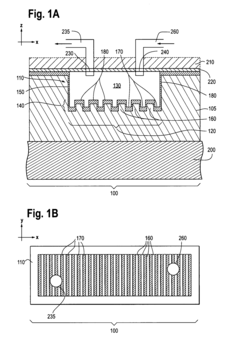
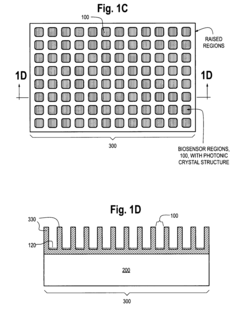
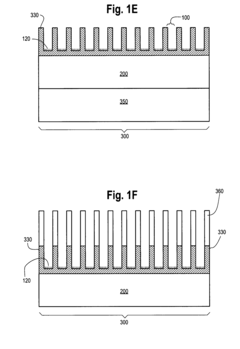
Photonic Crystal Biosensors: Working Principles, Sensitivity Metrics and Design Choices
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Photonic Crystal Biosensor Fundamentals and Objectives
Photonic crystal biosensors represent a cutting-edge technology in the field of biosensing, leveraging the unique optical properties of photonic crystals to detect and quantify biological analytes with high sensitivity and specificity. These sensors have emerged as a promising platform for various applications in healthcare, environmental monitoring, and biomedical research.
The fundamental principle behind photonic crystal biosensors lies in their periodic nanostructures, which can manipulate light propagation and create photonic bandgaps. When biological molecules interact with the sensor surface, they induce changes in the local refractive index, leading to measurable shifts in the optical properties of the photonic crystal. This forms the basis for label-free, real-time detection of biomolecular interactions.
Over the past two decades, photonic crystal biosensors have undergone significant advancements, driven by progress in nanofabrication techniques, materials science, and optical detection methods. The evolution of these sensors has been marked by improvements in sensitivity, miniaturization, and integration with microfluidic systems, enabling more sophisticated and versatile biosensing platforms.
The primary objectives in the development of photonic crystal biosensors include enhancing sensitivity to detect low concentrations of analytes, improving specificity to minimize false positives, and expanding the range of detectable biomolecules. Researchers aim to achieve these goals through innovative design strategies, such as optimizing the photonic crystal structure, exploring novel materials, and developing advanced signal processing algorithms.
Another crucial aspect of photonic crystal biosensor development is the pursuit of multiplexed detection capabilities, allowing simultaneous analysis of multiple analytes in a single assay. This objective aligns with the growing demand for comprehensive diagnostic tools in personalized medicine and point-of-care testing.
The integration of photonic crystal biosensors with other technologies, such as microfluidics and artificial intelligence, represents a key trend in the field. These hybrid systems aim to enhance overall performance, automate sample handling, and improve data interpretation, ultimately leading to more robust and user-friendly biosensing platforms.
As the technology continues to mature, there is an increasing focus on translating laboratory prototypes into commercially viable products. This transition involves addressing challenges related to scalability, reproducibility, and cost-effectiveness, which are essential for widespread adoption in clinical and industrial settings.
The fundamental principle behind photonic crystal biosensors lies in their periodic nanostructures, which can manipulate light propagation and create photonic bandgaps. When biological molecules interact with the sensor surface, they induce changes in the local refractive index, leading to measurable shifts in the optical properties of the photonic crystal. This forms the basis for label-free, real-time detection of biomolecular interactions.
Over the past two decades, photonic crystal biosensors have undergone significant advancements, driven by progress in nanofabrication techniques, materials science, and optical detection methods. The evolution of these sensors has been marked by improvements in sensitivity, miniaturization, and integration with microfluidic systems, enabling more sophisticated and versatile biosensing platforms.
The primary objectives in the development of photonic crystal biosensors include enhancing sensitivity to detect low concentrations of analytes, improving specificity to minimize false positives, and expanding the range of detectable biomolecules. Researchers aim to achieve these goals through innovative design strategies, such as optimizing the photonic crystal structure, exploring novel materials, and developing advanced signal processing algorithms.
