Case Study: CV Characterization of a Metal-oxide Catalyst — Data, Interpretation and Conclusions
AUG 21, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Metal-oxide Catalyst CV Characterization Background and Objectives
Cyclic voltammetry (CV) has emerged as a cornerstone analytical technique in the field of catalysis research, offering invaluable insights into the electrochemical properties of catalytic materials. Metal-oxide catalysts, in particular, have garnered significant attention due to their versatility, stability, and efficiency in various industrial applications including energy conversion, environmental remediation, and chemical synthesis.
The evolution of metal-oxide catalyst characterization techniques has witnessed remarkable advancement over the past decades. Initially limited to basic surface area measurements and rudimentary activity testing, the field has progressively incorporated sophisticated electrochemical methods that provide deeper understanding of reaction mechanisms at the molecular level. Cyclic voltammetry stands out as a powerful technique that has revolutionized our ability to probe the redox behavior, active site distribution, and reaction kinetics of these catalytic materials.
The primary objective of this CV characterization study is to establish a comprehensive understanding of the electrochemical behavior of selected metal-oxide catalysts under various operational conditions. By systematically analyzing the voltammetric responses, we aim to elucidate the fundamental relationships between catalyst structure, composition, and catalytic performance. This knowledge is crucial for rational catalyst design and optimization strategies.
Furthermore, this investigation seeks to address several persistent challenges in metal-oxide catalyst development, including surface reconstruction phenomena during catalytic cycles, the influence of support materials on catalytic activity, and the identification of rate-determining steps in complex reaction networks. Through detailed CV analysis, we anticipate gaining insights that could lead to enhanced catalyst stability, selectivity, and activity.
The technological trajectory in this field points toward integrating CV characterization with complementary techniques such as in-situ spectroscopy and advanced microscopy to create a multi-dimensional understanding of catalyst behavior under realistic operating conditions. This synergistic approach represents the frontier of catalyst characterization methodology and aligns with the broader industry trend toward precision engineering of catalytic materials.
By establishing clear correlations between CV signatures and catalytic performance metrics, this study aims to develop predictive models that can accelerate the discovery and optimization of next-generation metal-oxide catalysts. Such models would significantly reduce the empirical trial-and-error approach that has historically dominated catalyst development, potentially leading to substantial time and resource savings in industrial R&D processes.
The evolution of metal-oxide catalyst characterization techniques has witnessed remarkable advancement over the past decades. Initially limited to basic surface area measurements and rudimentary activity testing, the field has progressively incorporated sophisticated electrochemical methods that provide deeper understanding of reaction mechanisms at the molecular level. Cyclic voltammetry stands out as a powerful technique that has revolutionized our ability to probe the redox behavior, active site distribution, and reaction kinetics of these catalytic materials.
The primary objective of this CV characterization study is to establish a comprehensive understanding of the electrochemical behavior of selected metal-oxide catalysts under various operational conditions. By systematically analyzing the voltammetric responses, we aim to elucidate the fundamental relationships between catalyst structure, composition, and catalytic performance. This knowledge is crucial for rational catalyst design and optimization strategies.
Furthermore, this investigation seeks to address several persistent challenges in metal-oxide catalyst development, including surface reconstruction phenomena during catalytic cycles, the influence of support materials on catalytic activity, and the identification of rate-determining steps in complex reaction networks. Through detailed CV analysis, we anticipate gaining insights that could lead to enhanced catalyst stability, selectivity, and activity.
The technological trajectory in this field points toward integrating CV characterization with complementary techniques such as in-situ spectroscopy and advanced microscopy to create a multi-dimensional understanding of catalyst behavior under realistic operating conditions. This synergistic approach represents the frontier of catalyst characterization methodology and aligns with the broader industry trend toward precision engineering of catalytic materials.
By establishing clear correlations between CV signatures and catalytic performance metrics, this study aims to develop predictive models that can accelerate the discovery and optimization of next-generation metal-oxide catalysts. Such models would significantly reduce the empirical trial-and-error approach that has historically dominated catalyst development, potentially leading to substantial time and resource savings in industrial R&D processes.
