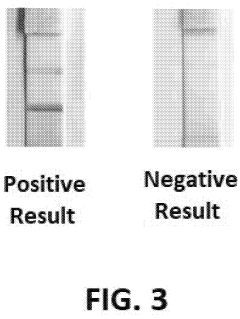
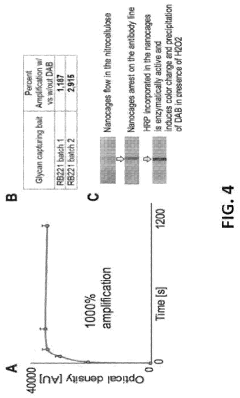
How to Validate Photonic Biosensor Performance Against ELISA/Gold Standards — Protocols
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Photonic Biosensor Validation Goals
Photonic biosensors represent a cutting-edge technology in the field of biomedical diagnostics, offering the potential for rapid, sensitive, and label-free detection of various biomolecules. The primary goal of validating photonic biosensor performance against established gold standards, such as ELISA (Enzyme-Linked Immunosorbent Assay), is to ensure the reliability, accuracy, and clinical relevance of these novel sensing platforms.
One of the key objectives is to establish the analytical performance characteristics of photonic biosensors, including sensitivity, specificity, dynamic range, and limit of detection. These parameters must be rigorously compared to those of ELISA to demonstrate the biosensor's capability to match or exceed the gold standard's performance. Additionally, the validation process aims to assess the reproducibility and robustness of photonic biosensor measurements across different experimental conditions and sample types.
Another crucial goal is to evaluate the clinical utility of photonic biosensors in real-world scenarios. This involves testing the sensors with complex biological samples, such as blood, serum, or tissue extracts, to determine their ability to accurately detect target analytes in the presence of potential interfering substances. The validation should also address the biosensor's performance in detecting clinically relevant biomarker concentrations and its capacity to distinguish between healthy and diseased states.
Developing standardized protocols for photonic biosensor validation is essential to ensure consistency and comparability across different research groups and potential commercial applications. These protocols should outline detailed procedures for sample preparation, measurement techniques, data analysis, and quality control measures. Furthermore, they should incorporate guidelines for calibration, normalization, and interpretation of results to facilitate direct comparison with ELISA outcomes.
The validation process also aims to assess the practical advantages of photonic biosensors over traditional methods. This includes evaluating factors such as assay time, sample volume requirements, multiplexing capabilities, and ease of use. Demonstrating improvements in these areas can highlight the potential of photonic biosensors to address current limitations in clinical diagnostics and point-of-care testing.
Ultimately, the overarching goal of photonic biosensor validation is to pave the way for their integration into clinical practice and regulatory approval. This requires comprehensive documentation of the validation process, including statistical analyses, method comparison studies, and inter-laboratory validations. By meeting these validation goals, photonic biosensors can establish their credibility as reliable alternatives or complementary tools to existing gold standard methods, potentially revolutionizing the field of biomedical diagnostics.
One of the key objectives is to establish the analytical performance characteristics of photonic biosensors, including sensitivity, specificity, dynamic range, and limit of detection. These parameters must be rigorously compared to those of ELISA to demonstrate the biosensor's capability to match or exceed the gold standard's performance. Additionally, the validation process aims to assess the reproducibility and robustness of photonic biosensor measurements across different experimental conditions and sample types.
Another crucial goal is to evaluate the clinical utility of photonic biosensors in real-world scenarios. This involves testing the sensors with complex biological samples, such as blood, serum, or tissue extracts, to determine their ability to accurately detect target analytes in the presence of potential interfering substances. The validation should also address the biosensor's performance in detecting clinically relevant biomarker concentrations and its capacity to distinguish between healthy and diseased states.
Developing standardized protocols for photonic biosensor validation is essential to ensure consistency and comparability across different research groups and potential commercial applications. These protocols should outline detailed procedures for sample preparation, measurement techniques, data analysis, and quality control measures. Furthermore, they should incorporate guidelines for calibration, normalization, and interpretation of results to facilitate direct comparison with ELISA outcomes.
The validation process also aims to assess the practical advantages of photonic biosensors over traditional methods. This includes evaluating factors such as assay time, sample volume requirements, multiplexing capabilities, and ease of use. Demonstrating improvements in these areas can highlight the potential of photonic biosensors to address current limitations in clinical diagnostics and point-of-care testing.