Another crucial aspect of photonic crystal biosensor development is the pursuit of multiplexed detection capabilities, allowing simultaneous analysis of multiple analytes in a single assay. This objective aligns with the growing demand for comprehensive diagnostic tools in personalized medicine and point-of-care testing.
The integration of photonic crystal biosensors with other technologies, such as microfluidics and artificial intelligence, represents a key trend in the field. These hybrid systems aim to enhance overall performance, automate sample handling, and improve data interpretation, ultimately leading to more robust and user-friendly biosensing platforms.
As the technology continues to mature, there is an increasing focus on translating laboratory prototypes into commercially viable products. This transition involves addressing challenges related to scalability, reproducibility, and cost-effectiveness, which are essential for widespread adoption in clinical and industrial settings.
Market Analysis for Photonic Crystal Biosensors
The market for photonic crystal biosensors is experiencing significant growth, driven by increasing demand for rapid, sensitive, and label-free detection methods in various fields, including healthcare, environmental monitoring, and food safety. These biosensors offer unique advantages over traditional sensing technologies, such as high sensitivity, real-time detection capabilities, and miniaturization potential, making them attractive for a wide range of applications.
In the healthcare sector, photonic crystal biosensors are finding applications in early disease detection, drug discovery, and personalized medicine. The growing emphasis on point-of-care diagnostics and the need for rapid, accurate testing methods are fueling market expansion. The COVID-19 pandemic has further accelerated the demand for advanced biosensing technologies, highlighting the potential of photonic crystal biosensors in infectious disease detection and management.
Environmental monitoring represents another significant market opportunity for photonic crystal biosensors. With increasing concerns about water and air quality, these sensors are being developed for detecting pollutants, toxins, and pathogens in environmental samples. Their ability to provide real-time, on-site measurements makes them valuable tools for environmental agencies and industries seeking to comply with stringent regulations.
The food and beverage industry is also adopting photonic crystal biosensors for quality control and safety assurance. These sensors can detect contaminants, allergens, and pathogens in food products, addressing the growing consumer demand for food safety and transparency in the supply chain.
Market analysis indicates that North America and Europe currently dominate the photonic crystal biosensor market, owing to advanced healthcare infrastructure, substantial research and development investments, and early adoption of novel technologies. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by increasing healthcare expenditure, rising awareness about early disease detection, and government initiatives to promote biotechnology research.
Key market players in the photonic crystal biosensor space include established biotechnology companies, innovative startups, and academic institutions collaborating with industry partners. These entities are focusing on developing more sensitive, specific, and cost-effective biosensor platforms to address unmet needs in various application areas.
Challenges facing the market include the need for standardization of biosensor performance metrics, integration with existing diagnostic platforms, and scaling up production for commercial applications. However, ongoing advancements in nanofabrication techniques, materials science, and data analysis algorithms are expected to address these challenges and further expand the market potential of photonic crystal biosensors.
In the healthcare sector, photonic crystal biosensors are finding applications in early disease detection, drug discovery, and personalized medicine. The growing emphasis on point-of-care diagnostics and the need for rapid, accurate testing methods are fueling market expansion. The COVID-19 pandemic has further accelerated the demand for advanced biosensing technologies, highlighting the potential of photonic crystal biosensors in infectious disease detection and management.
Environmental monitoring represents another significant market opportunity for photonic crystal biosensors. With increasing concerns about water and air quality, these sensors are being developed for detecting pollutants, toxins, and pathogens in environmental samples. Their ability to provide real-time, on-site measurements makes them valuable tools for environmental agencies and industries seeking to comply with stringent regulations.
The food and beverage industry is also adopting photonic crystal biosensors for quality control and safety assurance. These sensors can detect contaminants, allergens, and pathogens in food products, addressing the growing consumer demand for food safety and transparency in the supply chain.
Market analysis indicates that North America and Europe currently dominate the photonic crystal biosensor market, owing to advanced healthcare infrastructure, substantial research and development investments, and early adoption of novel technologies. However, the Asia-Pacific region is expected to witness the highest growth rate in the coming years, driven by increasing healthcare expenditure, rising awareness about early disease detection, and government initiatives to promote biotechnology research.
