Dynamic Light Scattering in Vaccine Formulation Optimization
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
DLS Technology Background and Objectives
Dynamic Light Scattering (DLS) has emerged as a pivotal analytical technique in the biopharmaceutical industry, particularly in vaccine development and formulation optimization. The technology's evolution traces back to the 1960s when it was primarily used for polymer characterization, but has since undergone significant refinement to become an essential tool in vaccine research and development.
DLS operates on the principle of Rayleigh scattering, measuring the Brownian motion of particles in suspension to determine their size distribution. This capability is crucial for vaccine formulation, as particle size directly impacts stability, immunogenicity, and safety profiles of vaccine candidates. The technology has evolved from basic particle sizing to sophisticated characterization of complex biological systems, including protein aggregates, liposomes, virus-like particles, and adjuvant components.
The current technological landscape of DLS encompasses both traditional cuvette-based systems and more advanced microfluidic platforms, with increasing integration of artificial intelligence for data interpretation. Recent advancements have significantly improved resolution, allowing for more precise differentiation between monomeric proteins and small aggregates—a critical factor in vaccine quality control.
In the context of vaccine formulation optimization, DLS addresses several key challenges: detection of subvisible particles that may trigger immunogenicity concerns, monitoring of stability during storage, characterization of adjuvant particle size for optimal immune response, and quality control during manufacturing scale-up. The COVID-19 pandemic has further accelerated DLS technology development, particularly for mRNA vaccine formulations where lipid nanoparticle characterization is essential.
The primary objectives of DLS technology in vaccine development include: enhancing formulation stability across various environmental conditions, optimizing particle size distribution for targeted immune responses, reducing aggregation during manufacturing and storage, streamlining analytical workflows for faster development cycles, and establishing robust quality control parameters for regulatory compliance.
Future technological goals focus on improving sensitivity for detection of low concentration aggregates, developing high-throughput capabilities for parallel formulation screening, integrating complementary techniques for comprehensive characterization, and establishing standardized protocols for consistent data interpretation across the industry. Additionally, miniaturization of DLS systems aims to enable real-time monitoring during manufacturing processes, potentially revolutionizing quality assurance in vaccine production.
DLS operates on the principle of Rayleigh scattering, measuring the Brownian motion of particles in suspension to determine their size distribution. This capability is crucial for vaccine formulation, as particle size directly impacts stability, immunogenicity, and safety profiles of vaccine candidates. The technology has evolved from basic particle sizing to sophisticated characterization of complex biological systems, including protein aggregates, liposomes, virus-like particles, and adjuvant components.
The current technological landscape of DLS encompasses both traditional cuvette-based systems and more advanced microfluidic platforms, with increasing integration of artificial intelligence for data interpretation. Recent advancements have significantly improved resolution, allowing for more precise differentiation between monomeric proteins and small aggregates—a critical factor in vaccine quality control.
In the context of vaccine formulation optimization, DLS addresses several key challenges: detection of subvisible particles that may trigger immunogenicity concerns, monitoring of stability during storage, characterization of adjuvant particle size for optimal immune response, and quality control during manufacturing scale-up. The COVID-19 pandemic has further accelerated DLS technology development, particularly for mRNA vaccine formulations where lipid nanoparticle characterization is essential.
The primary objectives of DLS technology in vaccine development include: enhancing formulation stability across various environmental conditions, optimizing particle size distribution for targeted immune responses, reducing aggregation during manufacturing and storage, streamlining analytical workflows for faster development cycles, and establishing robust quality control parameters for regulatory compliance.
Future technological goals focus on improving sensitivity for detection of low concentration aggregates, developing high-throughput capabilities for parallel formulation screening, integrating complementary techniques for comprehensive characterization, and establishing standardized protocols for consistent data interpretation across the industry. Additionally, miniaturization of DLS systems aims to enable real-time monitoring during manufacturing processes, potentially revolutionizing quality assurance in vaccine production.
Vaccine Formulation Market Analysis
The global vaccine formulation market has witnessed substantial growth in recent years, reaching approximately $94.6 billion in 2022, with projections indicating a compound annual growth rate (CAGR) of 10.5% through 2030. This growth is primarily driven by increasing immunization programs worldwide, rising prevalence of infectious diseases, and technological advancements in vaccine development methodologies.
