Label-free vs Label-based Readouts on Photonic Platforms: Which to Use and When
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Photonic Readout Evolution
The evolution of photonic readout technologies has been a critical factor in advancing biosensing and diagnostic capabilities. Initially, label-based readouts dominated the field, offering high sensitivity and specificity. These methods relied on fluorescent or colorimetric labels attached to target molecules, enabling their detection through optical signals. However, the labeling process often introduced complexities and potential interference with the analyte's natural behavior.
As photonic technologies progressed, label-free readouts emerged as a promising alternative. These techniques leverage intrinsic properties of biomolecules, such as refractive index changes or molecular vibrations, to detect and quantify targets without the need for labels. Early label-free methods included surface plasmon resonance (SPR) and interferometric techniques, which gained traction due to their ability to provide real-time, quantitative measurements of biomolecular interactions.
The development of integrated photonic circuits marked a significant milestone in readout evolution. These miniaturized platforms allowed for the integration of multiple sensing elements on a single chip, enhancing multiplexing capabilities and reducing sample volumes. Advances in nanofabrication techniques further improved the sensitivity and scalability of both label-free and label-based readouts on photonic platforms.
Recent years have seen a convergence of label-free and label-based approaches. Hybrid systems combining the strengths of both methodologies have emerged, offering enhanced performance and versatility. For instance, plasmonic-enhanced fluorescence techniques leverage the sensitivity of label-free plasmonic sensing with the specificity of fluorescent labels.
The ongoing evolution of photonic readouts has been driven by the demand for higher sensitivity, specificity, and throughput in various applications, from point-of-care diagnostics to environmental monitoring. Innovations in light sources, detectors, and data processing algorithms have continuously pushed the boundaries of what is achievable with photonic sensing platforms.
Looking forward, the field is moving towards more integrated, multifunctional photonic readout systems. These advanced platforms aim to combine multiple sensing modalities, enabling simultaneous label-free and label-based measurements on a single chip. Such developments promise to provide comprehensive, high-content information about complex biological systems, further expanding the capabilities and applications of photonic readout technologies.
As photonic technologies progressed, label-free readouts emerged as a promising alternative. These techniques leverage intrinsic properties of biomolecules, such as refractive index changes or molecular vibrations, to detect and quantify targets without the need for labels. Early label-free methods included surface plasmon resonance (SPR) and interferometric techniques, which gained traction due to their ability to provide real-time, quantitative measurements of biomolecular interactions.
The development of integrated photonic circuits marked a significant milestone in readout evolution. These miniaturized platforms allowed for the integration of multiple sensing elements on a single chip, enhancing multiplexing capabilities and reducing sample volumes. Advances in nanofabrication techniques further improved the sensitivity and scalability of both label-free and label-based readouts on photonic platforms.
Recent years have seen a convergence of label-free and label-based approaches. Hybrid systems combining the strengths of both methodologies have emerged, offering enhanced performance and versatility. For instance, plasmonic-enhanced fluorescence techniques leverage the sensitivity of label-free plasmonic sensing with the specificity of fluorescent labels.
The ongoing evolution of photonic readouts has been driven by the demand for higher sensitivity, specificity, and throughput in various applications, from point-of-care diagnostics to environmental monitoring. Innovations in light sources, detectors, and data processing algorithms have continuously pushed the boundaries of what is achievable with photonic sensing platforms.
Looking forward, the field is moving towards more integrated, multifunctional photonic readout systems. These advanced platforms aim to combine multiple sensing modalities, enabling simultaneous label-free and label-based measurements on a single chip. Such developments promise to provide comprehensive, high-content information about complex biological systems, further expanding the capabilities and applications of photonic readout technologies.
Market Demand Analysis
The market demand for photonic platforms utilizing label-free and label-based readouts has been steadily growing, driven by advancements in biosensing, medical diagnostics, and environmental monitoring. Label-free detection methods have gained significant traction due to their ability to provide real-time, quantitative measurements without the need for additional reagents or sample preparation steps. This has led to increased adoption in point-of-care diagnostics and continuous monitoring applications.
Label-based readouts, on the other hand, continue to dominate in areas requiring high sensitivity and specificity, such as genomics and proteomics research. The global biosensors market, which encompasses both label-free and label-based technologies, is projected to experience substantial growth in the coming years, with photonic platforms playing a crucial role in this expansion.
In the pharmaceutical industry, there is a growing demand for label-free technologies in drug discovery and development processes. These methods allow for the study of biomolecular interactions in their native state, providing more physiologically relevant data. This has led to increased investment in label-free photonic platforms by major pharmaceutical companies and contract research organizations.
The healthcare sector has shown particular interest in label-free photonic biosensors for rapid disease diagnosis and monitoring. The COVID-19 pandemic has further accelerated this trend, highlighting the need for quick, accurate, and easily deployable diagnostic tools. As a result, there has been a surge in research and development activities focused on label-free photonic platforms for viral detection and immune response monitoring.
Environmental monitoring applications have also contributed to the growing market demand for both label-free and label-based photonic platforms. Water quality assessment, air pollution monitoring, and food safety testing are areas where these technologies are finding increasing application. The ability of photonic platforms to provide rapid, on-site analysis has made them particularly attractive for environmental regulatory agencies and industries subject to strict environmental compliance requirements.
Despite the advantages of label-free methods, label-based readouts remain essential in many research and clinical applications due to their superior sensitivity and multiplexing capabilities. This has led to a parallel growth in the market for advanced fluorescent labels and nanoparticle-based tags compatible with photonic platforms. The choice between label-free and label-based approaches often depends on the specific application requirements, leading to a diverse and dynamic market landscape.
Label-based readouts, on the other hand, continue to dominate in areas requiring high sensitivity and specificity, such as genomics and proteomics research. The global biosensors market, which encompasses both label-free and label-based technologies, is projected to experience substantial growth in the coming years, with photonic platforms playing a crucial role in this expansion.