Ultimately, the overarching goal of photonic biosensor validation is to pave the way for their integration into clinical practice and regulatory approval. This requires comprehensive documentation of the validation process, including statistical analyses, method comparison studies, and inter-laboratory validations. By meeting these validation goals, photonic biosensors can establish their credibility as reliable alternatives or complementary tools to existing gold standard methods, potentially revolutionizing the field of biomedical diagnostics.
Market Demand for Biosensor Technologies
The market demand for biosensor technologies has been experiencing significant growth, driven by various factors across multiple industries. In the healthcare sector, there is an increasing need for rapid, accurate, and cost-effective diagnostic tools, particularly for point-of-care testing and personalized medicine. Photonic biosensors offer advantages in terms of sensitivity, specificity, and real-time monitoring capabilities, making them attractive for applications such as disease diagnosis, drug discovery, and therapeutic monitoring.
The pharmaceutical industry is another major driver of biosensor demand, as these technologies can streamline drug development processes and enhance the efficiency of clinical trials. Photonic biosensors enable researchers to study molecular interactions and cellular responses with high precision, potentially reducing the time and cost associated with bringing new drugs to market.
Environmental monitoring and food safety are also contributing to the growing market for biosensor technologies. With increasing concerns about pollution, water quality, and foodborne illnesses, there is a rising demand for rapid and reliable detection methods. Photonic biosensors offer the potential for on-site, real-time monitoring of contaminants and pathogens, addressing critical needs in these sectors.
The agriculture industry is emerging as a significant market for biosensor technologies, with applications in crop management, livestock health monitoring, and food quality control. Photonic biosensors can provide farmers and food producers with valuable data on soil conditions, plant health, and animal well-being, enabling more efficient and sustainable agricultural practices.
In the field of biodefense and security, there is a growing demand for biosensors capable of detecting biological threats quickly and accurately. Photonic biosensors show promise in this area due to their ability to detect minute quantities of biological agents with high specificity.
The global biosensor market is projected to continue its upward trajectory, with photonic biosensors playing an increasingly important role. As the technology matures and becomes more accessible, new applications are likely to emerge, further expanding the market potential. However, to fully capitalize on this demand, developers of photonic biosensors must address challenges related to standardization, scalability, and integration with existing diagnostic platforms.
Validating photonic biosensor performance against established gold standards like ELISA is crucial for market acceptance and regulatory approval. The development of robust validation protocols will be essential in demonstrating the reliability and accuracy of these new technologies, thereby accelerating their adoption across various industries.
The pharmaceutical industry is another major driver of biosensor demand, as these technologies can streamline drug development processes and enhance the efficiency of clinical trials. Photonic biosensors enable researchers to study molecular interactions and cellular responses with high precision, potentially reducing the time and cost associated with bringing new drugs to market.
Environmental monitoring and food safety are also contributing to the growing market for biosensor technologies. With increasing concerns about pollution, water quality, and foodborne illnesses, there is a rising demand for rapid and reliable detection methods. Photonic biosensors offer the potential for on-site, real-time monitoring of contaminants and pathogens, addressing critical needs in these sectors.
The agriculture industry is emerging as a significant market for biosensor technologies, with applications in crop management, livestock health monitoring, and food quality control. Photonic biosensors can provide farmers and food producers with valuable data on soil conditions, plant health, and animal well-being, enabling more efficient and sustainable agricultural practices.
In the field of biodefense and security, there is a growing demand for biosensors capable of detecting biological threats quickly and accurately. Photonic biosensors show promise in this area due to their ability to detect minute quantities of biological agents with high specificity.
The global biosensor market is projected to continue its upward trajectory, with photonic biosensors playing an increasingly important role. As the technology matures and becomes more accessible, new applications are likely to emerge, further expanding the market potential. However, to fully capitalize on this demand, developers of photonic biosensors must address challenges related to standardization, scalability, and integration with existing diagnostic platforms.
Validating photonic biosensor performance against established gold standards like ELISA is crucial for market acceptance and regulatory approval. The development of robust validation protocols will be essential in demonstrating the reliability and accuracy of these new technologies, thereby accelerating their adoption across various industries.
Current Challenges in Biosensor Validation
The validation of photonic biosensors against established gold standards like ELISA presents several significant challenges. One of the primary difficulties lies in the inherent differences between these technologies. Photonic biosensors operate on principles of light interaction with biological samples, while ELISA relies on enzyme-linked antibody reactions. This fundamental disparity in detection mechanisms can lead to discrepancies in sensitivity, specificity, and dynamic range, making direct comparisons challenging.
