Surface Functionalization Strategies for Photonic Biosensors: Chemistries and Stability Tests
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Photonic Biosensor Functionalization Background
Photonic biosensors have emerged as powerful tools for detecting and analyzing biological and chemical substances with high sensitivity and specificity. These sensors leverage the interaction between light and matter to detect changes in the surrounding environment, making them ideal for a wide range of applications in healthcare, environmental monitoring, and food safety. The foundation of photonic biosensor technology lies in the manipulation of light at the nanoscale, often utilizing phenomena such as surface plasmon resonance, photonic crystals, or optical waveguides.
The concept of photonic biosensors dates back to the 1980s, with significant advancements occurring in the 1990s and early 2000s. The development of these sensors has been driven by the need for rapid, label-free, and real-time detection methods in various fields. As the technology progressed, researchers focused on improving sensor sensitivity, selectivity, and miniaturization, leading to the creation of more sophisticated and versatile devices.
Surface functionalization plays a crucial role in the performance and applicability of photonic biosensors. This process involves modifying the sensor surface to enable specific and efficient binding of target molecules. The evolution of surface functionalization strategies has been closely tied to advancements in materials science, chemistry, and nanotechnology. Early approaches relied on simple physical adsorption or covalent attachment of biomolecules to the sensor surface. However, these methods often suffered from limitations such as non-specific binding and poor stability.
Over time, more advanced functionalization techniques have been developed to address these challenges. These include the use of self-assembled monolayers (SAMs), polymer brushes, and various bioconjugation chemistries. Each of these approaches aims to create a stable and well-defined interface between the sensor surface and the biological recognition elements, such as antibodies, aptamers, or enzymes.
The stability of the functionalized surface is a critical factor in the performance and longevity of photonic biosensors. Researchers have devoted considerable effort to developing robust functionalization strategies that can withstand various environmental conditions, including changes in pH, temperature, and ionic strength. This focus on stability has led to the exploration of novel materials and surface modification techniques, as well as the development of standardized testing protocols to evaluate the durability of functionalized surfaces.
As the field of photonic biosensors continues to advance, surface functionalization remains a key area of research and development. Current efforts are focused on creating multifunctional surfaces that can enhance sensor performance while also addressing challenges such as non-specific binding and fouling. The integration of stimuli-responsive materials and smart coatings is also being explored to enable dynamic control over surface properties and expand the capabilities of photonic biosensors.
The concept of photonic biosensors dates back to the 1980s, with significant advancements occurring in the 1990s and early 2000s. The development of these sensors has been driven by the need for rapid, label-free, and real-time detection methods in various fields. As the technology progressed, researchers focused on improving sensor sensitivity, selectivity, and miniaturization, leading to the creation of more sophisticated and versatile devices.
Surface functionalization plays a crucial role in the performance and applicability of photonic biosensors. This process involves modifying the sensor surface to enable specific and efficient binding of target molecules. The evolution of surface functionalization strategies has been closely tied to advancements in materials science, chemistry, and nanotechnology. Early approaches relied on simple physical adsorption or covalent attachment of biomolecules to the sensor surface. However, these methods often suffered from limitations such as non-specific binding and poor stability.
Over time, more advanced functionalization techniques have been developed to address these challenges. These include the use of self-assembled monolayers (SAMs), polymer brushes, and various bioconjugation chemistries. Each of these approaches aims to create a stable and well-defined interface between the sensor surface and the biological recognition elements, such as antibodies, aptamers, or enzymes.
The stability of the functionalized surface is a critical factor in the performance and longevity of photonic biosensors. Researchers have devoted considerable effort to developing robust functionalization strategies that can withstand various environmental conditions, including changes in pH, temperature, and ionic strength. This focus on stability has led to the exploration of novel materials and surface modification techniques, as well as the development of standardized testing protocols to evaluate the durability of functionalized surfaces.
