Benchmarking Dynamic Light Scattering for Bioassay Application
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
DLS Technology Background and Objectives
Dynamic Light Scattering (DLS) emerged in the 1960s as a technique for measuring particle size in colloidal suspensions. The technology evolved from theoretical work on light scattering by Einstein and Smoluchowski in the early 20th century, with significant advancements in laser technology during the 1970s and 1980s enabling more precise measurements. DLS operates on the principle that particles in suspension undergo Brownian motion, causing fluctuations in scattered light intensity that correlate with particle size.
In bioassay applications, DLS has gained prominence over the past two decades as a non-invasive, rapid method for characterizing biomolecules and their interactions. The technique offers unique advantages in analyzing proteins, nucleic acids, viruses, and other biological nanoparticles in their native states without requiring labeling or extensive sample preparation.
The evolution of DLS technology has been marked by increasing sensitivity, resolution, and automation. Modern instruments can detect particles ranging from sub-nanometer to several micrometers, making them versatile tools across various biomedical research and pharmaceutical development applications. Recent technological trends include miniaturization of DLS systems, integration with other analytical techniques, and development of specialized algorithms for complex biological sample analysis.
The primary objective of benchmarking DLS for bioassay applications is to establish standardized protocols and performance metrics that enable reliable comparison between different DLS platforms and methodologies. This standardization is crucial as DLS increasingly serves as a quality control tool in biopharmaceutical production, clinical diagnostics, and research settings where reproducibility and accuracy are paramount.
Additional objectives include identifying optimal operational parameters for specific bioassay types, determining detection limits and dynamic ranges for various biomolecules, and establishing validation criteria for DLS-based bioassays. These benchmarks will facilitate the transition of DLS from primarily a research tool to a routine analytical method in regulated environments such as pharmaceutical manufacturing and clinical laboratories.
The technology trajectory suggests continued refinement in data processing algorithms, particularly in addressing challenges related to polydisperse samples and complex biological matrices. Machine learning approaches are increasingly being incorporated to improve data interpretation and extract more meaningful biological information from DLS measurements. Future developments are likely to focus on real-time monitoring capabilities and integration with microfluidic systems for high-throughput bioassay applications.
In bioassay applications, DLS has gained prominence over the past two decades as a non-invasive, rapid method for characterizing biomolecules and their interactions. The technique offers unique advantages in analyzing proteins, nucleic acids, viruses, and other biological nanoparticles in their native states without requiring labeling or extensive sample preparation.
The evolution of DLS technology has been marked by increasing sensitivity, resolution, and automation. Modern instruments can detect particles ranging from sub-nanometer to several micrometers, making them versatile tools across various biomedical research and pharmaceutical development applications. Recent technological trends include miniaturization of DLS systems, integration with other analytical techniques, and development of specialized algorithms for complex biological sample analysis.
The primary objective of benchmarking DLS for bioassay applications is to establish standardized protocols and performance metrics that enable reliable comparison between different DLS platforms and methodologies. This standardization is crucial as DLS increasingly serves as a quality control tool in biopharmaceutical production, clinical diagnostics, and research settings where reproducibility and accuracy are paramount.
Additional objectives include identifying optimal operational parameters for specific bioassay types, determining detection limits and dynamic ranges for various biomolecules, and establishing validation criteria for DLS-based bioassays. These benchmarks will facilitate the transition of DLS from primarily a research tool to a routine analytical method in regulated environments such as pharmaceutical manufacturing and clinical laboratories.
The technology trajectory suggests continued refinement in data processing algorithms, particularly in addressing challenges related to polydisperse samples and complex biological matrices. Machine learning approaches are increasingly being incorporated to improve data interpretation and extract more meaningful biological information from DLS measurements. Future developments are likely to focus on real-time monitoring capabilities and integration with microfluidic systems for high-throughput bioassay applications.
Bioassay Market Demand Analysis
The global bioassay market has witnessed substantial growth in recent years, driven by increasing demand for accurate and efficient diagnostic tools across pharmaceutical, biotechnology, and clinical research sectors. The market size for bioassay technologies reached approximately $3.5 billion in 2022 and is projected to grow at a compound annual growth rate of 8.2% through 2028, highlighting the expanding opportunities for Dynamic Light Scattering (DLS) applications.
Pharmaceutical and biotechnology companies represent the largest segment of bioassay market consumers, accounting for nearly 45% of the total market share. These organizations require high-throughput screening methods for drug discovery and development processes, where DLS offers significant advantages in characterizing biomolecular interactions and stability assessments. The ability of DLS to provide real-time, non-destructive analysis of biological samples makes it particularly valuable in this sector.