Key market players in the photonic crystal biosensor space include established biotechnology companies, innovative startups, and academic institutions collaborating with industry partners. These entities are focusing on developing more sensitive, specific, and cost-effective biosensor platforms to address unmet needs in various application areas.
Challenges facing the market include the need for standardization of biosensor performance metrics, integration with existing diagnostic platforms, and scaling up production for commercial applications. However, ongoing advancements in nanofabrication techniques, materials science, and data analysis algorithms are expected to address these challenges and further expand the market potential of photonic crystal biosensors.
Current Challenges in Photonic Crystal Biosensor Technology
Despite the significant advancements in photonic crystal biosensor technology, several challenges persist that hinder their widespread adoption and optimal performance. One of the primary obstacles is the complexity of fabrication processes, which often require sophisticated and expensive equipment. The precise control of nanostructures and the need for high-quality, defect-free photonic crystals pose significant manufacturing hurdles, limiting large-scale production and increasing costs.
Sensitivity and specificity remain critical challenges in photonic crystal biosensors. While these sensors have demonstrated high sensitivity, achieving consistent and reliable detection of low-concentration analytes in complex biological samples continues to be problematic. The presence of non-specific binding and interference from other biomolecules in real-world samples can lead to false positives or reduced sensitivity, necessitating improved surface functionalization strategies and more robust detection algorithms.
The integration of photonic crystal biosensors into practical, user-friendly devices presents another significant challenge. Miniaturization and portability are crucial for point-of-care applications, but reducing the size of optical components without compromising performance is technically demanding. Additionally, the development of efficient light coupling mechanisms and the integration of microfluidic systems for sample handling add layers of complexity to the overall sensor design.
Stability and reproducibility issues also plague current photonic crystal biosensor technology. Environmental factors such as temperature fluctuations and mechanical stress can affect the optical properties of the photonic crystals, leading to drift in sensor response over time. Ensuring long-term stability and consistent performance across different batches of sensors remains a significant hurdle for commercial viability.
The development of multiplexed sensing capabilities is another area that requires further advancement. While photonic crystal biosensors have shown promise in detecting multiple analytes simultaneously, optimizing the design to accommodate a wide range of biomarkers without cross-reactivity or signal interference is challenging. This limitation restricts the application of these sensors in complex diagnostic scenarios where multiple biomarkers need to be monitored concurrently.
Lastly, the interpretation and analysis of sensor data present ongoing challenges. As the sensitivity of photonic crystal biosensors increases, so does the complexity of the generated data. Developing robust algorithms for real-time data processing, noise reduction, and accurate quantification of analytes is crucial for translating sensor outputs into meaningful clinical or research results. The integration of machine learning and artificial intelligence techniques for data analysis is an emerging area that holds promise but requires further development and validation.
Sensitivity and specificity remain critical challenges in photonic crystal biosensors. While these sensors have demonstrated high sensitivity, achieving consistent and reliable detection of low-concentration analytes in complex biological samples continues to be problematic. The presence of non-specific binding and interference from other biomolecules in real-world samples can lead to false positives or reduced sensitivity, necessitating improved surface functionalization strategies and more robust detection algorithms.
The integration of photonic crystal biosensors into practical, user-friendly devices presents another significant challenge. Miniaturization and portability are crucial for point-of-care applications, but reducing the size of optical components without compromising performance is technically demanding. Additionally, the development of efficient light coupling mechanisms and the integration of microfluidic systems for sample handling add layers of complexity to the overall sensor design.
Stability and reproducibility issues also plague current photonic crystal biosensor technology. Environmental factors such as temperature fluctuations and mechanical stress can affect the optical properties of the photonic crystals, leading to drift in sensor response over time. Ensuring long-term stability and consistent performance across different batches of sensors remains a significant hurdle for commercial viability.