As the field of photonic biosensors continues to advance, surface functionalization remains a key area of research and development. Current efforts are focused on creating multifunctional surfaces that can enhance sensor performance while also addressing challenges such as non-specific binding and fouling. The integration of stimuli-responsive materials and smart coatings is also being explored to enable dynamic control over surface properties and expand the capabilities of photonic biosensors.
Market Analysis for Functionalized Biosensors
The market for functionalized biosensors has experienced significant growth in recent years, driven by increasing demand for rapid, sensitive, and specific detection methods across various industries. The global biosensors market, which includes functionalized biosensors, was valued at approximately $21 billion in 2020 and is projected to reach $33 billion by 2026, with a compound annual growth rate (CAGR) of around 7.5%.
Surface functionalization strategies for photonic biosensors play a crucial role in this market expansion. These strategies enhance the sensitivity, selectivity, and stability of biosensors, making them more attractive for applications in healthcare, environmental monitoring, food safety, and biodefense. The healthcare sector, in particular, has been a major driver of market growth, with increasing adoption of biosensors for point-of-care diagnostics, continuous glucose monitoring, and drug discovery.
The market for functionalized photonic biosensors is characterized by a high degree of innovation and technological advancement. Key players in this space are continuously developing new surface functionalization chemistries and stability testing methods to improve sensor performance and reliability. This ongoing research and development activity is expected to fuel market growth and create new opportunities for both established companies and emerging startups.
Geographically, North America and Europe currently dominate the market for functionalized biosensors, owing to their advanced healthcare infrastructure, strong research and development capabilities, and favorable regulatory environments. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by increasing healthcare expenditure, rising awareness about early disease diagnosis, and growing investment in biotechnology research.
The COVID-19 pandemic has further accelerated market growth, highlighting the importance of rapid and accurate diagnostic tools. This has led to increased funding and research efforts in the development of biosensors for virus detection, creating new opportunities for surface functionalization technologies.
Despite the positive market outlook, challenges remain. These include the high cost of advanced biosensors, stringent regulatory requirements, and the need for skilled personnel to operate and interpret results from these devices. Addressing these challenges will be crucial for sustained market growth and wider adoption of functionalized photonic biosensors across various industries.
Surface functionalization strategies for photonic biosensors play a crucial role in this market expansion. These strategies enhance the sensitivity, selectivity, and stability of biosensors, making them more attractive for applications in healthcare, environmental monitoring, food safety, and biodefense. The healthcare sector, in particular, has been a major driver of market growth, with increasing adoption of biosensors for point-of-care diagnostics, continuous glucose monitoring, and drug discovery.
The market for functionalized photonic biosensors is characterized by a high degree of innovation and technological advancement. Key players in this space are continuously developing new surface functionalization chemistries and stability testing methods to improve sensor performance and reliability. This ongoing research and development activity is expected to fuel market growth and create new opportunities for both established companies and emerging startups.
Geographically, North America and Europe currently dominate the market for functionalized biosensors, owing to their advanced healthcare infrastructure, strong research and development capabilities, and favorable regulatory environments. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by increasing healthcare expenditure, rising awareness about early disease diagnosis, and growing investment in biotechnology research.
The COVID-19 pandemic has further accelerated market growth, highlighting the importance of rapid and accurate diagnostic tools. This has led to increased funding and research efforts in the development of biosensors for virus detection, creating new opportunities for surface functionalization technologies.
Despite the positive market outlook, challenges remain. These include the high cost of advanced biosensors, stringent regulatory requirements, and the need for skilled personnel to operate and interpret results from these devices. Addressing these challenges will be crucial for sustained market growth and wider adoption of functionalized photonic biosensors across various industries.
Current Challenges in Surface Chemistry
Surface functionalization is a critical aspect of photonic biosensor development, yet it presents several significant challenges in the realm of surface chemistry. One of the primary issues is achieving uniform and reproducible surface modifications across different batches of sensors. The heterogeneity of surface properties, even within a single batch, can lead to inconsistent sensor performance and reduced reliability in biosensing applications.