Clinical diagnostics represents another rapidly growing segment, with increasing adoption of bioassay technologies for disease detection and monitoring. The global pandemic has further accelerated this trend, with healthcare providers seeking faster and more reliable diagnostic tools. DLS-based bioassays offer advantages in detecting protein aggregation, virus particles, and other biomarkers relevant to disease states, driving demand in clinical settings.
Academic and research institutions constitute approximately 25% of the bioassay market, with growing interest in advanced analytical techniques for fundamental biological research. These organizations often serve as early adopters of innovative technologies like DLS applications in bioassay development, creating a testing ground for methods that may later be adopted by commercial entities.
Regional analysis reveals North America as the dominant market for bioassay technologies, holding approximately 40% of the global market share, followed by Europe at 30% and Asia-Pacific at 22%. However, the Asia-Pacific region demonstrates the highest growth rate at 10.5% annually, driven by expanding biotechnology sectors in China, India, and South Korea.
Key market drivers include increasing prevalence of chronic diseases, growing focus on personalized medicine, rising R&D investments in life sciences, and technological advancements in bioassay methodologies. Specifically for DLS applications, there is rising demand for techniques that can analyze biomolecular interactions in native-like conditions with minimal sample preparation and consumption.
Market challenges include high equipment costs, technical complexity requiring specialized training, and competition from alternative technologies such as Surface Plasmon Resonance and Bio-Layer Interferometry. However, the unique capabilities of DLS in analyzing particle size distributions and molecular interactions in solution position it favorably for continued market penetration in bioassay applications.
Pharmaceutical and biotechnology companies represent the largest segment of bioassay market consumers, accounting for nearly 45% of the total market share. These organizations require high-throughput screening methods for drug discovery and development processes, where DLS offers significant advantages in characterizing biomolecular interactions and stability assessments. The ability of DLS to provide real-time, non-destructive analysis of biological samples makes it particularly valuable in this sector.
Clinical diagnostics represents another rapidly growing segment, with increasing adoption of bioassay technologies for disease detection and monitoring. The global pandemic has further accelerated this trend, with healthcare providers seeking faster and more reliable diagnostic tools. DLS-based bioassays offer advantages in detecting protein aggregation, virus particles, and other biomarkers relevant to disease states, driving demand in clinical settings.
Academic and research institutions constitute approximately 25% of the bioassay market, with growing interest in advanced analytical techniques for fundamental biological research. These organizations often serve as early adopters of innovative technologies like DLS applications in bioassay development, creating a testing ground for methods that may later be adopted by commercial entities.
Regional analysis reveals North America as the dominant market for bioassay technologies, holding approximately 40% of the global market share, followed by Europe at 30% and Asia-Pacific at 22%. However, the Asia-Pacific region demonstrates the highest growth rate at 10.5% annually, driven by expanding biotechnology sectors in China, India, and South Korea.
Key market drivers include increasing prevalence of chronic diseases, growing focus on personalized medicine, rising R&D investments in life sciences, and technological advancements in bioassay methodologies. Specifically for DLS applications, there is rising demand for techniques that can analyze biomolecular interactions in native-like conditions with minimal sample preparation and consumption.
Market challenges include high equipment costs, technical complexity requiring specialized training, and competition from alternative technologies such as Surface Plasmon Resonance and Bio-Layer Interferometry. However, the unique capabilities of DLS in analyzing particle size distributions and molecular interactions in solution position it favorably for continued market penetration in bioassay applications.
DLS Technical Challenges in Bioassays
Despite significant advancements in Dynamic Light Scattering (DLS) technology for bioassay applications, several technical challenges continue to impede its widespread adoption and optimal performance in biological research settings. The primary challenge remains the accurate measurement of polydisperse biological samples, which often contain particles of varying sizes and shapes. Unlike monodisperse samples, biological specimens frequently present complex mixtures of proteins, vesicles, and aggregates, making size distribution analysis particularly challenging.
Signal-to-noise ratio limitations represent another significant hurdle, especially when analyzing low concentration biomolecules or samples with weak scattering properties. This becomes particularly problematic when attempting to detect early-stage protein aggregation or when working with dilute biological samples where the scattering intensity may be barely distinguishable from background noise.
Temperature control and stability pose considerable technical difficulties in DLS bioassays. Biological samples are notoriously sensitive to thermal fluctuations, which can induce conformational changes or aggregation. Even minor temperature variations during measurement can significantly alter the diffusion coefficients of biomolecules, leading to erroneous size determinations and inconsistent results across experiments.