Dynamic Light Scattering (DLS) technology has emerged as a critical analytical tool in the vaccine formulation sector, particularly for characterizing nanoparticle-based vaccines and adjuvants. The market segment specifically for analytical technologies in vaccine formulation, including DLS, was valued at $2.8 billion in 2022 and is expected to grow at a CAGR of 12.3% through 2028.
The COVID-19 pandemic has significantly accelerated market demand for advanced vaccine formulation technologies. The unprecedented speed of mRNA vaccine development highlighted the importance of rapid analytical methods like DLS in formulation optimization. Post-pandemic, this market segment continues to expand as manufacturers invest in technologies that can reduce time-to-market while ensuring product quality and stability.
Geographically, North America dominates the vaccine formulation market with approximately 42% market share, followed by Europe (28%) and Asia-Pacific (21%). However, the Asia-Pacific region is experiencing the fastest growth rate due to increasing healthcare expenditure, government initiatives for immunization programs, and expanding contract manufacturing capabilities in countries like India and China.
By vaccine type, the market segmentation reveals that recombinant/conjugate vaccines hold the largest market share (38%), followed by inactivated vaccines (27%), live attenuated vaccines (21%), and toxoid vaccines (14%). The newest category, mRNA vaccines, though currently representing only about 8% of the market, is projected to be the fastest-growing segment with a CAGR exceeding 18% through 2030.
Key demand drivers for DLS technology in vaccine formulation include the growing focus on particle size characterization, increasing regulatory requirements for vaccine stability studies, and the rise of complex delivery systems such as lipid nanoparticles. Additionally, the trend toward personalized medicine and the development of combination vaccines are creating new market opportunities for advanced analytical technologies.
The market also shows increasing demand for integrated analytical platforms that combine DLS with other characterization techniques, offering comprehensive formulation optimization solutions. This integration trend is expected to continue as manufacturers seek more efficient and cost-effective development processes.
Dynamic Light Scattering (DLS) technology has emerged as a critical analytical tool in the vaccine formulation sector, particularly for characterizing nanoparticle-based vaccines and adjuvants. The market segment specifically for analytical technologies in vaccine formulation, including DLS, was valued at $2.8 billion in 2022 and is expected to grow at a CAGR of 12.3% through 2028.
The COVID-19 pandemic has significantly accelerated market demand for advanced vaccine formulation technologies. The unprecedented speed of mRNA vaccine development highlighted the importance of rapid analytical methods like DLS in formulation optimization. Post-pandemic, this market segment continues to expand as manufacturers invest in technologies that can reduce time-to-market while ensuring product quality and stability.
Geographically, North America dominates the vaccine formulation market with approximately 42% market share, followed by Europe (28%) and Asia-Pacific (21%). However, the Asia-Pacific region is experiencing the fastest growth rate due to increasing healthcare expenditure, government initiatives for immunization programs, and expanding contract manufacturing capabilities in countries like India and China.
By vaccine type, the market segmentation reveals that recombinant/conjugate vaccines hold the largest market share (38%), followed by inactivated vaccines (27%), live attenuated vaccines (21%), and toxoid vaccines (14%). The newest category, mRNA vaccines, though currently representing only about 8% of the market, is projected to be the fastest-growing segment with a CAGR exceeding 18% through 2030.
Key demand drivers for DLS technology in vaccine formulation include the growing focus on particle size characterization, increasing regulatory requirements for vaccine stability studies, and the rise of complex delivery systems such as lipid nanoparticles. Additionally, the trend toward personalized medicine and the development of combination vaccines are creating new market opportunities for advanced analytical technologies.
The market also shows increasing demand for integrated analytical platforms that combine DLS with other characterization techniques, offering comprehensive formulation optimization solutions. This integration trend is expected to continue as manufacturers seek more efficient and cost-effective development processes.
Current DLS Applications and Challenges
Dynamic Light Scattering (DLS) has emerged as a critical analytical technique in vaccine formulation optimization, offering unique capabilities for characterizing nanoparticle-based vaccine components. Currently, DLS is widely employed across pharmaceutical research and development to assess particle size distribution, stability, and aggregation behavior of vaccine formulations under various conditions.
