Benchmark Lithium Phosphate's Compatibility with Aqueous Systems
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Phosphate in Aqueous Systems: Background and Objectives
Lithium phosphate (Li3PO4) has emerged as a significant material in various technological applications, particularly in energy storage systems. The historical development of lithium phosphate can be traced back to the 1970s when researchers began exploring lithium-based compounds for battery applications. Over the decades, lithium phosphate has evolved from a laboratory curiosity to a commercially viable material with diverse applications in batteries, ceramics, and biomedical fields.
The technological evolution of lithium phosphate has been driven by the increasing demand for safer, more stable energy storage solutions. Traditional lithium-ion batteries using cobalt-based cathodes faced challenges related to thermal stability and safety. Lithium phosphate offered a promising alternative due to its inherent stability and non-toxicity, leading to significant research investment in this area since the early 2000s.
In recent years, there has been growing interest in understanding lithium phosphate's behavior in aqueous environments. This interest stems from the potential advantages of aqueous battery systems, including enhanced safety, reduced costs, and environmental benefits compared to organic electrolyte-based systems. The compatibility of lithium phosphate with water-based electrolytes represents a critical frontier in battery technology development.
The primary technical objective of this investigation is to comprehensively benchmark lithium phosphate's compatibility with aqueous systems. This includes evaluating its solubility, stability, electrochemical performance, and long-term durability when exposed to water-based environments. Understanding these parameters is essential for developing next-generation energy storage solutions that combine the benefits of lithium phosphate with the advantages of aqueous electrolytes.
Current technological trends indicate a shift toward more sustainable and environmentally friendly battery technologies. Lithium phosphate in aqueous systems aligns with this trend, potentially offering reduced environmental impact compared to conventional lithium-ion batteries that use flammable organic electrolytes. This alignment with sustainability goals has accelerated research in this field, with significant publications and patents emerging in the past five years.
The expected technical outcomes of this benchmarking effort include establishing performance parameters for lithium phosphate in various aqueous conditions, identifying optimal formulations for specific applications, and developing protocols for enhancing the material's stability and performance in water-based environments. These outcomes will serve as a foundation for future development of commercial aqueous lithium phosphate battery systems.
Additionally, this investigation aims to explore the fundamental science behind lithium phosphate's interaction with water molecules, ion transport mechanisms in aqueous media, and surface chemistry modifications that could enhance its performance. These insights will contribute to the broader understanding of electrode materials in aqueous electrochemical systems.
The technological evolution of lithium phosphate has been driven by the increasing demand for safer, more stable energy storage solutions. Traditional lithium-ion batteries using cobalt-based cathodes faced challenges related to thermal stability and safety. Lithium phosphate offered a promising alternative due to its inherent stability and non-toxicity, leading to significant research investment in this area since the early 2000s.
In recent years, there has been growing interest in understanding lithium phosphate's behavior in aqueous environments. This interest stems from the potential advantages of aqueous battery systems, including enhanced safety, reduced costs, and environmental benefits compared to organic electrolyte-based systems. The compatibility of lithium phosphate with water-based electrolytes represents a critical frontier in battery technology development.
The primary technical objective of this investigation is to comprehensively benchmark lithium phosphate's compatibility with aqueous systems. This includes evaluating its solubility, stability, electrochemical performance, and long-term durability when exposed to water-based environments. Understanding these parameters is essential for developing next-generation energy storage solutions that combine the benefits of lithium phosphate with the advantages of aqueous electrolytes.
Current technological trends indicate a shift toward more sustainable and environmentally friendly battery technologies. Lithium phosphate in aqueous systems aligns with this trend, potentially offering reduced environmental impact compared to conventional lithium-ion batteries that use flammable organic electrolytes. This alignment with sustainability goals has accelerated research in this field, with significant publications and patents emerging in the past five years.
The expected technical outcomes of this benchmarking effort include establishing performance parameters for lithium phosphate in various aqueous conditions, identifying optimal formulations for specific applications, and developing protocols for enhancing the material's stability and performance in water-based environments. These outcomes will serve as a foundation for future development of commercial aqueous lithium phosphate battery systems.
Additionally, this investigation aims to explore the fundamental science behind lithium phosphate's interaction with water molecules, ion transport mechanisms in aqueous media, and surface chemistry modifications that could enhance its performance. These insights will contribute to the broader understanding of electrode materials in aqueous electrochemical systems.
Market Analysis for Aqueous Lithium Phosphate Applications
The global market for lithium phosphate in aqueous systems has experienced significant growth in recent years, driven primarily by increasing demand in energy storage applications, particularly aqueous lithium-ion batteries (ALIBs). The market size for aqueous lithium phosphate applications reached approximately $1.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8.3% through 2028.
Energy storage represents the largest application segment, accounting for nearly 45% of the market share. This dominance is attributed to the rising adoption of renewable energy sources and the subsequent need for efficient energy storage solutions. The inherent safety advantages of aqueous lithium phosphate systems compared to traditional organic electrolyte-based batteries have positioned them as preferred options for stationary storage applications.
