Cyclic Voltammetry vs Chronoamperometry for Electrocatalyst Screening — When to Use Which
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Electrochemical Techniques Background and Objectives
Electrochemical techniques have evolved significantly over the past century, transforming from rudimentary experimental setups to sophisticated analytical methods essential for modern materials science and catalysis research. The development trajectory of these techniques has been closely tied to advancements in electronics, computing capabilities, and theoretical understanding of electrode processes. Early electrochemical experiments in the late 19th and early 20th centuries laid the groundwork, while the mid-20th century saw the formalization of many techniques still used today.
Cyclic voltammetry (CV) and chronoamperometry (CA) represent two cornerstone techniques in electrochemical analysis that emerged during this evolution. CV was first described in detail by Randles and Ševčík in the 1940s, while chronoamperometric methods were developed around the same period. Both techniques have undergone continuous refinement in methodology and instrumentation, particularly with the advent of digital potentiostats and data processing capabilities in the 1970s and 1980s.
The current technological landscape shows a growing emphasis on high-throughput screening methods for electrocatalyst discovery and optimization. This trend is driven by the urgent need for efficient catalysts in renewable energy applications, including water splitting, CO2 reduction, and fuel cells. The selection between CV and CA for catalyst screening represents a critical decision point that can significantly impact research outcomes and efficiency.
Recent technological trends indicate a move toward integrated multi-technique approaches, combining electrochemical measurements with spectroscopic methods (such as in-situ Raman or FTIR) and advanced computational modeling. This integration allows for more comprehensive understanding of catalytic mechanisms and performance determinants. Additionally, there is growing interest in miniaturization and automation of electrochemical testing platforms to enable parallel screening of multiple catalyst candidates.
The primary objective of this technical research is to establish clear guidelines for when researchers should employ cyclic voltammetry versus chronoamperometry in electrocatalyst screening applications. This includes identifying the specific advantages, limitations, and optimal use cases for each technique across different catalyst classes and reaction systems. The research aims to develop a decision framework that considers factors such as reaction kinetics, mass transport effects, catalyst stability, and specific information requirements.
Secondary objectives include exploring complementary relationships between these techniques, identifying scenarios where combined approaches yield superior insights, and evaluating emerging methodological innovations that may enhance the utility of both CV and CA for catalyst characterization. The ultimate goal is to optimize experimental design strategies for accelerated discovery and development of next-generation electrocatalysts for sustainable energy applications.
Cyclic voltammetry (CV) and chronoamperometry (CA) represent two cornerstone techniques in electrochemical analysis that emerged during this evolution. CV was first described in detail by Randles and Ševčík in the 1940s, while chronoamperometric methods were developed around the same period. Both techniques have undergone continuous refinement in methodology and instrumentation, particularly with the advent of digital potentiostats and data processing capabilities in the 1970s and 1980s.
The current technological landscape shows a growing emphasis on high-throughput screening methods for electrocatalyst discovery and optimization. This trend is driven by the urgent need for efficient catalysts in renewable energy applications, including water splitting, CO2 reduction, and fuel cells. The selection between CV and CA for catalyst screening represents a critical decision point that can significantly impact research outcomes and efficiency.
Recent technological trends indicate a move toward integrated multi-technique approaches, combining electrochemical measurements with spectroscopic methods (such as in-situ Raman or FTIR) and advanced computational modeling. This integration allows for more comprehensive understanding of catalytic mechanisms and performance determinants. Additionally, there is growing interest in miniaturization and automation of electrochemical testing platforms to enable parallel screening of multiple catalyst candidates.
The primary objective of this technical research is to establish clear guidelines for when researchers should employ cyclic voltammetry versus chronoamperometry in electrocatalyst screening applications. This includes identifying the specific advantages, limitations, and optimal use cases for each technique across different catalyst classes and reaction systems. The research aims to develop a decision framework that considers factors such as reaction kinetics, mass transport effects, catalyst stability, and specific information requirements.