In the pharmaceutical industry, there is a growing demand for label-free technologies in drug discovery and development processes. These methods allow for the study of biomolecular interactions in their native state, providing more physiologically relevant data. This has led to increased investment in label-free photonic platforms by major pharmaceutical companies and contract research organizations.
The healthcare sector has shown particular interest in label-free photonic biosensors for rapid disease diagnosis and monitoring. The COVID-19 pandemic has further accelerated this trend, highlighting the need for quick, accurate, and easily deployable diagnostic tools. As a result, there has been a surge in research and development activities focused on label-free photonic platforms for viral detection and immune response monitoring.
Environmental monitoring applications have also contributed to the growing market demand for both label-free and label-based photonic platforms. Water quality assessment, air pollution monitoring, and food safety testing are areas where these technologies are finding increasing application. The ability of photonic platforms to provide rapid, on-site analysis has made them particularly attractive for environmental regulatory agencies and industries subject to strict environmental compliance requirements.
Despite the advantages of label-free methods, label-based readouts remain essential in many research and clinical applications due to their superior sensitivity and multiplexing capabilities. This has led to a parallel growth in the market for advanced fluorescent labels and nanoparticle-based tags compatible with photonic platforms. The choice between label-free and label-based approaches often depends on the specific application requirements, leading to a diverse and dynamic market landscape.
Current Challenges
The field of photonic platforms for biosensing faces several significant challenges, particularly when comparing label-free and label-based readout methods. One of the primary obstacles is achieving high sensitivity and specificity simultaneously. Label-free methods often struggle with specificity, as they rely on detecting minute changes in physical properties without the aid of specific markers. Conversely, label-based techniques may introduce additional complexity and potential interference from the labels themselves.
Another challenge lies in the miniaturization and integration of photonic biosensors. As the demand for portable and point-of-care devices grows, researchers must overcome the difficulties of scaling down optical components while maintaining performance. This is particularly challenging for label-based systems, which may require additional space for label storage and handling.
The cost-effectiveness of photonic platforms remains a significant hurdle. Label-free methods often require sophisticated and expensive equipment for precise measurements, while label-based approaches incur ongoing costs for reagents and labels. Striking a balance between performance and affordability is crucial for widespread adoption in both research and clinical settings.
Reproducibility and standardization pose challenges for both label-free and label-based readouts. Label-free methods may be more susceptible to environmental factors and sample variations, leading to inconsistent results across different laboratories. Label-based techniques, while potentially more robust, face issues with label stability, conjugation efficiency, and batch-to-batch variability.
The development of multiplexed assays presents another obstacle. Label-free methods often struggle to distinguish between multiple analytes simultaneously, whereas label-based approaches may encounter issues with spectral overlap and cross-reactivity when using multiple labels.
Real-time monitoring capabilities are increasingly important in biosensing applications. Label-free methods generally excel in this area, providing continuous data on binding events. However, they may lack the ability to provide detailed information about the nature of the interactions. Label-based techniques, while potentially offering more specific information, often require discrete time points for measurement, limiting real-time capabilities.
Lastly, the challenge of non-specific binding affects both label-free and label-based readouts. Label-free methods are particularly susceptible to this issue, as they detect all changes in the sensing environment. Label-based approaches, while potentially more specific, may still suffer from non-specific interactions between labels and non-target molecules. Developing effective surface chemistries and blocking strategies to minimize these effects remains an ongoing challenge in the field.
Another challenge lies in the miniaturization and integration of photonic biosensors. As the demand for portable and point-of-care devices grows, researchers must overcome the difficulties of scaling down optical components while maintaining performance. This is particularly challenging for label-based systems, which may require additional space for label storage and handling.
The cost-effectiveness of photonic platforms remains a significant hurdle. Label-free methods often require sophisticated and expensive equipment for precise measurements, while label-based approaches incur ongoing costs for reagents and labels. Striking a balance between performance and affordability is crucial for widespread adoption in both research and clinical settings.
Reproducibility and standardization pose challenges for both label-free and label-based readouts. Label-free methods may be more susceptible to environmental factors and sample variations, leading to inconsistent results across different laboratories. Label-based techniques, while potentially more robust, face issues with label stability, conjugation efficiency, and batch-to-batch variability.
The development of multiplexed assays presents another obstacle. Label-free methods often struggle to distinguish between multiple analytes simultaneously, whereas label-based approaches may encounter issues with spectral overlap and cross-reactivity when using multiple labels.
Real-time monitoring capabilities are increasingly important in biosensing applications. Label-free methods generally excel in this area, providing continuous data on binding events. However, they may lack the ability to provide detailed information about the nature of the interactions. Label-based techniques, while potentially offering more specific information, often require discrete time points for measurement, limiting real-time capabilities.
Lastly, the challenge of non-specific binding affects both label-free and label-based readouts. Label-free methods are particularly susceptible to this issue, as they detect all changes in the sensing environment. Label-based approaches, while potentially more specific, may still suffer from non-specific interactions between labels and non-target molecules. Developing effective surface chemistries and blocking strategies to minimize these effects remains an ongoing challenge in the field.
Existing Solutions
01 Integrated photonic readout platforms
Integrated photonic platforms are used for readout applications, combining optical and electronic components on a single chip. These platforms can enhance sensitivity, reduce noise, and improve overall performance in various sensing and measurement systems.- Integrated photonic readout platforms: Integrated photonic platforms are used for readout applications, combining optical and electronic components on a single chip. These platforms can enhance sensitivity, reduce noise, and improve overall performance in various sensing and detection systems.
- Optical waveguide-based readout systems: Optical waveguides are utilized in photonic platforms for efficient light transmission and signal readout. These systems can be designed for specific wavelengths and can be integrated with other optical components for enhanced functionality.