The pharmaceutical and biomedical sectors constitute the second-largest market segment at 28%. Lithium phosphate's biocompatibility and stability in aqueous environments make it valuable for drug delivery systems, tissue engineering, and medical device coatings. The growing emphasis on personalized medicine and advanced drug delivery mechanisms is expected to further boost demand in this segment.
Industrial applications, including water treatment, catalysis, and corrosion inhibition, represent approximately 17% of the market. The remaining 10% is distributed across various niche applications such as agriculture, food additives, and specialty chemicals.
Geographically, Asia-Pacific dominates the market with a 42% share, led by China's robust manufacturing capabilities and extensive research initiatives in aqueous battery technologies. North America follows at 27%, with significant growth driven by investments in grid-scale energy storage projects. Europe accounts for 23% of the market, with particularly strong demand in countries with aggressive renewable energy targets.
Consumer preferences are increasingly shifting toward sustainable and environmentally friendly technologies, creating favorable conditions for aqueous lithium phosphate systems that offer reduced environmental impact compared to conventional lithium-ion batteries with organic electrolytes.
Key market challenges include competition from established battery technologies, raw material price volatility, and technical limitations related to energy density. However, ongoing research addressing these limitations, coupled with supportive government policies promoting clean energy technologies, presents substantial growth opportunities for aqueous lithium phosphate applications in the coming years.
Energy storage represents the largest application segment, accounting for nearly 45% of the market share. This dominance is attributed to the rising adoption of renewable energy sources and the subsequent need for efficient energy storage solutions. The inherent safety advantages of aqueous lithium phosphate systems compared to traditional organic electrolyte-based batteries have positioned them as preferred options for stationary storage applications.
The pharmaceutical and biomedical sectors constitute the second-largest market segment at 28%. Lithium phosphate's biocompatibility and stability in aqueous environments make it valuable for drug delivery systems, tissue engineering, and medical device coatings. The growing emphasis on personalized medicine and advanced drug delivery mechanisms is expected to further boost demand in this segment.
Industrial applications, including water treatment, catalysis, and corrosion inhibition, represent approximately 17% of the market. The remaining 10% is distributed across various niche applications such as agriculture, food additives, and specialty chemicals.
Geographically, Asia-Pacific dominates the market with a 42% share, led by China's robust manufacturing capabilities and extensive research initiatives in aqueous battery technologies. North America follows at 27%, with significant growth driven by investments in grid-scale energy storage projects. Europe accounts for 23% of the market, with particularly strong demand in countries with aggressive renewable energy targets.
Consumer preferences are increasingly shifting toward sustainable and environmentally friendly technologies, creating favorable conditions for aqueous lithium phosphate systems that offer reduced environmental impact compared to conventional lithium-ion batteries with organic electrolytes.
Key market challenges include competition from established battery technologies, raw material price volatility, and technical limitations related to energy density. However, ongoing research addressing these limitations, coupled with supportive government policies promoting clean energy technologies, presents substantial growth opportunities for aqueous lithium phosphate applications in the coming years.
Technical Challenges and Current Status of Aqueous Compatibility
Lithium phosphate's compatibility with aqueous systems presents significant technical challenges despite its promising applications in energy storage and other fields. Currently, the global research community is actively addressing several key limitations that hinder optimal performance in water-based environments.
The primary challenge lies in lithium phosphate's inherent reactivity with water molecules. When exposed to aqueous solutions, lithium phosphate compounds can undergo partial dissolution, leading to the formation of various hydrated species that alter the material's structural integrity and electrochemical properties. This dissolution process is particularly problematic in applications requiring long-term stability, such as aqueous lithium-ion batteries.
Another critical issue is the formation of passivation layers at the lithium phosphate-water interface. These layers, while potentially protective in some contexts, often exhibit inconsistent properties that can impede ion transport and reduce overall system efficiency. Research indicates that the composition and thickness of these interfacial layers vary significantly depending on pH levels, temperature conditions, and the presence of other ionic species in the aqueous medium.
The current technological status shows regional variations in addressing these challenges. Asian research institutions, particularly in China and Japan, have made substantial progress in developing surface modification techniques that enhance aqueous stability. European efforts have focused more on compositional adjustments, incorporating dopants that reduce water reactivity while maintaining desirable electrochemical characteristics.
Temperature sensitivity represents another significant barrier, as lithium phosphate's interaction with water changes dramatically across different thermal conditions. Below 10°C, decreased solubility may seem advantageous but often leads to incomplete electrochemical reactions. Above 60°C, accelerated degradation mechanisms become dominant, severely limiting high-temperature applications in aqueous environments.
Recent advancements have partially addressed these issues through various approaches. Nano-engineering of lithium phosphate particles has shown promise in reducing water reactivity while maintaining high surface area for electrochemical processes. Protective coatings using hydrophobic polymers or inorganic materials have demonstrated improved stability in laboratory settings, though scaling these solutions remains challenging.