Market Applications and Demand Analysis for Metal-oxide Catalysts
Metal-oxide catalysts have witnessed substantial market growth due to their versatile applications across multiple industries. The global catalyst market, valued at approximately $33.5 billion in 2022, is projected to reach $47.2 billion by 2027, with metal-oxide catalysts representing a significant segment of this expansion. This growth is primarily driven by increasing demand in petrochemical processing, environmental remediation, and emerging clean energy technologies.
In the petrochemical sector, metal-oxide catalysts are essential for refining processes, including hydrocracking, fluid catalytic cracking, and reforming. The industry's continuous pursuit of higher efficiency and lower environmental impact has intensified research into advanced metal-oxide catalyst formulations that demonstrate superior selectivity and stability under harsh reaction conditions, as evidenced in the CV characterization studies.
Environmental applications represent another major market driver, with metal-oxide catalysts playing a crucial role in emission control systems. Stringent environmental regulations worldwide have accelerated the adoption of catalytic converters in automotive applications and industrial emission control systems. The market for environmental catalysts alone is expected to grow at a CAGR of 6.8% through 2028.
The renewable energy sector presents emerging opportunities for metal-oxide catalysts. Their application in fuel cells, water splitting for hydrogen production, and CO2 conversion technologies aligns with global decarbonization efforts. The electrochemical properties revealed through cyclic voltammetry characterization directly inform the development of more efficient catalysts for these applications.
Regional analysis indicates that Asia-Pacific dominates the metal-oxide catalyst market, accounting for approximately 40% of global demand, followed by North America and Europe. China's rapid industrialization and increasing environmental concerns have positioned it as the fastest-growing market for advanced catalyst technologies.
Customer requirements are evolving toward catalysts with enhanced durability, reduced precious metal content, and improved performance under variable operating conditions. The detailed electrochemical characterization methods highlighted in the case study are becoming standard industry practices for quality assurance and performance validation.
Market challenges include raw material price volatility, particularly for precious metals often used in conjunction with metal oxides, and increasing competition from alternative catalyst technologies. However, the superior stability and cost-effectiveness of properly engineered metal-oxide catalysts, as demonstrated through comprehensive CV characterization, continue to secure their market position.
Future market growth will likely be driven by innovations in catalyst design informed by advanced characterization techniques, enabling tailored solutions for specific industrial processes and emerging applications in green chemistry and sustainable manufacturing.
In the petrochemical sector, metal-oxide catalysts are essential for refining processes, including hydrocracking, fluid catalytic cracking, and reforming. The industry's continuous pursuit of higher efficiency and lower environmental impact has intensified research into advanced metal-oxide catalyst formulations that demonstrate superior selectivity and stability under harsh reaction conditions, as evidenced in the CV characterization studies.
Environmental applications represent another major market driver, with metal-oxide catalysts playing a crucial role in emission control systems. Stringent environmental regulations worldwide have accelerated the adoption of catalytic converters in automotive applications and industrial emission control systems. The market for environmental catalysts alone is expected to grow at a CAGR of 6.8% through 2028.
The renewable energy sector presents emerging opportunities for metal-oxide catalysts. Their application in fuel cells, water splitting for hydrogen production, and CO2 conversion technologies aligns with global decarbonization efforts. The electrochemical properties revealed through cyclic voltammetry characterization directly inform the development of more efficient catalysts for these applications.
Regional analysis indicates that Asia-Pacific dominates the metal-oxide catalyst market, accounting for approximately 40% of global demand, followed by North America and Europe. China's rapid industrialization and increasing environmental concerns have positioned it as the fastest-growing market for advanced catalyst technologies.
Customer requirements are evolving toward catalysts with enhanced durability, reduced precious metal content, and improved performance under variable operating conditions. The detailed electrochemical characterization methods highlighted in the case study are becoming standard industry practices for quality assurance and performance validation.
Market challenges include raw material price volatility, particularly for precious metals often used in conjunction with metal oxides, and increasing competition from alternative catalyst technologies. However, the superior stability and cost-effectiveness of properly engineered metal-oxide catalysts, as demonstrated through comprehensive CV characterization, continue to secure their market position.