Another major challenge lies in maintaining the stability of the functionalized surface over time and under various environmental conditions. Many surface modifications are susceptible to degradation due to factors such as pH changes, temperature fluctuations, and exposure to biological fluids. This instability can result in a loss of sensor sensitivity and specificity, limiting the long-term usability of the biosensor devices.
The complexity of biomolecule immobilization on sensor surfaces also poses significant hurdles. Ensuring proper orientation and density of capture molecules, such as antibodies or aptamers, is crucial for optimal biosensor performance. However, controlling these parameters precisely during the functionalization process remains challenging, often leading to suboptimal sensor responses and reduced detection limits.
Non-specific binding is another persistent issue in surface chemistry for photonic biosensors. Despite advances in surface passivation techniques, completely eliminating unwanted interactions between the sensor surface and non-target molecules in complex biological samples remains elusive. This can result in false positive signals and decreased sensor specificity, particularly in real-world applications where sample matrices are complex and variable.
The development of multifunctional surfaces that can simultaneously perform multiple tasks, such as analyte capture, signal enhancement, and anti-fouling, presents additional challenges. Integrating these diverse functionalities while maintaining the overall performance and stability of the sensor surface requires sophisticated chemical strategies and often involves trade-offs between different desired properties.
Scalability and cost-effectiveness of surface functionalization processes also remain significant hurdles, particularly for commercial applications. Many current functionalization methods are labor-intensive, time-consuming, or require expensive reagents, making large-scale production of photonic biosensors challenging from both economic and practical standpoints.
Lastly, the environmental impact and biocompatibility of surface modification chemistries are becoming increasingly important considerations. Developing green chemistry approaches for surface functionalization that minimize the use of hazardous reagents and reduce waste generation, while still achieving the desired surface properties, is an ongoing challenge in the field.
Another major challenge lies in maintaining the stability of the functionalized surface over time and under various environmental conditions. Many surface modifications are susceptible to degradation due to factors such as pH changes, temperature fluctuations, and exposure to biological fluids. This instability can result in a loss of sensor sensitivity and specificity, limiting the long-term usability of the biosensor devices.
The complexity of biomolecule immobilization on sensor surfaces also poses significant hurdles. Ensuring proper orientation and density of capture molecules, such as antibodies or aptamers, is crucial for optimal biosensor performance. However, controlling these parameters precisely during the functionalization process remains challenging, often leading to suboptimal sensor responses and reduced detection limits.
Non-specific binding is another persistent issue in surface chemistry for photonic biosensors. Despite advances in surface passivation techniques, completely eliminating unwanted interactions between the sensor surface and non-target molecules in complex biological samples remains elusive. This can result in false positive signals and decreased sensor specificity, particularly in real-world applications where sample matrices are complex and variable.
The development of multifunctional surfaces that can simultaneously perform multiple tasks, such as analyte capture, signal enhancement, and anti-fouling, presents additional challenges. Integrating these diverse functionalities while maintaining the overall performance and stability of the sensor surface requires sophisticated chemical strategies and often involves trade-offs between different desired properties.
Scalability and cost-effectiveness of surface functionalization processes also remain significant hurdles, particularly for commercial applications. Many current functionalization methods are labor-intensive, time-consuming, or require expensive reagents, making large-scale production of photonic biosensors challenging from both economic and practical standpoints.
Lastly, the environmental impact and biocompatibility of surface modification chemistries are becoming increasingly important considerations. Developing green chemistry approaches for surface functionalization that minimize the use of hazardous reagents and reduce waste generation, while still achieving the desired surface properties, is an ongoing challenge in the field.
Existing Surface Modification Techniques
01 Surface modification techniques for biosensors
Various surface modification techniques are employed to functionalize photonic biosensors. These methods enhance the sensitivity and specificity of the biosensors by creating a suitable interface for biomolecule attachment. Common techniques include chemical modification, plasma treatment, and self-assembled monolayers, which allow for the immobilization of specific recognition elements on the sensor surface.- Surface functionalization techniques for photonic biosensors: Various surface functionalization techniques are employed to enhance the sensitivity and specificity of photonic biosensors. These methods involve modifying the sensor surface with specific biomolecules or chemical groups to enable selective binding of target analytes. Common approaches include covalent attachment, physical adsorption, and self-assembled monolayers, which can be tailored to different types of biosensors and target molecules.