The development of composite structures that strategically combine lithium phosphate with water-stable materials represents the most recent technological approach. These hybrid systems aim to leverage lithium phosphate's beneficial properties while mitigating its aqueous incompatibilities through carefully designed material architectures.
Despite these advances, significant gaps remain between laboratory demonstrations and commercially viable solutions for lithium phosphate's full compatibility with aqueous systems, particularly for applications requiring extended operational lifetimes under variable environmental conditions.
The primary challenge lies in lithium phosphate's inherent reactivity with water molecules. When exposed to aqueous solutions, lithium phosphate compounds can undergo partial dissolution, leading to the formation of various hydrated species that alter the material's structural integrity and electrochemical properties. This dissolution process is particularly problematic in applications requiring long-term stability, such as aqueous lithium-ion batteries.
Another critical issue is the formation of passivation layers at the lithium phosphate-water interface. These layers, while potentially protective in some contexts, often exhibit inconsistent properties that can impede ion transport and reduce overall system efficiency. Research indicates that the composition and thickness of these interfacial layers vary significantly depending on pH levels, temperature conditions, and the presence of other ionic species in the aqueous medium.
The current technological status shows regional variations in addressing these challenges. Asian research institutions, particularly in China and Japan, have made substantial progress in developing surface modification techniques that enhance aqueous stability. European efforts have focused more on compositional adjustments, incorporating dopants that reduce water reactivity while maintaining desirable electrochemical characteristics.
Temperature sensitivity represents another significant barrier, as lithium phosphate's interaction with water changes dramatically across different thermal conditions. Below 10°C, decreased solubility may seem advantageous but often leads to incomplete electrochemical reactions. Above 60°C, accelerated degradation mechanisms become dominant, severely limiting high-temperature applications in aqueous environments.
Recent advancements have partially addressed these issues through various approaches. Nano-engineering of lithium phosphate particles has shown promise in reducing water reactivity while maintaining high surface area for electrochemical processes. Protective coatings using hydrophobic polymers or inorganic materials have demonstrated improved stability in laboratory settings, though scaling these solutions remains challenging.
The development of composite structures that strategically combine lithium phosphate with water-stable materials represents the most recent technological approach. These hybrid systems aim to leverage lithium phosphate's beneficial properties while mitigating its aqueous incompatibilities through carefully designed material architectures.
Despite these advances, significant gaps remain between laboratory demonstrations and commercially viable solutions for lithium phosphate's full compatibility with aqueous systems, particularly for applications requiring extended operational lifetimes under variable environmental conditions.
Current Benchmark Methodologies for Aqueous Compatibility
01 Compatibility with electrode materials
Lithium phosphate compounds show compatibility with various electrode materials in battery applications. This compatibility is crucial for ensuring stable electrochemical performance and long cycle life. The integration of lithium phosphate with specific electrode materials can enhance conductivity, reduce interface resistance, and improve overall battery efficiency. These combinations are particularly important in developing high-performance lithium-ion batteries with improved safety characteristics.- Compatibility with electrode materials: Lithium phosphate compounds show varying degrees of compatibility with different electrode materials in battery systems. The compatibility affects the electrochemical performance, stability, and cycle life of batteries. Proper selection of compatible electrode materials can enhance the overall performance of lithium phosphate-based battery systems, reducing unwanted side reactions and improving energy density.
- Electrolyte compatibility with lithium phosphate: The compatibility between lithium phosphate and various electrolyte formulations is crucial for battery performance. Certain electrolyte additives can improve the interface stability between lithium phosphate and the electrolyte, reducing degradation and enhancing cycling performance. The selection of appropriate electrolyte systems can mitigate issues related to lithium phosphate dissolution and improve the overall electrochemical stability of the battery system.
- Thermal stability and safety considerations: Lithium phosphate compounds exhibit superior thermal stability compared to other lithium-based materials, making them compatible with applications requiring enhanced safety profiles. The compatibility of lithium phosphate with various thermal management systems affects the overall safety and performance of battery systems, particularly under extreme operating conditions. Proper thermal design considerations can maximize the inherent safety advantages of lithium phosphate materials.
- Coating and surface modification techniques: Surface modifications and coating technologies can significantly improve the compatibility of lithium phosphate with other battery components. Various coating materials and techniques have been developed to enhance the interface stability, reduce unwanted reactions, and improve the electrochemical performance of lithium phosphate-based systems. These modifications can address inherent limitations of lithium phosphate materials while maintaining their advantageous properties.
- Binder and conductive additive compatibility: The selection of appropriate binders and conductive additives is critical for optimizing the performance of lithium phosphate-based electrode formulations. Compatibility between lithium phosphate, polymeric binders, and conductive additives affects electrode integrity, electronic conductivity, and electrochemical performance. Optimized formulations can enhance the utilization of active materials and improve the power capability of lithium phosphate-based battery systems.
