Cyclic Voltammetry for Corrosion Assessments — Protocol, Metrics and Reporting Template
AUG 21, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Cyclic Voltammetry Corrosion Assessment Background and Objectives
Cyclic voltammetry (CV) has evolved significantly since its inception in the early 20th century as an electroanalytical technique. Initially developed for studying redox reactions, its application has expanded dramatically into materials science, particularly in corrosion assessment. The technique's ability to provide detailed information about electrochemical processes at material surfaces has made it invaluable for understanding corrosion mechanisms across various industries including aerospace, maritime, infrastructure, and electronics.
The evolution of CV technology has been marked by several key advancements. Early implementations were limited by rudimentary potentiostats and recording equipment, but modern digital systems now offer unprecedented precision and data processing capabilities. This technological progression has transformed CV from a specialized laboratory technique to an essential tool in industrial corrosion monitoring and prevention strategies.
Recent developments have focused on standardizing CV protocols for corrosion assessment, addressing the historical challenge of inconsistent methodologies that hindered result comparability across different research groups and industries. The need for standardization has become increasingly critical as materials complexity grows and as industries face stricter safety and durability requirements.
The primary objective of developing comprehensive CV protocols for corrosion assessment is to establish a unified framework that ensures reproducibility, reliability, and comparability of results. This includes standardizing experimental parameters such as scan rates, potential windows, reference electrodes, and electrolyte compositions specific to different material systems and corrosion environments.
Additionally, there is a pressing need to define universal metrics for quantifying corrosion susceptibility from CV data. Current practices vary widely, with different industries and researchers employing diverse parameters such as corrosion potential, polarization resistance, and passive current density. A standardized set of metrics would facilitate more meaningful comparisons and more accurate predictive models.
The reporting template component aims to address the inconsistency in data presentation that has hampered knowledge transfer and meta-analysis in the field. A structured reporting format would ensure that all critical experimental conditions, material characteristics, and analytical methods are documented, enabling better reproduction of results and more effective knowledge sharing across the scientific and engineering communities.
Looking forward, the integration of CV with other analytical techniques and computational modeling represents a significant trend, potentially offering more comprehensive corrosion assessment capabilities. Machine learning approaches are increasingly being applied to CV data interpretation, promising more accurate prediction of long-term corrosion behavior from short-term electrochemical measurements.
The evolution of CV technology has been marked by several key advancements. Early implementations were limited by rudimentary potentiostats and recording equipment, but modern digital systems now offer unprecedented precision and data processing capabilities. This technological progression has transformed CV from a specialized laboratory technique to an essential tool in industrial corrosion monitoring and prevention strategies.
Recent developments have focused on standardizing CV protocols for corrosion assessment, addressing the historical challenge of inconsistent methodologies that hindered result comparability across different research groups and industries. The need for standardization has become increasingly critical as materials complexity grows and as industries face stricter safety and durability requirements.
The primary objective of developing comprehensive CV protocols for corrosion assessment is to establish a unified framework that ensures reproducibility, reliability, and comparability of results. This includes standardizing experimental parameters such as scan rates, potential windows, reference electrodes, and electrolyte compositions specific to different material systems and corrosion environments.
Additionally, there is a pressing need to define universal metrics for quantifying corrosion susceptibility from CV data. Current practices vary widely, with different industries and researchers employing diverse parameters such as corrosion potential, polarization resistance, and passive current density. A standardized set of metrics would facilitate more meaningful comparisons and more accurate predictive models.
The reporting template component aims to address the inconsistency in data presentation that has hampered knowledge transfer and meta-analysis in the field. A structured reporting format would ensure that all critical experimental conditions, material characteristics, and analytical methods are documented, enabling better reproduction of results and more effective knowledge sharing across the scientific and engineering communities.
Looking forward, the integration of CV with other analytical techniques and computational modeling represents a significant trend, potentially offering more comprehensive corrosion assessment capabilities. Machine learning approaches are increasingly being applied to CV data interpretation, promising more accurate prediction of long-term corrosion behavior from short-term electrochemical measurements.
Market Applications and Demand for Corrosion Testing Methods
The global market for corrosion testing methods has experienced significant growth in recent years, driven primarily by increasing awareness of the economic impact of corrosion across various industries. The annual cost of corrosion worldwide is estimated at $2.5 trillion, representing approximately 3.4% of global GDP, according to NACE International (now AMPP). This substantial economic burden has intensified the demand for reliable and efficient corrosion assessment techniques, with Cyclic Voltammetry (CV) emerging as a particularly valuable method.
The oil and gas industry represents the largest market segment for corrosion testing methods, accounting for approximately 25% of the total market share. In this sector, CV techniques are increasingly adopted for monitoring pipeline integrity, offshore platform components, and refinery equipment. The ability of CV to detect localized corrosion phenomena in their early stages provides significant value in preventing catastrophic failures and unplanned shutdowns.
The aerospace and defense sectors have also demonstrated growing demand for advanced electrochemical corrosion assessment methods. With safety-critical components operating in diverse and often extreme environments, these industries require precise and reliable corrosion data. CV techniques offer particular advantages in evaluating protective coatings and surface treatments on aluminum alloys and other lightweight materials used in aircraft construction.