Secondary objectives include exploring complementary relationships between these techniques, identifying scenarios where combined approaches yield superior insights, and evaluating emerging methodological innovations that may enhance the utility of both CV and CA for catalyst characterization. The ultimate goal is to optimize experimental design strategies for accelerated discovery and development of next-generation electrocatalysts for sustainable energy applications.
Market Applications and Demand Analysis
The market for electrochemical analysis techniques, particularly Cyclic Voltammetry (CV) and Chronoamperometry (CA), has experienced significant growth driven by increasing demand for efficient electrocatalyst screening methods across multiple industries. The global electrochemical instruments market was valued at approximately $2.3 billion in 2022 and is projected to grow at a CAGR of 5.8% through 2030, with electrocatalyst research representing a substantial segment.
Energy sector applications constitute the largest market share, with fuel cell development, battery research, and renewable energy storage solutions driving demand for precise electrochemical characterization techniques. The hydrogen economy's expansion has particularly accelerated the need for efficient electrocatalyst screening, as companies seek materials that can reduce the cost and improve the efficiency of hydrogen production through water electrolysis.
Environmental applications represent another significant market segment, where electrocatalysts are being developed for wastewater treatment, pollution control, and CO2 reduction technologies. These applications require precise screening methods to identify catalysts that operate efficiently under specific environmental conditions.
The pharmaceutical and fine chemicals industries are increasingly adopting electrochemical techniques for catalyst development in green chemistry applications. The shift toward sustainable manufacturing processes has created demand for electrocatalysts that can replace traditional chemical processes with electrochemical alternatives, reducing waste and energy consumption.
Academic and research institutions remain major consumers of electrochemical analysis equipment, with government funding for renewable energy research driving significant investment in advanced characterization techniques. The market shows regional variations, with North America and Europe leading in terms of technology adoption, while Asia-Pacific demonstrates the fastest growth rate due to expanding industrial research capabilities in China, Japan, and South Korea.
End-users increasingly demand integrated systems that combine multiple electrochemical techniques with automated data analysis capabilities. This trend has led to the development of comprehensive electrochemical workstations that incorporate both CV and CA functionalities, allowing researchers to seamlessly transition between techniques based on specific screening requirements.
The market also shows growing demand for miniaturized and portable electrochemical systems, particularly for field testing and point-of-use applications. This trend is creating opportunities for equipment manufacturers to develop compact, multi-functional devices that maintain high analytical precision while offering greater flexibility in research environments.
Energy sector applications constitute the largest market share, with fuel cell development, battery research, and renewable energy storage solutions driving demand for precise electrochemical characterization techniques. The hydrogen economy's expansion has particularly accelerated the need for efficient electrocatalyst screening, as companies seek materials that can reduce the cost and improve the efficiency of hydrogen production through water electrolysis.
Environmental applications represent another significant market segment, where electrocatalysts are being developed for wastewater treatment, pollution control, and CO2 reduction technologies. These applications require precise screening methods to identify catalysts that operate efficiently under specific environmental conditions.
The pharmaceutical and fine chemicals industries are increasingly adopting electrochemical techniques for catalyst development in green chemistry applications. The shift toward sustainable manufacturing processes has created demand for electrocatalysts that can replace traditional chemical processes with electrochemical alternatives, reducing waste and energy consumption.
Academic and research institutions remain major consumers of electrochemical analysis equipment, with government funding for renewable energy research driving significant investment in advanced characterization techniques. The market shows regional variations, with North America and Europe leading in terms of technology adoption, while Asia-Pacific demonstrates the fastest growth rate due to expanding industrial research capabilities in China, Japan, and South Korea.
End-users increasingly demand integrated systems that combine multiple electrochemical techniques with automated data analysis capabilities. This trend has led to the development of comprehensive electrochemical workstations that incorporate both CV and CA functionalities, allowing researchers to seamlessly transition between techniques based on specific screening requirements.