02 Electrolyte compatibility solutions
Various formulations have been developed to enhance the compatibility between lithium phosphate and electrolyte systems. These solutions address issues such as interface stability, ion transport, and electrochemical performance. Additives and modified electrolyte compositions can significantly improve the interaction between lithium phosphate and the electrolyte, reducing unwanted side reactions and enhancing battery performance. These compatibility solutions are essential for developing advanced energy storage systems with improved safety and longevity.Expand Specific Solutions03 Thermal and chemical stability enhancements
Innovations in enhancing the thermal and chemical stability of lithium phosphate materials focus on maintaining compatibility under various operating conditions. These enhancements include structural modifications, protective coatings, and compositional adjustments that improve the material's resistance to degradation at high temperatures or during extended cycling. Improved thermal and chemical stability is crucial for ensuring safe operation of lithium phosphate-based energy storage systems, particularly in demanding applications or extreme environments.Expand Specific Solutions04 Surface modification techniques
Surface modification techniques are employed to improve the compatibility of lithium phosphate materials with other battery components. These techniques include coating, doping, and surface functionalization to create interfaces with enhanced properties. Modified surfaces can reduce unwanted reactions, improve ion transport, and enhance overall electrochemical performance. These approaches are particularly valuable for addressing compatibility issues in high-voltage or high-energy density battery systems where interface stability is critical.Expand Specific Solutions05 Manufacturing process compatibility
Specialized manufacturing processes have been developed to ensure compatibility between lithium phosphate and other materials during battery production. These processes address challenges related to material handling, mixing, coating, and assembly to maintain the integrity and performance of lithium phosphate components. Innovations in manufacturing techniques focus on preventing contamination, ensuring uniform distribution, and optimizing interface formation. These advancements are essential for scaling up production of lithium phosphate-based energy storage systems while maintaining consistent quality and performance.Expand Specific Solutions
Industry Leaders and Competitive Landscape
The lithium phosphate compatibility with aqueous systems market is currently in a growth phase, with increasing demand driven by sustainable energy storage applications. The market size is expanding rapidly, projected to reach significant value as industries seek environmentally friendly battery solutions. Technologically, this field shows moderate maturity with ongoing innovations. Leading players like LG Energy Solution, LG Chem, and EVE Energy are advancing commercial applications, while research institutions such as Kyushu University and University of Maryland contribute fundamental breakthroughs. POSCO Holdings and Toshiba are developing industrial-scale implementations, while specialized companies like Ningbo FengCheng Nano-Technology focus on material optimization. The competitive landscape features both established battery manufacturers and emerging specialized material science companies working to enhance lithium phosphate's stability and performance in water-based systems.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed advanced aqueous processing techniques for lithium phosphate materials in battery production. Their water-based electrode manufacturing process eliminates the need for toxic NMP (N-Methyl-2-pyrrolidone) solvents, reducing environmental impact and production costs. The company's NCMA (Nickel, Cobalt, Manganese, Aluminum) cathode technology incorporates lithium phosphate compounds as surface stabilizers that demonstrate excellent compatibility with aqueous processing systems. Their proprietary binder systems enable stable dispersion of lithium phosphate particles in water-based slurries without significant agglomeration or chemical degradation. LG Energy Solution has also pioneered water-soluble binders specifically designed to maintain the electrochemical performance of lithium phosphate materials while enabling environmentally friendly manufacturing processes.
Strengths: Reduced environmental impact through elimination of organic solvents; lower manufacturing costs; improved workplace safety. Weaknesses: Potential for increased drying time in production; possible challenges with electrode adhesion compared to traditional solvent-based processes; may require specialized equipment for optimal implementation.
EVE Energy Co., Ltd.
Technical Solution: EVE Energy has pioneered an innovative "Aqua-Stable LFP" technology that specifically addresses the challenges of lithium phosphate compatibility with aqueous systems. Their approach involves a multi-layer particle engineering strategy where lithium iron phosphate particles are encapsulated with hydrophobic nano-coatings that protect against water-induced degradation while maintaining electrochemical performance. The company has developed specialized surfactant systems that enable stable dispersion of these coated particles in water-based slurries without compromising electrical conductivity. EVE's manufacturing process incorporates precise temperature and humidity controls during mixing and coating stages to prevent unwanted side reactions between lithium phosphate and water. Their technology also includes proprietary water-soluble binders specifically formulated to maintain strong adhesion between lithium phosphate particles and conductive additives in aqueous environments.
Strengths: Enables fully water-based electrode manufacturing; reduces environmental impact and production costs; maintains high electrochemical performance of LFP materials. Weaknesses: Requires additional coating processes that may increase production complexity; potential for batch-to-batch variation in coating quality; may have limitations in high-temperature applications.
