How to Couple Microfluidics to Photonic Resonators for Real-Time Biomarker Detection
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Microfluidic-Photonic Integration Background
The integration of microfluidics and photonic resonators represents a significant advancement in the field of biosensing and biomarker detection. This convergence of technologies has its roots in the early 2000s when researchers began exploring ways to combine the precise fluid handling capabilities of microfluidics with the high sensitivity of optical sensing methods.
Microfluidics, a technology that manipulates small volumes of fluids in channels with dimensions of tens to hundreds of micrometers, emerged in the 1990s. It offered the ability to handle minute sample volumes, reduce reagent consumption, and enable rapid analysis. Concurrently, photonic resonators, particularly whispering gallery mode (WGM) resonators, were being developed as highly sensitive optical sensors capable of detecting single molecules.
The motivation for coupling these technologies stemmed from the need for real-time, label-free detection of biomarkers in complex biological samples. Traditional biomarker detection methods often required time-consuming laboratory procedures and large sample volumes. By integrating microfluidics with photonic resonators, researchers aimed to create compact, sensitive, and rapid biosensing platforms.
Early attempts at integration faced challenges in aligning the microfluidic channels with the optical sensing elements while maintaining the high quality factor of the resonators. Researchers experimented with various fabrication techniques, including soft lithography for microfluidics and etching processes for photonic structures, to achieve seamless integration.
A significant milestone was reached in the mid-2000s when several research groups successfully demonstrated the first integrated microfluidic-photonic resonator systems. These early prototypes showed promise in detecting proteins and nucleic acids with high sensitivity. However, they often suffered from issues such as non-specific binding and limited reproducibility.
Over the past decade, advancements in nanofabrication techniques, surface chemistry, and optical detection methods have led to more sophisticated integrated systems. The development of on-chip lasers and detectors has further miniaturized these devices, paving the way for portable, point-of-care diagnostic tools.
The potential applications of microfluidic-photonic integrated systems have expanded beyond biomarker detection to include environmental monitoring, food safety testing, and drug discovery. This versatility has attracted significant interest from both academia and industry, driving further research and development in the field.
Microfluidics, a technology that manipulates small volumes of fluids in channels with dimensions of tens to hundreds of micrometers, emerged in the 1990s. It offered the ability to handle minute sample volumes, reduce reagent consumption, and enable rapid analysis. Concurrently, photonic resonators, particularly whispering gallery mode (WGM) resonators, were being developed as highly sensitive optical sensors capable of detecting single molecules.
The motivation for coupling these technologies stemmed from the need for real-time, label-free detection of biomarkers in complex biological samples. Traditional biomarker detection methods often required time-consuming laboratory procedures and large sample volumes. By integrating microfluidics with photonic resonators, researchers aimed to create compact, sensitive, and rapid biosensing platforms.
Early attempts at integration faced challenges in aligning the microfluidic channels with the optical sensing elements while maintaining the high quality factor of the resonators. Researchers experimented with various fabrication techniques, including soft lithography for microfluidics and etching processes for photonic structures, to achieve seamless integration.
A significant milestone was reached in the mid-2000s when several research groups successfully demonstrated the first integrated microfluidic-photonic resonator systems. These early prototypes showed promise in detecting proteins and nucleic acids with high sensitivity. However, they often suffered from issues such as non-specific binding and limited reproducibility.
Over the past decade, advancements in nanofabrication techniques, surface chemistry, and optical detection methods have led to more sophisticated integrated systems. The development of on-chip lasers and detectors has further miniaturized these devices, paving the way for portable, point-of-care diagnostic tools.
The potential applications of microfluidic-photonic integrated systems have expanded beyond biomarker detection to include environmental monitoring, food safety testing, and drug discovery. This versatility has attracted significant interest from both academia and industry, driving further research and development in the field.