Sample preparation inconsistencies further complicate DLS applications in bioassays. Factors such as buffer composition, pH variations, and ionic strength can dramatically influence the hydrodynamic behavior of biomolecules. Additionally, the presence of dust particles or air bubbles, which are difficult to eliminate completely in laboratory settings, can introduce artifacts that skew measurement results.
Data interpretation challenges persist due to the mathematical complexities involved in converting correlation functions to size distributions. The inherent assumptions in algorithms like CONTIN or NNLS (non-negative least squares) may not always accurately represent biological reality, particularly for non-spherical particles or interacting systems common in biological environments.
Reproducibility across different DLS instruments remains problematic, with variations in laser wavelength, detector positioning, and optical configurations leading to discrepancies in results between different platforms. This lack of standardization complicates multi-center studies and technology transfer in biomedical research and pharmaceutical development.
Lastly, the integration of DLS with other analytical techniques presents both an opportunity and a challenge. While combining DLS with techniques like size exclusion chromatography or multi-angle light scattering can provide more comprehensive characterization, achieving seamless integration and correlation between different data streams requires sophisticated hardware and software solutions that are not yet fully optimized for routine bioassay applications.
Signal-to-noise ratio limitations represent another significant hurdle, especially when analyzing low concentration biomolecules or samples with weak scattering properties. This becomes particularly problematic when attempting to detect early-stage protein aggregation or when working with dilute biological samples where the scattering intensity may be barely distinguishable from background noise.
Temperature control and stability pose considerable technical difficulties in DLS bioassays. Biological samples are notoriously sensitive to thermal fluctuations, which can induce conformational changes or aggregation. Even minor temperature variations during measurement can significantly alter the diffusion coefficients of biomolecules, leading to erroneous size determinations and inconsistent results across experiments.
Sample preparation inconsistencies further complicate DLS applications in bioassays. Factors such as buffer composition, pH variations, and ionic strength can dramatically influence the hydrodynamic behavior of biomolecules. Additionally, the presence of dust particles or air bubbles, which are difficult to eliminate completely in laboratory settings, can introduce artifacts that skew measurement results.
Data interpretation challenges persist due to the mathematical complexities involved in converting correlation functions to size distributions. The inherent assumptions in algorithms like CONTIN or NNLS (non-negative least squares) may not always accurately represent biological reality, particularly for non-spherical particles or interacting systems common in biological environments.
Reproducibility across different DLS instruments remains problematic, with variations in laser wavelength, detector positioning, and optical configurations leading to discrepancies in results between different platforms. This lack of standardization complicates multi-center studies and technology transfer in biomedical research and pharmaceutical development.
Lastly, the integration of DLS with other analytical techniques presents both an opportunity and a challenge. While combining DLS with techniques like size exclusion chromatography or multi-angle light scattering can provide more comprehensive characterization, achieving seamless integration and correlation between different data streams requires sophisticated hardware and software solutions that are not yet fully optimized for routine bioassay applications.
Current DLS Benchmarking Methodologies
01 DLS instrumentation and apparatus design
Various designs and improvements in dynamic light scattering instruments focus on enhancing measurement accuracy and reliability. These include specialized optical arrangements, detection systems, and sample holders that optimize the collection of scattered light signals. Advanced instrumentation may incorporate features such as temperature control, multiple angle detection, and automated alignment systems to ensure consistent and reproducible measurements across different sample types.- DLS instrumentation and apparatus design: Dynamic Light Scattering (DLS) instrumentation has evolved with various apparatus designs to improve measurement accuracy and reliability. These designs include specialized optical arrangements, detection systems, and sample holders that optimize the collection of scattered light. Advanced DLS instruments incorporate features such as temperature control, multiple angle detection, and automated calibration systems to ensure consistent benchmarking results across different samples and experimental conditions.
- DLS data processing and analysis methods: Various data processing and analysis methods have been developed to enhance the interpretation of Dynamic Light Scattering measurements. These methods include algorithms for correlation function analysis, particle size distribution calculations, and statistical approaches to improve signal-to-noise ratios. Advanced computational techniques help in benchmarking DLS performance by filtering artifacts, addressing polydispersity issues, and enabling more accurate characterization of complex samples with multiple particle populations.
- DLS calibration and standardization protocols: Standardized protocols for calibrating DLS systems are essential for reliable benchmarking. These protocols involve the use of reference materials with known particle sizes and distributions to validate instrument performance. Calibration procedures may include systematic verification of laser stability, detector response, and correlation time accuracy. Regular standardization ensures that DLS measurements remain comparable across different instruments, laboratories, and time periods, establishing a foundation for meaningful benchmarking studies.