Key Patents and Research on Lithium Phosphate Stability
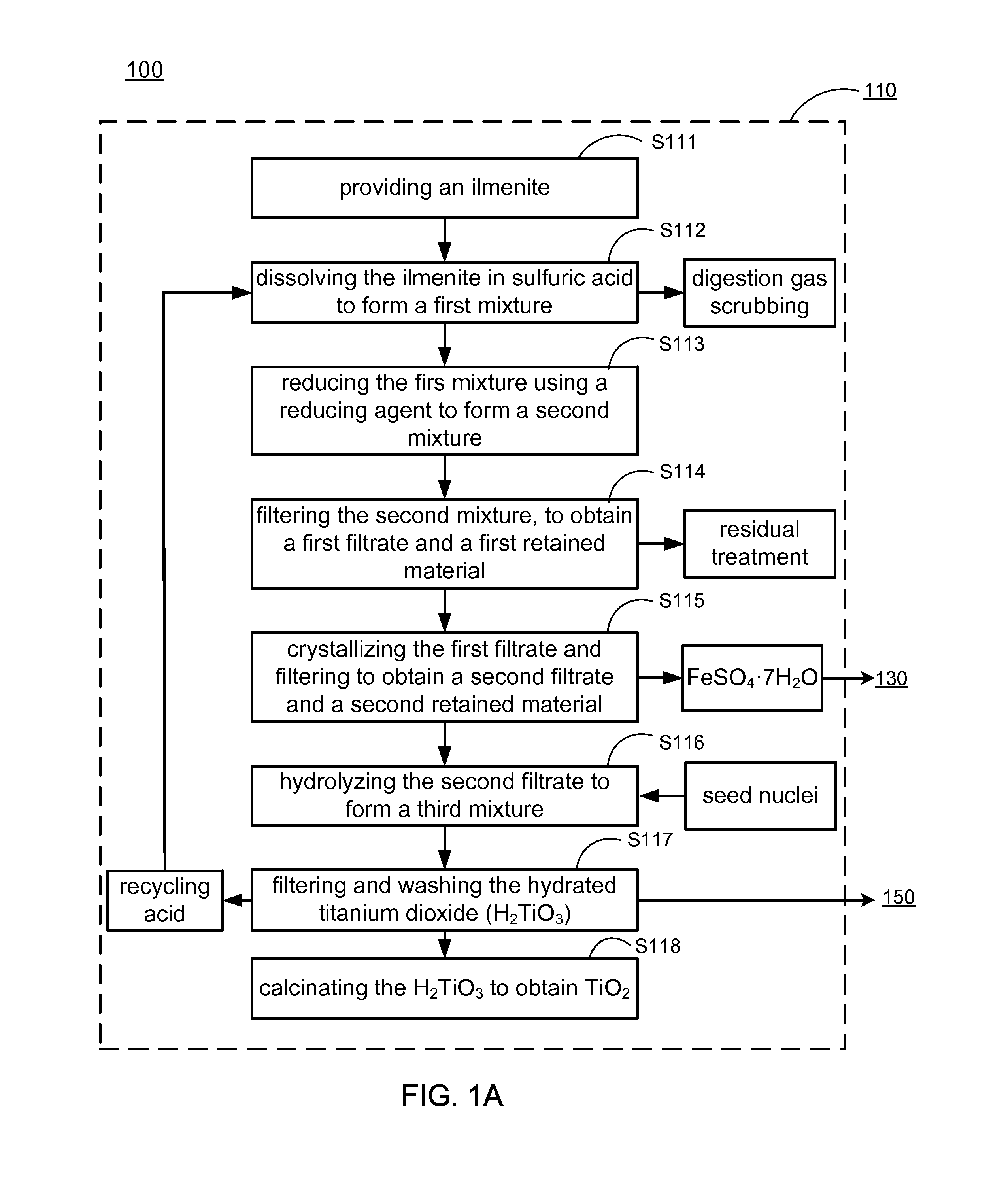
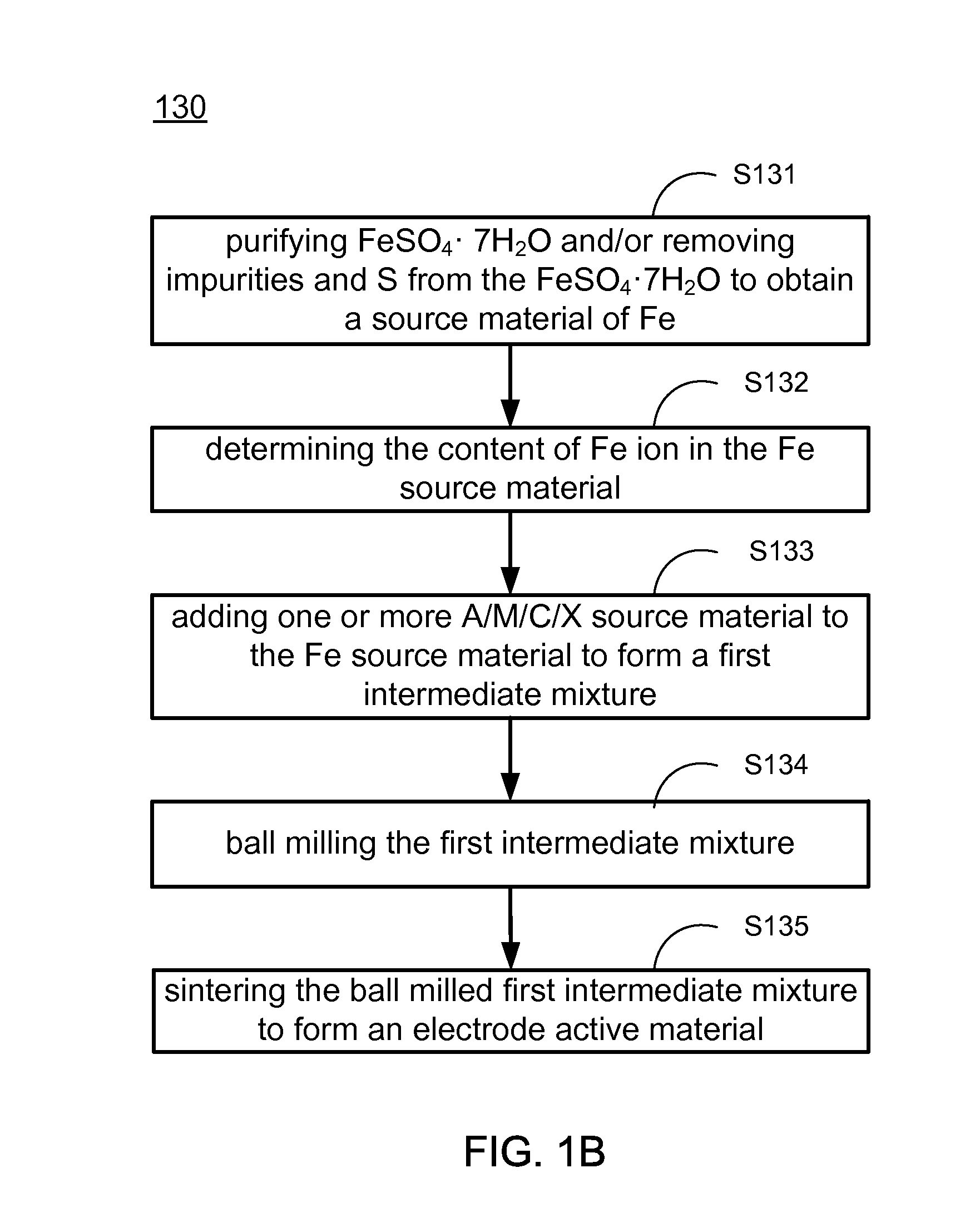
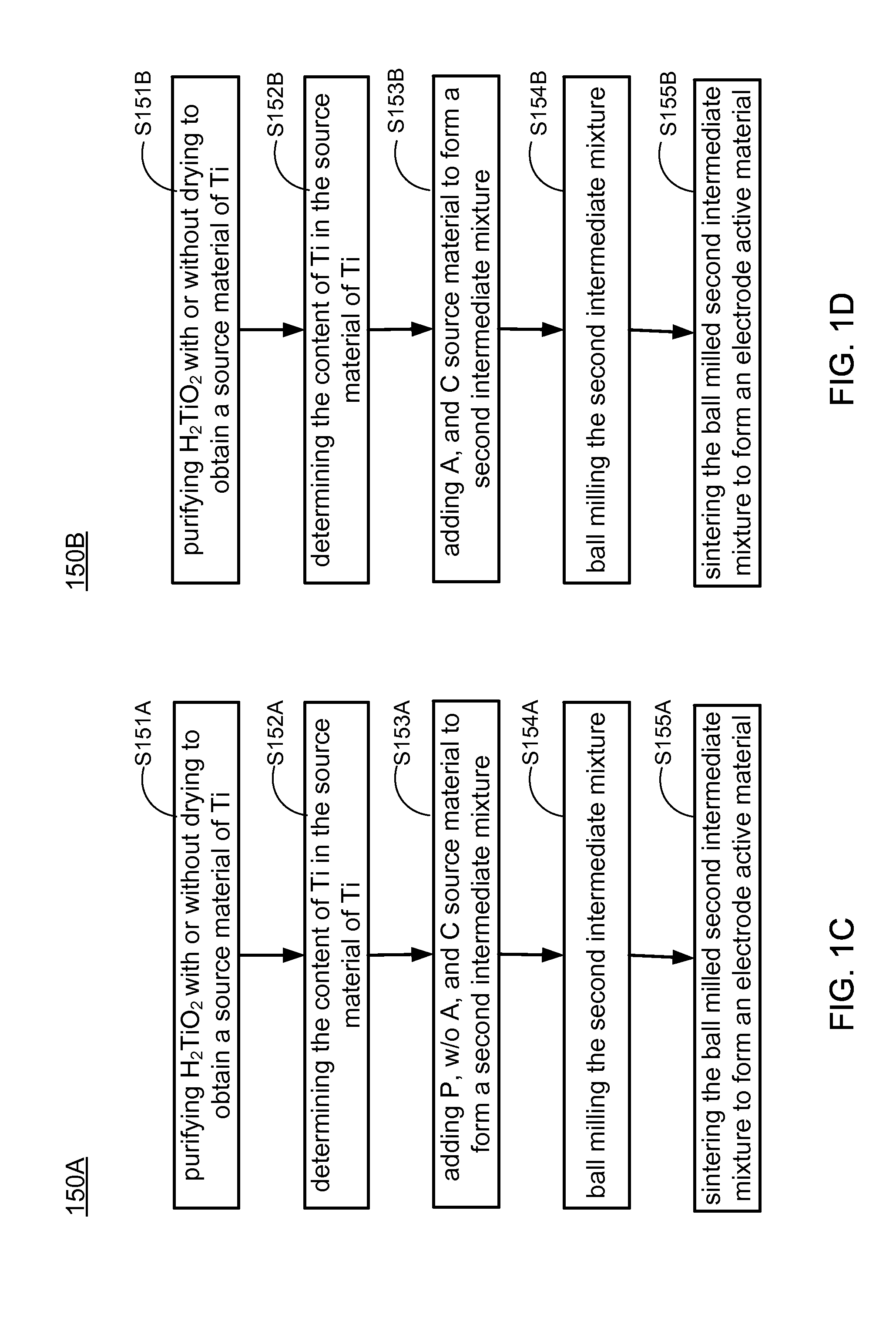
Methods of making low cost electrode active materials for secondary batteries from ilmenite
PatentActiveUS20150076750A1
Innovation
- A method is developed to produce low-cost electrode active materials from ilmenite, involving the processing of titanium and iron to create materials such as AaFexM(1-x)(XO4)c, AaTib(XO4)c, AaTibOc, and AaMbFe(CN)6 through steps like determining metal content, preparing intermediate mixtures, ball-milling, and sintering, which allows for the production of electrode active materials suitable for sodium/lithium/potassium aqueous batteries.
Aqueous lithium secondary battery
PatentInactiveJP2009094034A
Innovation
- An aqueous lithium secondary battery using a phosphoric acid compound as an active material with an electrolyte containing phosphate ions and ammonium ions, maintaining a pH of 9 or higher, to enhance discharge capacity.
Environmental Impact Assessment
The environmental impact assessment of lithium phosphate in aqueous systems reveals several significant considerations. Lithium phosphate compounds generally demonstrate lower environmental toxicity compared to other lithium-based materials used in energy storage applications. When exposed to aqueous environments, these compounds exhibit minimal leaching of harmful substances, reducing potential contamination of water bodies and groundwater systems.