In vaccine development, DLS serves multiple essential functions. It enables real-time monitoring of antigen stability during formulation development, allowing researchers to detect early signs of protein aggregation that could compromise vaccine efficacy or safety. The technique provides valuable insights into the colloidal stability of adjuvants and carrier systems, which directly impacts immunogenicity and shelf-life of final products.
The non-destructive nature of DLS makes it particularly valuable for analyzing precious vaccine samples during early development stages when material quantities are limited. Furthermore, its ability to perform measurements across a wide temperature range helps evaluate formulation behavior under various storage conditions, supporting stability studies essential for vaccine deployment in diverse environments.
Despite these advantages, DLS faces several significant technical challenges in vaccine applications. The technique shows limitations when analyzing highly polydisperse samples, which are common in complex vaccine formulations containing multiple components. The presence of large aggregates can disproportionately influence results, potentially masking smaller particles that may be critical for vaccine efficacy.
Sensitivity issues arise when measuring low concentration samples, particularly problematic during early-stage development when antigen availability is restricted. Additionally, DLS struggles to differentiate between particles of similar size but different composition, limiting its ability to characterize heterogeneous vaccine formulations without complementary techniques.
Data interpretation presents another challenge, as converting intensity-weighted distributions to volume or number distributions involves mathematical assumptions that may introduce errors. This becomes particularly problematic when analyzing multimodal distributions typical in vaccine formulations containing adjuvants, antigens, and stabilizers.
Regulatory considerations further complicate DLS implementation, as standardization across different instruments and laboratories remains challenging. Method validation for DLS in vaccine quality control requires robust protocols that can reliably detect subtle changes in formulation characteristics, a requirement not always easily met with current technology.
Recent technological advances are addressing these limitations through multi-angle DLS systems, improved algorithms for polydisperse sample analysis, and integration with complementary techniques like nanoparticle tracking analysis. These developments are gradually expanding DLS capabilities for more comprehensive characterization of increasingly complex vaccine formulations.
In vaccine development, DLS serves multiple essential functions. It enables real-time monitoring of antigen stability during formulation development, allowing researchers to detect early signs of protein aggregation that could compromise vaccine efficacy or safety. The technique provides valuable insights into the colloidal stability of adjuvants and carrier systems, which directly impacts immunogenicity and shelf-life of final products.
The non-destructive nature of DLS makes it particularly valuable for analyzing precious vaccine samples during early development stages when material quantities are limited. Furthermore, its ability to perform measurements across a wide temperature range helps evaluate formulation behavior under various storage conditions, supporting stability studies essential for vaccine deployment in diverse environments.
Despite these advantages, DLS faces several significant technical challenges in vaccine applications. The technique shows limitations when analyzing highly polydisperse samples, which are common in complex vaccine formulations containing multiple components. The presence of large aggregates can disproportionately influence results, potentially masking smaller particles that may be critical for vaccine efficacy.
Sensitivity issues arise when measuring low concentration samples, particularly problematic during early-stage development when antigen availability is restricted. Additionally, DLS struggles to differentiate between particles of similar size but different composition, limiting its ability to characterize heterogeneous vaccine formulations without complementary techniques.
Data interpretation presents another challenge, as converting intensity-weighted distributions to volume or number distributions involves mathematical assumptions that may introduce errors. This becomes particularly problematic when analyzing multimodal distributions typical in vaccine formulations containing adjuvants, antigens, and stabilizers.
Regulatory considerations further complicate DLS implementation, as standardization across different instruments and laboratories remains challenging. Method validation for DLS in vaccine quality control requires robust protocols that can reliably detect subtle changes in formulation characteristics, a requirement not always easily met with current technology.
Recent technological advances are addressing these limitations through multi-angle DLS systems, improved algorithms for polydisperse sample analysis, and integration with complementary techniques like nanoparticle tracking analysis. These developments are gradually expanding DLS capabilities for more comprehensive characterization of increasingly complex vaccine formulations.