Biomarker Detection Market Analysis
The biomarker detection market has experienced significant growth in recent years, driven by increasing demand for early disease diagnosis, personalized medicine, and point-of-care testing. The global biomarker detection market was valued at approximately $30 billion in 2020 and is projected to reach over $70 billion by 2026, with a compound annual growth rate (CAGR) of around 15%.
Several factors contribute to this market expansion. The rising prevalence of chronic diseases, such as cancer, cardiovascular disorders, and neurological conditions, has intensified the need for accurate and timely biomarker detection. Additionally, advancements in genomics and proteomics have led to the discovery of novel biomarkers, expanding the potential applications of biomarker-based diagnostics.
The integration of microfluidics and photonic resonators for real-time biomarker detection represents a promising technological advancement in this field. This approach offers several advantages, including high sensitivity, rapid analysis, and minimal sample volume requirements. These features align well with the growing demand for point-of-care diagnostics and personalized medicine.
The market for microfluidic-based biomarker detection systems is expected to grow at a faster rate than the overall biomarker detection market, with a CAGR of approximately 20% through 2026. This growth is fueled by the increasing adoption of lab-on-a-chip technologies and the need for portable, user-friendly diagnostic devices.
Key market segments for biomarker detection include oncology, cardiovascular diseases, neurological disorders, and infectious diseases. Oncology remains the largest segment, accounting for about 30% of the market share, due to the critical role of biomarkers in cancer diagnosis, prognosis, and treatment monitoring.
Geographically, North America dominates the biomarker detection market, followed by Europe and Asia-Pacific. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by improving healthcare infrastructure, increasing research and development activities, and rising healthcare expenditure in countries like China and India.
The competitive landscape of the biomarker detection market is characterized by the presence of both established players and innovative start-ups. Major companies in this space include Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, and Thermo Fisher Scientific. These companies are investing heavily in research and development to maintain their market position and capitalize on emerging technologies like microfluidics-coupled photonic resonators.
Several factors contribute to this market expansion. The rising prevalence of chronic diseases, such as cancer, cardiovascular disorders, and neurological conditions, has intensified the need for accurate and timely biomarker detection. Additionally, advancements in genomics and proteomics have led to the discovery of novel biomarkers, expanding the potential applications of biomarker-based diagnostics.
The integration of microfluidics and photonic resonators for real-time biomarker detection represents a promising technological advancement in this field. This approach offers several advantages, including high sensitivity, rapid analysis, and minimal sample volume requirements. These features align well with the growing demand for point-of-care diagnostics and personalized medicine.
The market for microfluidic-based biomarker detection systems is expected to grow at a faster rate than the overall biomarker detection market, with a CAGR of approximately 20% through 2026. This growth is fueled by the increasing adoption of lab-on-a-chip technologies and the need for portable, user-friendly diagnostic devices.
Key market segments for biomarker detection include oncology, cardiovascular diseases, neurological disorders, and infectious diseases. Oncology remains the largest segment, accounting for about 30% of the market share, due to the critical role of biomarkers in cancer diagnosis, prognosis, and treatment monitoring.
Geographically, North America dominates the biomarker detection market, followed by Europe and Asia-Pacific. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by improving healthcare infrastructure, increasing research and development activities, and rising healthcare expenditure in countries like China and India.
The competitive landscape of the biomarker detection market is characterized by the presence of both established players and innovative start-ups. Major companies in this space include Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, and Thermo Fisher Scientific. These companies are investing heavily in research and development to maintain their market position and capitalize on emerging technologies like microfluidics-coupled photonic resonators.
Challenges in Microfluidic-Photonic Coupling
Coupling microfluidics to photonic resonators for real-time biomarker detection presents several significant challenges that researchers and engineers must overcome. One of the primary obstacles is achieving precise alignment between the microfluidic channels and the photonic resonators. Even slight misalignments can lead to significant reductions in sensing performance and reliability.