- Photonic crystal-based readout devices: Photonic crystals are employed in readout platforms to manipulate light propagation and enhance sensitivity. These structures can be tailored to specific applications, such as biosensing or spectroscopy, by controlling their optical properties.
- Plasmonic readout techniques: Plasmonic effects are utilized in photonic readout platforms to enhance light-matter interactions and improve sensitivity. These techniques can be applied to various sensing applications, including chemical and biological detection.
- Quantum photonic readout systems: Quantum photonic platforms are developed for high-precision readout applications. These systems leverage quantum effects to achieve enhanced sensitivity and resolution, particularly in areas such as quantum sensing and quantum information processing.
02 Optical waveguide-based readout systems
Optical waveguides are utilized in photonic platforms for efficient light transmission and signal readout. These systems can be designed for specific wavelengths and can be integrated with other optical components for enhanced functionality.Expand Specific Solutions03 Photonic crystal-based sensing and readout
Photonic crystals are employed in readout platforms to manipulate light propagation and enhance sensitivity. These structures can be tailored for specific applications, such as biosensing or environmental monitoring, by controlling their optical properties.Expand Specific Solutions04 Plasmonic readout techniques
Plasmonic structures are incorporated into photonic platforms to enhance light-matter interactions and improve readout sensitivity. These techniques can be particularly useful for detecting small molecules or low-concentration analytes in various applications.Expand Specific Solutions05 Multi-modal photonic readout systems
Multi-modal photonic platforms combine different readout techniques, such as fluorescence, Raman spectroscopy, and interferometry, to provide comprehensive and versatile sensing capabilities. These systems can offer improved accuracy and broader applicability in complex environments.Expand Specific Solutions
Key Industry Players
The competition landscape for label-free vs label-based readouts on photonic platforms is evolving rapidly. The industry is in a growth phase, with increasing market size driven by applications in drug discovery, diagnostics, and biosensing. Technological maturity varies, with label-free methods gaining traction due to their simplicity and real-time capabilities. Companies like Novartis, Corning, and Siemens are investing heavily in both approaches, while specialized firms such as X-Body and SRU Biosystems focus on innovative label-free technologies. Academic institutions like Cornell University and Xidian University are also contributing to advancements in this field, indicating a dynamic ecosystem of established players and emerging technologies.