Current DLS Methods for Vaccine Development
01 DLS for particle size analysis in formulation optimization
Dynamic Light Scattering (DLS) is utilized for precise particle size analysis in various formulations. This technique enables researchers to measure particle dimensions in nanometer to micrometer ranges, which is crucial for optimizing formulation stability and performance. By monitoring particle size distribution, formulators can adjust composition parameters to achieve desired physical properties and prevent aggregation or sedimentation issues in the final product.- DLS for particle size characterization in formulations: Dynamic Light Scattering (DLS) is utilized for precise characterization of particle size distribution in various formulations. This technique enables researchers to measure particles in the nanometer to micrometer range, providing critical data for formulation stability and performance. By monitoring particle size, formulators can optimize compositions to prevent aggregation and ensure consistent product quality. The technique is particularly valuable for pharmaceutical, cosmetic, and colloidal formulations where particle size directly impacts efficacy and shelf-life.
- Real-time formulation monitoring and process control: Dynamic Light Scattering enables real-time monitoring of formulation processes, allowing for immediate adjustments to optimize product quality. This application involves continuous measurement of particle characteristics during manufacturing, providing feedback for process control systems. By implementing DLS in production lines, manufacturers can detect deviations from target specifications early, reducing batch failures and improving consistency. The technology supports quality-by-design approaches by establishing correlations between process parameters and formulation attributes.
- Advanced algorithms for DLS data analysis and interpretation: Sophisticated algorithms have been developed to enhance the analysis and interpretation of Dynamic Light Scattering data for formulation optimization. These computational methods improve the resolution of multimodal particle distributions and extract meaningful information from complex scattering patterns. Machine learning approaches can identify patterns in DLS data that correlate with formulation performance, enabling predictive modeling. Advanced signal processing techniques reduce noise and increase sensitivity, allowing for detection of subtle changes in formulation characteristics that might impact stability or efficacy.
- Integration of DLS with other analytical techniques: Combining Dynamic Light Scattering with complementary analytical methods creates powerful integrated approaches for comprehensive formulation characterization. These hybrid systems may incorporate spectroscopic techniques, rheological measurements, or microscopy to provide multidimensional data on formulation properties. The synergistic use of multiple techniques overcomes the limitations of individual methods and provides more complete understanding of complex formulations. This integrated approach is particularly valuable for challenging systems such as biologics, nanoparticle formulations, and complex suspensions where multiple attributes must be optimized simultaneously.
- High-throughput DLS for formulation screening: High-throughput Dynamic Light Scattering platforms enable rapid screening of multiple formulation variables to identify optimal compositions. These systems typically feature automated sample handling, parallel measurement capabilities, and integrated data analysis software. By testing numerous formulation parameters simultaneously, developers can efficiently explore design spaces and identify robust formulation regions. This approach accelerates development timelines and reduces material requirements during formulation optimization. The high-throughput methodology is particularly valuable in early-stage development when numerous excipients and concentrations must be evaluated.
02 Real-time monitoring of formulation stability using DLS
Dynamic Light Scattering enables real-time monitoring of formulation stability by tracking changes in particle size distribution over time. This application is particularly valuable during formulation development as it allows scientists to observe how various environmental conditions (temperature, pH, ionic strength) affect the stability of dispersed systems. The technique provides immediate feedback on formulation modifications, accelerating the optimization process and reducing development time.Expand Specific Solutions03 Advanced DLS algorithms for complex formulation analysis
Advanced algorithms have been developed to enhance DLS capabilities for analyzing complex formulations containing multiple particle populations. These computational approaches improve data interpretation by deconvoluting overlapping signals, enabling more accurate characterization of polydisperse systems. Machine learning and artificial intelligence integration with DLS technology allows for automated optimization of formulation parameters based on particle behavior patterns observed during analysis.Expand Specific Solutions04 DLS-based high-throughput screening for formulation development
High-throughput DLS screening platforms enable rapid evaluation of multiple formulation candidates simultaneously. This approach significantly accelerates the formulation optimization process by allowing parallel testing of various excipient combinations and processing conditions. Automated DLS systems can analyze hundreds of samples per day, generating comprehensive datasets that help identify optimal formulation parameters while minimizing material consumption and development time.Expand Specific Solutions05 Integration of DLS with other analytical techniques for comprehensive formulation characterization
Combining DLS with complementary analytical techniques provides comprehensive characterization of formulations beyond particle size alone. Integration with techniques such as zeta potential measurement, rheology, spectroscopy, or microscopy creates a more complete understanding of formulation properties. This multi-analytical approach enables formulators to correlate particle characteristics with other physical properties, leading to more robust optimization strategies and better prediction of formulation performance.Expand Specific Solutions
Key Players in DLS Instrumentation
Dynamic Light Scattering (DLS) technology in vaccine formulation optimization is currently in a growth phase, with increasing market adoption driven by the need for more efficient vaccine development processes. The global market for DLS in biopharmaceutical applications is expanding rapidly, estimated to reach significant value as vaccine development accelerates post-pandemic. Technologically, DLS has reached moderate maturity with established applications in particle size analysis and stability assessment. Leading players include Sanofi Pasteur, which has integrated DLS into its vaccine development pipeline, alongside research contributions from academic institutions like the University of Oslo and Sun Yat-Sen University. Pharmaceutical companies such as Kowa Co. and medical technology firms like Honeywell International Technologies are advancing practical applications, while specialized entities like NanoClear Technologies and Sonata Scientific are developing complementary technologies enhancing DLS capabilities in formulation analysis.