Another major challenge lies in managing the different scale regimes of microfluidics and photonics. Microfluidic channels typically operate on the scale of tens to hundreds of micrometers, while photonic resonators often have features on the nanometer scale. Bridging this gap requires innovative design approaches and fabrication techniques to ensure effective coupling between the two systems.
Material compatibility is a crucial consideration in microfluidic-photonic coupling. The materials used for microfluidic devices, such as polydimethylsiloxane (PDMS), may not always be compatible with the high-quality optical materials required for photonic resonators. This incompatibility can lead to issues with device integration, optical performance, and long-term stability.
Minimizing optical losses at the interface between the microfluidic and photonic components is another significant challenge. Refractive index mismatches and surface roughness can cause scattering and reflection, reducing the overall sensitivity of the biosensing system. Developing strategies to mitigate these losses while maintaining the integrity of both the microfluidic and photonic elements is crucial.
The integration of microfluidics with photonic resonators also faces challenges related to fluid dynamics. Ensuring uniform flow and avoiding bubble formation within the sensing region is critical for accurate and reproducible biomarker detection. The design of microfluidic channels must carefully consider flow patterns and potential turbulence that could affect the interaction between the analyte and the photonic sensing elements.
Temperature control and stability present another hurdle in microfluidic-photonic coupling. Photonic resonators are often sensitive to temperature fluctuations, which can affect their resonance characteristics. Simultaneously, fluid flow in microfluidic channels can introduce thermal gradients. Developing effective thermal management strategies is essential to maintain consistent and reliable sensing performance.
Lastly, the challenge of scalability and manufacturability must be addressed. While proof-of-concept devices can be fabricated in research settings, translating these designs into commercially viable products requires consideration of mass production techniques, cost-effectiveness, and reproducibility. Developing fabrication processes that can reliably produce integrated microfluidic-photonic devices at scale remains an ongoing challenge in the field.
Another major challenge lies in managing the different scale regimes of microfluidics and photonics. Microfluidic channels typically operate on the scale of tens to hundreds of micrometers, while photonic resonators often have features on the nanometer scale. Bridging this gap requires innovative design approaches and fabrication techniques to ensure effective coupling between the two systems.
Material compatibility is a crucial consideration in microfluidic-photonic coupling. The materials used for microfluidic devices, such as polydimethylsiloxane (PDMS), may not always be compatible with the high-quality optical materials required for photonic resonators. This incompatibility can lead to issues with device integration, optical performance, and long-term stability.
Minimizing optical losses at the interface between the microfluidic and photonic components is another significant challenge. Refractive index mismatches and surface roughness can cause scattering and reflection, reducing the overall sensitivity of the biosensing system. Developing strategies to mitigate these losses while maintaining the integrity of both the microfluidic and photonic elements is crucial.
The integration of microfluidics with photonic resonators also faces challenges related to fluid dynamics. Ensuring uniform flow and avoiding bubble formation within the sensing region is critical for accurate and reproducible biomarker detection. The design of microfluidic channels must carefully consider flow patterns and potential turbulence that could affect the interaction between the analyte and the photonic sensing elements.
Temperature control and stability present another hurdle in microfluidic-photonic coupling. Photonic resonators are often sensitive to temperature fluctuations, which can affect their resonance characteristics. Simultaneously, fluid flow in microfluidic channels can introduce thermal gradients. Developing effective thermal management strategies is essential to maintain consistent and reliable sensing performance.
Lastly, the challenge of scalability and manufacturability must be addressed. While proof-of-concept devices can be fabricated in research settings, translating these designs into commercially viable products requires consideration of mass production techniques, cost-effectiveness, and reproducibility. Developing fabrication processes that can reliably produce integrated microfluidic-photonic devices at scale remains an ongoing challenge in the field.