Another major hurdle is the lack of standardized protocols for photonic biosensor validation. Unlike ELISA, which has well-established and widely accepted procedures, photonic biosensors are still an emerging technology with diverse designs and operating principles. This variability makes it difficult to develop universal validation protocols, leading to inconsistencies in performance evaluation across different research groups and manufacturers.
The complexity of biological samples also poses a significant challenge. Real-world samples often contain interfering substances that can affect biosensor performance differently than they would impact ELISA results. These matrix effects can lead to false positives or negatives, complicating the validation process and potentially undermining the reliability of photonic biosensors in practical applications.
Reproducibility and robustness are additional concerns in biosensor validation. Photonic biosensors can be sensitive to environmental factors such as temperature fluctuations, pH changes, and mechanical vibrations. Ensuring consistent performance across different batches, users, and laboratory conditions is crucial for validation but often proves difficult due to these sensitivities.
The time and resource-intensive nature of comprehensive validation studies presents another obstacle. Thorough comparison with gold standards requires extensive testing across a wide range of analytes, concentrations, and sample types. This process can be prohibitively expensive and time-consuming, particularly for smaller research groups or start-ups developing new biosensor technologies.
Lastly, the rapid pace of technological advancement in the field of photonic biosensors creates a moving target for validation efforts. As new materials, designs, and detection strategies emerge, validation protocols must evolve to keep pace. This constant evolution makes it challenging to establish long-term, widely accepted standards for performance comparison against traditional methods like ELISA.
Another major hurdle is the lack of standardized protocols for photonic biosensor validation. Unlike ELISA, which has well-established and widely accepted procedures, photonic biosensors are still an emerging technology with diverse designs and operating principles. This variability makes it difficult to develop universal validation protocols, leading to inconsistencies in performance evaluation across different research groups and manufacturers.
The complexity of biological samples also poses a significant challenge. Real-world samples often contain interfering substances that can affect biosensor performance differently than they would impact ELISA results. These matrix effects can lead to false positives or negatives, complicating the validation process and potentially undermining the reliability of photonic biosensors in practical applications.
Reproducibility and robustness are additional concerns in biosensor validation. Photonic biosensors can be sensitive to environmental factors such as temperature fluctuations, pH changes, and mechanical vibrations. Ensuring consistent performance across different batches, users, and laboratory conditions is crucial for validation but often proves difficult due to these sensitivities.
The time and resource-intensive nature of comprehensive validation studies presents another obstacle. Thorough comparison with gold standards requires extensive testing across a wide range of analytes, concentrations, and sample types. This process can be prohibitively expensive and time-consuming, particularly for smaller research groups or start-ups developing new biosensor technologies.
Lastly, the rapid pace of technological advancement in the field of photonic biosensors creates a moving target for validation efforts. As new materials, designs, and detection strategies emerge, validation protocols must evolve to keep pace. This constant evolution makes it challenging to establish long-term, widely accepted standards for performance comparison against traditional methods like ELISA.
Existing Validation Protocols vs ELISA
01 Optical design and performance enhancement
Photonic biosensors' performance can be improved through advanced optical designs. This includes optimizing light propagation, enhancing sensitivity, and reducing noise. Techniques such as plasmonic resonance, waveguide structures, and photonic crystals are employed to increase the sensor's efficiency and detection capabilities.- Optical design and performance enhancement: Photonic biosensors' performance can be improved through advanced optical designs. This includes optimizing light propagation, enhancing sensitivity, and reducing noise. Techniques such as plasmonic structures, photonic crystals, and waveguide designs are employed to increase the interaction between light and the analyte, resulting in better detection capabilities.
- Integration of nanomaterials: Incorporating nanomaterials into photonic biosensors can significantly enhance their performance. Nanomaterials such as quantum dots, nanoparticles, and nanostructured surfaces can increase sensitivity, selectivity, and signal-to-noise ratio. These materials can also enable multiplexed sensing and improve the overall efficiency of the biosensor.
- Signal processing and data analysis: Advanced signal processing techniques and data analysis methods play a crucial role in improving the performance of photonic biosensors. This includes developing algorithms for noise reduction, signal amplification, and pattern recognition. Machine learning and artificial intelligence approaches are also being applied to enhance data interpretation and sensor calibration.