Future market growth will likely be driven by innovations in catalyst design informed by advanced characterization techniques, enabling tailored solutions for specific industrial processes and emerging applications in green chemistry and sustainable manufacturing.
Current CV Characterization Techniques and Challenges
Cyclic Voltammetry (CV) has emerged as a cornerstone technique in electrochemical characterization of catalysts, particularly metal-oxide systems. Current CV characterization methodologies employ potentiostats that apply controlled potential sweeps while measuring resultant current responses. The technique's popularity stems from its ability to provide rich information about redox processes, reaction kinetics, and surface phenomena with relatively simple instrumentation.
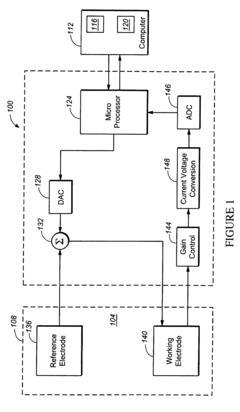
Standard CV characterization typically involves a three-electrode setup consisting of working, reference, and counter electrodes immersed in an electrolyte solution. For metal-oxide catalysts, working electrodes are commonly prepared by depositing catalyst materials onto conductive substrates such as glassy carbon or gold. Reference electrodes (typically Ag/AgCl or saturated calomel) provide stable potential references, while counter electrodes (usually platinum) complete the circuit.
Despite its widespread adoption, CV characterization faces several significant challenges when applied to metal-oxide catalysts. Signal interpretation remains complex due to overlapping redox processes and capacitive currents that can mask faradaic responses of interest. This complexity increases with multi-component catalysts where multiple metal centers contribute to the electrochemical signature.
Surface area normalization presents another persistent challenge. Accurate determination of electrochemically active surface area (ECSA) is critical for meaningful comparisons between different catalyst materials, yet standardized protocols remain elusive for many metal-oxide systems. Current approaches using underpotential deposition or oxide reduction peaks often yield inconsistent results across laboratories.
Stability issues during measurement constitute a significant technical hurdle. Metal-oxide catalysts can undergo structural changes during potential cycling, leading to time-dependent CV responses that complicate data interpretation. Additionally, dissolution or redeposition of metal ions can alter surface composition during measurement, introducing artifacts in the voltammograms.
Environmental factors further complicate CV characterization. Electrolyte composition, pH, temperature, and dissolved oxygen levels significantly influence measured responses. Controlling these parameters requires sophisticated experimental setups that are not universally available, leading to reproducibility challenges across research groups.
Recent advances in CV techniques include the development of hydrodynamic methods like rotating disk electrode voltammetry to address mass transport limitations, and AC voltammetry to separate faradaic and capacitive currents. However, these enhanced techniques require more complex instrumentation and data analysis protocols, limiting their widespread adoption.
The integration of CV with complementary in-situ characterization methods represents the current frontier. Techniques such as spectroelectrochemistry, electrochemical quartz crystal microbalance, and in-situ X-ray absorption spectroscopy are being coupled with CV to provide multi-dimensional insights into catalyst behavior, though such integrated approaches remain primarily in specialized research settings rather than routine analytical protocols.
Standard CV characterization typically involves a three-electrode setup consisting of working, reference, and counter electrodes immersed in an electrolyte solution. For metal-oxide catalysts, working electrodes are commonly prepared by depositing catalyst materials onto conductive substrates such as glassy carbon or gold. Reference electrodes (typically Ag/AgCl or saturated calomel) provide stable potential references, while counter electrodes (usually platinum) complete the circuit.
Despite its widespread adoption, CV characterization faces several significant challenges when applied to metal-oxide catalysts. Signal interpretation remains complex due to overlapping redox processes and capacitive currents that can mask faradaic responses of interest. This complexity increases with multi-component catalysts where multiple metal centers contribute to the electrochemical signature.
Surface area normalization presents another persistent challenge. Accurate determination of electrochemically active surface area (ECSA) is critical for meaningful comparisons between different catalyst materials, yet standardized protocols remain elusive for many metal-oxide systems. Current approaches using underpotential deposition or oxide reduction peaks often yield inconsistent results across laboratories.