Ares Trading SA
Technical Solution: Ares Trading SA has developed an advanced Dynamic Light Scattering (DLS) platform specifically for vaccine formulation optimization. Their technology utilizes multi-angle DLS measurements to characterize particle size distributions with enhanced resolution, particularly valuable for detecting aggregation in protein-based vaccines. The system incorporates temperature-controlled sample chambers (4-60°C) that allow real-time monitoring of formulation stability under various thermal conditions, simulating storage and transport scenarios. Their proprietary algorithms can distinguish between monomeric proteins, oligomers, and larger aggregates in complex vaccine formulations, providing critical data for stability assessment. The platform also features automated high-throughput screening capabilities, enabling simultaneous evaluation of multiple formulation candidates with different adjuvants, preservatives, and buffer systems to rapidly identify optimal compositions that maintain antigen integrity while maximizing immunogenicity.
Strengths: Superior resolution for detecting subtle changes in particle size distribution; integrated temperature control for real-world stability testing; high-throughput capabilities for efficient formulation screening. Weaknesses: Requires significant expertise for data interpretation; higher cost compared to conventional DLS systems; may require complementary techniques for complete characterization of complex formulations.
Sanofi Pasteur SA
Technical Solution: Sanofi Pasteur has pioneered an integrated DLS-based vaccine formulation platform called "VaxStable" that combines dynamic light scattering with artificial intelligence for predictive stability analysis. Their system employs multi-temperature DLS measurements to generate comprehensive stability profiles across different environmental conditions. The technology incorporates specialized sample cells designed to minimize sample volume requirements (as low as 20μL), which is particularly valuable during early-stage vaccine development when material availability is limited. Sanofi's approach includes correlation of DLS data with immunogenicity outcomes, establishing predictive models that can forecast in vivo performance based on physical stability parameters. Their system also features proprietary algorithms that can detect subtle changes in particle populations before visible aggregation occurs, allowing for early intervention in formulation development. The platform has been successfully applied across their vaccine portfolio, including influenza, meningococcal, and COVID-19 vaccine candidates, demonstrating its versatility and reliability.
Strengths: AI-enhanced predictive capabilities linking physical stability to biological performance; minimal sample requirements suitable for precious antigens; extensive validation across diverse vaccine types. Weaknesses: Proprietary nature limits accessibility to other developers; complex data integration systems require specialized training; initial investment costs are substantial for smaller organizations.
Critical DLS Patents and Technical Literature
Dynamic light scattering based optical microrheology in non-aqueous solutions
PatentInactiveGB2537550A
Innovation
- The use of hydrophobic probe particles with specific surface chemistries, such as aromatic or aliphatic groups, to effectively disperse in non-aqueous solvents, allowing for the measurement of rheological properties through DLS by embedding these particles in the sample and detecting scattered photons.
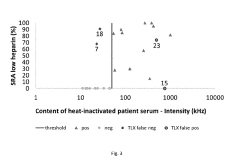
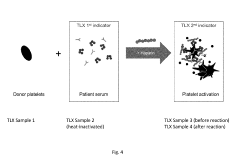
Methods and Apparatus for Predicting and Confirming Drug-Induced Thrombocytopenia Through Particle Detection with Dynamic Light Scattering
PatentInactiveUS20190250088A1
Innovation
- Dynamic Light Scattering (DLS) techniques are used to measure all particles in a patient sample, including platelets, microparticles, and aggregates, to predict and confirm DIT by analyzing changes in particle size distribution before and after exposure to suspected drugs, providing a more comprehensive assessment than existing methods.