- Integration of nanomaterials for improved biosensor performance: Nanomaterials such as nanoparticles, quantum dots, and carbon nanotubes are incorporated into photonic biosensors to enhance their performance. These materials can increase the surface area for biomolecule immobilization, improve light-matter interactions, and provide unique optical properties. The integration of nanomaterials often results in higher sensitivity, lower detection limits, and improved signal-to-noise ratios in biosensing applications.
- Novel photonic structures for biosensing: Advanced photonic structures are developed to improve the performance of biosensors. These include photonic crystals, plasmonic nanostructures, and resonant cavities. Such structures can enhance light-analyte interactions, leading to improved sensitivity and detection limits. The design and fabrication of these structures often involve precise control of material properties and geometries to achieve optimal biosensing performance.
- Microfluidic integration for sample handling and analysis: Microfluidic systems are integrated with photonic biosensors to facilitate sample handling, reduce sample volume requirements, and enable multiplexed analysis. These systems can incorporate various functions such as sample preparation, analyte concentration, and waste removal. The combination of microfluidics and photonic sensing enables the development of lab-on-a-chip devices for point-of-care diagnostics and high-throughput screening applications.
- Signal processing and data analysis for improved biosensor performance: Advanced signal processing and data analysis techniques are employed to enhance the performance of photonic biosensors. These methods can include noise reduction algorithms, pattern recognition, and machine learning approaches. By implementing sophisticated data processing techniques, researchers can improve the sensitivity, specificity, and reliability of biosensor measurements, enabling more accurate and robust detection of target analytes.
02 Nanostructured surfaces for improved sensitivity
Nanostructured surfaces are utilized to increase the surface area and enhance the sensitivity of photonic biosensors. These structures can include nanoparticles, nanopillars, or nanogrooves, which provide a larger number of binding sites for target molecules. The increased surface area leads to improved signal-to-noise ratios and lower detection limits in biosensing applications.Expand Specific Solutions03 Biomolecule immobilization strategies
Various strategies are employed for immobilizing biomolecules on photonic biosensor surfaces. These include covalent attachment, physical adsorption, and affinity-based immobilization. The choice of immobilization method depends on the nature of the biomolecule and the desired sensor performance. Proper immobilization ensures optimal orientation and activity of the recognition elements.Expand Specific Solutions04 Integration of microfluidics with surface functionalization
Microfluidic systems are integrated with surface functionalization techniques to enhance the performance of photonic biosensors. This combination allows for precise control over sample delivery, reagent mixing, and surface modification processes. Microfluidic channels can be designed to optimize the interaction between analytes and functionalized surfaces, improving sensor efficiency and reproducibility.Expand Specific Solutions05 Multi-analyte detection through surface patterning
Surface patterning techniques are used to create multiple sensing regions on a single photonic biosensor platform. This approach enables the simultaneous detection of multiple analytes by functionalizing different areas with specific recognition elements. Patterning methods include photolithography, microcontact printing, and inkjet printing, allowing for the development of multiplexed biosensing devices.Expand Specific Solutions
Key Players in Biosensor Industry
The field of surface functionalization for photonic biosensors is in a growth phase, with increasing market size and technological advancements. The global biosensors market, valued at $21.2 billion in 2020, is projected to reach $38.7 billion by 2027, driven by healthcare and environmental monitoring applications. Key players like FUJIFILM Corp., Element Biosciences, and InSilixa are advancing the technology, focusing on improving sensor sensitivity, stability, and biocompatibility. Academic institutions such as Duke University and the University of Illinois are contributing to fundamental research, while companies like SRU Biosystems and X-Body are developing innovative commercial applications. The technology is maturing rapidly, with a focus on enhancing surface chemistry, improving signal-to-noise ratios, and expanding the range of detectable biomolecules.