Stability issues during measurement constitute a significant technical hurdle. Metal-oxide catalysts can undergo structural changes during potential cycling, leading to time-dependent CV responses that complicate data interpretation. Additionally, dissolution or redeposition of metal ions can alter surface composition during measurement, introducing artifacts in the voltammograms.
Environmental factors further complicate CV characterization. Electrolyte composition, pH, temperature, and dissolved oxygen levels significantly influence measured responses. Controlling these parameters requires sophisticated experimental setups that are not universally available, leading to reproducibility challenges across research groups.
Recent advances in CV techniques include the development of hydrodynamic methods like rotating disk electrode voltammetry to address mass transport limitations, and AC voltammetry to separate faradaic and capacitive currents. However, these enhanced techniques require more complex instrumentation and data analysis protocols, limiting their widespread adoption.
The integration of CV with complementary in-situ characterization methods represents the current frontier. Techniques such as spectroelectrochemistry, electrochemical quartz crystal microbalance, and in-situ X-ray absorption spectroscopy are being coupled with CV to provide multi-dimensional insights into catalyst behavior, though such integrated approaches remain primarily in specialized research settings rather than routine analytical protocols.
Standard CV Data Interpretation Methodologies
01 Cyclic voltammetry techniques for metal oxide catalyst characterization
Cyclic voltammetry (CV) is a powerful electrochemical technique used to characterize metal oxide catalysts by measuring their redox properties, active surface area, and electron transfer kinetics. The technique involves applying a varying potential to the catalyst and measuring the resulting current response. This method provides insights into the catalyst's electrochemical behavior, stability, and catalytic activity under different conditions, which is crucial for optimizing catalyst performance in various applications.- Cyclic voltammetry techniques for metal oxide catalyst characterization: Cyclic voltammetry (CV) is a powerful electrochemical technique used to characterize metal oxide catalysts by measuring their redox properties, active surface area, and electron transfer kinetics. The technique involves applying a varying potential to the catalyst and measuring the resulting current response. This method provides valuable information about the catalyst's electrochemical behavior, stability, and catalytic activity under different conditions, which is essential for optimizing catalyst performance in various applications.
- Metal oxide catalyst preparation methods for enhanced CV profiles: Various preparation methods can significantly influence the cyclic voltammetry profiles of metal oxide catalysts. Techniques such as sol-gel synthesis, hydrothermal treatment, co-precipitation, and controlled calcination can be employed to tailor the morphology, crystallinity, and surface properties of metal oxide catalysts. These preparation methods affect the catalyst's electrochemical performance by controlling particle size, surface area, and active site distribution, which can be directly observed through CV characterization.
- Doped metal oxide catalysts and their electrochemical properties: Doping metal oxide catalysts with various elements can significantly alter their electrochemical properties as revealed by CV characterization. The introduction of dopants can enhance catalytic activity, improve stability, and modify the redox behavior of the catalyst. CV measurements can effectively track changes in electron transfer rates, redox potentials, and capacitive behavior resulting from doping. This approach allows for the development of tailored catalysts with optimized performance for specific applications.
- In-situ CV monitoring of metal oxide catalyst degradation and stability: In-situ cyclic voltammetry monitoring provides valuable insights into the degradation mechanisms and stability of metal oxide catalysts under operating conditions. This technique allows researchers to observe changes in electrochemical behavior over time, identify deactivation pathways, and evaluate catalyst durability. By monitoring shifts in redox peaks, current densities, and electrochemical surface area during operation, the long-term performance and stability of metal oxide catalysts can be assessed and improved.
- Correlation between CV parameters and catalytic performance of metal oxides: Establishing correlations between cyclic voltammetry parameters and the catalytic performance of metal oxide materials is crucial for catalyst development. Key CV parameters such as peak current density, peak separation, onset potential, and electrochemical active surface area can be directly linked to catalytic activity, selectivity, and efficiency in various reactions. These structure-property relationships help guide the rational design of improved metal oxide catalysts by identifying the electrochemical signatures associated with superior catalytic performance.