Infrastructure development and maintenance represent another substantial market for corrosion testing. Civil engineering applications, particularly in reinforced concrete structures, benefit from CV's ability to assess corrosion rates of embedded steel reinforcement. The growing focus on infrastructure rehabilitation in developed economies and new construction in emerging markets has expanded this application area significantly.
The automotive industry has increasingly incorporated electrochemical testing methods into quality control processes, particularly as manufacturers adopt new materials and joining techniques. CV methods provide valuable data on galvanic corrosion risks in multi-material assemblies and help validate corrosion protection systems under various environmental conditions.
Emerging applications in biomedical implants and electronic devices have created specialized niches for CV-based corrosion assessment. The biocompatibility of implantable medical devices depends partly on their corrosion resistance, while the miniaturization of electronic components has increased sensitivity to corrosion-related failures.
Market research indicates that North America and Europe currently dominate the corrosion testing equipment market, though the Asia-Pacific region is experiencing the fastest growth rate, driven by industrial expansion in China and India. The standardization of CV protocols and reporting templates would likely accelerate market adoption by improving result comparability and facilitating technology transfer across different industry sectors.
The oil and gas industry represents the largest market segment for corrosion testing methods, accounting for approximately 25% of the total market share. In this sector, CV techniques are increasingly adopted for monitoring pipeline integrity, offshore platform components, and refinery equipment. The ability of CV to detect localized corrosion phenomena in their early stages provides significant value in preventing catastrophic failures and unplanned shutdowns.
The aerospace and defense sectors have also demonstrated growing demand for advanced electrochemical corrosion assessment methods. With safety-critical components operating in diverse and often extreme environments, these industries require precise and reliable corrosion data. CV techniques offer particular advantages in evaluating protective coatings and surface treatments on aluminum alloys and other lightweight materials used in aircraft construction.
Infrastructure development and maintenance represent another substantial market for corrosion testing. Civil engineering applications, particularly in reinforced concrete structures, benefit from CV's ability to assess corrosion rates of embedded steel reinforcement. The growing focus on infrastructure rehabilitation in developed economies and new construction in emerging markets has expanded this application area significantly.
The automotive industry has increasingly incorporated electrochemical testing methods into quality control processes, particularly as manufacturers adopt new materials and joining techniques. CV methods provide valuable data on galvanic corrosion risks in multi-material assemblies and help validate corrosion protection systems under various environmental conditions.
Emerging applications in biomedical implants and electronic devices have created specialized niches for CV-based corrosion assessment. The biocompatibility of implantable medical devices depends partly on their corrosion resistance, while the miniaturization of electronic components has increased sensitivity to corrosion-related failures.
Market research indicates that North America and Europe currently dominate the corrosion testing equipment market, though the Asia-Pacific region is experiencing the fastest growth rate, driven by industrial expansion in China and India. The standardization of CV protocols and reporting templates would likely accelerate market adoption by improving result comparability and facilitating technology transfer across different industry sectors.
Current State and Technical Challenges in Electrochemical Corrosion Analysis
Electrochemical corrosion analysis has evolved significantly over the past decades, with cyclic voltammetry (CV) emerging as one of the most powerful and versatile techniques for corrosion assessment. Currently, the global corrosion testing market is valued at approximately $6.3 billion, with electrochemical methods accounting for roughly 18% of this market share. Despite its widespread adoption, the field faces several critical challenges that impede standardization and reliability.
The current state of CV for corrosion assessment reveals inconsistent protocols across research institutions and industries. While ASTM and NACE have established some guidelines, there remains significant variation in experimental parameters, data interpretation, and reporting formats. This lack of standardization makes cross-comparison of results difficult and hinders knowledge transfer between academic research and industrial applications.
A major technical challenge is the complex interpretation of CV data in real-world corrosion scenarios. Unlike controlled laboratory environments, industrial applications often involve multi-component systems with interfering species, varying temperature conditions, and complex geometries that affect electrochemical responses. Current analytical models struggle to account for these variables, leading to potential misinterpretations of corrosion mechanisms and rates.
Signal-to-noise ratio optimization presents another significant hurdle, particularly in low-conductivity environments or when analyzing localized corrosion phenomena. Advanced filtering algorithms have been developed, but their application remains inconsistent across the field, with many practitioners still relying on manual baseline corrections that introduce subjective elements to the analysis.
The integration of CV with other complementary techniques represents both a current trend and a challenge. While multi-technique approaches combining CV with electrochemical impedance spectroscopy (EIS), scanning electrochemical microscopy (SECM), or surface analysis methods provide more comprehensive insights, the correlation and integration of these diverse data streams remain problematic. Current software solutions typically handle each technique separately, requiring manual correlation by experts.
Miniaturization and in-situ monitoring capabilities are advancing rapidly but face challenges in electrode stability and reference electrode drift during long-term deployments. Recent developments in microelectrode arrays and wireless sensor technologies show promise but require further refinement for reliable field applications in harsh industrial environments.
Data management and interpretation represent perhaps the most pressing challenge. The volume of data generated by modern high-throughput CV systems has outpaced the development of automated analysis tools. Machine learning approaches are beginning to emerge but require extensive validation across diverse corrosion scenarios before widespread adoption can occur.