Duke University
Technical Solution: Duke University has made significant contributions to surface functionalization strategies for photonic biosensors, particularly in the development of nanoplasmonic sensors. Their research focuses on creating stable and biocompatible surface chemistries that can withstand harsh environments while maintaining high sensitivity. One of their key innovations is the use of mixed self-assembled monolayers (SAMs) incorporating both functional and passivating molecules to reduce non-specific binding and improve sensor stability [4]. Duke researchers have also explored the use of zwitterionic polymers for antifouling coatings, which significantly enhance the long-term stability and performance of biosensors in complex biological media [5]. Additionally, they have developed novel click chemistry approaches for the attachment of biomolecules to sensor surfaces, allowing for rapid and efficient functionalization with a wide range of recognition elements [6].
Strengths: Enhanced stability in harsh environments, reduced non-specific binding, improved long-term performance. Weaknesses: Potential limitations in sensitivity compared to less stable but more responsive surface chemistries.
SRU Biosystems, Inc.
Technical Solution: SRU Biosystems has developed proprietary surface functionalization strategies for their label-free photonic crystal biosensor technology. Their approach focuses on creating high-density, uniform protein-binding surfaces that maximize sensitivity while minimizing non-specific interactions. SRU's technology utilizes a combination of hydrogel-based coatings and specific linker chemistries to immobilize capture molecules on the sensor surface [7]. The company has also developed methods for patterning multiple distinct capture regions on a single sensor surface, enabling multiplexed detection of various analytes simultaneously [8]. SRU's surface functionalization protocols are designed to be compatible with automated liquid handling systems, facilitating high-throughput screening applications in drug discovery and life science research [9].
Strengths: High-density, uniform protein-binding surfaces; multiplexing capabilities; compatibility with automated systems. Weaknesses: Potential limitations in customization for specific research applications compared to academic solutions.
Innovative Chemistries for Biosensor Surfaces
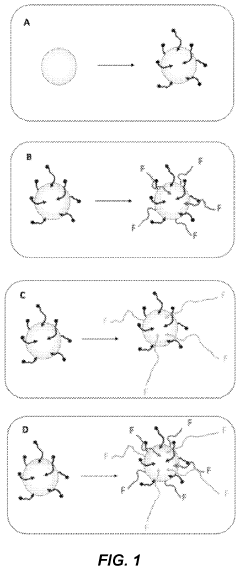
Nanoparticles pre-functionalized using a self-assembled monolayer and method for preparing same
PatentActiveUS11925692B2
Innovation
- Pre-functionalization of nanoparticles using a self-assembled monolayer of low molecular weight polyethylene glycol (PEG) molecules with a partial coverage rate, which inhibits non-specific interactions and optimizes specific interactions, allowing for controlled immobilization of biomolecules and improved stability in buffer and complex media.
Low binding supports for improved solid-phase DNA hybridization and amplification
PatentWO2020102594A1
Innovation
- The development of low nonspecific binding supports with hydrophilic polymer coatings and high-density oligonucleotide attachment, enabling improved nucleic acid amplification and sequencing by increasing signal magnitude, reducing background noise, and enhancing CNR, thus improving base-calling accuracy and reducing instrumentation dependence.
Stability Testing Protocols
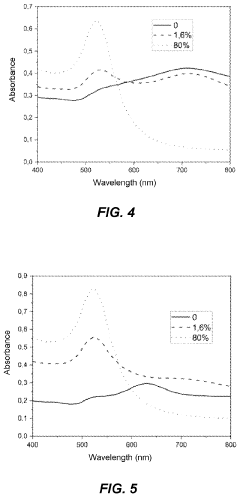
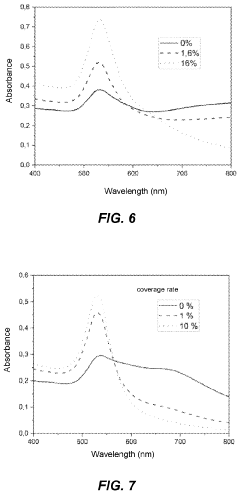
Stability testing protocols are crucial for evaluating the long-term performance and reliability of surface functionalization strategies in photonic biosensors. These protocols typically involve subjecting the functionalized surfaces to various environmental conditions and stressors to assess their durability and maintain their biorecognition capabilities over time.