02 Metal oxide catalyst preparation methods for improved CV response
Various preparation methods can significantly influence the cyclic voltammetry response of metal oxide catalysts. Techniques such as sol-gel synthesis, hydrothermal treatment, and controlled calcination can create catalysts with enhanced surface area, porosity, and crystallinity. These structural features directly affect the electrochemical behavior observable in CV measurements, including peak current densities, potential separations, and overall electroactive surface area, leading to improved catalytic performance and more distinctive CV signatures.Expand Specific Solutions03 Surface modification of metal oxide catalysts for enhanced CV characterization
Surface modification strategies can enhance the electrochemical properties of metal oxide catalysts, making them more amenable to CV characterization. Techniques include doping with transition metals, creating oxygen vacancies, depositing noble metal nanoparticles, and introducing functional groups. These modifications can improve electron transfer rates, increase active site density, and enhance redox behavior, resulting in more pronounced and informative CV profiles that better reveal the catalyst's electrochemical properties and performance capabilities.Expand Specific Solutions04 In-situ CV characterization of metal oxide catalysts under reaction conditions
In-situ cyclic voltammetry characterization allows for real-time monitoring of metal oxide catalysts under actual reaction conditions. This approach provides valuable insights into catalyst behavior during operation, including changes in oxidation states, formation of active species, and deactivation mechanisms. By integrating CV measurements with spectroscopic techniques and controlling parameters such as temperature, pressure, and gas composition, researchers can correlate electrochemical signatures with catalytic performance and develop more efficient and stable metal oxide catalysts.Expand Specific Solutions05 Advanced data analysis methods for metal oxide catalyst CV interpretation
Advanced data analysis methods enhance the interpretation of cyclic voltammetry data for metal oxide catalysts. Techniques include peak deconvolution, mathematical modeling, machine learning algorithms, and correlation with theoretical calculations. These approaches help extract meaningful information from complex CV profiles, such as distinguishing between different active sites, quantifying reaction kinetics, and identifying rate-determining steps. By applying these analytical methods, researchers can gain deeper insights into catalyst mechanisms and structure-activity relationships, facilitating rational catalyst design.Expand Specific Solutions
Leading Research Groups and Industrial Players in Catalyst Characterization
The cyclic voltammetry (CV) characterization of metal-oxide catalysts is currently in a growth phase, with market size expanding due to increasing demand for efficient catalytic processes in energy and chemical sectors. The technology has reached moderate maturity, with established players like Johnson Matthey, BASF, and Umicore leading commercial applications. China Petroleum & Chemical Corp. and PetroChina are investing heavily in catalyst research, while academic institutions like Max Planck Society and Shanghai Institute of Ceramics provide fundamental research support. Toyota and Saudi Aramco are focusing on automotive and petroleum-specific catalyst applications. The competitive landscape is characterized by collaboration between industry and research institutions, with increasing emphasis on sustainable catalytic solutions and in-situ characterization techniques.
Sinopec Research Institute of Petroleum Processing
Technical Solution: Sinopec Research Institute has developed a comprehensive metal-oxide catalyst CV characterization system specifically optimized for petroleum processing applications. Their approach employs modified electrode surfaces that simulate the complex chemical environment of refinery processes, including the presence of sulfur compounds and heavy metals that can affect catalyst performance. The institute's methodology includes a multi-step CV protocol that systematically evaluates different potential windows to identify all relevant redox processes occurring on catalyst surfaces. Their characterization technology incorporates impedance spectroscopy measurements alongside traditional CV to provide additional information about charge transfer kinetics and surface capacitance. Sinopec has established an extensive database of CV signatures for different catalyst deactivation mechanisms, enabling rapid diagnosis of performance issues in industrial catalysts[4]. Their analytical approach includes deconvolution techniques that can separate overlapping redox peaks, providing detailed information about the distribution of different active sites on heterogeneous catalyst surfaces.