- Microfluidic integration: Integrating microfluidic systems with photonic biosensors can enhance their performance by improving sample handling, reducing reagent consumption, and enabling real-time monitoring. Microfluidic channels can be designed to optimize analyte delivery to the sensing area, increasing sensitivity and reducing response time.
- Multi-parameter sensing and miniaturization: Developing photonic biosensors capable of multi-parameter sensing and miniaturization can improve overall performance. This involves designing compact, integrated systems that can simultaneously detect multiple analytes or biomarkers. Miniaturization techniques, such as lab-on-a-chip approaches, can lead to portable and high-throughput biosensing platforms with enhanced sensitivity and specificity.
02 Integration of nanomaterials
Incorporating nanomaterials into photonic biosensors can significantly enhance their performance. Nanomaterials such as quantum dots, nanoparticles, and nanostructured surfaces can increase sensitivity, specificity, and signal-to-noise ratio. These materials enable the detection of smaller analyte concentrations and improve overall sensor reliability.Expand Specific Solutions03 Signal processing and data analysis
Advanced signal processing techniques and data analysis algorithms play a crucial role in improving photonic biosensor performance. Machine learning, artificial intelligence, and statistical methods are used to enhance signal interpretation, reduce false positives, and increase the accuracy of analyte detection. These computational approaches enable real-time analysis and improve the overall reliability of biosensor measurements.Expand Specific Solutions04 Microfluidic integration
Integrating microfluidic systems with photonic biosensors can enhance their performance by improving sample handling, reducing reagent consumption, and enabling multiplexed detection. Microfluidic channels and structures allow for precise control of fluid flow, which can increase the interaction between analytes and sensing elements, leading to improved sensitivity and faster response times.Expand Specific Solutions05 Multi-parameter sensing and miniaturization
Developing photonic biosensors capable of multi-parameter sensing and miniaturization can significantly improve their performance and applicability. These advancements allow for simultaneous detection of multiple analytes, increased portability, and reduced sample volume requirements. Miniaturization techniques, such as lab-on-a-chip designs, enable the integration of multiple sensing elements and functionalities into a single compact device.Expand Specific Solutions
Key Players in Biosensor Development
The photonic biosensor performance validation market is in a growth phase, driven by increasing demand for rapid and accurate diagnostic tools. The market size is expanding, with potential applications in healthcare, environmental monitoring, and food safety. Technologically, photonic biosensors are advancing, but still require validation against established methods like ELISA. Companies such as Korea Research Institute of Bioscience & Biotechnology, i-SENS, and ARKRAY are actively developing and refining these technologies. Research institutions like Agency for Science, Technology & Research and Electronics & Telecommunications Research Institute are contributing to the field's progression. While promising, the technology's maturity varies across different applications, necessitating ongoing research and development to enhance reliability and widespread adoption.
Corning, Inc.
Technical Solution: Corning has developed advanced photonic biosensor platforms for validating performance against ELISA standards. Their Epic® label-free detection system utilizes optical biosensors based on resonant waveguide grating technology[1]. This system enables real-time, high-throughput screening and characterization of biomolecular interactions without the need for labels. The protocol involves immobilizing target molecules on the sensor surface, introducing analytes, and measuring binding-induced changes in the resonant wavelength. Corning has optimized surface chemistry and assay protocols to achieve high sensitivity and reproducibility comparable to gold standard ELISA methods[2].
Strengths: High-throughput capability, real-time measurements, and label-free detection. Weaknesses: May require specialized equipment and expertise for operation and data interpretation.
The Scripps Research Institute
Technical Solution: The Scripps Research Institute has developed a validation protocol for photonic biosensors based on surface plasmon resonance (SPR) imaging. Their approach involves creating a microarray of capture molecules on the sensor surface and comparing the binding kinetics and affinity measurements with those obtained from traditional ELISA methods. The protocol includes optimizing surface chemistry, determining the dynamic range, and assessing cross-reactivity. Scripps researchers have also developed algorithms for data analysis that account for mass transport limitations and heterogeneous binding[9]. Their method has been successfully applied to validate biosensors for detecting various biomarkers and drug-target interactions[10].
Strengths: Provides detailed kinetic information and is suitable for a wide range of biomolecular interactions. Weaknesses: May be less sensitive than some other methods for detecting low-abundance analytes.