The development of multiplexed sensing capabilities is another area that requires further advancement. While photonic crystal biosensors have shown promise in detecting multiple analytes simultaneously, optimizing the design to accommodate a wide range of biomarkers without cross-reactivity or signal interference is challenging. This limitation restricts the application of these sensors in complex diagnostic scenarios where multiple biomarkers need to be monitored concurrently.
Lastly, the interpretation and analysis of sensor data present ongoing challenges. As the sensitivity of photonic crystal biosensors increases, so does the complexity of the generated data. Developing robust algorithms for real-time data processing, noise reduction, and accurate quantification of analytes is crucial for translating sensor outputs into meaningful clinical or research results. The integration of machine learning and artificial intelligence techniques for data analysis is an emerging area that holds promise but requires further development and validation.
Existing Photonic Crystal Biosensor Architectures
01 Structural design for enhanced sensitivity
Photonic crystal biosensors can be designed with specific structural features to enhance sensitivity. This includes optimizing the crystal lattice, introducing defects, or creating multi-layer structures. These design modifications can increase light-matter interactions and improve the detection of small changes in the refractive index, resulting in higher sensitivity for biosensing applications.- Structural design for enhanced sensitivity: Photonic crystal biosensors can be designed with specific structural features to enhance sensitivity. This includes optimizing the crystal lattice, introducing defects, or creating multi-layer structures. These design modifications can increase light-matter interactions and improve the detection of small changes in the refractive index, resulting in higher sensitivity for biosensing applications.
- Surface functionalization techniques: The sensitivity of photonic crystal biosensors can be improved through various surface functionalization techniques. These methods involve modifying the sensor surface with specific biomolecules or chemical groups to enhance target analyte binding. Proper surface functionalization can increase the selectivity and sensitivity of the biosensor, allowing for detection of lower concentrations of target molecules.
- Integration of plasmonic materials: Incorporating plasmonic materials into photonic crystal structures can significantly enhance the sensitivity of biosensors. The combination of photonic and plasmonic effects can lead to stronger light confinement and field enhancement, resulting in improved detection capabilities. This approach allows for the development of highly sensitive hybrid photonic-plasmonic biosensors.
- Signal processing and data analysis techniques: Advanced signal processing and data analysis techniques can be employed to improve the sensitivity of photonic crystal biosensors. These methods may include noise reduction algorithms, machine learning approaches, or statistical analysis to extract meaningful information from sensor outputs. By enhancing signal-to-noise ratios and minimizing false positives, these techniques can significantly increase the overall sensitivity of the biosensing system.
- Temperature control and environmental stability: Implementing precise temperature control and ensuring environmental stability can enhance the sensitivity of photonic crystal biosensors. Temperature fluctuations and environmental factors can affect the sensor's performance and lead to false readings. By incorporating temperature compensation mechanisms and improving the sensor's robustness against external disturbances, the overall sensitivity and reliability of the biosensor can be significantly improved.
02 Surface functionalization techniques
The sensitivity of photonic crystal biosensors can be improved through various surface functionalization techniques. These methods involve modifying the sensor surface with specific biomolecules or chemical groups to enhance target analyte binding. Proper surface functionalization can increase the specificity and efficiency of biosensing, leading to improved sensitivity and lower detection limits.Expand Specific Solutions03 Integration of plasmonic materials
Incorporating plasmonic materials into photonic crystal structures can significantly enhance biosensor sensitivity. The combination of photonic and plasmonic effects can lead to stronger light confinement and field enhancement, resulting in improved detection capabilities. This approach allows for the development of highly sensitive hybrid photonic-plasmonic biosensors.Expand Specific Solutions04 Signal processing and data analysis techniques
Advanced signal processing and data analysis techniques can be employed to improve the sensitivity of photonic crystal biosensors. These methods may include noise reduction algorithms, machine learning approaches, or statistical analysis tools. By enhancing the signal-to-noise ratio and extracting meaningful information from raw data, the overall sensitivity and reliability of the biosensing system can be significantly improved.Expand Specific Solutions05 Microfluidic integration for sample handling
Integrating microfluidic systems with photonic crystal biosensors can enhance sensitivity by improving sample handling and delivery. Microfluidic channels can be designed to control sample flow, concentrate analytes, and reduce background noise. This integration allows for more efficient and precise biosensing, ultimately leading to increased sensitivity and lower detection limits.Expand Specific Solutions
Key Players in Photonic Crystal Biosensor Industry
The field of Photonic Crystal Biosensors is in a dynamic growth phase, with increasing market potential and technological advancements. The global biosensors market, including photonic crystal-based devices, is expanding rapidly due to rising healthcare demands and environmental monitoring needs. While the technology is maturing, there's still significant room for innovation and improvement in sensitivity, specificity, and miniaturization. Key players like Novartis AG, Hitachi Ltd., and Fujitsu Ltd. are investing in R&D to enhance sensor performance and broaden applications. Academic institutions such as Zhejiang University and the University of Rochester are contributing fundamental research, while companies like SRU Biosystems and Omega Optics are developing commercial applications, indicating a collaborative ecosystem driving progress in this field.