The current state of CV for corrosion assessment reveals inconsistent protocols across research institutions and industries. While ASTM and NACE have established some guidelines, there remains significant variation in experimental parameters, data interpretation, and reporting formats. This lack of standardization makes cross-comparison of results difficult and hinders knowledge transfer between academic research and industrial applications.
A major technical challenge is the complex interpretation of CV data in real-world corrosion scenarios. Unlike controlled laboratory environments, industrial applications often involve multi-component systems with interfering species, varying temperature conditions, and complex geometries that affect electrochemical responses. Current analytical models struggle to account for these variables, leading to potential misinterpretations of corrosion mechanisms and rates.
Signal-to-noise ratio optimization presents another significant hurdle, particularly in low-conductivity environments or when analyzing localized corrosion phenomena. Advanced filtering algorithms have been developed, but their application remains inconsistent across the field, with many practitioners still relying on manual baseline corrections that introduce subjective elements to the analysis.
The integration of CV with other complementary techniques represents both a current trend and a challenge. While multi-technique approaches combining CV with electrochemical impedance spectroscopy (EIS), scanning electrochemical microscopy (SECM), or surface analysis methods provide more comprehensive insights, the correlation and integration of these diverse data streams remain problematic. Current software solutions typically handle each technique separately, requiring manual correlation by experts.
