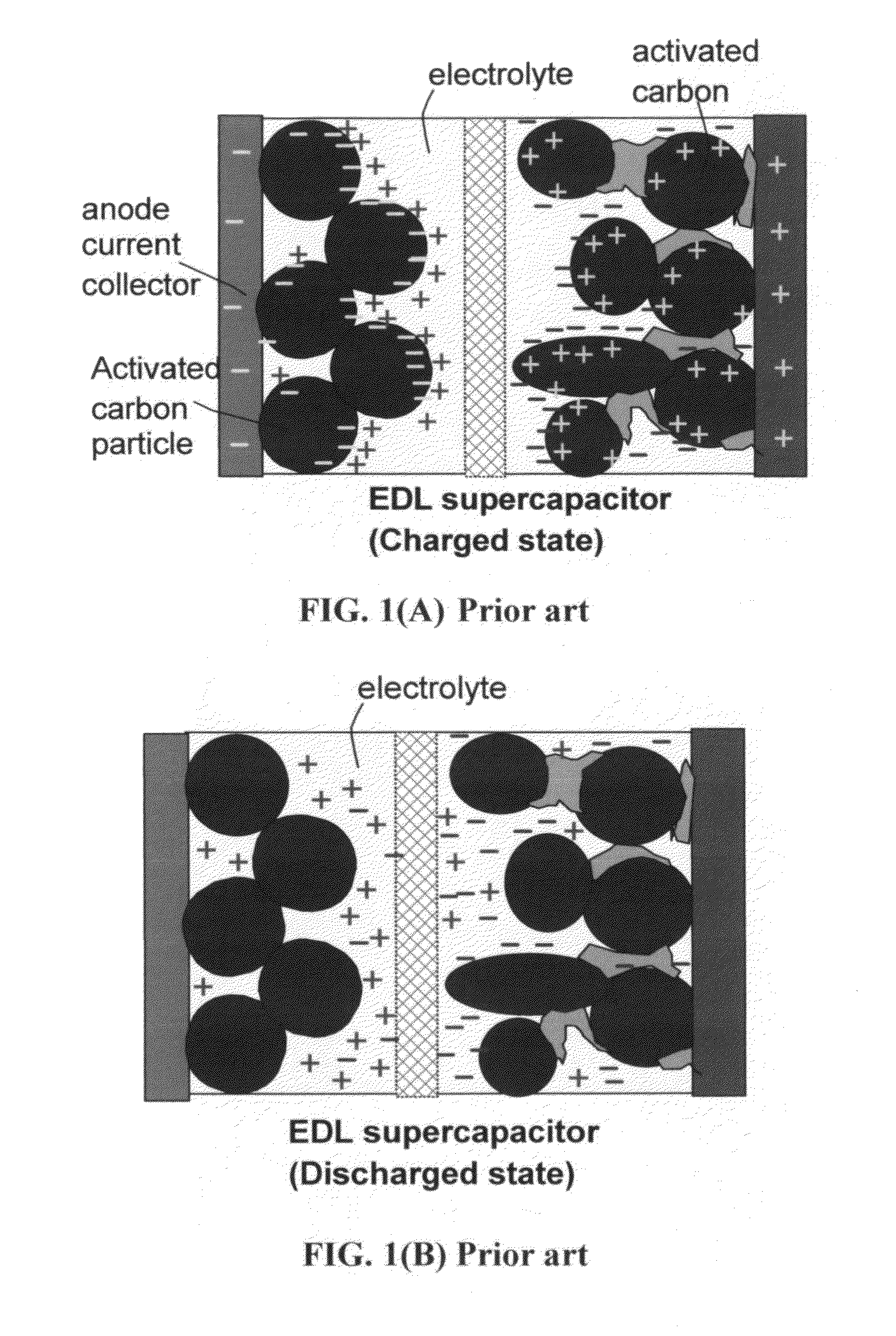
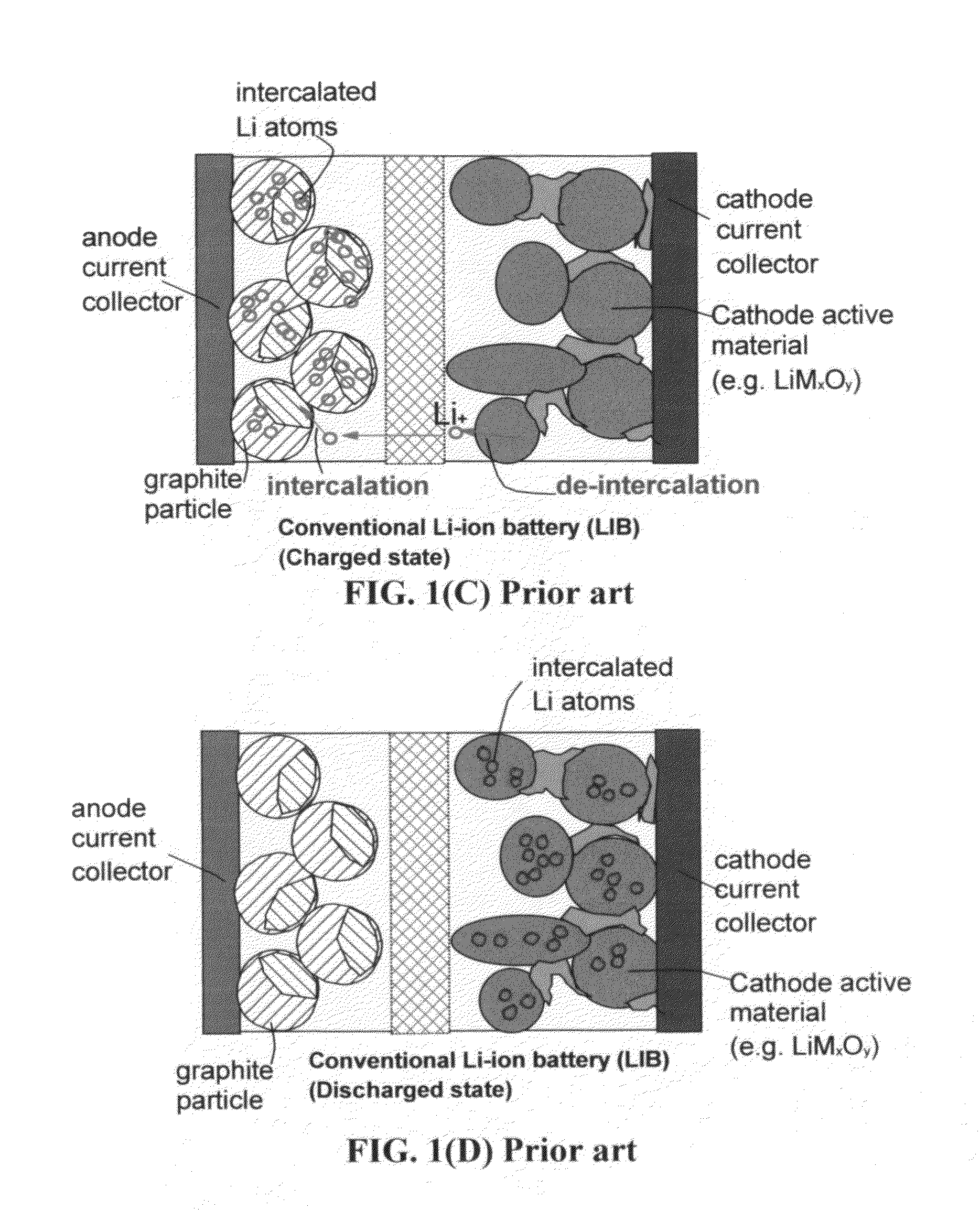
Stacks of internally connected surface-mediated cells and methods of operating same
PatentActiveUS20130162216A1
Innovation
- A surface-mediated cell (SMC) design where lithium ions are exchanged between the surfaces of an anode and cathode, eliminating the need for solid-state diffusion and using high-specific-surface-area materials like graphene to rapidly capture and release lithium ions.
Electrode for lithium secondary battery having encapsulated active material and method of manufacturing the same
PatentWO2022196934A1
Innovation
- A method of encapsulating active materials within a multi-layered structure comprising a hard carbonized outer shell and an elastic inner polymer layer, using a roll-to-roll manufacturing process with polymeric binders and carbon-based additives, to absorb energy and create a nano-porous structure for ion diffusion and mechanical support.