SRU Biosystems, Inc.
Technical Solution: SRU Biosystems has developed a proprietary photonic crystal biosensor technology called BIND (Biomolecular Interaction Detection). Their approach utilizes a nanostructured optical surface that reflects only a narrow band of light. When biomolecules bind to the sensor surface, the reflected wavelength shifts, allowing for label-free, real-time detection of molecular interactions. The BIND platform incorporates a microplate-based format, enabling high-throughput screening capabilities[1]. SRU's photonic crystal biosensors achieve high sensitivity through optimization of the crystal structure and surface chemistry, allowing detection of small molecules, proteins, and cells with minimal sample preparation[2].
Strengths: High sensitivity, label-free detection, real-time monitoring, and high-throughput capabilities. Weaknesses: May require specialized equipment and expertise for operation and data interpretation.
Hitachi Ltd.
Technical Solution: Hitachi has developed photonic crystal biosensors utilizing their expertise in nanofabrication and optoelectronics. Their approach focuses on creating highly sensitive sensors through precise control of the photonic crystal structure. Hitachi's biosensors employ a unique double-layer photonic crystal design that enhances the interaction between light and analytes. This configuration allows for improved sensitivity and specificity in detecting biomolecules. The company has also developed advanced surface functionalization techniques to optimize biomolecule capture and reduce non-specific binding. Hitachi's photonic crystal biosensors have been applied in various fields, including medical diagnostics and environmental monitoring[7].
Strengths: Advanced nanofabrication capabilities, unique double-layer design for enhanced sensitivity, and diverse application potential. Weaknesses: Potentially high production costs and complexity in sensor operation.
Core Innovations in Photonic Crystal Biosensor Sensitivity
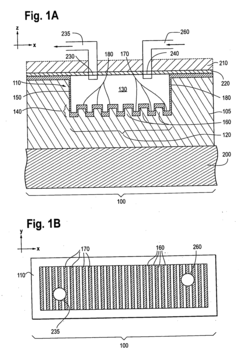
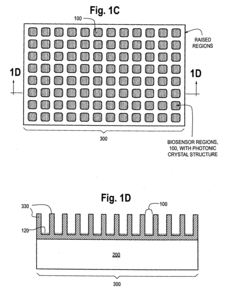
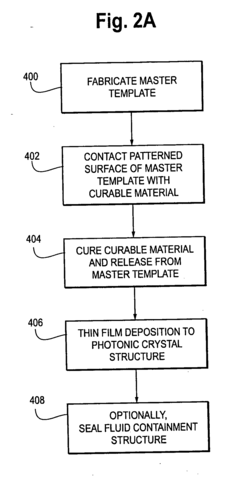
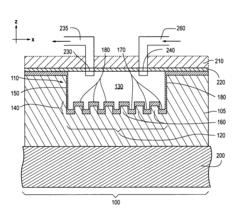
Photonic crystal sensors with integrated fluid containment structure, sample handling devices incorporating same, and uses thereof for biomolecular interaction analysis
PatentInactiveUS20100221847A1
Innovation
- The integration of photonic crystal sensors with fluid containment structures in a monolithic structure, where the photonic crystal surface grating is part of the internal surface of the fluid containment structure, allowing for simultaneous fabrication and alignment, reducing the complexity and cost of the fabrication process while preventing fluid leakage and maintaining sensor performance.