Core Innovations in Photonic Biosensors
Enzyme linked immunosorbent assay sensor based on intelligent mobile terminal and using method
PatentActiveCN111830252A
Innovation
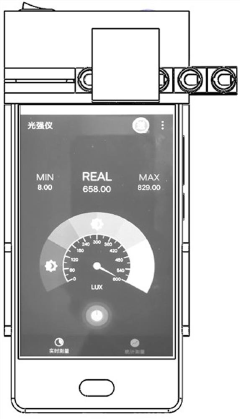
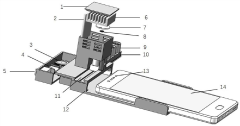
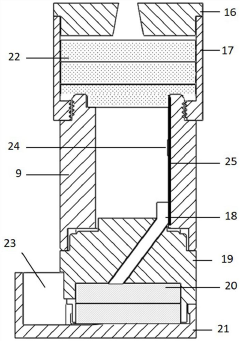
- An enzyme-linked immunochromatography sensor based on a smart mobile terminal is designed. It combines the immune detection process with the ambient light sensor of a smartphone. The light sensor detects the light intensity of the chromogenic solution to achieve instant interpretation of the test results without the need for professional equipment and operations. personnel, using LED lamp beads and optical fibers to improve signal transmission stability, and manufacturing structures through 3D printing to reduce costs.
Simultaneous Parallel Signal Amplification and Analyte-Ligand Capture Functions
PatentInactiveUS20210132049A1
Innovation
- An electrical, paper-based immunoassay using high affinity capture hydrogel particles with enzymatic amplification and amperometric sensors to detect TB biomarkers in urine samples, providing a sensitive, specific, and quantitative method for LTBI diagnosis.
Regulatory Requirements for Biosensors
Regulatory requirements for biosensors are critical to ensure their safety, efficacy, and reliability in various applications, including medical diagnostics and environmental monitoring. These requirements are established by regulatory bodies such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in Europe.
For photonic biosensors, the validation process against ELISA or other gold standard methods must adhere to specific regulatory guidelines. The FDA's guidance on bioanalytical method validation provides a framework for demonstrating the performance of new biosensor technologies. This includes requirements for accuracy, precision, selectivity, sensitivity, reproducibility, and stability.
In the context of comparing photonic biosensors to ELISA, regulatory bodies typically require comprehensive comparative studies. These studies must demonstrate that the new technology performs at least as well as the established gold standard. Key performance metrics such as limit of detection, dynamic range, and specificity must be thoroughly evaluated and documented.
Regulatory agencies also emphasize the importance of standardization in biosensor development and validation. This includes the use of certified reference materials and participation in interlaboratory comparison studies to ensure consistency across different laboratories and platforms.
Quality control measures are another crucial aspect of regulatory compliance for biosensors. Manufacturers must implement robust quality management systems that cover all aspects of biosensor production, from raw materials to final product testing. This includes maintaining detailed records of manufacturing processes, quality control tests, and any deviations or corrective actions taken.
For medical applications, biosensors may be classified as in vitro diagnostic devices (IVDs) and must comply with specific regulations such as the EU's In Vitro Diagnostic Regulation (IVDR) or the FDA's regulations for medical devices. These regulations often require clinical performance studies to demonstrate the biosensor's effectiveness in real-world settings.
Data integrity and security are increasingly important aspects of regulatory compliance, especially for biosensors that generate and transmit sensitive health information. Manufacturers must implement appropriate measures to protect data throughout its lifecycle, from collection to storage and analysis.
For photonic biosensors, the validation process against ELISA or other gold standard methods must adhere to specific regulatory guidelines. The FDA's guidance on bioanalytical method validation provides a framework for demonstrating the performance of new biosensor technologies. This includes requirements for accuracy, precision, selectivity, sensitivity, reproducibility, and stability.
In the context of comparing photonic biosensors to ELISA, regulatory bodies typically require comprehensive comparative studies. These studies must demonstrate that the new technology performs at least as well as the established gold standard. Key performance metrics such as limit of detection, dynamic range, and specificity must be thoroughly evaluated and documented.
Regulatory agencies also emphasize the importance of standardization in biosensor development and validation. This includes the use of certified reference materials and participation in interlaboratory comparison studies to ensure consistency across different laboratories and platforms.
Quality control measures are another crucial aspect of regulatory compliance for biosensors. Manufacturers must implement robust quality management systems that cover all aspects of biosensor production, from raw materials to final product testing. This includes maintaining detailed records of manufacturing processes, quality control tests, and any deviations or corrective actions taken.