Miniaturization and in-situ monitoring capabilities are advancing rapidly but face challenges in electrode stability and reference electrode drift during long-term deployments. Recent developments in microelectrode arrays and wireless sensor technologies show promise but require further refinement for reliable field applications in harsh industrial environments.
Data management and interpretation represent perhaps the most pressing challenge. The volume of data generated by modern high-throughput CV systems has outpaced the development of automated analysis tools. Machine learning approaches are beginning to emerge but require extensive validation across diverse corrosion scenarios before widespread adoption can occur.
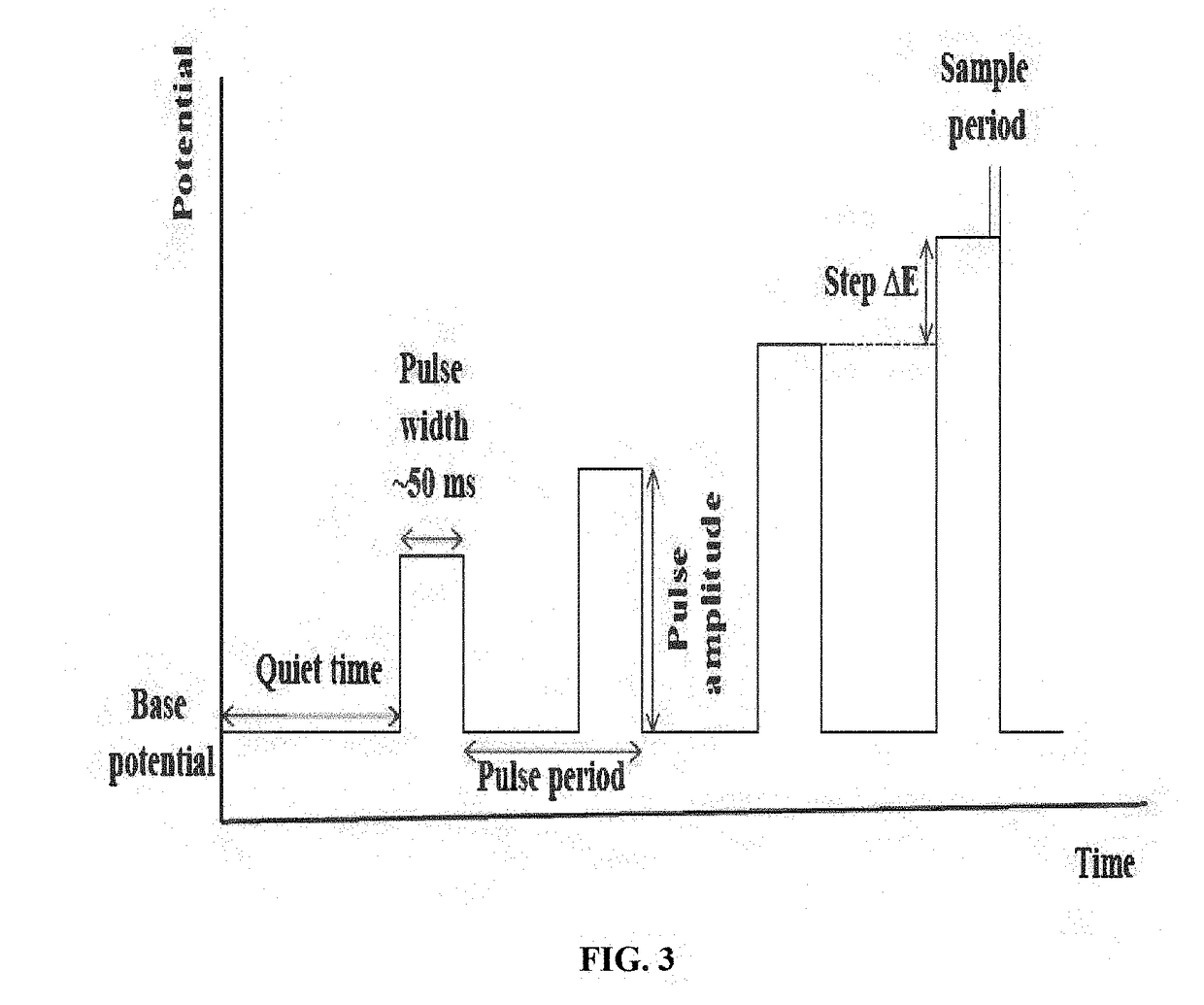
Standard Protocols and Methodologies for CV Corrosion Measurements
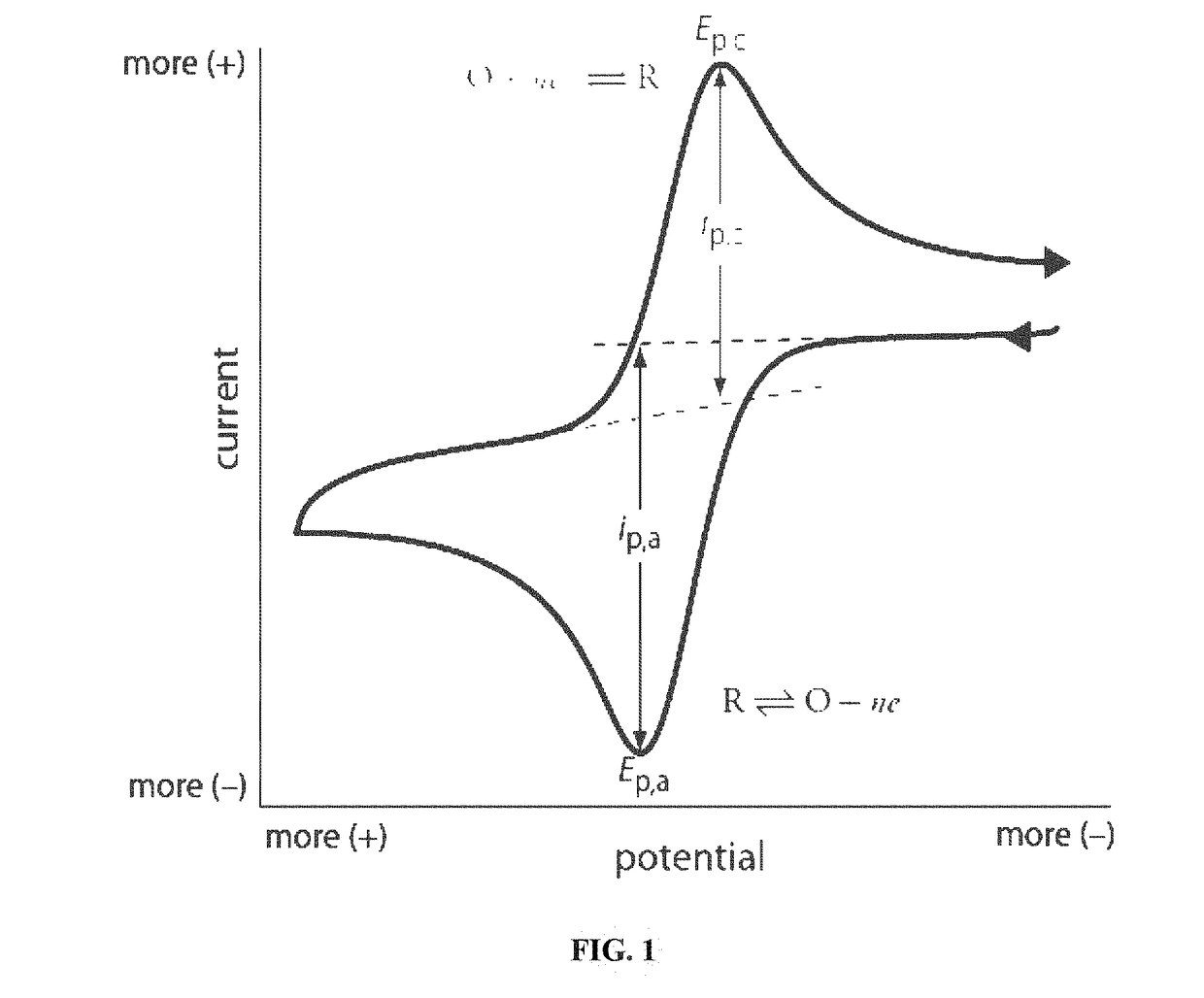
01 Cyclic voltammetry techniques for corrosion rate measurement
Cyclic voltammetry can be used to measure corrosion rates by analyzing the electrochemical behavior of materials in corrosive environments. This technique involves applying a varying potential to a sample and measuring the resulting current response. The data obtained can be used to calculate corrosion rates, identify corrosion mechanisms, and evaluate the effectiveness of corrosion inhibitors. This method provides quantitative information about the corrosion behavior of materials under different conditions.- Cyclic voltammetry techniques for corrosion rate measurement: Cyclic voltammetry can be used to measure and evaluate corrosion rates of various materials by analyzing the electrochemical behavior during potential cycling. This technique allows for the determination of corrosion current density, which directly correlates to corrosion rate. The method involves scanning the potential in both anodic and cathodic directions while measuring the resulting current, providing information about the kinetics of corrosion processes and the effectiveness of corrosion inhibitors.