Current Microfluidic-Photonic Coupling Methods
01 Integration of microfluidics and photonic resonators
Combining microfluidic systems with photonic resonators enables real-time detection of various analytes. This integration allows for precise control of fluid flow and highly sensitive optical sensing, making it possible to detect small changes in the sample composition or concentration.- Integration of microfluidics and photonic resonators: Combining microfluidic systems with photonic resonators enables real-time detection of various analytes. This integration allows for precise control of fluid flow and enhanced sensitivity in optical sensing, making it suitable for applications in biosensing and chemical analysis.
- Real-time detection using whispering gallery mode resonators: Whispering gallery mode resonators, a type of photonic resonator, are utilized in microfluidic systems for real-time detection. These resonators offer high sensitivity and can detect minute changes in the surrounding medium, making them ideal for monitoring biological and chemical processes in real-time.
- Lab-on-a-chip devices with integrated optical sensing: Lab-on-a-chip devices incorporating microfluidics and photonic resonators enable miniaturized, portable systems for real-time detection. These integrated devices offer advantages such as reduced sample volume, faster analysis times, and improved sensitivity compared to traditional methods.
- Surface plasmon resonance-based detection in microfluidic systems: Surface plasmon resonance (SPR) is utilized in microfluidic systems for real-time detection of biomolecules and chemical compounds. This technique offers label-free detection with high sensitivity and is particularly useful for studying molecular interactions and kinetics in real-time.
- Microfluidic photonic crystal sensors for real-time detection: Photonic crystal sensors integrated with microfluidic systems enable real-time detection of various analytes. These sensors utilize the unique optical properties of photonic crystals to achieve high sensitivity and specificity in detecting changes in the local environment, making them suitable for applications in environmental monitoring and biomedical diagnostics.
02 Real-time detection using optical sensing techniques
Photonic resonators coupled with microfluidics enable real-time detection through various optical sensing techniques. These may include surface plasmon resonance, whispering gallery mode resonators, or interferometric methods, allowing for continuous monitoring of chemical or biological processes within the microfluidic channels.Expand Specific Solutions03 Enhanced sensitivity and specificity in detection
The combination of microfluidics and photonic resonators significantly improves the sensitivity and specificity of detection. By confining light and analytes in small volumes, the interaction between them is enhanced, leading to lower detection limits and improved signal-to-noise ratios.Expand Specific Solutions04 Miniaturization and lab-on-a-chip applications
Microfluidics coupled with photonic resonators enables the development of compact, portable lab-on-a-chip devices. These miniaturized systems integrate multiple laboratory functions on a single chip, allowing for point-of-care diagnostics and on-site environmental monitoring.Expand Specific Solutions05 Multi-analyte detection and parallel processing
The integration of microfluidics and photonic resonators facilitates multi-analyte detection and parallel processing. By incorporating multiple sensing elements or channels, these systems can simultaneously detect and analyze various target molecules or particles in a single sample, increasing throughput and efficiency.Expand Specific Solutions
Key Players in Integrated Biosensors
The field of coupling microfluidics to photonic resonators for real-time biomarker detection is in an early growth stage, with significant potential for expansion. The market size is projected to increase substantially as this technology addresses critical needs in rapid, sensitive diagnostics. While still evolving, the technology is progressing towards maturity, with key players like Massachusetts Institute of Technology, École Polytechnique Fédérale de Lausanne, and Tianjin University leading academic research. Companies such as Intel Corp. and Robert Bosch GmbH are leveraging their expertise in microelectronics to advance commercial applications. Startups like Xsensio SA and SeLux Diagnostics are also emerging with innovative approaches, indicating a dynamic and competitive landscape poised for further development and market penetration.
École Polytechnique Fédérale de Lausanne
Technical Solution: EPFL has developed an innovative platform that combines microfluidics with photonic crystal cavities for biomarker detection. Their approach uses a slotted photonic crystal cavity design, which enhances light-matter interaction and improves sensitivity. The microfluidic channel is precisely aligned with the cavity to maximize the overlap between the optical mode and the analyte. EPFL's system incorporates on-chip reference channels to compensate for environmental fluctuations, ensuring reliable measurements[2]. They have also implemented advanced surface chemistry techniques to enable multiplexed detection of multiple biomarkers simultaneously. The platform utilizes a tunable laser source and high-resolution spectrometer for precise readout of resonance shifts[4].