The market also shows growing demand for miniaturized and portable electrochemical systems, particularly for field testing and point-of-use applications. This trend is creating opportunities for equipment manufacturers to develop compact, multi-functional devices that maintain high analytical precision while offering greater flexibility in research environments.
Current Challenges in Electrocatalyst Screening Methods
The electrocatalyst screening landscape faces significant methodological challenges that impede efficient catalyst discovery and optimization. Traditional screening approaches often suffer from reproducibility issues due to variations in experimental conditions, electrode preparation, and measurement protocols. This inconsistency makes direct comparison between different research groups problematic and slows down the validation of promising catalysts.
Sensitivity limitations represent another critical challenge, particularly when evaluating catalysts with subtle performance differences. Current methods frequently lack the resolution needed to distinguish small but potentially significant variations in catalytic activity, especially in the early stages of material development where performance improvements may be incremental.
Time efficiency remains a major bottleneck in electrocatalyst screening. Cyclic voltammetry (CV), while information-rich, requires multiple scan rates and cycles for comprehensive analysis, making high-throughput screening challenging. Chronoamperometry (CA) offers faster data acquisition but sacrifices certain mechanistic insights, creating a persistent trade-off between speed and depth of analysis.
Data interpretation complexities further complicate the screening process. Both CV and CA generate substantial datasets that require sophisticated analysis to extract meaningful performance metrics. The field lacks standardized data processing protocols, leading to subjective interpretations and difficulty in establishing universal performance benchmarks for emerging catalysts.
Electrode stability assessment presents unique challenges, as degradation mechanisms vary widely among catalyst types. Current screening methods often fail to capture long-term stability characteristics within practical timeframes, necessitating extrapolation that may not accurately predict real-world performance.
The translation gap between laboratory screening and industrial application remains substantial. Many screening protocols utilize idealized conditions that poorly represent industrial environments, leading to catalysts that perform well in laboratories but disappoint in practical applications. This disconnect undermines the practical value of screening efforts.
Emerging complex reaction systems, particularly those involving multiple electron transfers or competing pathways, pose additional challenges. Traditional screening methods may oversimplify reaction mechanisms, missing critical aspects of catalyst behavior that become apparent only under specific conditions or extended operation.
Instrumentation limitations also constrain screening capabilities, with many laboratories lacking advanced equipment for simultaneous multi-parameter analysis. This forces researchers to make methodological compromises that may not capture the full performance profile of novel catalysts.
Sensitivity limitations represent another critical challenge, particularly when evaluating catalysts with subtle performance differences. Current methods frequently lack the resolution needed to distinguish small but potentially significant variations in catalytic activity, especially in the early stages of material development where performance improvements may be incremental.
Time efficiency remains a major bottleneck in electrocatalyst screening. Cyclic voltammetry (CV), while information-rich, requires multiple scan rates and cycles for comprehensive analysis, making high-throughput screening challenging. Chronoamperometry (CA) offers faster data acquisition but sacrifices certain mechanistic insights, creating a persistent trade-off between speed and depth of analysis.
Data interpretation complexities further complicate the screening process. Both CV and CA generate substantial datasets that require sophisticated analysis to extract meaningful performance metrics. The field lacks standardized data processing protocols, leading to subjective interpretations and difficulty in establishing universal performance benchmarks for emerging catalysts.
Electrode stability assessment presents unique challenges, as degradation mechanisms vary widely among catalyst types. Current screening methods often fail to capture long-term stability characteristics within practical timeframes, necessitating extrapolation that may not accurately predict real-world performance.
The translation gap between laboratory screening and industrial application remains substantial. Many screening protocols utilize idealized conditions that poorly represent industrial environments, leading to catalysts that perform well in laboratories but disappoint in practical applications. This disconnect undermines the practical value of screening efforts.