- Corrosion assessment of metal alloys and coatings: Cyclic voltammetry is employed to assess the corrosion resistance of various metal alloys and protective coatings. The technique can evaluate the performance of different coating compositions and thicknesses, as well as the corrosion behavior of specialized alloys in aggressive environments. By analyzing the voltammetric curves, researchers can identify the formation of passive films, pitting susceptibility, and overall corrosion protection mechanisms, enabling the development of more corrosion-resistant materials.
- Environmental and solution effects on corrosion behavior: The influence of environmental factors and solution composition on corrosion behavior can be systematically studied using cyclic voltammetry. This includes the effects of pH, temperature, ion concentration, and presence of specific aggressive species on the corrosion mechanisms. The technique allows for comparative analysis of corrosion behavior under different conditions, helping to identify critical environmental parameters that accelerate or inhibit corrosion processes.
- Advanced cyclic voltammetry methodologies for corrosion inhibitor evaluation: Advanced cyclic voltammetry methodologies have been developed specifically for evaluating the effectiveness of corrosion inhibitors. These techniques can quantify inhibition efficiency, determine optimal inhibitor concentrations, and elucidate inhibition mechanisms. By comparing voltammograms obtained with and without inhibitors, researchers can assess how these compounds affect anodic and cathodic reactions, adsorption processes, and the formation of protective films on metal surfaces.
- Integration of cyclic voltammetry with other analytical techniques: Cyclic voltammetry can be integrated with other analytical techniques to provide comprehensive corrosion assessments. This includes combining voltammetry with electrochemical impedance spectroscopy, surface analysis methods, computational modeling, and machine learning approaches. Such integrated methodologies enable more detailed characterization of corrosion mechanisms, improved prediction of long-term corrosion behavior, and development of more effective corrosion mitigation strategies for complex systems.
02 Corrosion assessment of metal alloys and coatings
Cyclic voltammetry is employed to assess the corrosion resistance of various metal alloys and protective coatings. By analyzing the voltammetric curves, researchers can determine the susceptibility of different materials to corrosion in specific environments. This technique helps in comparing the performance of various alloys and coatings, enabling the selection of optimal materials for corrosion-resistant applications. The method is particularly useful for evaluating new alloy compositions and coating technologies.Expand Specific Solutions03 In-situ corrosion monitoring systems
In-situ corrosion monitoring systems utilize cyclic voltammetry to provide real-time assessment of corrosion processes in operational environments. These systems incorporate electrochemical sensors that can be deployed in industrial settings to continuously monitor corrosion activity. The data collected can be used for early detection of corrosion issues, allowing for timely intervention and prevention of equipment failure. These monitoring systems are particularly valuable in high-risk environments where corrosion can lead to significant safety hazards or economic losses.Expand Specific Solutions04 Electrochemical impedance analysis combined with cyclic voltammetry
The combination of electrochemical impedance spectroscopy (EIS) with cyclic voltammetry provides comprehensive corrosion assessment capabilities. This integrated approach allows for the characterization of both the kinetic and mechanistic aspects of corrosion processes. By analyzing the impedance data alongside voltammetric measurements, researchers can gain insights into the formation and properties of passive films, the influence of surface treatments, and the effectiveness of corrosion inhibitors. This combined methodology enhances the accuracy and reliability of corrosion assessments.Expand Specific Solutions05 Advanced data analysis methods for cyclic voltammetry corrosion data
Advanced computational methods and algorithms are being developed to analyze cyclic voltammetry data for corrosion assessment. These include machine learning approaches, statistical analysis techniques, and mathematical modeling to extract meaningful information from complex electrochemical data. By applying these advanced analytical methods, researchers can identify subtle patterns in corrosion behavior, predict long-term corrosion performance, and optimize corrosion protection strategies. These techniques enhance the sensitivity and specificity of cyclic voltammetry for corrosion assessment applications.Expand Specific Solutions
Leading Organizations and Researchers in Electrochemical Corrosion Testing
The cyclic voltammetry corrosion assessment field is currently in a growth phase, with an expanding market driven by increasing industrial demands for reliable corrosion monitoring. The technology has reached moderate maturity, with established protocols emerging but significant room for innovation remains. Key players include major energy corporations like China Petroleum & Chemical Corp. and Schlumberger, who are investing in advanced corrosion assessment technologies. Equipment manufacturers such as Yokogawa Electric and Teledyne Instruments are developing specialized instrumentation, while research institutions like Johns Hopkins University and USC contribute to protocol standardization. Baker Hughes and Halliburton are integrating these technologies into comprehensive asset integrity solutions, particularly for challenging environments in oil and gas applications.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed an advanced cyclic voltammetry (CV) protocol specifically tailored for oil and gas infrastructure corrosion assessment. Their approach integrates multi-frequency electrochemical impedance spectroscopy with CV to create a comprehensive corrosion evaluation system. The protocol employs specialized three-electrode cells with reference electrodes calibrated to specific environments (sour gas, high-temperature crude, etc.) and utilizes scan rates optimized between 0.1-10 mV/s to capture both fast and slow corrosion processes. Sinopec's system incorporates automated data processing algorithms that extract critical parameters including corrosion current density, passivation potential, and pitting susceptibility indices. Their reporting template standardizes results across different field conditions, enabling comparative analysis across their vast pipeline network. The system has been validated across various hydrocarbon environments and has demonstrated superior sensitivity in detecting localized corrosion compared to traditional weight-loss methods.