Strengths: Ultra-high sensitivity, multiplexing capability, environmental drift compensation. Weaknesses: Complex fabrication process, potential for clogging in narrow slot regions, higher cost due to specialized components.
The Johns Hopkins University
Technical Solution: Johns Hopkins University has developed a microfluidic-photonic platform for biomarker detection using plasmonic nanoantennas integrated with microfluidic channels. Their system utilizes arrays of gold nanoantennas fabricated on a glass substrate, which are then sealed with a PDMS microfluidic layer. The plasmonic resonances of the nanoantennas are highly sensitive to changes in the local refractive index, allowing for detection of biomarker binding events. JHU researchers have implemented a dual-wavelength interrogation scheme to improve the signal-to-noise ratio and reduce false positives[7]. The platform also incorporates on-chip sample preparation modules, including mixing and filtration units, to enable analysis of complex biological samples. Advanced data analysis techniques, including machine learning algorithms, are employed for real-time biomarker quantification[8].
Strengths: High sensitivity due to plasmonic enhancement, integrated sample preparation, advanced data analysis capabilities. Weaknesses: Potential for non-specific adsorption on metal surfaces, limited dynamic range, higher fabrication costs.
Innovations in Resonator-based Biosensing
Integrated microfluidic device for preconcentration and detection of multiple biomarkers
PatentWO2008043040A3
Innovation
- Integration of a nanofluidic concentrator with a suspended microchannel resonator for enhanced biomarker detection.
- Amplification of biomarker concentration to approach the dissociation constant of biomarker/antibody complex for improved sensitivity.
- Use of suspended microchannel resonator for signal generation related to biomarker quantity.
Surface measurement sensing-based real time nucleic acid amplification measuring device
PatentWO2017119639A1
Innovation
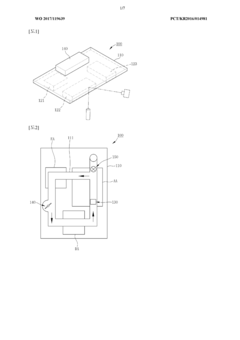
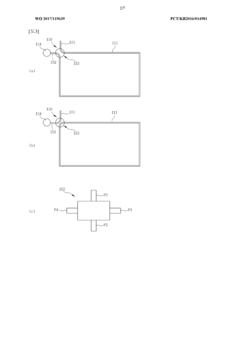
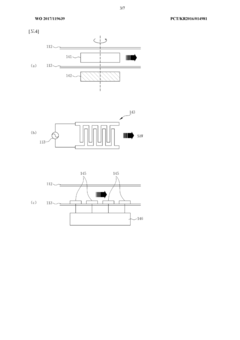
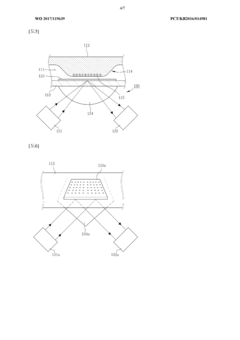
- A real-time nucleic acid amplification measurement device using a microfluidic chip with surface measurement sensing, such as surface plasmon resonance, that reduces reagent use, eliminates air bubble errors, and allows for simultaneous detection of hundreds to thousands of biomarkers through a closed-loop microfluidic channel with circulating sample flow and multiple heating units for PCR.
Fabrication Techniques for Integrated Devices
The fabrication of integrated devices that couple microfluidics to photonic resonators for real-time biomarker detection requires a combination of advanced manufacturing techniques. These techniques must address the challenges of creating both microfluidic channels and photonic structures on a single platform while maintaining high precision and compatibility.