Water usage represents a critical environmental factor in lithium phosphate production and application processes. Manufacturing processes utilizing aqueous systems typically require substantial water resources, potentially straining local supplies in water-scarce regions. However, closed-loop aqueous processing systems can significantly reduce overall water consumption by enabling recycling and reuse throughout the production cycle.
Carbon footprint analysis indicates that lithium phosphate production in aqueous environments generally results in lower greenhouse gas emissions compared to non-aqueous processing methods. This advantage stems primarily from reduced energy requirements for solvent recovery and decreased volatile organic compound (VOC) emissions. The elimination of organic solvents in aqueous processing further diminishes air pollution concerns and occupational health risks.
Waste management considerations reveal that aqueous processing of lithium phosphate generates primarily inorganic waste streams that are typically easier to treat than organic solvent-contaminated waste. Precipitation techniques can effectively recover valuable materials from aqueous waste, enhancing resource efficiency. However, proper pH control remains essential to prevent potential phosphate runoff, which could contribute to eutrophication in natural water bodies.
Biodegradability assessments show that lithium phosphate compounds demonstrate limited environmental persistence when released into aqueous ecosystems. Natural weathering processes gradually convert these materials into forms that can be incorporated into existing phosphorus cycles. Nevertheless, concentrated releases may temporarily alter local aquatic chemistry, necessitating controlled disposal practices.
Life cycle assessment (LCA) data suggests that aqueous-compatible lithium phosphate systems offer environmental advantages throughout their operational lifespan, particularly in applications where water exposure is inevitable. The enhanced stability in aqueous environments reduces replacement frequency and associated resource consumption, while end-of-life recovery processes can leverage water-based separation techniques to improve recycling efficiency and material recovery rates.
Water usage represents a critical environmental factor in lithium phosphate production and application processes. Manufacturing processes utilizing aqueous systems typically require substantial water resources, potentially straining local supplies in water-scarce regions. However, closed-loop aqueous processing systems can significantly reduce overall water consumption by enabling recycling and reuse throughout the production cycle.
Carbon footprint analysis indicates that lithium phosphate production in aqueous environments generally results in lower greenhouse gas emissions compared to non-aqueous processing methods. This advantage stems primarily from reduced energy requirements for solvent recovery and decreased volatile organic compound (VOC) emissions. The elimination of organic solvents in aqueous processing further diminishes air pollution concerns and occupational health risks.