Strengths: Highly specialized for oil and gas environments with proven field implementation across diverse conditions. The integration with EIS provides multi-dimensional corrosion data. Weaknesses: The system requires specialized training and equipment, making deployment costly in remote locations. The protocol may be overly tailored to petroleum environments, limiting transferability to other industries.
Baker Hughes Co.
Technical Solution: Baker Hughes has pioneered a high-pressure, high-temperature (HPHT) cyclic voltammetry system specifically designed for downhole corrosion monitoring in extreme well environments. Their protocol utilizes miniaturized three-electrode arrays with proprietary reference electrodes stable at temperatures up to 200°C and pressures to 20,000 psi. The system employs adaptive scan rates that automatically adjust based on detected electrochemical response, typically ranging from 0.5-50 mV/s, with higher rates used for rapid field assessments and lower rates for detailed laboratory analysis. Baker Hughes' approach incorporates machine learning algorithms that analyze voltammograms to distinguish between different corrosion mechanisms (uniform, pitting, stress corrosion cracking) and predict remaining component life. Their reporting template includes standardized corrosion rate calculations alongside confidence intervals based on historical data patterns. The system transmits data in real-time via their WellLink™ monitoring platform, allowing for immediate intervention when corrosion parameters exceed predefined thresholds.
Strengths: Exceptional performance in extreme environments with real-time monitoring capabilities. The integration with predictive analytics provides actionable insights beyond simple corrosion rates. Weaknesses: The proprietary nature of their system creates vendor lock-in, and the sophisticated electronics require regular calibration and maintenance, which can be challenging in remote operations.
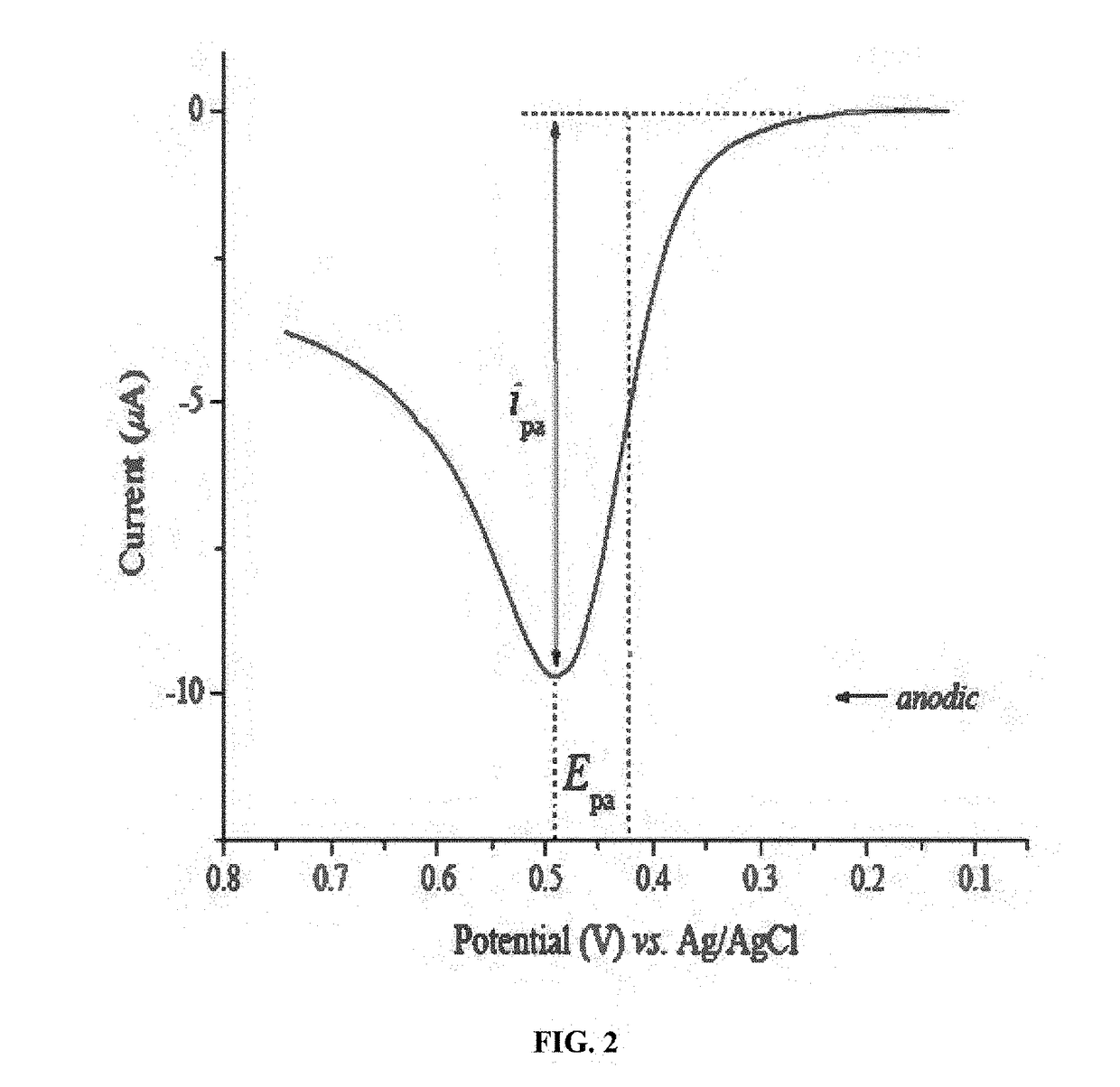
Key Metrics and Data Interpretation in CV Corrosion Analysis
Rare earth metal incorporated zeolite modified electrodes for detection and quantification of heavy metal ions in aqueous solution
PatentInactiveUS20170315079A1
Innovation
- Development of rare earth metal impregnated zeolite modified carbon paste electrodes, specifically lanthanum or cerium impregnated mordenite electrodes, for use in square wave anodic stripping voltammetry, enhancing electroactive surface area and detection limits.