Regulatory Considerations for Vaccine Analytics
Regulatory frameworks governing vaccine development and approval processes significantly impact the implementation of Dynamic Light Scattering (DLS) in vaccine formulation optimization. The FDA, EMA, and WHO have established specific guidelines for analytical methods used in vaccine characterization, with DLS increasingly recognized as a valuable tool for particle size analysis and stability assessment.
The ICH Q2(R1) guideline on validation of analytical procedures provides the foundation for validating DLS methods in vaccine development. This framework requires demonstration of accuracy, precision, specificity, linearity, range, and robustness—parameters that must be carefully established when implementing DLS for vaccine formulation optimization. Additionally, the FDA's Guidance for Industry on Analytical Procedures and Methods Validation for Drugs and Biologics specifically addresses considerations for particle size analysis techniques.
Regulatory bodies increasingly emphasize Quality by Design (QbD) approaches in vaccine development, where DLS serves as a critical analytical tool for establishing design space and control strategies. The ICH Q8(R2) guideline on pharmaceutical development explicitly recognizes the importance of particle characterization methods in defining critical quality attributes (CQAs) for vaccine formulations.
Method validation represents a significant regulatory hurdle for DLS implementation. Developers must demonstrate that DLS measurements are reproducible across different instruments, operators, and laboratories. The FDA's Process Validation guidance emphasizes the need for robust analytical methods that can reliably detect changes in product quality throughout the manufacturing process.
Regulatory submissions for vaccines utilizing DLS must include comprehensive method validation reports, reference standards, and control strategies. The level of detail required has increased in recent years, with regulators requesting more information on sample preparation procedures, instrument qualification, data processing algorithms, and statistical analysis methods used in DLS measurements.
Harmonization efforts between major regulatory agencies have improved consistency in expectations for analytical methods like DLS. The International Council for Harmonisation (ICH) continues to develop guidelines that standardize requirements across regions, facilitating global vaccine development programs that incorporate advanced analytical techniques.
Looking forward, regulatory trends indicate increasing acceptance of DLS as a compendial method for vaccine characterization, with pharmacopeias beginning to include specific monographs for DLS applications. This evolution will likely streamline regulatory approval processes for vaccines developed using DLS-optimized formulations, provided that developers maintain rigorous validation standards and documentation practices.
The ICH Q2(R1) guideline on validation of analytical procedures provides the foundation for validating DLS methods in vaccine development. This framework requires demonstration of accuracy, precision, specificity, linearity, range, and robustness—parameters that must be carefully established when implementing DLS for vaccine formulation optimization. Additionally, the FDA's Guidance for Industry on Analytical Procedures and Methods Validation for Drugs and Biologics specifically addresses considerations for particle size analysis techniques.
Regulatory bodies increasingly emphasize Quality by Design (QbD) approaches in vaccine development, where DLS serves as a critical analytical tool for establishing design space and control strategies. The ICH Q8(R2) guideline on pharmaceutical development explicitly recognizes the importance of particle characterization methods in defining critical quality attributes (CQAs) for vaccine formulations.
Method validation represents a significant regulatory hurdle for DLS implementation. Developers must demonstrate that DLS measurements are reproducible across different instruments, operators, and laboratories. The FDA's Process Validation guidance emphasizes the need for robust analytical methods that can reliably detect changes in product quality throughout the manufacturing process.
Regulatory submissions for vaccines utilizing DLS must include comprehensive method validation reports, reference standards, and control strategies. The level of detail required has increased in recent years, with regulators requesting more information on sample preparation procedures, instrument qualification, data processing algorithms, and statistical analysis methods used in DLS measurements.
Harmonization efforts between major regulatory agencies have improved consistency in expectations for analytical methods like DLS. The International Council for Harmonisation (ICH) continues to develop guidelines that standardize requirements across regions, facilitating global vaccine development programs that incorporate advanced analytical techniques.
Looking forward, regulatory trends indicate increasing acceptance of DLS as a compendial method for vaccine characterization, with pharmacopeias beginning to include specific monographs for DLS applications. This evolution will likely streamline regulatory approval processes for vaccines developed using DLS-optimized formulations, provided that developers maintain rigorous validation standards and documentation practices.