One of the primary stability tests is thermal stability, which examines the sensor's performance under different temperature conditions. This test often involves exposing the functionalized surface to a range of temperatures, from sub-zero to elevated temperatures, for extended periods. The sensor's response and sensitivity are monitored throughout the process to determine any degradation in performance.
pH stability testing is another essential protocol, particularly for biosensors that may be exposed to varying pH environments in biological samples. The functionalized surface is subjected to different pH solutions, ranging from acidic to alkaline, to evaluate its ability to maintain its structural integrity and biorecognition properties across a spectrum of pH values.
Storage stability is a critical aspect of biosensor development, as it determines the shelf-life of the device. This test involves storing the functionalized sensors under controlled conditions for extended periods, typically several months to a year, and periodically assessing their performance to ensure consistent results over time.
Humidity and oxidative stress tests are also commonly employed to simulate real-world environmental conditions. These tests expose the sensors to high humidity levels and oxidizing agents to evaluate their resistance to moisture-induced degradation and oxidative damage, which can affect the stability of biomolecule immobilization.
Mechanical stability testing is particularly relevant for biosensors that may experience physical stress during handling or operation. This protocol may include subjecting the functionalized surface to controlled mechanical forces, such as shear stress or vibration, to assess its ability to withstand physical disturbances without compromising its sensing capabilities.
Chemical resistance tests are designed to evaluate the stability of the functionalized surface when exposed to various chemical agents that may be present in sample matrices or cleaning solutions. This involves immersing the sensor in different solvents, detergents, or other relevant chemicals and monitoring any changes in its performance or surface characteristics.
Lastly, biofouling resistance tests are crucial for biosensors intended for use in complex biological environments. These protocols assess the sensor's ability to maintain its specificity and sensitivity in the presence of non-specific biomolecules that may adhere to the surface and interfere with target detection.
One of the primary stability tests is thermal stability, which examines the sensor's performance under different temperature conditions. This test often involves exposing the functionalized surface to a range of temperatures, from sub-zero to elevated temperatures, for extended periods. The sensor's response and sensitivity are monitored throughout the process to determine any degradation in performance.
pH stability testing is another essential protocol, particularly for biosensors that may be exposed to varying pH environments in biological samples. The functionalized surface is subjected to different pH solutions, ranging from acidic to alkaline, to evaluate its ability to maintain its structural integrity and biorecognition properties across a spectrum of pH values.
Storage stability is a critical aspect of biosensor development, as it determines the shelf-life of the device. This test involves storing the functionalized sensors under controlled conditions for extended periods, typically several months to a year, and periodically assessing their performance to ensure consistent results over time.
Humidity and oxidative stress tests are also commonly employed to simulate real-world environmental conditions. These tests expose the sensors to high humidity levels and oxidizing agents to evaluate their resistance to moisture-induced degradation and oxidative damage, which can affect the stability of biomolecule immobilization.
Mechanical stability testing is particularly relevant for biosensors that may experience physical stress during handling or operation. This protocol may include subjecting the functionalized surface to controlled mechanical forces, such as shear stress or vibration, to assess its ability to withstand physical disturbances without compromising its sensing capabilities.
Chemical resistance tests are designed to evaluate the stability of the functionalized surface when exposed to various chemical agents that may be present in sample matrices or cleaning solutions. This involves immersing the sensor in different solvents, detergents, or other relevant chemicals and monitoring any changes in its performance or surface characteristics.
Lastly, biofouling resistance tests are crucial for biosensors intended for use in complex biological environments. These protocols assess the sensor's ability to maintain its specificity and sensitivity in the presence of non-specific biomolecules that may adhere to the surface and interfere with target detection.