- DLS performance evaluation metrics: Various metrics have been established to evaluate and benchmark the performance of Dynamic Light Scattering systems. These metrics include measurement reproducibility, resolution limits for particle size discrimination, sensitivity to concentration variations, and accuracy in characterizing polydisperse samples. Performance evaluation frameworks may also assess factors such as measurement speed, minimum detectable particle size, and robustness against contaminants or aggregates, providing comprehensive benchmarking criteria for DLS technology.
- DLS applications in quality control and process monitoring: Dynamic Light Scattering has been implemented in quality control and process monitoring systems across various industries. These applications leverage DLS for real-time particle size analysis, stability assessment, and detection of aggregation or degradation. Benchmarking studies in this context focus on evaluating how DLS performance correlates with product quality attributes or process parameters. Integration of DLS into automated monitoring systems enables continuous benchmarking against established quality standards and facilitates process optimization based on particle characterization data.
02 Calibration and reference standards for DLS
Benchmarking dynamic light scattering systems requires reliable calibration methods and reference standards. These standards typically consist of monodisperse particles with well-characterized size distributions that allow for system validation and performance verification. Calibration protocols may include procedures for verifying detector response, laser stability, and correlation accuracy, ensuring that measurements across different instruments or laboratories can be meaningfully compared.Expand Specific Solutions03 Data processing and analysis algorithms
Advanced algorithms for processing dynamic light scattering data improve the accuracy of particle size distribution analysis. These computational methods may include noise reduction techniques, correlation function analysis, and mathematical models for converting raw scattering data into meaningful size distributions. Machine learning approaches can enhance data interpretation by recognizing patterns in complex scattering profiles and improving the resolution of multimodal distributions.Expand Specific Solutions04 Quality control and performance metrics
Standardized performance metrics and quality control procedures ensure reliable dynamic light scattering measurements. These benchmarking approaches may include statistical methods for evaluating measurement reproducibility, sensitivity limits, and resolution capabilities. Regular performance testing using defined protocols helps identify instrument drift or degradation and ensures consistent results over time, which is particularly important for regulated environments and comparative studies.Expand Specific Solutions05 Integration with other analytical techniques
Combining dynamic light scattering with complementary analytical methods creates powerful hybrid approaches for comprehensive particle characterization. These integrated systems may pair DLS with techniques such as Raman spectroscopy, microscopy, or chromatography to provide correlated information about particle size, composition, and behavior. Benchmarking these multi-modal systems requires specialized reference materials and protocols that address the unique challenges of data integration and cross-validation.Expand Specific Solutions
Key Industry Players in DLS Instrumentation
Dynamic Light Scattering (DLS) for bioassay applications is in a growth phase, with the market expanding due to increasing demand for precise biomolecular characterization. The global market is estimated at approximately $300-400 million annually, with steady growth projected as pharmaceutical and biotechnology sectors advance. Technologically, DLS has reached moderate maturity with established players like Malvern Panalytical and Wyatt Technology leading instrumentation development, while companies such as Roche Diagnostics, Abbott Laboratories, and Illumina are integrating DLS into their bioanalytical platforms. Academic institutions including UMBC and Auburn University are advancing fundamental research, while industrial players like FUJIFILM and Sony are exploring novel applications. The competitive landscape shows a mix of specialized instrument manufacturers and larger life science corporations developing application-specific implementations.
Malvern Panalytical Ltd.
Technical Solution: Malvern Panalytical has developed comprehensive DLS platforms specifically optimized for bioassay applications, featuring their flagship Zetasizer series. Their technology employs non-invasive back scatter (NIBS) detection with 173° optics that significantly reduces multiple scattering effects and minimizes dust interference in biological samples[1]. The company's systems incorporate adaptive correlation algorithms that automatically optimize measurement parameters based on sample characteristics, enabling accurate size determination of biomolecules ranging from 0.3nm to 10μm[2]. Their Multi-Angle Dynamic Light Scattering (MADLS) technology allows for improved resolution of polydisperse biological samples by simultaneously collecting data at multiple angles, providing more comprehensive characterization of complex protein formulations and nanoparticle-based drug delivery systems[3].
Strengths: Industry-leading sensitivity for protein and biomolecule characterization; robust validation protocols specifically designed for regulatory compliance in pharmaceutical applications; extensive software capabilities for data interpretation. Weaknesses: Higher cost compared to academic-focused systems; requires significant expertise for advanced applications; some limitations in extremely concentrated samples.