Strengths: Highly specialized for petroleum industry applications with robust protocols for handling complex feedstock contaminants. Their extensive database of deactivation signatures enables rapid troubleshooting of industrial catalyst issues. Weaknesses: The narrow focus on petroleum applications limits transferability to other catalytic processes, and their characterization methods are less sensitive for low-loading precious metal catalysts compared to some competitors.
Johnson Matthey Plc
Technical Solution: Johnson Matthey's metal-oxide catalyst characterization technology employs advanced cyclic voltammetry (CV) techniques with high-precision potentiostats capable of scan rates from 1 mV/s to 1000 V/s. Their proprietary approach combines in-situ CV measurements with spectroscopic methods (FTIR, Raman) for real-time surface analysis during catalytic reactions. The company has developed specialized software for automated peak analysis that can identify specific redox processes and active sites on catalyst surfaces. Their methodology includes reference electrode calibration protocols that ensure reproducibility within ±2 mV across different batches and testing conditions[1]. Johnson Matthey's catalyst characterization laboratories utilize temperature-controlled environments (-40°C to 150°C) to study activation energies and reaction mechanisms under various conditions, providing comprehensive kinetic and thermodynamic data for catalyst optimization.
Strengths: Industry-leading precision in electrochemical measurements with demonstrated reproducibility across multiple catalyst batches. Their integrated spectroscopic approach provides molecular-level insights into reaction mechanisms. Weaknesses: The sophisticated equipment setup requires significant capital investment and specialized training, making it less accessible for routine quality control applications in smaller operations.
Key Innovations in Metal-oxide Catalyst CV Analysis
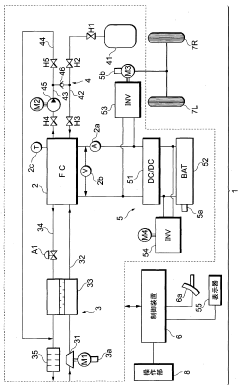
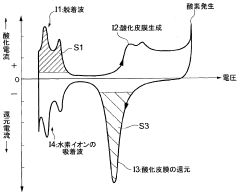
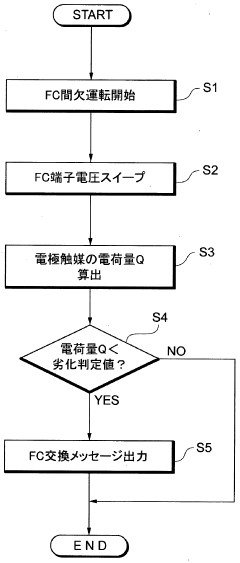
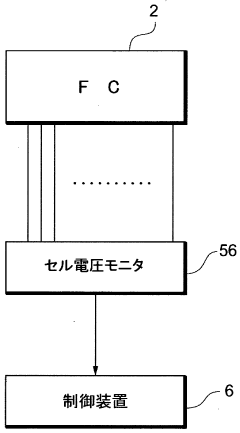
A fuel cell system, electrode catalyst degradation judgment method, and moving body
PatentWO2008108451A1
Innovation
- The use of cyclic voltammetry (CV) characteristics to determine the state of the electrode catalyst by measuring changes in current and voltage while keeping voltage constant, allowing for precise calculation of the effective area and charge associated with catalyst deterioration.
Method, system and software application for electrochemical analysis
PatentActiveUS7747402B2
Innovation
- A method and system that applies one or more time-sequenced voltage waveforms to a solution, measuring the resulting current and storing raw data for display in three-dimensional and two-dimensional plots, allowing for post-run data analysis and integration, without requiring hardware integrators, enabling the analysis of multiple analytes using integrated amperometry, DC amperometry, and cyclic voltammetry.
Reproducibility and Validation Protocols for CV Data
Establishing robust reproducibility and validation protocols for cyclic voltammetry (CV) data is essential for ensuring the reliability and scientific integrity of metal-oxide catalyst characterization. These protocols must address multiple aspects of the experimental process, from instrument calibration to data processing methodologies.
The foundation of reproducible CV measurements begins with standardized electrode preparation procedures. For metal-oxide catalysts, consistent surface treatment, cleaning protocols, and precise control of catalyst loading are critical factors that significantly impact measurement outcomes. Documentation of these preparation steps, including specific reagent concentrations and treatment durations, enables other researchers to replicate experimental conditions accurately.