Emerging complex reaction systems, particularly those involving multiple electron transfers or competing pathways, pose additional challenges. Traditional screening methods may oversimplify reaction mechanisms, missing critical aspects of catalyst behavior that become apparent only under specific conditions or extended operation.
Instrumentation limitations also constrain screening capabilities, with many laboratories lacking advanced equipment for simultaneous multi-parameter analysis. This forces researchers to make methodological compromises that may not capture the full performance profile of novel catalysts.
Comparative Analysis of CV and CA Methodologies
01 Electrochemical methods for catalyst screening
Cyclic voltammetry and chronoamperometry are powerful electrochemical techniques used for screening electrocatalysts. These methods allow for rapid evaluation of catalytic activity, stability, and selectivity by measuring current responses under controlled potential conditions. The techniques provide valuable insights into reaction kinetics, electron transfer processes, and catalytic mechanisms, enabling researchers to efficiently identify promising catalyst candidates for various electrochemical applications.- Electrochemical methods for catalyst screening: Cyclic voltammetry and chronoamperometry are powerful electrochemical techniques used for screening electrocatalysts. These methods allow for rapid evaluation of catalytic activity, stability, and selectivity by measuring current responses under controlled potential conditions. The techniques can be applied to various catalyst materials and provide insights into reaction mechanisms, kinetics, and efficiency of electrocatalytic processes.
- Novel electrocatalyst materials characterization: Various novel materials are being developed and characterized as electrocatalysts using cyclic voltammetry and chronoamperometry. These include metal-organic frameworks, nanostructured metals, carbon-based materials, and composite structures. The electrochemical screening methods help identify promising candidates with enhanced catalytic activity, selectivity, and durability for applications in energy conversion, storage, and environmental remediation.
- High-throughput screening systems: Advanced high-throughput screening systems have been developed to accelerate electrocatalyst discovery and optimization. These systems integrate cyclic voltammetry and chronoamperometry with automated sample handling, parallel testing capabilities, and data analysis algorithms. Such approaches enable rapid screening of large catalyst libraries, composition gradients, and processing conditions to identify optimal electrocatalyst formulations with minimal time and resource investment.
- Computational methods integrated with electrochemical screening: Integration of computational methods with experimental electrochemical screening techniques enhances the efficiency of electrocatalyst development. Machine learning algorithms, density functional theory calculations, and molecular modeling are used to predict catalyst performance, guide experimental design, and interpret cyclic voltammetry and chronoamperometry data. This combined approach accelerates the discovery of high-performance electrocatalysts by reducing the experimental search space.
- Application-specific electrocatalyst screening protocols: Specialized screening protocols using cyclic voltammetry and chronoamperometry have been developed for specific applications such as fuel cells, water splitting, CO2 reduction, and nitrogen fixation. These protocols involve tailored electrode configurations, electrolyte compositions, and measurement parameters optimized for the target reaction. Application-specific screening enables more accurate prediction of real-world catalyst performance and facilitates the development of electrocatalysts for practical energy and environmental technologies.