Wyatt Technology LLC
Technical Solution: Wyatt Technology has pioneered multi-angle light scattering (MALS) technology integrated with DLS for bioassay applications through their DynaPro® NanoStar® and DynaPro® Plate Reader platforms. Their approach combines batch DLS measurements with automated sample handling systems specifically designed for high-throughput protein formulation screening[1]. The company's DYNAMICS® software incorporates machine learning algorithms to detect subtle changes in protein aggregation states, enabling early detection of stability issues in biopharmaceutical formulations[2]. Wyatt's technology employs fiber-optic-based detection systems that achieve exceptional signal-to-noise ratios even with minimal sample volumes (as low as 4μL), making it particularly valuable for precious biological samples[3]. Their systems feature temperature control from 4°C to 85°C with 0.1°C precision, allowing for detailed thermal stability studies of biomolecules under precisely controlled conditions[4].
Strengths: Superior performance with extremely small sample volumes; excellent integration with chromatography systems for online measurements; highly automated workflows for pharmaceutical screening applications. Weaknesses: More specialized for protein characterization than for general nanoparticle analysis; higher learning curve for advanced applications; premium pricing compared to basic DLS systems.
Critical Patents and Literature in DLS Bioassays
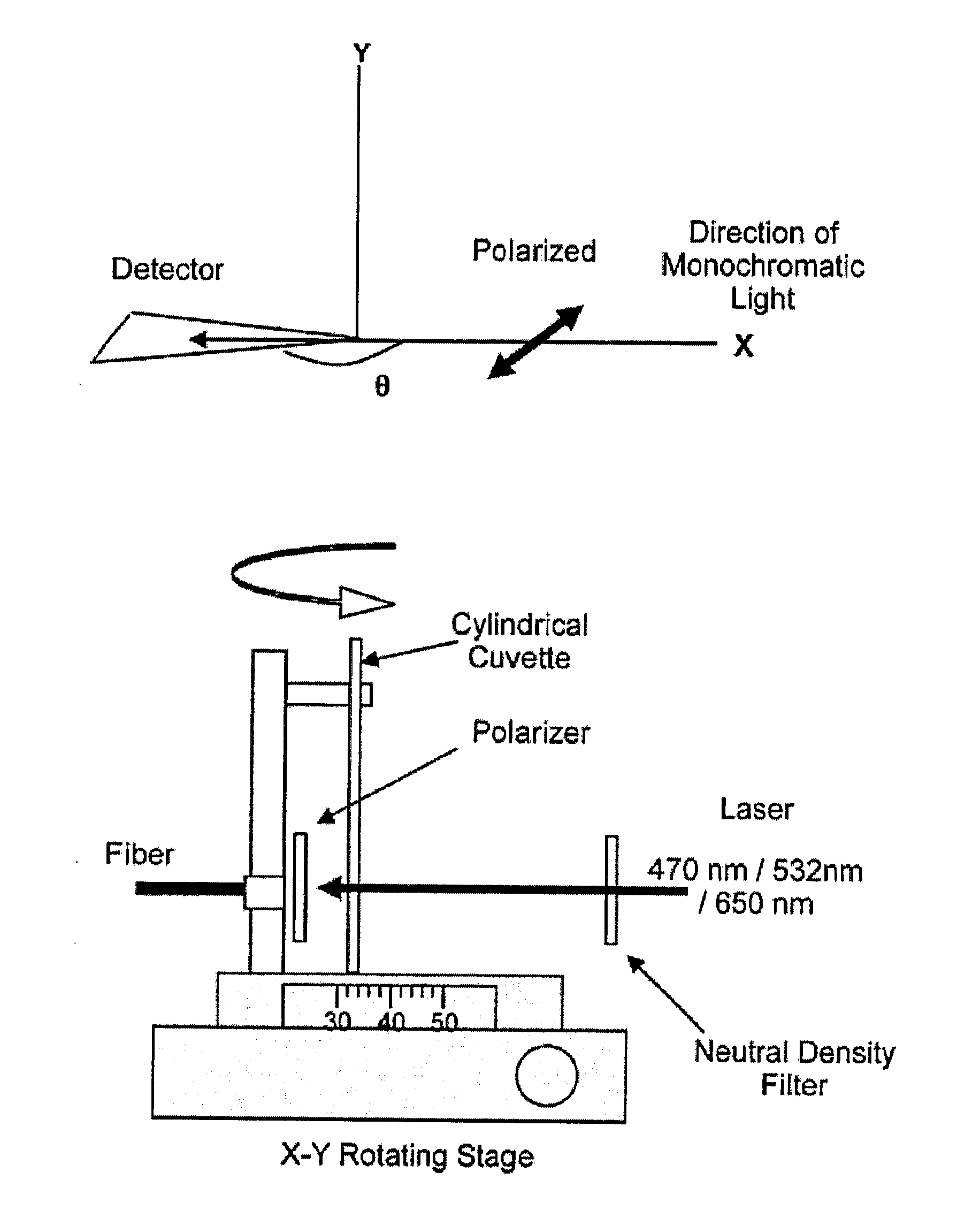
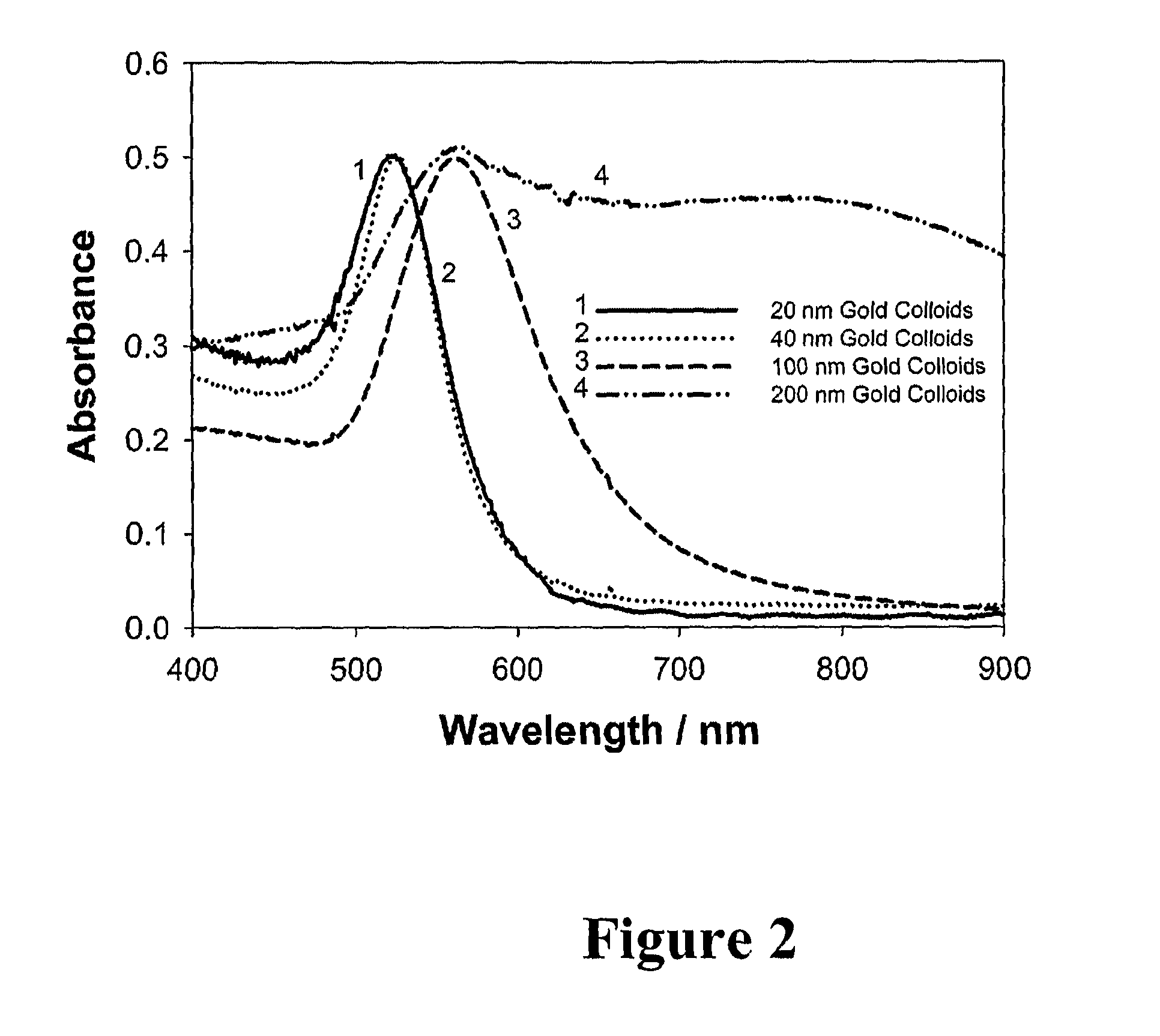
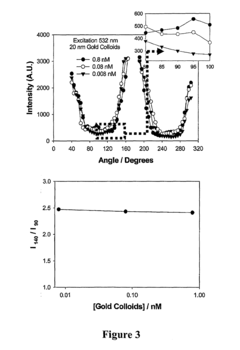
Bioassays using plasmonic scattering from noble metal nanostructures
PatentInactiveUS20100062545A1
Innovation
- The use of surface plasmons from metallic nanoparticles to measure scattering effects at different angles and wavelengths, allowing for the detection of analyte concentration through changes in intensity and polarization, which is more stable and sensitive than traditional methods by utilizing the superior scattering properties of noble metal nanostructures.