Photonic crystal sensors with intergrated fluid containment structure
PatentActiveUS7742662B2
Innovation
- The integration of fluid containment structures, such as wells or flow channels, directly with the photonic crystal sensors in a monolithic device, allowing for simultaneous fabrication and alignment, reducing the complexity of alignment and bonding processes and preventing fluid leakage between adjacent sensors.
Regulatory Framework for Biosensor Applications
The regulatory framework for biosensor applications plays a crucial role in ensuring the safety, efficacy, and ethical use of photonic crystal biosensors. In the United States, the Food and Drug Administration (FDA) is the primary regulatory body overseeing biosensor technologies. The FDA classifies biosensors based on their intended use and potential risk, with most falling under Class II medical devices requiring premarket notification (510(k)) or premarket approval (PMA).
The European Union employs the In Vitro Diagnostic Regulation (IVDR) and Medical Device Regulation (MDR) to govern biosensor applications. These regulations emphasize risk-based classification, post-market surveillance, and clinical evidence requirements. Manufacturers must obtain CE marking to demonstrate compliance with EU safety, health, and environmental protection standards.
In Asia, countries like Japan and China have their own regulatory frameworks. Japan's Pharmaceuticals and Medical Devices Agency (PMDA) oversees the approval process for biosensors, while China's National Medical Products Administration (NMPA) regulates medical devices, including biosensors.
Globally, the International Organization for Standardization (ISO) provides standards relevant to biosensor development and manufacturing, such as ISO 13485 for quality management systems and ISO 14971 for risk management in medical devices.
Regulatory considerations for photonic crystal biosensors include performance validation, sensitivity and specificity metrics, and potential interference from environmental factors. Manufacturers must demonstrate the analytical and clinical performance of their devices, as well as address potential risks associated with their use.
Data privacy and security regulations, such as the General Data Protection Regulation (GDPR) in the EU and the Health Insurance Portability and Accountability Act (HIPAA) in the US, also impact biosensor applications, particularly when dealing with patient health information.
As the field of photonic crystal biosensors advances, regulatory frameworks are evolving to keep pace with technological innovations. Emerging areas of focus include the regulation of artificial intelligence and machine learning algorithms used in biosensor data analysis, as well as the development of standards for nanomaterials used in sensor fabrication.
The European Union employs the In Vitro Diagnostic Regulation (IVDR) and Medical Device Regulation (MDR) to govern biosensor applications. These regulations emphasize risk-based classification, post-market surveillance, and clinical evidence requirements. Manufacturers must obtain CE marking to demonstrate compliance with EU safety, health, and environmental protection standards.
In Asia, countries like Japan and China have their own regulatory frameworks. Japan's Pharmaceuticals and Medical Devices Agency (PMDA) oversees the approval process for biosensors, while China's National Medical Products Administration (NMPA) regulates medical devices, including biosensors.
Globally, the International Organization for Standardization (ISO) provides standards relevant to biosensor development and manufacturing, such as ISO 13485 for quality management systems and ISO 14971 for risk management in medical devices.
Regulatory considerations for photonic crystal biosensors include performance validation, sensitivity and specificity metrics, and potential interference from environmental factors. Manufacturers must demonstrate the analytical and clinical performance of their devices, as well as address potential risks associated with their use.
Data privacy and security regulations, such as the General Data Protection Regulation (GDPR) in the EU and the Health Insurance Portability and Accountability Act (HIPAA) in the US, also impact biosensor applications, particularly when dealing with patient health information.
As the field of photonic crystal biosensors advances, regulatory frameworks are evolving to keep pace with technological innovations. Emerging areas of focus include the regulation of artificial intelligence and machine learning algorithms used in biosensor data analysis, as well as the development of standards for nanomaterials used in sensor fabrication.