02 Novel electrocatalyst materials characterization
Various novel electrocatalyst materials can be characterized using cyclic voltammetry and chronoamperometry techniques. These include metal-based catalysts, carbon-supported materials, metal oxides, and composite structures. The electrochemical screening methods help determine key performance parameters such as onset potential, current density, and Tafel slope, which are crucial for evaluating catalytic efficiency and comparing different catalyst formulations for specific electrochemical reactions.Expand Specific Solutions03 High-throughput screening systems
Advanced high-throughput screening systems combine cyclic voltammetry and chronoamperometry with automated testing platforms to rapidly evaluate multiple electrocatalyst candidates. These systems often incorporate parallel electrochemical cells, robotic sample handling, and data analysis software to accelerate the discovery and optimization of electrocatalysts. The high-throughput approach significantly reduces the time and resources required for catalyst development while generating comprehensive electrochemical performance data.Expand Specific Solutions04 Electrocatalyst screening for specific reactions
Cyclic voltammetry and chronoamperometry techniques are tailored for screening electrocatalysts for specific electrochemical reactions such as oxygen reduction reaction (ORR), hydrogen evolution reaction (HER), oxygen evolution reaction (OER), and CO2 reduction. The screening protocols involve specific potential ranges, scan rates, and electrolyte compositions optimized for each reaction type. These targeted screening approaches help identify the most effective catalysts for particular applications like fuel cells, water electrolyzers, and CO2 conversion systems.Expand Specific Solutions05 Data analysis and interpretation methods
Advanced data analysis and interpretation methods are essential for extracting meaningful insights from cyclic voltammetry and chronoamperometry measurements in electrocatalyst screening. These include mathematical modeling, statistical analysis, machine learning algorithms, and computational approaches that help correlate electrochemical responses with catalyst structure and composition. The analysis methods enable researchers to identify structure-activity relationships, predict catalyst performance, and guide the rational design of improved electrocatalyst materials.Expand Specific Solutions
Leading Research Groups and Industrial Players
The electrochemical analysis techniques of Cyclic Voltammetry (CV) and Chronoamperometry (CA) for electrocatalyst screening are in a mature development stage, with growing market applications across energy, materials science, and biomedical sectors. The global electrochemical instrumentation market is expanding rapidly, driven by renewable energy research and industrial applications. Leading players include established analytical instrument manufacturers like Dionex Corp. and specialized research institutions such as Johns Hopkins University and Caltech. Industrial giants Toyota Motor Corp., Siemens AG, and Infineon Technologies AG are investing heavily in electrocatalyst development for energy applications, while research organizations like CNRS and RISE are advancing fundamental understanding. The technology ecosystem shows a balanced distribution between academic research centers, specialized instrumentation companies, and industrial end-users implementing these techniques for product development.
The Johns Hopkins University
Technical Solution: The Johns Hopkins University has developed a comprehensive electrocatalyst evaluation framework that strategically employs both cyclic voltammetry (CV) and chronoamperometry (CA) techniques. Their approach begins with rapid CV screening to identify promising catalysts based on peak positions, current densities, and reversibility characteristics. For materials showing favorable initial performance, they employ a multi-step CA protocol at various potentials to evaluate stability, activity, and selectivity under operating conditions. Johns Hopkins researchers have pioneered the integration of these electrochemical techniques with in-situ spectroscopic methods (including surface-enhanced Raman spectroscopy and ATR-FTIR) to correlate electrochemical behavior with molecular-level surface processes. Their methodology includes specialized protocols for distinguishing faradaic processes from capacitive currents, enabling more accurate determination of catalytic activity. Additionally, they've developed computational models that correlate CV features with long-term stability metrics from CA, allowing for predictive screening that reduces experimental time requirements by approximately 60%. This integrated approach has been particularly successful in developing catalysts for energy conversion applications, including water splitting and CO2 reduction.
Strengths: Their combined spectroelectrochemical approach provides molecular-level insights into catalyst mechanisms that neither technique alone could achieve. The predictive modeling capabilities significantly reduce experimental time requirements. Weaknesses: The sophisticated instrumentation required for their integrated approach limits accessibility. The computational models require extensive validation across different catalyst classes to ensure broad applicability.
California Institute of Technology
Technical Solution: California Institute of Technology (Caltech) has pioneered a systematic approach to electrocatalyst evaluation that strategically combines cyclic voltammetry (CV) and chronoamperometry (CA). Their methodology begins with rapid CV screening to identify promising catalyst candidates based on redox behavior, followed by targeted CA studies for performance validation. Caltech researchers have developed specialized microelectrode arrays that enable high-throughput parallel testing of multiple catalysts under identical conditions. Their proprietary data analysis framework incorporates machine learning algorithms to correlate CV features with long-term CA performance metrics, enabling predictive modeling of catalyst durability from initial CV signatures. This approach has been particularly successful in identifying optimal catalysts for CO2 reduction and water splitting applications. Caltech has also developed protocols for distinguishing between mass transport limitations and intrinsic catalytic activity using a combination of rotating disk electrode measurements with CV and CA techniques, allowing for more accurate comparison between different catalyst materials.