Quality Control Standards and Compliance
The implementation of Dynamic Light Scattering (DLS) in vaccine formulation optimization necessitates adherence to stringent quality control standards and regulatory compliance frameworks. Pharmaceutical manufacturers must follow guidelines established by regulatory bodies such as the FDA, EMA, and WHO, which provide specific requirements for particle size analysis methods used in vaccine development and production.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) have established specific monographs that detail the acceptable parameters for DLS measurements in vaccine formulations. USP <729> and Ph. Eur. 2.9.31 provide standardized protocols for particle sizing methods, including DLS, with defined acceptance criteria for measurement accuracy, precision, and reproducibility. These standards typically require system suitability tests using certified reference materials to verify instrument performance before analyzing vaccine samples.
International Conference on Harmonisation (ICH) guidelines, particularly ICH Q2(R1) for analytical method validation, apply directly to DLS implementation in vaccine quality control. These guidelines mandate validation of DLS methods through assessment of specificity, linearity, range, accuracy, precision, detection limit, and robustness. For vaccine applications, method validation must demonstrate that DLS can reliably detect changes in particle size distribution that could impact vaccine stability and efficacy.
Good Manufacturing Practice (GMP) regulations require comprehensive documentation of DLS procedures, including Standard Operating Procedures (SOPs), equipment qualification (IQ/OQ/PQ), and regular calibration schedules. Manufacturers must establish control charts with defined action and alert limits for particle size measurements to ensure consistent quality across production batches.
The FDA's Process Analytical Technology (PAT) initiative encourages the implementation of real-time DLS monitoring during vaccine manufacturing. This approach enables continuous quality verification rather than relying solely on end-product testing, allowing for immediate corrective actions when measurements deviate from specifications.
Data integrity requirements for DLS in vaccine quality control follow ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). Electronic data systems used for DLS measurements must comply with 21 CFR Part 11 or equivalent regulations, implementing appropriate access controls, audit trails, and data security measures.
Emerging global harmonization efforts, such as those led by the International Organization for Standardization (ISO), are working to standardize DLS methodologies across different regulatory jurisdictions, potentially simplifying compliance for manufacturers operating in multiple markets. ISO 22412:2017 specifically addresses DLS methods and provides internationally recognized standards for measurement protocols and data analysis.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) have established specific monographs that detail the acceptable parameters for DLS measurements in vaccine formulations. USP <729> and Ph. Eur. 2.9.31 provide standardized protocols for particle sizing methods, including DLS, with defined acceptance criteria for measurement accuracy, precision, and reproducibility. These standards typically require system suitability tests using certified reference materials to verify instrument performance before analyzing vaccine samples.
International Conference on Harmonisation (ICH) guidelines, particularly ICH Q2(R1) for analytical method validation, apply directly to DLS implementation in vaccine quality control. These guidelines mandate validation of DLS methods through assessment of specificity, linearity, range, accuracy, precision, detection limit, and robustness. For vaccine applications, method validation must demonstrate that DLS can reliably detect changes in particle size distribution that could impact vaccine stability and efficacy.
Good Manufacturing Practice (GMP) regulations require comprehensive documentation of DLS procedures, including Standard Operating Procedures (SOPs), equipment qualification (IQ/OQ/PQ), and regular calibration schedules. Manufacturers must establish control charts with defined action and alert limits for particle size measurements to ensure consistent quality across production batches.
The FDA's Process Analytical Technology (PAT) initiative encourages the implementation of real-time DLS monitoring during vaccine manufacturing. This approach enables continuous quality verification rather than relying solely on end-product testing, allowing for immediate corrective actions when measurements deviate from specifications.
Data integrity requirements for DLS in vaccine quality control follow ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available). Electronic data systems used for DLS measurements must comply with 21 CFR Part 11 or equivalent regulations, implementing appropriate access controls, audit trails, and data security measures.
Emerging global harmonization efforts, such as those led by the International Organization for Standardization (ISO), are working to standardize DLS methodologies across different regulatory jurisdictions, potentially simplifying compliance for manufacturers operating in multiple markets. ISO 22412:2017 specifically addresses DLS methods and provides internationally recognized standards for measurement protocols and data analysis.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!