Cobalt chemistry for smooth topology
PatentWO2021118972A1
Innovation
- The use of cyclic voltammetry to screen and evaluate additives in cobalt electroplating baths, combined with a cobalt electrolyte composition including boric acid, organic suppressors, and optional additives, to achieve a seamless, seam-free, and void-free cobalt deposit with controlled overburden thickness.
Standardization and Quality Control in CV Corrosion Testing
Standardization and quality control in cyclic voltammetry (CV) for corrosion assessments are critical for ensuring reliable, reproducible, and comparable results across different laboratories and research institutions. The current landscape reveals significant variability in testing protocols, data interpretation, and reporting formats, which hampers the advancement of corrosion science and engineering applications.
Establishing standardized protocols begins with electrode preparation, where surface conditions must be precisely controlled. This includes standardized polishing procedures, cleaning protocols, and surface characterization methods. The lack of uniformity in these preparatory steps often leads to inconsistent baseline measurements and compromises the validity of comparative studies.
Reference electrode selection and maintenance represent another critical aspect requiring standardization. Variations in reference electrode potential can significantly alter interpretation of corrosion parameters. Industry consensus is emerging around specific reference electrodes for particular environments, with silver/silver chloride and saturated calomel electrodes being predominant choices for aqueous systems.
Quality control measures must include regular calibration of potentiostats using certified reference materials. These calibrations should be performed at frequencies determined by usage patterns and environmental conditions. Documentation of calibration history provides essential metadata for result validation and inter-laboratory comparisons.
Temperature control during CV measurements has been identified as a major source of variability. Standardized protocols increasingly specify temperature ranges of ±0.5°C, with continuous monitoring and reporting throughout the measurement process. This level of control is particularly important for corrosion processes with high temperature sensitivity.
Scan rate standardization represents another key parameter requiring quality control. Research indicates that optimal scan rates vary by material system and corrosion mechanism, necessitating application-specific guidelines rather than universal standards. However, consistency within comparative studies remains paramount.
Data processing algorithms also require standardization, particularly for baseline correction, peak identification, and calculation of corrosion parameters. Open-source software solutions with transparent algorithms are gaining traction as they enable reproducibility verification and eliminate proprietary "black box" processing issues.
Reporting templates must include comprehensive metadata about experimental conditions, equipment specifications, and data processing methods. The emerging consensus favors machine-readable formats that facilitate automated analysis and database integration, supporting broader meta-analyses and artificial intelligence applications in corrosion science.
Proficiency testing programs and round-robin studies are increasingly being implemented to validate laboratory competence and identify systematic errors in CV corrosion assessment methodologies. These collaborative efforts are essential for refining standards and establishing confidence intervals for critical corrosion parameters.
Establishing standardized protocols begins with electrode preparation, where surface conditions must be precisely controlled. This includes standardized polishing procedures, cleaning protocols, and surface characterization methods. The lack of uniformity in these preparatory steps often leads to inconsistent baseline measurements and compromises the validity of comparative studies.
Reference electrode selection and maintenance represent another critical aspect requiring standardization. Variations in reference electrode potential can significantly alter interpretation of corrosion parameters. Industry consensus is emerging around specific reference electrodes for particular environments, with silver/silver chloride and saturated calomel electrodes being predominant choices for aqueous systems.
Quality control measures must include regular calibration of potentiostats using certified reference materials. These calibrations should be performed at frequencies determined by usage patterns and environmental conditions. Documentation of calibration history provides essential metadata for result validation and inter-laboratory comparisons.
Temperature control during CV measurements has been identified as a major source of variability. Standardized protocols increasingly specify temperature ranges of ±0.5°C, with continuous monitoring and reporting throughout the measurement process. This level of control is particularly important for corrosion processes with high temperature sensitivity.
Scan rate standardization represents another key parameter requiring quality control. Research indicates that optimal scan rates vary by material system and corrosion mechanism, necessitating application-specific guidelines rather than universal standards. However, consistency within comparative studies remains paramount.