Strengths: Their integrated machine learning approach enables predictive modeling of long-term performance from initial screening data, significantly accelerating discovery. The microelectrode array technology allows for highly efficient parallel testing. Weaknesses: The sophisticated computational models require extensive training datasets to be effective, limiting application to well-studied reaction systems. The approach is more resource-intensive than conventional sequential testing methods.
Technical Principles and Theoretical Foundations
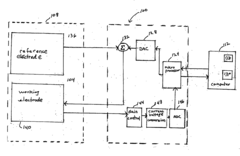
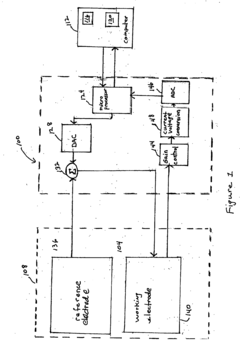
Method, system and software application for electrochemical analysis
PatentActiveUS20060180478A1
Innovation
- A method and system that applies one or more time-sequenced voltage waveforms to a solution, measures the resulting current, and stores raw data for display in three-dimensional and two-dimensional plots, allowing for baseline correction and arithmetic operations, with data transmission and storage rates between 100 Hz and 10 KHz, enabling analysis of integrated and raw data post-run.
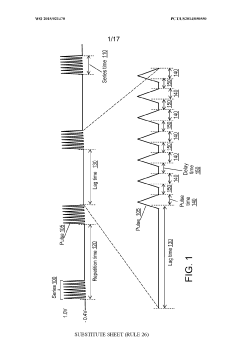
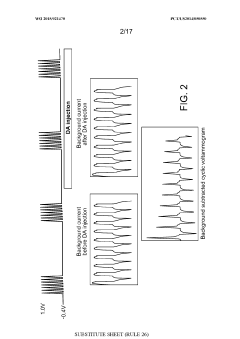
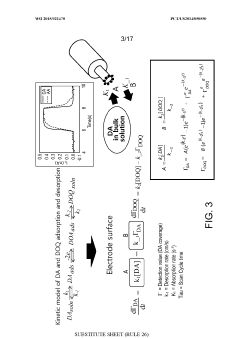
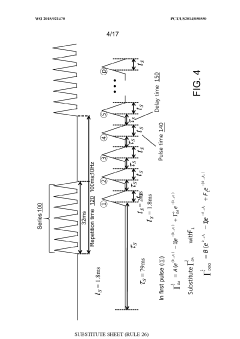
Using kinetic cyclic voltammetry to evaluate analyte kinetics and concentrations
PatentWO2015021470A1
Innovation
- Kinetic cyclic voltammetry (KCV) is developed, which involves multi-pulse cyclic voltammetry at various voltages to generate kinetic and concentration maps (K-maps and A-maps) without background subtraction, enabling absolute quantification of analytes and real-time signal analysis, suitable for use in smart neuromodulation systems.
Data Processing and Interpretation Strategies
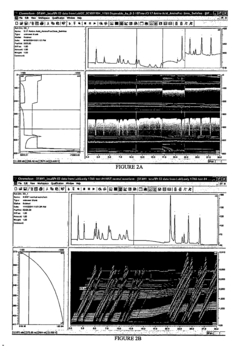
Effective data processing and interpretation are critical components in electrochemical analysis, particularly when comparing cyclic voltammetry (CV) and chronoamperometry (CA) techniques for electrocatalyst screening. The raw data obtained from these methods requires systematic processing to extract meaningful insights about catalyst performance.
For cyclic voltammetry data, baseline correction represents the initial crucial step to eliminate background currents that may obscure catalytic signals. This typically involves subtracting capacitive currents or applying mathematical algorithms to isolate faradaic processes. Normalization by electrode surface area follows, enabling fair comparison between different catalyst samples regardless of loading variations.