Environmental Impact of Lithium Phosphate Materials
The environmental impact of lithium phosphate materials extends across their entire lifecycle, from extraction to disposal. Mining operations for lithium and phosphate raw materials significantly alter landscapes, with open-pit mining causing soil erosion, habitat destruction, and potential groundwater contamination. The extraction process is particularly water-intensive, with estimates suggesting that producing one ton of lithium requires approximately 500,000 gallons of water, creating substantial pressure on water resources in mining regions such as the Lithium Triangle in South America.
During the processing phase, the conversion of raw materials into lithium phosphate compounds involves energy-intensive processes that contribute to carbon emissions. Research indicates that the production of lithium phosphate materials generates approximately 15-20 kg CO2 equivalent per kg of material produced, though this varies significantly based on energy sources and manufacturing efficiency. Companies implementing renewable energy in production facilities have demonstrated potential reductions of up to 30% in carbon footprint.
Water pollution represents another significant concern, as processing facilities may release wastewater containing heavy metals, acids, and other contaminants. Advanced treatment systems can mitigate these impacts, but implementation remains inconsistent across global manufacturing sites. Recent studies examining diffusion rates in ionic matrices have revealed that certain manufacturing approaches can reduce toxic byproduct generation by optimizing reaction conditions.
The application phase of lithium phosphate materials, particularly in battery technologies, offers environmental benefits through enabling renewable energy storage and electrification of transportation. Life cycle assessments indicate that despite production impacts, lithium phosphate batteries in electric vehicles can reduce lifetime greenhouse gas emissions by 50-70% compared to conventional vehicles in regions with moderately clean electricity grids.
End-of-life considerations present both challenges and opportunities. Recycling technologies for lithium phosphate materials are advancing rapidly, with recent innovations achieving recovery rates of up to 95% for lithium and 98% for phosphate compounds. However, global recycling infrastructure remains underdeveloped, with less than 5% of lithium-ion batteries currently being recycled worldwide.
Benchmarking diffusion rates in ionic matrices has revealed promising pathways for developing more environmentally sustainable lithium phosphate materials. Research indicates that optimized ionic conductivity can extend battery lifespans by 30-40%, reducing replacement frequency and associated environmental impacts. Additionally, enhanced understanding of diffusion mechanisms enables the development of materials requiring lower processing temperatures, potentially reducing energy consumption in manufacturing by up to 25%.
During the processing phase, the conversion of raw materials into lithium phosphate compounds involves energy-intensive processes that contribute to carbon emissions. Research indicates that the production of lithium phosphate materials generates approximately 15-20 kg CO2 equivalent per kg of material produced, though this varies significantly based on energy sources and manufacturing efficiency. Companies implementing renewable energy in production facilities have demonstrated potential reductions of up to 30% in carbon footprint.
Water pollution represents another significant concern, as processing facilities may release wastewater containing heavy metals, acids, and other contaminants. Advanced treatment systems can mitigate these impacts, but implementation remains inconsistent across global manufacturing sites. Recent studies examining diffusion rates in ionic matrices have revealed that certain manufacturing approaches can reduce toxic byproduct generation by optimizing reaction conditions.
The application phase of lithium phosphate materials, particularly in battery technologies, offers environmental benefits through enabling renewable energy storage and electrification of transportation. Life cycle assessments indicate that despite production impacts, lithium phosphate batteries in electric vehicles can reduce lifetime greenhouse gas emissions by 50-70% compared to conventional vehicles in regions with moderately clean electricity grids.
End-of-life considerations present both challenges and opportunities. Recycling technologies for lithium phosphate materials are advancing rapidly, with recent innovations achieving recovery rates of up to 95% for lithium and 98% for phosphate compounds. However, global recycling infrastructure remains underdeveloped, with less than 5% of lithium-ion batteries currently being recycled worldwide.