Biocompatibility and Safety Considerations
Biocompatibility and safety considerations are paramount in the development and application of photonic crystal biosensors. These devices, which interface directly with biological systems, must be designed and manufactured with utmost care to ensure they do not cause adverse reactions or compromise the integrity of the samples being analyzed.
The materials used in photonic crystal biosensors must be carefully selected to minimize potential toxicity or immunogenicity. Common materials include silicon, glass, and various polymers, which have been extensively studied for their biocompatibility. However, the specific surface chemistry and nanostructures of photonic crystals may introduce unique interactions with biological systems that require thorough investigation.
Surface modifications play a crucial role in enhancing biocompatibility and reducing non-specific binding. Techniques such as PEGylation or the application of self-assembled monolayers can create a bio-inert surface that resists protein adsorption and cell adhesion. These modifications not only improve the safety profile of the biosensor but also enhance its specificity and sensitivity by reducing background noise.
The potential for nanoparticle shedding from photonic crystal structures must be carefully evaluated. While the rigid nature of most photonic crystals minimizes this risk, long-term stability studies are essential to ensure that no potentially harmful particles are released into the biological environment during extended use or under various environmental conditions.
Sterilization and cleaning protocols for photonic crystal biosensors must be developed with consideration for both efficacy and material compatibility. Traditional sterilization methods such as autoclaving may not be suitable for all photonic crystal materials, necessitating the exploration of alternative techniques like ethylene oxide treatment or gamma irradiation.
Long-term biocompatibility studies are crucial for biosensors intended for in vivo applications or prolonged contact with biological samples. These studies should assess potential inflammatory responses, thrombogenicity, and any changes in local tissue morphology or function. Additionally, the impact of the biosensor on the analyte of interest must be carefully evaluated to ensure that the measurement process does not alter the biological system being studied.
Regulatory compliance is a critical aspect of biosensor development, with agencies such as the FDA imposing stringent requirements for devices used in medical applications. Manufacturers must conduct comprehensive safety assessments and provide extensive documentation to obtain necessary approvals. This process often involves a series of in vitro and in vivo studies to demonstrate both short-term and long-term safety profiles.
The materials used in photonic crystal biosensors must be carefully selected to minimize potential toxicity or immunogenicity. Common materials include silicon, glass, and various polymers, which have been extensively studied for their biocompatibility. However, the specific surface chemistry and nanostructures of photonic crystals may introduce unique interactions with biological systems that require thorough investigation.
Surface modifications play a crucial role in enhancing biocompatibility and reducing non-specific binding. Techniques such as PEGylation or the application of self-assembled monolayers can create a bio-inert surface that resists protein adsorption and cell adhesion. These modifications not only improve the safety profile of the biosensor but also enhance its specificity and sensitivity by reducing background noise.
The potential for nanoparticle shedding from photonic crystal structures must be carefully evaluated. While the rigid nature of most photonic crystals minimizes this risk, long-term stability studies are essential to ensure that no potentially harmful particles are released into the biological environment during extended use or under various environmental conditions.
Sterilization and cleaning protocols for photonic crystal biosensors must be developed with consideration for both efficacy and material compatibility. Traditional sterilization methods such as autoclaving may not be suitable for all photonic crystal materials, necessitating the exploration of alternative techniques like ethylene oxide treatment or gamma irradiation.
Long-term biocompatibility studies are crucial for biosensors intended for in vivo applications or prolonged contact with biological samples. These studies should assess potential inflammatory responses, thrombogenicity, and any changes in local tissue morphology or function. Additionally, the impact of the biosensor on the analyte of interest must be carefully evaluated to ensure that the measurement process does not alter the biological system being studied.
Regulatory compliance is a critical aspect of biosensor development, with agencies such as the FDA imposing stringent requirements for devices used in medical applications. Manufacturers must conduct comprehensive safety assessments and provide extensive documentation to obtain necessary approvals. This process often involves a series of in vitro and in vivo studies to demonstrate both short-term and long-term safety profiles.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!