Chronoamperometry data processing begins with transient response analysis, focusing on current stabilization periods that indicate when steady-state catalytic behavior is achieved. Time-dependent decay patterns provide valuable information about reaction kinetics and mass transport limitations that may not be evident in CV measurements.
Both techniques benefit from advanced statistical methods to enhance signal-to-noise ratios and ensure reproducibility. Principal component analysis (PCA) and other multivariate approaches have emerged as powerful tools for identifying subtle patterns in electrochemical responses that might indicate superior catalytic activity.
Interpretation strategies differ significantly between the techniques. CV data interpretation centers on peak position analysis, with potential differences between anodic and cathodic peaks (ΔEp) offering insights into electron transfer kinetics. The shape and symmetry of voltammograms provide qualitative information about reaction mechanisms, while peak current ratios help determine reversibility characteristics.
For chronoamperometry, steady-state current values serve as direct indicators of catalytic activity, with higher currents generally suggesting better performance. The Cottrell equation application enables diffusion coefficient determination, while chronoamperometric response to potential steps reveals information about activation barriers and reaction pathways.
Integration of computational modeling with experimental data has revolutionized interpretation approaches. Digital simulations of electrochemical processes allow researchers to fit experimental data to theoretical models, extracting kinetic parameters that would be difficult to determine through direct measurement alone. Machine learning algorithms increasingly assist in identifying complex relationships between electrochemical signatures and catalyst performance metrics.
Cross-validation between CV and CA results strengthens data reliability, with complementary information from both techniques providing a more comprehensive understanding of electrocatalyst behavior than either method alone could achieve.
For cyclic voltammetry data, baseline correction represents the initial crucial step to eliminate background currents that may obscure catalytic signals. This typically involves subtracting capacitive currents or applying mathematical algorithms to isolate faradaic processes. Normalization by electrode surface area follows, enabling fair comparison between different catalyst samples regardless of loading variations.
Chronoamperometry data processing begins with transient response analysis, focusing on current stabilization periods that indicate when steady-state catalytic behavior is achieved. Time-dependent decay patterns provide valuable information about reaction kinetics and mass transport limitations that may not be evident in CV measurements.
Both techniques benefit from advanced statistical methods to enhance signal-to-noise ratios and ensure reproducibility. Principal component analysis (PCA) and other multivariate approaches have emerged as powerful tools for identifying subtle patterns in electrochemical responses that might indicate superior catalytic activity.
Interpretation strategies differ significantly between the techniques. CV data interpretation centers on peak position analysis, with potential differences between anodic and cathodic peaks (ΔEp) offering insights into electron transfer kinetics. The shape and symmetry of voltammograms provide qualitative information about reaction mechanisms, while peak current ratios help determine reversibility characteristics.
For chronoamperometry, steady-state current values serve as direct indicators of catalytic activity, with higher currents generally suggesting better performance. The Cottrell equation application enables diffusion coefficient determination, while chronoamperometric response to potential steps reveals information about activation barriers and reaction pathways.
Integration of computational modeling with experimental data has revolutionized interpretation approaches. Digital simulations of electrochemical processes allow researchers to fit experimental data to theoretical models, extracting kinetic parameters that would be difficult to determine through direct measurement alone. Machine learning algorithms increasingly assist in identifying complex relationships between electrochemical signatures and catalyst performance metrics.
Cross-validation between CV and CA results strengthens data reliability, with complementary information from both techniques providing a more comprehensive understanding of electrocatalyst behavior than either method alone could achieve.
Standardization and Reproducibility Considerations
Standardization and reproducibility represent critical challenges in electrocatalyst screening methodologies, particularly when comparing cyclic voltammetry (CV) and chronoamperometry (CA) techniques. The scientific community has increasingly recognized the need for robust protocols to ensure reliable and comparable results across different research groups and experimental setups.