Benchmarking diffusion rates in ionic matrices has revealed promising pathways for developing more environmentally sustainable lithium phosphate materials. Research indicates that optimized ionic conductivity can extend battery lifespans by 30-40%, reducing replacement frequency and associated environmental impacts. Additionally, enhanced understanding of diffusion mechanisms enables the development of materials requiring lower processing temperatures, potentially reducing energy consumption in manufacturing by up to 25%.
Standardization Requirements for Diffusion Rate Testing
The standardization of diffusion rate testing methodologies for lithium phosphate in ionic matrices represents a critical challenge in the advancement of battery technology. Current testing protocols exhibit significant variations across research institutions and industry laboratories, leading to inconsistent results that hinder technological progress and commercial application.
A comprehensive standardization framework must address multiple parameters that influence diffusion measurements. Temperature control protocols require precise definition, with recommended testing at multiple fixed points (typically 25°C, 40°C, and 60°C) to enable Arrhenius relationship calculations. Measurement duration standards should be established based on the specific ionic matrix being tested, with minimum observation periods defined to capture both initial rapid diffusion and subsequent steady-state behavior.
Sample preparation techniques demand particular attention, as variations in particle size, crystallinity, and surface characteristics significantly impact measured diffusion rates. The standardization should specify acceptable ranges for particle size distribution (preferably <5μm with <10% deviation), crystallinity verification methods, and surface treatment documentation requirements.
Analytical instrumentation specifications represent another crucial standardization component. Electrochemical impedance spectroscopy (EIS) parameters, nuclear magnetic resonance (NMR) relaxometry settings, and galvanostatic intermittent titration technique (GITT) protocols all require standardization to ensure cross-laboratory reproducibility. Calibration procedures using reference materials with known diffusion properties should be mandated at regular intervals.
Data reporting formats constitute an essential element of the standardization framework. Reports should include raw diffusion coefficients, temperature-normalized values, activation energies, and confidence intervals. Statistical analysis guidelines should specify minimum sample sizes and acceptable variance thresholds to ensure reliability.
Interlaboratory validation programs will be necessary to verify the effectiveness of these standards. A round-robin testing protocol involving at least five independent laboratories should be implemented, with statistical analysis of variance to identify and address remaining methodological inconsistencies.
The implementation timeline for these standardization requirements should follow a phased approach, beginning with a draft standard for industry comment, followed by pilot implementation in selected laboratories, and culminating in formal adoption by international standards organizations such as ISO, IEC, or ASTM within a 24-month timeframe.
A comprehensive standardization framework must address multiple parameters that influence diffusion measurements. Temperature control protocols require precise definition, with recommended testing at multiple fixed points (typically 25°C, 40°C, and 60°C) to enable Arrhenius relationship calculations. Measurement duration standards should be established based on the specific ionic matrix being tested, with minimum observation periods defined to capture both initial rapid diffusion and subsequent steady-state behavior.
Sample preparation techniques demand particular attention, as variations in particle size, crystallinity, and surface characteristics significantly impact measured diffusion rates. The standardization should specify acceptable ranges for particle size distribution (preferably <5μm with <10% deviation), crystallinity verification methods, and surface treatment documentation requirements.
Analytical instrumentation specifications represent another crucial standardization component. Electrochemical impedance spectroscopy (EIS) parameters, nuclear magnetic resonance (NMR) relaxometry settings, and galvanostatic intermittent titration technique (GITT) protocols all require standardization to ensure cross-laboratory reproducibility. Calibration procedures using reference materials with known diffusion properties should be mandated at regular intervals.
Data reporting formats constitute an essential element of the standardization framework. Reports should include raw diffusion coefficients, temperature-normalized values, activation energies, and confidence intervals. Statistical analysis guidelines should specify minimum sample sizes and acceptable variance thresholds to ensure reliability.
Interlaboratory validation programs will be necessary to verify the effectiveness of these standards. A round-robin testing protocol involving at least five independent laboratories should be implemented, with statistical analysis of variance to identify and address remaining methodological inconsistencies.
The implementation timeline for these standardization requirements should follow a phased approach, beginning with a draft standard for industry comment, followed by pilot implementation in selected laboratories, and culminating in formal adoption by international standards organizations such as ISO, IEC, or ASTM within a 24-month timeframe.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!