SRU Biosystems, Inc.
Technical Solution: SRU Biosystems specializes in label-free optical biosensing technologies, particularly their proprietary BIND® (Biomolecular Interaction Detection) platform. This technology uses photonic crystal biosensors to detect changes in the resonant reflected wavelength of light as biomolecules bind to the sensor surface[8]. The BIND® system allows for high-sensitivity, real-time monitoring of molecular interactions without the need for labels. SRU has also developed methods to enhance their label-free platform with optional label-based detection, such as fluorescence amplification techniques, providing a hybrid approach that can increase sensitivity for certain applications[9]. This flexibility allows users to optimize their assays based on specific experimental requirements.
Strengths: High sensitivity in label-free mode, flexibility to incorporate label-based methods, applicable to a wide range of biomolecular interactions. Weaknesses: May require specialized expertise for optimal use, potential limitations in multiplexing compared to some label-based methods.
Novartis AG
Technical Solution: Novartis has invested in both label-free and label-based photonic platforms for drug discovery and development. Their label-free approach utilizes surface plasmon resonance (SPR) and biolayer interferometry (BLI) for real-time, quantitative analysis of biomolecular interactions[6]. For label-based methods, Novartis employs fluorescence-based microarray technologies and time-resolved fluorescence resonance energy transfer (TR-FRET) assays[7]. The company has developed integrated workflows that combine these technologies, allowing for initial high-throughput screening using label-based methods followed by detailed kinetic analysis with label-free techniques. This approach maximizes the strengths of both methodologies in the drug discovery pipeline.
Strengths: Comprehensive approach integrating multiple technologies, strong application in drug discovery. Weaknesses: High initial investment costs, complexity in data interpretation across platforms.
Core Innovations
Label-free methods for performing assays using a colorimetric resonant reflectance optical biosensor
PatentInactiveUS7312090B2
Innovation
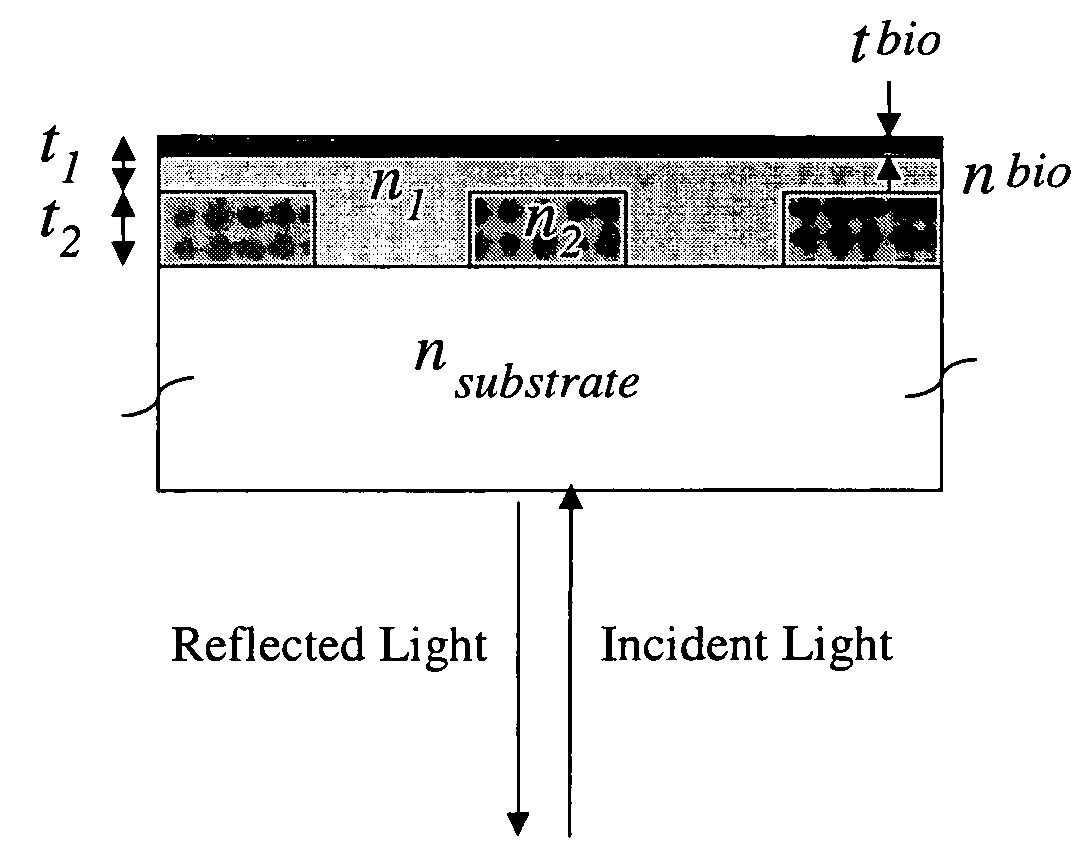
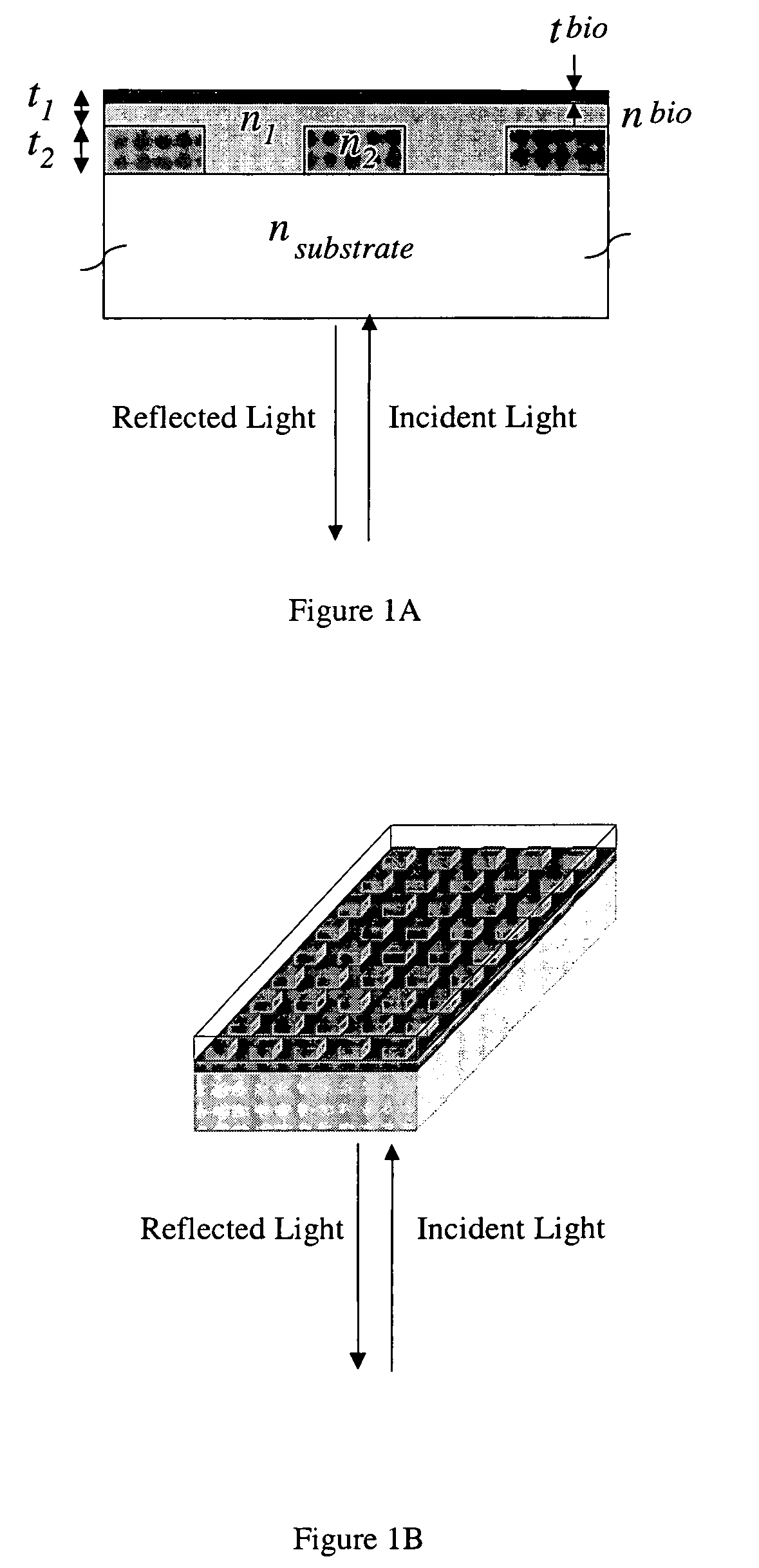
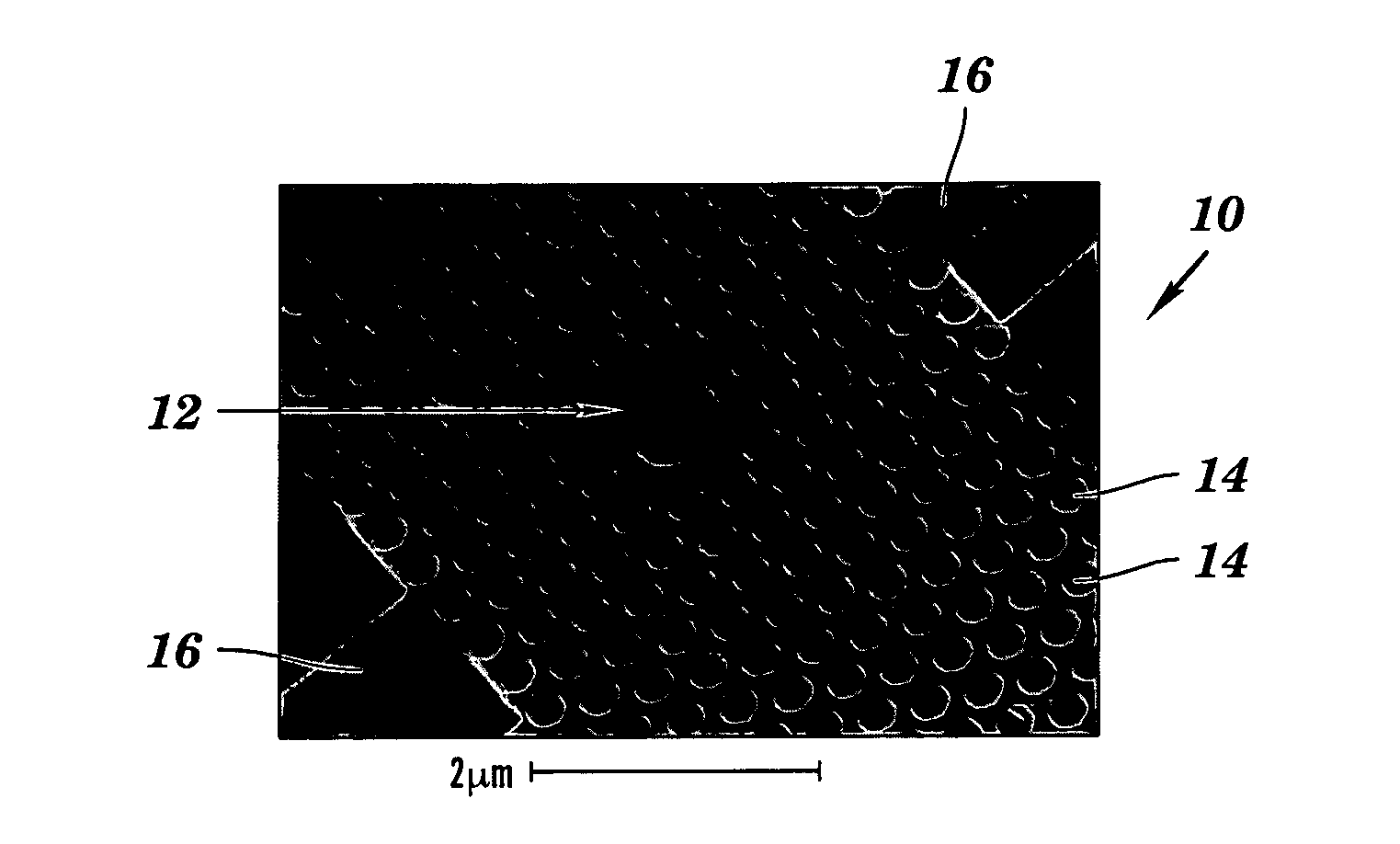
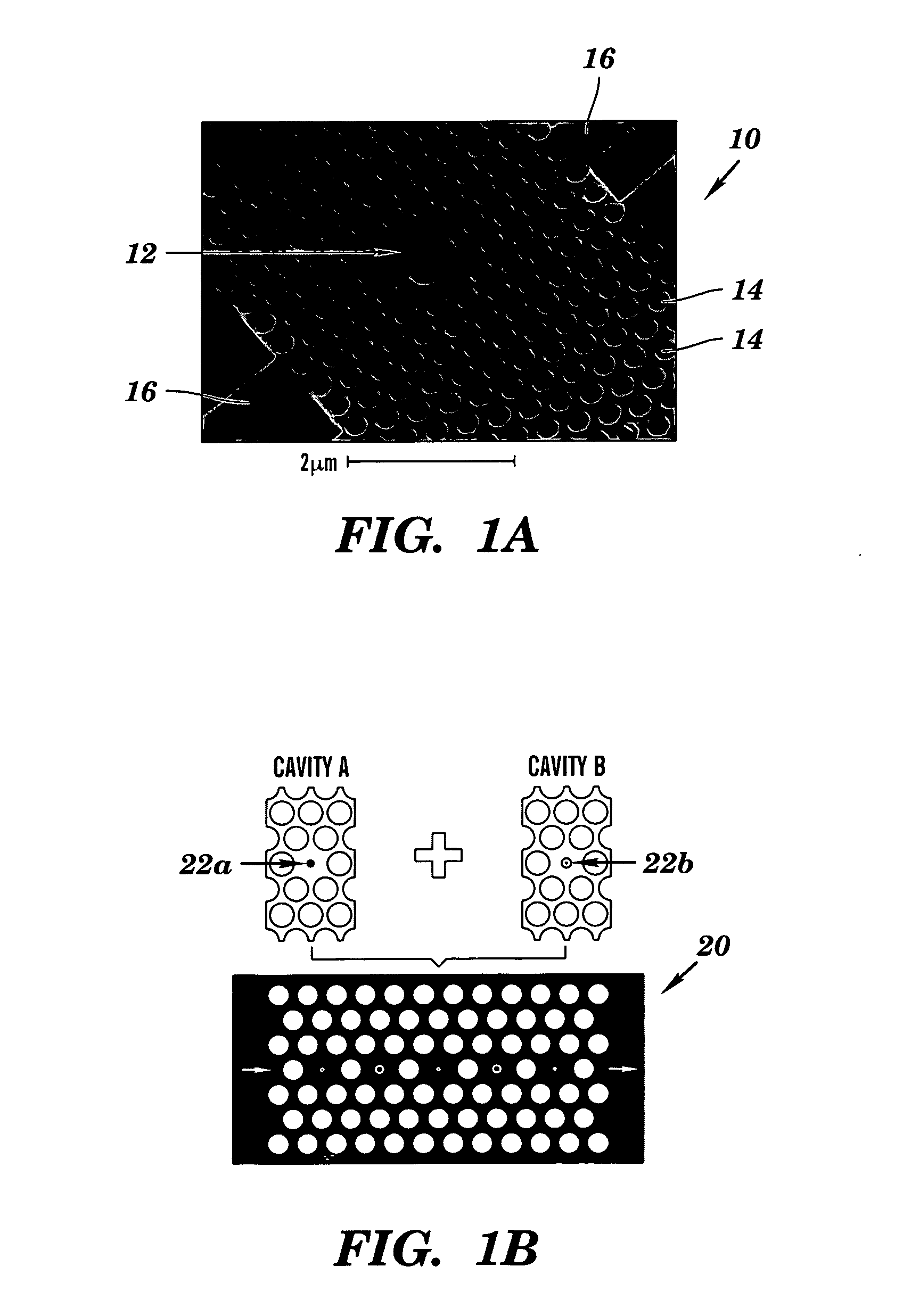
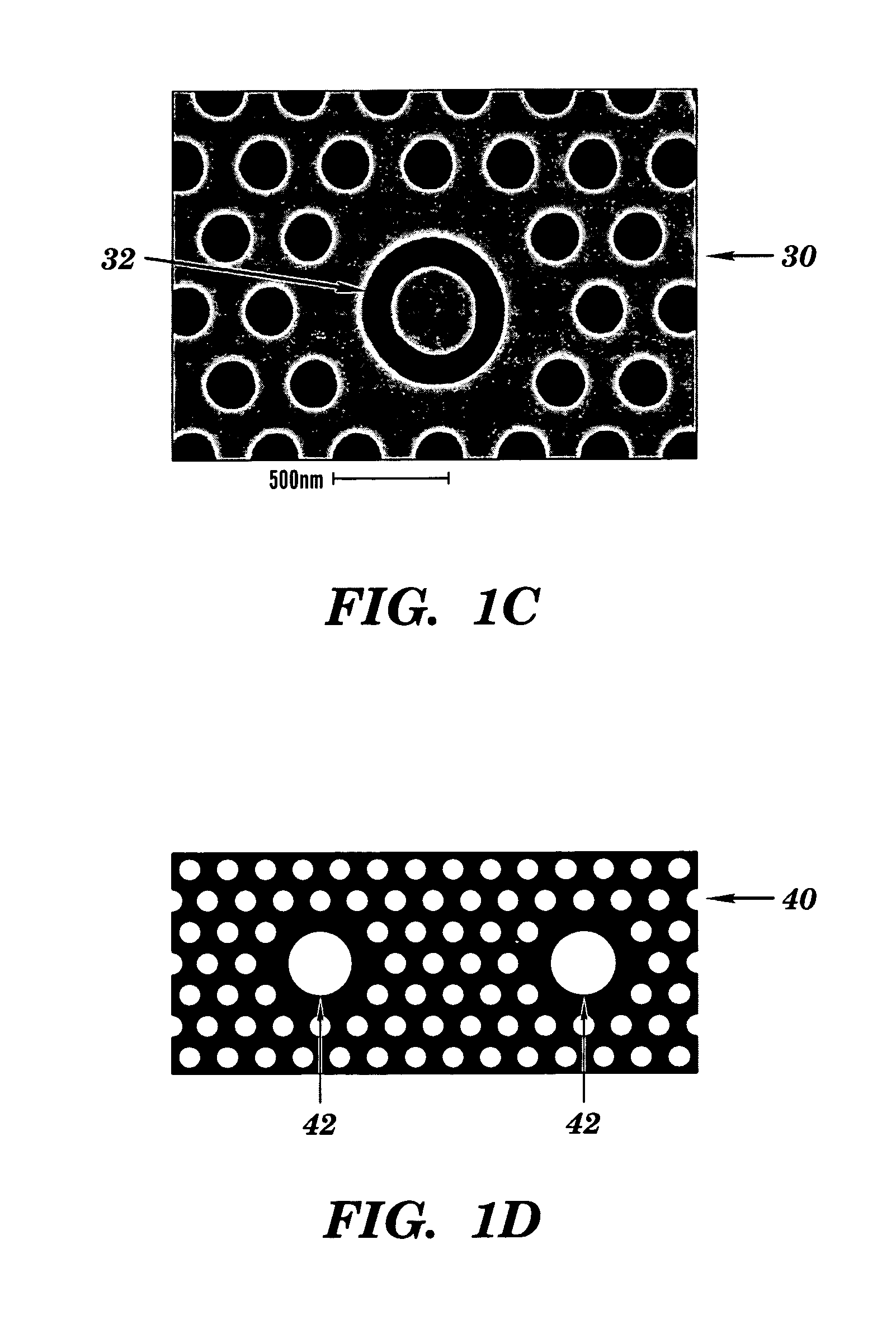
- A colorimetric resonant reflectance optical biosensor technology that allows for the detection of binding or cleavage of specific binding substances without labels, utilizing a surface with immobilized specific binding substances and cleaving molecules, and measuring peak wavelength values to detect interactions or changes on the biosensor surface.