Bioassays using plasmonic scattering from noble metal nanostructures
PatentActiveUS20120122240A1
Innovation
- The use of surface plasmons from metallic nanoparticles to measure scattering effects, including changes in intensity and polarization, for detecting analyte concentrations through colloidal aggregation, employing metallic nanostructures that do not photodegrade and have greater emissions than fluorophores, allowing for efficient light scattering at low concentrations and overcoming the limitations of traditional methods.
Standardization and Validation Protocols
The establishment of robust standardization and validation protocols is essential for ensuring the reliability and reproducibility of Dynamic Light Scattering (DLS) measurements in bioassay applications. These protocols must address the inherent variability in biological samples while maintaining scientific rigor across different laboratory settings and instrument configurations.
Standard operating procedures (SOPs) for DLS bioassay applications should include detailed specifications for sample preparation, measurement parameters, data analysis, and quality control metrics. Sample preparation protocols must address concentration ranges, buffer compositions, filtration requirements, and temperature equilibration periods specific to biological samples. Standardized measurement parameters should specify scattering angle, laser wavelength, acquisition time, and number of measurement repetitions to ensure consistency.
Validation of DLS methods for bioassay applications requires multi-level verification approaches. Primary validation should include linearity assessment across relevant concentration ranges, determination of detection and quantification limits, and evaluation of measurement precision through repeatability and intermediate precision studies. Secondary validation should address robustness by examining the method's stability under varying experimental conditions such as temperature fluctuations, sample aging, and minor procedural deviations.
Reference materials play a crucial role in DLS standardization for bioassays. While polystyrene latex beads serve as traditional size standards, protein-based reference materials that better mimic the optical and physical properties of biological analytes are increasingly important. Organizations such as NIST, NIBSC, and USP have developed certified reference materials specifically for biopharmaceutical applications, which should be incorporated into validation protocols.
Interlaboratory comparison studies represent a vital component of DLS validation for bioassays. These studies evaluate method transferability and identify sources of variability between different instruments and operators. Statistical approaches such as z-score analysis and Youden plots can help quantify measurement uncertainties and establish acceptance criteria for bioassay applications.
Quality control procedures must be integrated into routine DLS bioassay workflows. These should include regular system suitability tests using reference standards, blank measurements to detect contamination, and trending analysis to monitor instrument performance over time. Acceptance criteria should be established based on the specific requirements of the bioassay application, considering factors such as required precision, accuracy, and detection sensitivity.
Documentation requirements for validated DLS bioassay methods should follow regulatory guidelines such as ICH Q2(R1) for analytical method validation. This documentation should include validation protocols, raw data, statistical analyses, and conclusions regarding method suitability for the intended bioassay application.
Standard operating procedures (SOPs) for DLS bioassay applications should include detailed specifications for sample preparation, measurement parameters, data analysis, and quality control metrics. Sample preparation protocols must address concentration ranges, buffer compositions, filtration requirements, and temperature equilibration periods specific to biological samples. Standardized measurement parameters should specify scattering angle, laser wavelength, acquisition time, and number of measurement repetitions to ensure consistency.
Validation of DLS methods for bioassay applications requires multi-level verification approaches. Primary validation should include linearity assessment across relevant concentration ranges, determination of detection and quantification limits, and evaluation of measurement precision through repeatability and intermediate precision studies. Secondary validation should address robustness by examining the method's stability under varying experimental conditions such as temperature fluctuations, sample aging, and minor procedural deviations.
Reference materials play a crucial role in DLS standardization for bioassays. While polystyrene latex beads serve as traditional size standards, protein-based reference materials that better mimic the optical and physical properties of biological analytes are increasingly important. Organizations such as NIST, NIBSC, and USP have developed certified reference materials specifically for biopharmaceutical applications, which should be incorporated into validation protocols.