Photolithography remains a cornerstone technique in this field, allowing for the creation of intricate patterns on substrates. For photonic resonators, electron-beam lithography is often employed due to its ability to produce nanoscale features with high resolution. This technique is particularly crucial for creating optical waveguides and resonator structures that require sub-micron precision.
Soft lithography, particularly replica molding using polydimethylsiloxane (PDMS), is widely used for fabricating microfluidic channels. This method offers flexibility in design and rapid prototyping capabilities. The integration of PDMS-based microfluidics with photonic structures often involves careful alignment and bonding processes to ensure seamless coupling.
Etching techniques play a vital role in creating three-dimensional structures. Reactive ion etching (RIE) and deep reactive ion etching (DRIE) are commonly used to create high-aspect-ratio features in silicon or glass substrates. These techniques allow for the formation of vertical sidewalls in both microfluidic channels and photonic components.
Surface modification techniques are essential for optimizing the interface between microfluidic and photonic elements. Plasma treatment and chemical functionalization are frequently employed to alter surface properties, enhancing bonding strength and controlling wettability.
Advanced deposition methods such as atomic layer deposition (ALD) and chemical vapor deposition (CVD) are utilized to create thin films with precise thickness control. These techniques are crucial for depositing materials that form the core and cladding layers of waveguides and resonators.
3D printing technologies, particularly stereolithography and two-photon polymerization, are emerging as powerful tools for fabricating complex microfluidic-photonic integrated devices. These additive manufacturing techniques offer unprecedented design freedom and the ability to create truly three-dimensional structures.
Lastly, hybrid integration approaches are gaining traction, combining different materials and fabrication techniques to leverage their respective strengths. For instance, silicon photonics may be integrated with polymer microfluidics using wafer-level bonding techniques, enabling high-performance devices with cost-effective manufacturing.
Photolithography remains a cornerstone technique in this field, allowing for the creation of intricate patterns on substrates. For photonic resonators, electron-beam lithography is often employed due to its ability to produce nanoscale features with high resolution. This technique is particularly crucial for creating optical waveguides and resonator structures that require sub-micron precision.
Soft lithography, particularly replica molding using polydimethylsiloxane (PDMS), is widely used for fabricating microfluidic channels. This method offers flexibility in design and rapid prototyping capabilities. The integration of PDMS-based microfluidics with photonic structures often involves careful alignment and bonding processes to ensure seamless coupling.
Etching techniques play a vital role in creating three-dimensional structures. Reactive ion etching (RIE) and deep reactive ion etching (DRIE) are commonly used to create high-aspect-ratio features in silicon or glass substrates. These techniques allow for the formation of vertical sidewalls in both microfluidic channels and photonic components.
Surface modification techniques are essential for optimizing the interface between microfluidic and photonic elements. Plasma treatment and chemical functionalization are frequently employed to alter surface properties, enhancing bonding strength and controlling wettability.
Advanced deposition methods such as atomic layer deposition (ALD) and chemical vapor deposition (CVD) are utilized to create thin films with precise thickness control. These techniques are crucial for depositing materials that form the core and cladding layers of waveguides and resonators.
3D printing technologies, particularly stereolithography and two-photon polymerization, are emerging as powerful tools for fabricating complex microfluidic-photonic integrated devices. These additive manufacturing techniques offer unprecedented design freedom and the ability to create truly three-dimensional structures.
Lastly, hybrid integration approaches are gaining traction, combining different materials and fabrication techniques to leverage their respective strengths. For instance, silicon photonics may be integrated with polymer microfluidics using wafer-level bonding techniques, enabling high-performance devices with cost-effective manufacturing.
Regulatory Considerations for Biosensors
The integration of microfluidics and photonic resonators for real-time biomarker detection presents significant regulatory considerations that must be addressed to ensure compliance and market approval. These biosensors fall under the purview of medical device regulations in most jurisdictions, requiring careful navigation of the regulatory landscape.