Data processing algorithms also require standardization, particularly for baseline correction, peak identification, and calculation of corrosion parameters. Open-source software solutions with transparent algorithms are gaining traction as they enable reproducibility verification and eliminate proprietary "black box" processing issues.
Reporting templates must include comprehensive metadata about experimental conditions, equipment specifications, and data processing methods. The emerging consensus favors machine-readable formats that facilitate automated analysis and database integration, supporting broader meta-analyses and artificial intelligence applications in corrosion science.
Proficiency testing programs and round-robin studies are increasingly being implemented to validate laboratory competence and identify systematic errors in CV corrosion assessment methodologies. These collaborative efforts are essential for refining standards and establishing confidence intervals for critical corrosion parameters.
Environmental and Industry-Specific Considerations for CV Corrosion Protocols
Cyclic voltammetry (CV) protocols for corrosion assessment must be adapted to specific environmental conditions and industry requirements to ensure accurate and relevant results. Different industries face unique corrosion challenges based on their operational environments, materials used, and exposure conditions.
In marine environments, CV protocols must account for high chloride content, varying oxygen levels, and biological fouling factors. Testing parameters should include extended potential ranges to capture the complex redox reactions occurring in seawater, with scan rates adjusted to account for the slow diffusion processes in high-salinity conditions. Marine industry standards often require specific reference electrodes stable in chloride-rich environments, such as silver/silver chloride electrodes.
Oil and gas industries require CV protocols that address high-temperature, high-pressure conditions and the presence of hydrogen sulfide and carbon dioxide. These environments demand specialized electrochemical cells capable of withstanding extreme conditions while maintaining measurement accuracy. Scan rates must be optimized to detect sulfide-induced corrosion mechanisms, which often manifest at different potential ranges than oxygen-driven corrosion.
For aerospace applications, CV protocols must consider the effects of altitude, temperature cycling, and exposure to de-icing chemicals. The protocols should incorporate multiple scan cycles to simulate the repeated stress conditions experienced by aerospace components, with particular attention to potential ranges where aluminum and titanium alloys exhibit passivation breakdown.
Nuclear industry applications demand CV protocols that account for radiation effects on electrochemical processes and the presence of unique coolant chemistries. Testing must often be conducted remotely, requiring robust instrumentation and data acquisition systems capable of operating in high-radiation environments.
Electronic manufacturing requires micro-scale CV techniques to assess corrosion on circuit boards and components. Protocols must be adapted for low-current measurements and confined spaces, often utilizing microelectrodes and specialized cell designs to evaluate corrosion in the presence of flux residues and under condensing humidity conditions.
Standardization efforts across industries have led to the development of industry-specific CV reporting templates that include environmental parameters such as temperature, pH, dissolved oxygen content, and contaminant levels. These templates ensure that corrosion data can be properly contextualized and compared across different testing facilities.
The integration of environmental monitoring with CV measurements represents an emerging trend, allowing real-time correlation between environmental fluctuations and corrosion behavior. This approach is particularly valuable in industries where seasonal or operational variations significantly impact corrosion mechanisms.
In marine environments, CV protocols must account for high chloride content, varying oxygen levels, and biological fouling factors. Testing parameters should include extended potential ranges to capture the complex redox reactions occurring in seawater, with scan rates adjusted to account for the slow diffusion processes in high-salinity conditions. Marine industry standards often require specific reference electrodes stable in chloride-rich environments, such as silver/silver chloride electrodes.
Oil and gas industries require CV protocols that address high-temperature, high-pressure conditions and the presence of hydrogen sulfide and carbon dioxide. These environments demand specialized electrochemical cells capable of withstanding extreme conditions while maintaining measurement accuracy. Scan rates must be optimized to detect sulfide-induced corrosion mechanisms, which often manifest at different potential ranges than oxygen-driven corrosion.
For aerospace applications, CV protocols must consider the effects of altitude, temperature cycling, and exposure to de-icing chemicals. The protocols should incorporate multiple scan cycles to simulate the repeated stress conditions experienced by aerospace components, with particular attention to potential ranges where aluminum and titanium alloys exhibit passivation breakdown.
Nuclear industry applications demand CV protocols that account for radiation effects on electrochemical processes and the presence of unique coolant chemistries. Testing must often be conducted remotely, requiring robust instrumentation and data acquisition systems capable of operating in high-radiation environments.
Electronic manufacturing requires micro-scale CV techniques to assess corrosion on circuit boards and components. Protocols must be adapted for low-current measurements and confined spaces, often utilizing microelectrodes and specialized cell designs to evaluate corrosion in the presence of flux residues and under condensing humidity conditions.
Standardization efforts across industries have led to the development of industry-specific CV reporting templates that include environmental parameters such as temperature, pH, dissolved oxygen content, and contaminant levels. These templates ensure that corrosion data can be properly contextualized and compared across different testing facilities.
The integration of environmental monitoring with CV measurements represents an emerging trend, allowing real-time correlation between environmental fluctuations and corrosion behavior. This approach is particularly valuable in industries where seasonal or operational variations significantly impact corrosion mechanisms.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!