Interlaboratory comparison studies represent a vital component of DLS validation for bioassays. These studies evaluate method transferability and identify sources of variability between different instruments and operators. Statistical approaches such as z-score analysis and Youden plots can help quantify measurement uncertainties and establish acceptance criteria for bioassay applications.
Quality control procedures must be integrated into routine DLS bioassay workflows. These should include regular system suitability tests using reference standards, blank measurements to detect contamination, and trending analysis to monitor instrument performance over time. Acceptance criteria should be established based on the specific requirements of the bioassay application, considering factors such as required precision, accuracy, and detection sensitivity.
Documentation requirements for validated DLS bioassay methods should follow regulatory guidelines such as ICH Q2(R1) for analytical method validation. This documentation should include validation protocols, raw data, statistical analyses, and conclusions regarding method suitability for the intended bioassay application.
Regulatory Compliance for DLS-Based Diagnostic Tools
Dynamic Light Scattering (DLS) technologies employed in diagnostic and bioassay applications must adhere to stringent regulatory frameworks established by various international bodies. The FDA's regulatory pathway for DLS-based diagnostic tools typically follows either the 510(k) clearance process for devices substantially equivalent to legally marketed predecessors, or the more rigorous Premarket Approval (PMA) for novel technologies. These pathways necessitate comprehensive validation studies demonstrating accuracy, precision, and clinical utility.
In the European market, DLS diagnostic tools must comply with the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous IVD Directive in May 2022. This transition introduced a risk-based classification system requiring more extensive clinical evidence and post-market surveillance for higher-risk devices. Manufacturers must now implement more robust quality management systems and technical documentation to demonstrate compliance.
The International Organization for Standardization (ISO) provides several standards directly applicable to DLS technology in bioassay applications. ISO 22412:2017 specifically addresses particle size analysis by dynamic light scattering, establishing standardized measurement protocols and reporting requirements. Additionally, ISO 13485:2016 outlines quality management systems for medical devices, while ISO 14971:2019 provides a framework for risk management.
Regulatory bodies increasingly emphasize the validation of DLS methods for specific bioassay applications. This includes establishing performance characteristics such as linearity, detection limits, specificity, and robustness across different biological matrices. Interlaboratory comparison studies are often required to demonstrate reproducibility across different instruments and operators.
Data integrity requirements present another critical regulatory consideration. Electronic records from DLS instruments must comply with 21 CFR Part 11 (FDA) or Annex 11 (EU GMP), requiring audit trails, electronic signatures, and validated software systems. These regulations ensure the authenticity and traceability of all data generated during development and routine use.
Emerging regulatory trends indicate a shift toward harmonized global standards for DLS-based diagnostics. The Medical Device Single Audit Program (MDSAP) allows for a single regulatory audit to satisfy requirements of multiple regulatory jurisdictions. Additionally, regulatory bodies are developing frameworks for novel applications of DLS in point-of-care diagnostics and personalized medicine, potentially streamlining approval processes while maintaining safety standards.
In the European market, DLS diagnostic tools must comply with the In Vitro Diagnostic Regulation (IVDR 2017/746), which replaced the previous IVD Directive in May 2022. This transition introduced a risk-based classification system requiring more extensive clinical evidence and post-market surveillance for higher-risk devices. Manufacturers must now implement more robust quality management systems and technical documentation to demonstrate compliance.
The International Organization for Standardization (ISO) provides several standards directly applicable to DLS technology in bioassay applications. ISO 22412:2017 specifically addresses particle size analysis by dynamic light scattering, establishing standardized measurement protocols and reporting requirements. Additionally, ISO 13485:2016 outlines quality management systems for medical devices, while ISO 14971:2019 provides a framework for risk management.
Regulatory bodies increasingly emphasize the validation of DLS methods for specific bioassay applications. This includes establishing performance characteristics such as linearity, detection limits, specificity, and robustness across different biological matrices. Interlaboratory comparison studies are often required to demonstrate reproducibility across different instruments and operators.
Data integrity requirements present another critical regulatory consideration. Electronic records from DLS instruments must comply with 21 CFR Part 11 (FDA) or Annex 11 (EU GMP), requiring audit trails, electronic signatures, and validated software systems. These regulations ensure the authenticity and traceability of all data generated during development and routine use.
Emerging regulatory trends indicate a shift toward harmonized global standards for DLS-based diagnostics. The Medical Device Single Audit Program (MDSAP) allows for a single regulatory audit to satisfy requirements of multiple regulatory jurisdictions. Additionally, regulatory bodies are developing frameworks for novel applications of DLS in point-of-care diagnostics and personalized medicine, potentially streamlining approval processes while maintaining safety standards.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!