In the United States, the Food and Drug Administration (FDA) oversees the regulation of medical devices, including biosensors. The classification of these devices depends on their intended use and risk level. Microfluidic-photonic biosensors for real-time biomarker detection are likely to be classified as Class II or Class III devices, necessitating either a 510(k) premarket notification or a more rigorous premarket approval (PMA) process.
The European Union's regulatory framework for medical devices has recently undergone significant changes with the implementation of the Medical Device Regulation (MDR). Under the MDR, these biosensors would likely be classified as Class C or D devices, requiring conformity assessment by a notified body and the implementation of a comprehensive quality management system.
Data privacy and security regulations, such as the General Data Protection Regulation (GDPR) in the EU and the Health Insurance Portability and Accountability Act (HIPAA) in the US, must also be considered. These regulations impact the collection, storage, and transmission of patient data generated by the biosensors.
Clinical validation is a critical aspect of the regulatory process. Manufacturers must demonstrate the analytical and clinical performance of their devices through well-designed studies. This includes establishing sensitivity, specificity, and reproducibility of biomarker detection, as well as demonstrating the clinical utility of real-time monitoring.
Regulatory bodies also focus on the manufacturing processes and quality control measures. Good Manufacturing Practices (GMP) must be adhered to, ensuring consistent production of high-quality devices. This includes validation of the microfluidic and photonic components, as well as the integration process.
Post-market surveillance is another crucial regulatory consideration. Manufacturers must implement systems to monitor device performance, collect user feedback, and report adverse events. This ongoing process helps ensure the continued safety and efficacy of the biosensors in real-world clinical settings.
As the field of microfluidic-photonic biosensors evolves, regulatory frameworks may need to adapt. Collaboration between industry, academia, and regulatory bodies is essential to develop appropriate guidelines that balance innovation with patient safety. Early engagement with regulatory authorities can help navigate these complex requirements and streamline the path to market approval.
In the United States, the Food and Drug Administration (FDA) oversees the regulation of medical devices, including biosensors. The classification of these devices depends on their intended use and risk level. Microfluidic-photonic biosensors for real-time biomarker detection are likely to be classified as Class II or Class III devices, necessitating either a 510(k) premarket notification or a more rigorous premarket approval (PMA) process.
The European Union's regulatory framework for medical devices has recently undergone significant changes with the implementation of the Medical Device Regulation (MDR). Under the MDR, these biosensors would likely be classified as Class C or D devices, requiring conformity assessment by a notified body and the implementation of a comprehensive quality management system.
Data privacy and security regulations, such as the General Data Protection Regulation (GDPR) in the EU and the Health Insurance Portability and Accountability Act (HIPAA) in the US, must also be considered. These regulations impact the collection, storage, and transmission of patient data generated by the biosensors.
Clinical validation is a critical aspect of the regulatory process. Manufacturers must demonstrate the analytical and clinical performance of their devices through well-designed studies. This includes establishing sensitivity, specificity, and reproducibility of biomarker detection, as well as demonstrating the clinical utility of real-time monitoring.
Regulatory bodies also focus on the manufacturing processes and quality control measures. Good Manufacturing Practices (GMP) must be adhered to, ensuring consistent production of high-quality devices. This includes validation of the microfluidic and photonic components, as well as the integration process.
Post-market surveillance is another crucial regulatory consideration. Manufacturers must implement systems to monitor device performance, collect user feedback, and report adverse events. This ongoing process helps ensure the continued safety and efficacy of the biosensors in real-world clinical settings.
As the field of microfluidic-photonic biosensors evolves, regulatory frameworks may need to adapt. Collaboration between industry, academia, and regulatory bodies is essential to develop appropriate guidelines that balance innovation with patient safety. Early engagement with regulatory authorities can help navigate these complex requirements and streamline the path to market approval.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!