Electrode preparation procedures significantly impact measurement outcomes in both CV and CA. Variations in catalyst loading, distribution, and adhesion to electrode surfaces can lead to substantial differences in observed catalytic activity. Research has shown that even minor inconsistencies in ink formulation or deposition techniques can result in performance variations exceeding 20% for identical catalyst materials.
Reference electrode calibration emerges as another crucial factor affecting reproducibility. The potential drift in reference electrodes over time can introduce systematic errors in both CV and CA measurements. Standard protocols now recommend regular calibration against known redox couples and reporting potentials versus reversible hydrogen electrode (RHE) to facilitate cross-laboratory comparisons.
Electrolyte purity and preparation standardization significantly influence background currents and overall measurement stability. Trace impurities can act as promoters or poisons for electrocatalytic reactions, particularly affecting long-duration CA measurements where accumulation effects become pronounced. Established guidelines now specify electrolyte grade, purification methods, and pre-electrolysis procedures to minimize these variables.
Temperature control represents a frequently overlooked parameter that affects reaction kinetics in both techniques. Studies demonstrate that even 5°C variations can alter apparent catalytic activity by 15-30%, with CA measurements being particularly sensitive due to their longer duration. Standardized testing now increasingly specifies temperature control within ±1°C and reporting of actual cell temperatures.
Data processing and analysis protocols show considerable variation across the literature, complicating direct comparison of results. For CV, inconsistencies in background correction, iR compensation, and scan rate normalization create significant barriers to reproducibility. Similarly, for CA measurements, variations in current stabilization criteria and time-point selection for activity comparison introduce additional complexity.
International initiatives like the Benchmarking Protocol for Electrocatalysts (BPE) and the Electrocatalysis Network (ElecNet) are working to establish consensus standards for both CV and CA measurements. These efforts aim to develop reference materials, standardized cell designs, and unified reporting formats to enhance reproducibility across the field and accelerate reliable electrocatalyst development.
Electrode preparation procedures significantly impact measurement outcomes in both CV and CA. Variations in catalyst loading, distribution, and adhesion to electrode surfaces can lead to substantial differences in observed catalytic activity. Research has shown that even minor inconsistencies in ink formulation or deposition techniques can result in performance variations exceeding 20% for identical catalyst materials.
Reference electrode calibration emerges as another crucial factor affecting reproducibility. The potential drift in reference electrodes over time can introduce systematic errors in both CV and CA measurements. Standard protocols now recommend regular calibration against known redox couples and reporting potentials versus reversible hydrogen electrode (RHE) to facilitate cross-laboratory comparisons.
Electrolyte purity and preparation standardization significantly influence background currents and overall measurement stability. Trace impurities can act as promoters or poisons for electrocatalytic reactions, particularly affecting long-duration CA measurements where accumulation effects become pronounced. Established guidelines now specify electrolyte grade, purification methods, and pre-electrolysis procedures to minimize these variables.
Temperature control represents a frequently overlooked parameter that affects reaction kinetics in both techniques. Studies demonstrate that even 5°C variations can alter apparent catalytic activity by 15-30%, with CA measurements being particularly sensitive due to their longer duration. Standardized testing now increasingly specifies temperature control within ±1°C and reporting of actual cell temperatures.
Data processing and analysis protocols show considerable variation across the literature, complicating direct comparison of results. For CV, inconsistencies in background correction, iR compensation, and scan rate normalization create significant barriers to reproducibility. Similarly, for CA measurements, variations in current stabilization criteria and time-point selection for activity comparison introduce additional complexity.
International initiatives like the Benchmarking Protocol for Electrocatalysts (BPE) and the Electrocatalysis Network (ElecNet) are working to establish consensus standards for both CV and CA measurements. These efforts aim to develop reference materials, standardized cell designs, and unified reporting formats to enhance reproducibility across the field and accelerate reliable electrocatalyst development.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!