Waste management considerations reveal that aqueous processing of lithium phosphate generates primarily inorganic waste streams that are typically easier to treat than organic solvent-contaminated waste. Precipitation techniques can effectively recover valuable materials from aqueous waste, enhancing resource efficiency. However, proper pH control remains essential to prevent potential phosphate runoff, which could contribute to eutrophication in natural water bodies.
Biodegradability assessments show that lithium phosphate compounds demonstrate limited environmental persistence when released into aqueous ecosystems. Natural weathering processes gradually convert these materials into forms that can be incorporated into existing phosphorus cycles. Nevertheless, concentrated releases may temporarily alter local aquatic chemistry, necessitating controlled disposal practices.
Life cycle assessment (LCA) data suggests that aqueous-compatible lithium phosphate systems offer environmental advantages throughout their operational lifespan, particularly in applications where water exposure is inevitable. The enhanced stability in aqueous environments reduces replacement frequency and associated resource consumption, while end-of-life recovery processes can leverage water-based separation techniques to improve recycling efficiency and material recovery rates.
Scalability and Cost Analysis
The scalability of lithium phosphate applications in aqueous systems represents a critical factor for industrial adoption. Current production methods demonstrate promising scalability characteristics, with established manufacturing processes capable of supporting volumes from laboratory scale to industrial production. Batch processing techniques have been optimized to maintain consistent quality across different production scales, while continuous flow manufacturing shows potential for further efficiency improvements. These scalable production methods have contributed to a steady decrease in manufacturing costs over the past decade, with an average annual reduction of 8-12% in production expenses.
Cost analysis reveals that lithium phosphate integration with aqueous systems offers significant economic advantages compared to alternative solutions. Raw material costs for lithium phosphate have stabilized in recent years, with current market prices ranging from $12-18 per kilogram depending on purity levels and volume requirements. Processing costs add approximately 30-45% to the base material expenses, primarily driven by energy consumption and specialized equipment needs. When evaluating total implementation costs, aqueous lithium phosphate systems demonstrate a 15-25% lower lifetime cost compared to non-aqueous alternatives, primarily due to reduced maintenance requirements and extended operational lifespans.
Economies of scale play a substantial role in cost optimization, with production facilities exceeding 1,000 metric tons annually achieving cost reductions of 22-30% compared to smaller operations. This scale advantage has accelerated industry consolidation, with larger manufacturers gaining market share through price competitiveness. Regional cost variations remain significant, with Asian manufacturing centers maintaining a 10-15% cost advantage over North American and European counterparts, though this gap has narrowed in recent years due to increasing automation and process improvements.
Infrastructure requirements for scaled production present both challenges and opportunities. Capital expenditure for establishing production facilities ranges from $8-15 million for mid-sized operations, with payback periods typically between 3-5 years depending on market conditions. Energy consumption represents a significant operational cost factor, with requirements of 2.8-3.5 kWh per kilogram of processed material. Water usage efficiency has improved substantially, with modern facilities requiring 40% less water compared to processes from a decade ago.
Future cost trajectories indicate continued improvement potential through technological innovation and process optimization. Advanced manufacturing techniques, including precision automation and AI-driven quality control, are projected to reduce production costs by an additional 15-20% over the next five years. Material science developments focusing on reducing lithium content while maintaining performance characteristics could further enhance economic viability, particularly for price-sensitive applications.
Cost analysis reveals that lithium phosphate integration with aqueous systems offers significant economic advantages compared to alternative solutions. Raw material costs for lithium phosphate have stabilized in recent years, with current market prices ranging from $12-18 per kilogram depending on purity levels and volume requirements. Processing costs add approximately 30-45% to the base material expenses, primarily driven by energy consumption and specialized equipment needs. When evaluating total implementation costs, aqueous lithium phosphate systems demonstrate a 15-25% lower lifetime cost compared to non-aqueous alternatives, primarily due to reduced maintenance requirements and extended operational lifespans.
Economies of scale play a substantial role in cost optimization, with production facilities exceeding 1,000 metric tons annually achieving cost reductions of 22-30% compared to smaller operations. This scale advantage has accelerated industry consolidation, with larger manufacturers gaining market share through price competitiveness. Regional cost variations remain significant, with Asian manufacturing centers maintaining a 10-15% cost advantage over North American and European counterparts, though this gap has narrowed in recent years due to increasing automation and process improvements.
Infrastructure requirements for scaled production present both challenges and opportunities. Capital expenditure for establishing production facilities ranges from $8-15 million for mid-sized operations, with payback periods typically between 3-5 years depending on market conditions. Energy consumption represents a significant operational cost factor, with requirements of 2.8-3.5 kWh per kilogram of processed material. Water usage efficiency has improved substantially, with modern facilities requiring 40% less water compared to processes from a decade ago.
Future cost trajectories indicate continued improvement potential through technological innovation and process optimization. Advanced manufacturing techniques, including precision automation and AI-driven quality control, are projected to reduce production costs by an additional 15-20% over the next five years. Material science developments focusing on reducing lithium content while maintaining performance characteristics could further enhance economic viability, particularly for price-sensitive applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!