Two-dimensional photonic bandgap structures for ultrahigh-sensitivity biosensing
PatentInactiveUS20100279886A1
Innovation
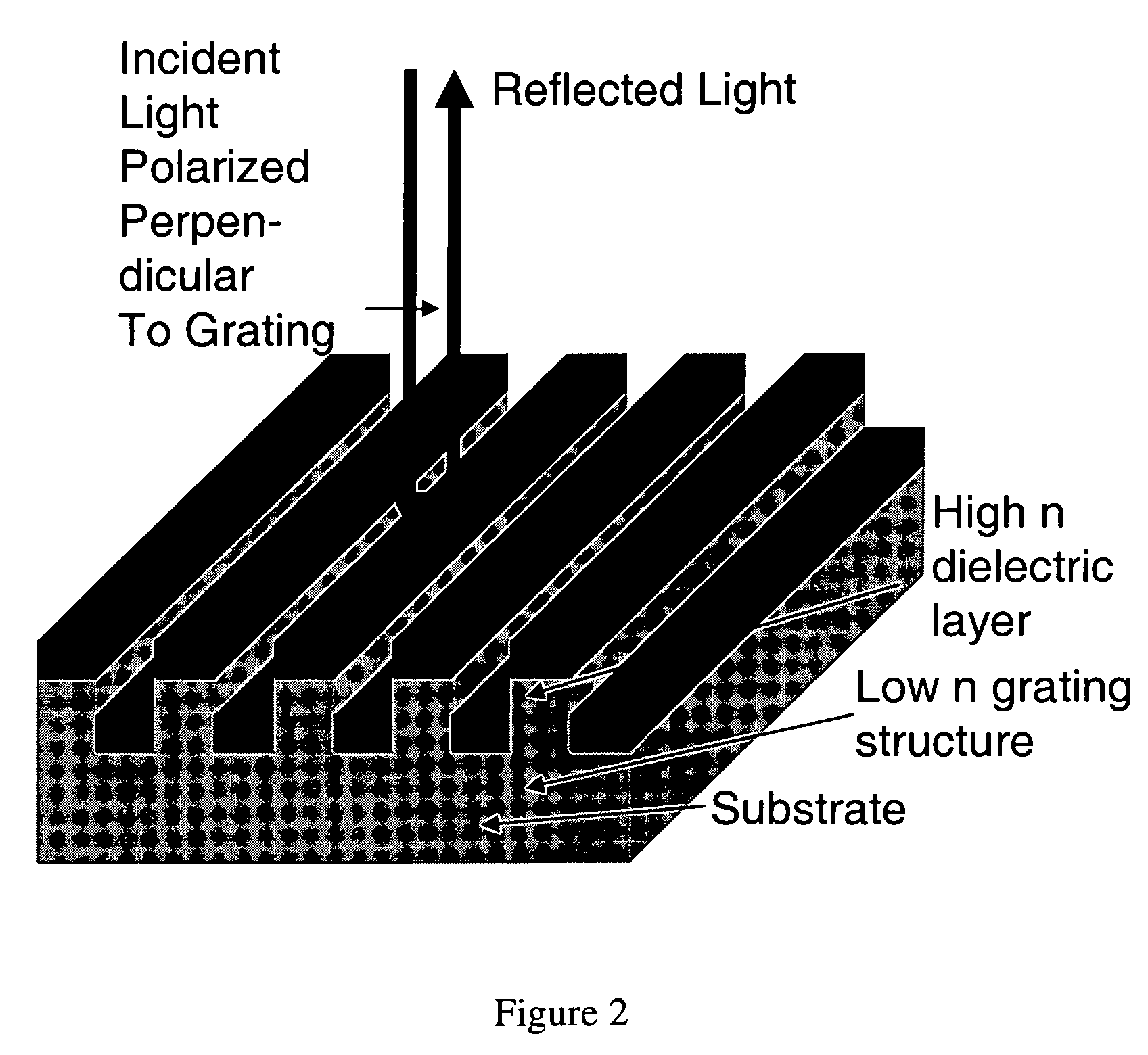
- A two-dimensional photonic crystal biosensor chip with a substrate having a lattice array of aligned pores and central defects, where probes bound to the substrate surface can detect refractive index changes caused by target binding, allowing for sensitive detection of biological targets.
Regulatory Considerations
Regulatory considerations play a crucial role in the development and implementation of label-free and label-based readouts on photonic platforms. These considerations are essential to ensure the safety, efficacy, and reliability of diagnostic and analytical tools in various fields, including healthcare, environmental monitoring, and food safety.
For label-free readouts, regulatory bodies often focus on the sensitivity and specificity of the detection methods. As these techniques do not rely on additional markers or labels, there is a heightened emphasis on validating the accuracy of direct measurements. Regulatory agencies may require extensive validation studies to demonstrate the robustness of label-free detection across different sample types and environmental conditions.
In contrast, label-based readouts face scrutiny regarding the safety and stability of the labels used. Regulatory bodies typically require comprehensive data on the biocompatibility of fluorescent dyes or other labeling agents, especially for in vivo applications. Additionally, the potential for label-induced artifacts or interference with the target analyte must be thoroughly addressed to meet regulatory standards.
Both approaches must adhere to good laboratory practices (GLP) and good manufacturing practices (GMP) guidelines. These standards ensure the consistency and reliability of results, which is particularly critical for diagnostic applications. Regulatory bodies such as the FDA in the United States or the EMA in Europe may require specific performance benchmarks and quality control measures for both label-free and label-based systems.
The choice between label-free and label-based readouts can also impact the regulatory pathway. Label-free methods may face fewer hurdles in terms of reagent approval but may require more extensive validation of the detection principle. Conversely, label-based methods might benefit from established regulatory frameworks for certain labels but may need additional approvals for novel labeling compounds.
Regulatory considerations also extend to data management and interpretation. Both approaches must comply with data integrity guidelines, ensuring that results are traceable, reproducible, and secure. This is particularly important for applications in clinical diagnostics or forensic analysis, where the reliability of results can have significant legal and ethical implications.