For medical applications, biosensors may be classified as in vitro diagnostic devices (IVDs) and must comply with specific regulations such as the EU's In Vitro Diagnostic Regulation (IVDR) or the FDA's regulations for medical devices. These regulations often require clinical performance studies to demonstrate the biosensor's effectiveness in real-world settings.
Data integrity and security are increasingly important aspects of regulatory compliance, especially for biosensors that generate and transmit sensitive health information. Manufacturers must implement appropriate measures to protect data throughout its lifecycle, from collection to storage and analysis.
Biosensor Data Analysis Techniques
Data analysis techniques play a crucial role in validating photonic biosensor performance against established gold standards like ELISA. These techniques involve processing and interpreting the raw data obtained from biosensor measurements to extract meaningful information about analyte detection and quantification.
One of the primary data analysis methods for photonic biosensors is signal processing. This involves filtering and smoothing the raw sensor output to reduce noise and enhance the signal-to-noise ratio. Common techniques include moving average filters, Savitzky-Golay filters, and wavelet transforms. These methods help to isolate the true sensor response from background fluctuations and interference.
Calibration curve analysis is another essential technique for biosensor data interpretation. This involves plotting the sensor response against known concentrations of the target analyte to establish a relationship between signal intensity and analyte concentration. Various mathematical models, such as linear regression, four-parameter logistic regression, or Hill equation fitting, can be applied to generate calibration curves that enable quantitative analysis of unknown samples.
Statistical analysis is fundamental in assessing biosensor performance and comparing it to ELISA results. This includes calculating parameters such as limit of detection (LOD), limit of quantification (LOQ), dynamic range, and coefficient of variation (CV). These metrics provide insights into the sensitivity, accuracy, and precision of the biosensor compared to the gold standard method.
Multivariate data analysis techniques are increasingly employed for complex biosensor systems. Principal component analysis (PCA) and partial least squares regression (PLSR) can be used to extract relevant information from multidimensional data sets, particularly when dealing with multiplexed biosensors or when analyzing spectral data from optical biosensors.
Machine learning algorithms are gaining traction in biosensor data analysis. Supervised learning methods like support vector machines (SVM) and artificial neural networks (ANN) can be trained on known samples to develop predictive models for analyte classification and quantification. These approaches can enhance the specificity and sensitivity of biosensor measurements, especially in complex sample matrices.
Time-series analysis is particularly relevant for real-time biosensing applications. Techniques such as autocorrelation, cross-correlation, and Fourier transforms can be applied to analyze temporal patterns in biosensor signals, enabling the detection of binding kinetics and dynamic changes in analyte concentrations.
One of the primary data analysis methods for photonic biosensors is signal processing. This involves filtering and smoothing the raw sensor output to reduce noise and enhance the signal-to-noise ratio. Common techniques include moving average filters, Savitzky-Golay filters, and wavelet transforms. These methods help to isolate the true sensor response from background fluctuations and interference.
Calibration curve analysis is another essential technique for biosensor data interpretation. This involves plotting the sensor response against known concentrations of the target analyte to establish a relationship between signal intensity and analyte concentration. Various mathematical models, such as linear regression, four-parameter logistic regression, or Hill equation fitting, can be applied to generate calibration curves that enable quantitative analysis of unknown samples.
Statistical analysis is fundamental in assessing biosensor performance and comparing it to ELISA results. This includes calculating parameters such as limit of detection (LOD), limit of quantification (LOQ), dynamic range, and coefficient of variation (CV). These metrics provide insights into the sensitivity, accuracy, and precision of the biosensor compared to the gold standard method.
Multivariate data analysis techniques are increasingly employed for complex biosensor systems. Principal component analysis (PCA) and partial least squares regression (PLSR) can be used to extract relevant information from multidimensional data sets, particularly when dealing with multiplexed biosensors or when analyzing spectral data from optical biosensors.
Machine learning algorithms are gaining traction in biosensor data analysis. Supervised learning methods like support vector machines (SVM) and artificial neural networks (ANN) can be trained on known samples to develop predictive models for analyte classification and quantification. These approaches can enhance the specificity and sensitivity of biosensor measurements, especially in complex sample matrices.
Time-series analysis is particularly relevant for real-time biosensing applications. Techniques such as autocorrelation, cross-correlation, and Fourier transforms can be applied to analyze temporal patterns in biosensor signals, enabling the detection of binding kinetics and dynamic changes in analyte concentrations.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!