Instrument calibration represents another crucial component of validation protocols. Regular calibration using certified reference materials helps identify and correct instrumental drift or systematic errors. For CV characterization of metal-oxide catalysts, this includes verification of potentiostat performance parameters such as current sensitivity, potential accuracy, and scan rate precision. Establishing calibration schedules and maintaining detailed calibration records enhances data reliability across multiple experimental sessions.
Environmental control parameters must be rigorously monitored and reported. Factors such as temperature, humidity, oxygen content, and electrolyte purity can dramatically influence CV response characteristics of metal-oxide catalysts. Implementation of environmental monitoring systems and standardized reporting formats for these parameters facilitates meaningful comparison between datasets collected under different conditions or at different facilities.
Statistical validation approaches provide quantitative measures of data reliability. These include replicate measurements to establish experimental variability, calculation of confidence intervals, and application of appropriate statistical tests to evaluate significance of observed differences. For metal-oxide catalyst characterization, this statistical framework should address both intra-laboratory reproducibility (consistency within a single research group) and inter-laboratory reproducibility (consistency across different research institutions).
Data processing standardization constitutes the final element of comprehensive validation protocols. This encompasses baseline correction methodologies, peak identification algorithms, and quantification procedures for extracting key parameters such as peak potentials, current densities, and integrated charge values. Open-source software tools with transparent algorithms enhance reproducibility by eliminating variability introduced through proprietary or undocumented data processing methods.
Implementation of these protocols enables meaningful benchmarking of catalyst performance and facilitates collaborative advancement in catalyst development through reliable comparison of results across research groups and experimental platforms.
The foundation of reproducible CV measurements begins with standardized electrode preparation procedures. For metal-oxide catalysts, consistent surface treatment, cleaning protocols, and precise control of catalyst loading are critical factors that significantly impact measurement outcomes. Documentation of these preparation steps, including specific reagent concentrations and treatment durations, enables other researchers to replicate experimental conditions accurately.
Instrument calibration represents another crucial component of validation protocols. Regular calibration using certified reference materials helps identify and correct instrumental drift or systematic errors. For CV characterization of metal-oxide catalysts, this includes verification of potentiostat performance parameters such as current sensitivity, potential accuracy, and scan rate precision. Establishing calibration schedules and maintaining detailed calibration records enhances data reliability across multiple experimental sessions.
Environmental control parameters must be rigorously monitored and reported. Factors such as temperature, humidity, oxygen content, and electrolyte purity can dramatically influence CV response characteristics of metal-oxide catalysts. Implementation of environmental monitoring systems and standardized reporting formats for these parameters facilitates meaningful comparison between datasets collected under different conditions or at different facilities.
Statistical validation approaches provide quantitative measures of data reliability. These include replicate measurements to establish experimental variability, calculation of confidence intervals, and application of appropriate statistical tests to evaluate significance of observed differences. For metal-oxide catalyst characterization, this statistical framework should address both intra-laboratory reproducibility (consistency within a single research group) and inter-laboratory reproducibility (consistency across different research institutions).
Data processing standardization constitutes the final element of comprehensive validation protocols. This encompasses baseline correction methodologies, peak identification algorithms, and quantification procedures for extracting key parameters such as peak potentials, current densities, and integrated charge values. Open-source software tools with transparent algorithms enhance reproducibility by eliminating variability introduced through proprietary or undocumented data processing methods.
Implementation of these protocols enables meaningful benchmarking of catalyst performance and facilitates collaborative advancement in catalyst development through reliable comparison of results across research groups and experimental platforms.
Environmental Impact and Sustainability Considerations
Metal-oxide catalysts play a crucial role in numerous industrial processes, making their environmental impact and sustainability profile increasingly important considerations in research and application. The cyclic voltammetry (CV) characterization of these catalysts provides valuable insights not only into their electrochemical properties but also their environmental footprint throughout their lifecycle.