Regulatory Considerations for Biosensor Development
The development and commercialization of photonic biosensors are subject to stringent regulatory oversight due to their potential impact on human health and safety. Regulatory bodies such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in Europe play crucial roles in ensuring the safety, efficacy, and quality of these devices.
For surface functionalization strategies in photonic biosensors, regulatory considerations primarily focus on the biocompatibility and stability of the materials used. Manufacturers must demonstrate that the chosen chemistries and functionalization methods do not introduce harmful substances or compromise the integrity of the biosensor over time. This often involves extensive toxicology studies and long-term stability tests.
The regulatory pathway for photonic biosensors typically depends on their intended use and level of risk. For instance, biosensors intended for diagnostic purposes may be classified as in vitro diagnostic devices (IVDs) and subject to specific regulations such as the EU's In Vitro Diagnostic Regulation (IVDR) or the FDA's regulations for medical devices.
Stability testing is a critical aspect of regulatory compliance for photonic biosensors. Manufacturers must provide data demonstrating the shelf life and operational stability of the functionalized surfaces under various environmental conditions. This includes temperature cycling, humidity exposure, and accelerated aging studies to predict long-term performance.
Quality control measures for surface functionalization processes are also scrutinized by regulatory bodies. Manufacturers are expected to implement robust quality management systems that ensure consistency and reproducibility in the functionalization process. This may involve validation of cleaning procedures, verification of surface chemistry, and ongoing monitoring of batch-to-batch variability.
Regulatory agencies also emphasize the importance of risk management throughout the development and production of photonic biosensors. This includes identifying potential failure modes related to surface functionalization and implementing appropriate mitigation strategies. Manufacturers must maintain comprehensive documentation of their risk assessment and management processes.
As the field of photonic biosensors continues to evolve, regulatory frameworks are adapting to keep pace with technological advancements. Emerging areas such as nanotechnology-based surface modifications and novel biomolecule immobilization techniques may require additional regulatory considerations. Manufacturers are encouraged to engage in early dialogue with regulatory agencies to address potential concerns and streamline the approval process.
For surface functionalization strategies in photonic biosensors, regulatory considerations primarily focus on the biocompatibility and stability of the materials used. Manufacturers must demonstrate that the chosen chemistries and functionalization methods do not introduce harmful substances or compromise the integrity of the biosensor over time. This often involves extensive toxicology studies and long-term stability tests.
The regulatory pathway for photonic biosensors typically depends on their intended use and level of risk. For instance, biosensors intended for diagnostic purposes may be classified as in vitro diagnostic devices (IVDs) and subject to specific regulations such as the EU's In Vitro Diagnostic Regulation (IVDR) or the FDA's regulations for medical devices.
Stability testing is a critical aspect of regulatory compliance for photonic biosensors. Manufacturers must provide data demonstrating the shelf life and operational stability of the functionalized surfaces under various environmental conditions. This includes temperature cycling, humidity exposure, and accelerated aging studies to predict long-term performance.
Quality control measures for surface functionalization processes are also scrutinized by regulatory bodies. Manufacturers are expected to implement robust quality management systems that ensure consistency and reproducibility in the functionalization process. This may involve validation of cleaning procedures, verification of surface chemistry, and ongoing monitoring of batch-to-batch variability.
Regulatory agencies also emphasize the importance of risk management throughout the development and production of photonic biosensors. This includes identifying potential failure modes related to surface functionalization and implementing appropriate mitigation strategies. Manufacturers must maintain comprehensive documentation of their risk assessment and management processes.
As the field of photonic biosensors continues to evolve, regulatory frameworks are adapting to keep pace with technological advancements. Emerging areas such as nanotechnology-based surface modifications and novel biomolecule immobilization techniques may require additional regulatory considerations. Manufacturers are encouraged to engage in early dialogue with regulatory agencies to address potential concerns and streamline the approval process.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!