As the field of photonic platforms continues to evolve, regulatory bodies are adapting their guidelines to keep pace with technological advancements. This dynamic regulatory landscape requires developers to maintain ongoing dialogue with regulatory agencies to ensure compliance throughout the development and commercialization process.
For label-free readouts, regulatory bodies often focus on the sensitivity and specificity of the detection methods. As these techniques do not rely on additional markers or labels, there is a heightened emphasis on validating the accuracy of direct measurements. Regulatory agencies may require extensive validation studies to demonstrate the robustness of label-free detection across different sample types and environmental conditions.
In contrast, label-based readouts face scrutiny regarding the safety and stability of the labels used. Regulatory bodies typically require comprehensive data on the biocompatibility of fluorescent dyes or other labeling agents, especially for in vivo applications. Additionally, the potential for label-induced artifacts or interference with the target analyte must be thoroughly addressed to meet regulatory standards.
Both approaches must adhere to good laboratory practices (GLP) and good manufacturing practices (GMP) guidelines. These standards ensure the consistency and reliability of results, which is particularly critical for diagnostic applications. Regulatory bodies such as the FDA in the United States or the EMA in Europe may require specific performance benchmarks and quality control measures for both label-free and label-based systems.
The choice between label-free and label-based readouts can also impact the regulatory pathway. Label-free methods may face fewer hurdles in terms of reagent approval but may require more extensive validation of the detection principle. Conversely, label-based methods might benefit from established regulatory frameworks for certain labels but may need additional approvals for novel labeling compounds.
Regulatory considerations also extend to data management and interpretation. Both approaches must comply with data integrity guidelines, ensuring that results are traceable, reproducible, and secure. This is particularly important for applications in clinical diagnostics or forensic analysis, where the reliability of results can have significant legal and ethical implications.
As the field of photonic platforms continues to evolve, regulatory bodies are adapting their guidelines to keep pace with technological advancements. This dynamic regulatory landscape requires developers to maintain ongoing dialogue with regulatory agencies to ensure compliance throughout the development and commercialization process.
Cost-Benefit Analysis
When considering the choice between label-free and label-based readouts on photonic platforms, a comprehensive cost-benefit analysis is crucial for making informed decisions. This analysis encompasses various factors, including financial considerations, technical performance, and operational efficiency.
From a financial perspective, label-free systems generally have lower ongoing costs due to the absence of expensive labeling reagents. The initial investment in label-free platforms may be higher, but the reduced consumable expenses over time can lead to significant long-term savings. Conversely, label-based systems often have lower upfront costs but require continuous investment in specific labels and reagents.
Technical performance is another critical aspect to consider. Label-free methods offer real-time, continuous monitoring capabilities, which can be advantageous for studying dynamic biological processes. They also maintain the native state of molecules, potentially providing more physiologically relevant data. However, label-based techniques often offer higher sensitivity and specificity, particularly when dealing with complex sample matrices or low-abundance analytes.
Operational efficiency is a key consideration in the cost-benefit analysis. Label-free systems typically require less sample preparation time and fewer handling steps, potentially increasing throughput and reducing labor costs. Label-based methods, while more time-consuming in terms of sample preparation, often provide more straightforward data interpretation due to their high specificity.
The choice between these approaches also impacts experimental design and data quality. Label-free methods allow for non-destructive, repeated measurements on the same sample, which can be beneficial for longitudinal studies. Label-based techniques, however, may offer multiplexing capabilities, enabling the simultaneous detection of multiple analytes, which can be more cost-effective and time-efficient in certain research scenarios.
Considering the long-term implications, label-free systems may offer greater flexibility and adaptability to different types of biomolecular interactions, potentially extending the lifespan and utility of the investment. Label-based systems, while more specific, may require updates or replacements as new targets of interest emerge.
In conclusion, the cost-benefit analysis of label-free versus label-based readouts on photonic platforms reveals a complex interplay of factors. The optimal choice depends on the specific research needs, budget constraints, and long-term objectives of the user. A thorough evaluation of these aspects is essential to maximize the return on investment and ensure the most suitable technology is employed for the intended applications.
From a financial perspective, label-free systems generally have lower ongoing costs due to the absence of expensive labeling reagents. The initial investment in label-free platforms may be higher, but the reduced consumable expenses over time can lead to significant long-term savings. Conversely, label-based systems often have lower upfront costs but require continuous investment in specific labels and reagents.
Technical performance is another critical aspect to consider. Label-free methods offer real-time, continuous monitoring capabilities, which can be advantageous for studying dynamic biological processes. They also maintain the native state of molecules, potentially providing more physiologically relevant data. However, label-based techniques often offer higher sensitivity and specificity, particularly when dealing with complex sample matrices or low-abundance analytes.
Operational efficiency is a key consideration in the cost-benefit analysis. Label-free systems typically require less sample preparation time and fewer handling steps, potentially increasing throughput and reducing labor costs. Label-based methods, while more time-consuming in terms of sample preparation, often provide more straightforward data interpretation due to their high specificity.
The choice between these approaches also impacts experimental design and data quality. Label-free methods allow for non-destructive, repeated measurements on the same sample, which can be beneficial for longitudinal studies. Label-based techniques, however, may offer multiplexing capabilities, enabling the simultaneous detection of multiple analytes, which can be more cost-effective and time-efficient in certain research scenarios.
Considering the long-term implications, label-free systems may offer greater flexibility and adaptability to different types of biomolecular interactions, potentially extending the lifespan and utility of the investment. Label-based systems, while more specific, may require updates or replacements as new targets of interest emerge.
In conclusion, the cost-benefit analysis of label-free versus label-based readouts on photonic platforms reveals a complex interplay of factors. The optimal choice depends on the specific research needs, budget constraints, and long-term objectives of the user. A thorough evaluation of these aspects is essential to maximize the return on investment and ensure the most suitable technology is employed for the intended applications.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!