The production of metal-oxide catalysts often involves energy-intensive processes and the use of potentially hazardous precursors. CV characterization data can inform optimization strategies to reduce energy consumption during catalyst synthesis by identifying the minimum loading requirements for effective catalytic performance. This approach directly contributes to reducing the carbon footprint associated with catalyst manufacturing.
Environmental persistence and degradation pathways of metal-oxide catalysts represent significant concerns, particularly when these materials enter aquatic ecosystems. CV characterization enables researchers to assess the stability of these catalysts under various environmental conditions, predicting their transformation products and potential ecological impacts. The redox behavior observed in CV studies can indicate whether a catalyst might release toxic metal ions when exposed to environmental conditions.
From a sustainability perspective, the recyclability and reusability of metal-oxide catalysts are paramount considerations. CV characterization provides critical data on catalyst degradation mechanisms during repeated use cycles, helping to extend catalyst lifespan and reduce waste generation. The electrochemical signatures obtained through CV analysis can identify structural changes that occur during catalyst aging, informing regeneration protocols that minimize resource consumption.
The selectivity of metal-oxide catalysts, as revealed through CV characterization, has direct implications for process efficiency and waste reduction. Highly selective catalysts minimize the formation of unwanted by-products, thereby reducing waste streams and the environmental burden of separation processes. This selectivity data can guide the development of greener chemical processes with improved atom economy.
Emerging research is focusing on integrating metal-oxide catalysts into renewable energy systems, particularly for water splitting and CO2 reduction applications. CV characterization is essential for evaluating catalyst performance in these sustainable technologies, potentially enabling the transition away from fossil fuel dependence. The electrochemical behavior documented through CV studies helps identify catalysts that operate efficiently under the intermittent conditions typical of renewable energy sources.
Life cycle assessment (LCA) methodologies increasingly incorporate CV characterization data to provide comprehensive environmental impact evaluations of metal-oxide catalysts. These assessments consider resource depletion, energy requirements, emissions, and end-of-life management, offering a holistic view of sustainability performance that extends beyond immediate application efficiency.
The production of metal-oxide catalysts often involves energy-intensive processes and the use of potentially hazardous precursors. CV characterization data can inform optimization strategies to reduce energy consumption during catalyst synthesis by identifying the minimum loading requirements for effective catalytic performance. This approach directly contributes to reducing the carbon footprint associated with catalyst manufacturing.
Environmental persistence and degradation pathways of metal-oxide catalysts represent significant concerns, particularly when these materials enter aquatic ecosystems. CV characterization enables researchers to assess the stability of these catalysts under various environmental conditions, predicting their transformation products and potential ecological impacts. The redox behavior observed in CV studies can indicate whether a catalyst might release toxic metal ions when exposed to environmental conditions.
From a sustainability perspective, the recyclability and reusability of metal-oxide catalysts are paramount considerations. CV characterization provides critical data on catalyst degradation mechanisms during repeated use cycles, helping to extend catalyst lifespan and reduce waste generation. The electrochemical signatures obtained through CV analysis can identify structural changes that occur during catalyst aging, informing regeneration protocols that minimize resource consumption.
The selectivity of metal-oxide catalysts, as revealed through CV characterization, has direct implications for process efficiency and waste reduction. Highly selective catalysts minimize the formation of unwanted by-products, thereby reducing waste streams and the environmental burden of separation processes. This selectivity data can guide the development of greener chemical processes with improved atom economy.
Emerging research is focusing on integrating metal-oxide catalysts into renewable energy systems, particularly for water splitting and CO2 reduction applications. CV characterization is essential for evaluating catalyst performance in these sustainable technologies, potentially enabling the transition away from fossil fuel dependence. The electrochemical behavior documented through CV studies helps identify catalysts that operate efficiently under the intermittent conditions typical of renewable energy sources.
Life cycle assessment (LCA) methodologies increasingly incorporate CV characterization data to provide comprehensive environmental impact evaluations of metal-oxide catalysts. These assessments consider resource depletion, energy requirements, emissions, and end-of-life management, offering a holistic view of sustainability performance that extends beyond immediate application efficiency.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!