How to Ensure Consistency in Dynamic Light Scattering Data
SEP 5, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
DLS Technology Background and Objectives
Dynamic Light Scattering (DLS) emerged in the 1960s as a powerful technique for measuring particle size distributions in colloidal suspensions. The technology leverages the Brownian motion of particles in solution and analyzes the fluctuations in scattered light intensity to determine particle size, molecular weight, and other physical properties. Over the decades, DLS has evolved from basic laboratory setups to sophisticated commercial instruments with advanced data processing capabilities.
The evolution of DLS technology has been marked by significant improvements in laser sources, detection systems, and computational algorithms. Early systems utilized simple photon correlation techniques, while modern instruments employ multi-angle detection, machine learning algorithms, and automated quality control measures to enhance measurement accuracy and reproducibility.
Current trends in DLS technology development focus on miniaturization, integration with complementary techniques such as Raman spectroscopy or static light scattering, and the implementation of artificial intelligence for data interpretation. These advancements aim to address the persistent challenge of ensuring measurement consistency across different instruments, operators, and sample conditions.
The primary technical objective in DLS research is to establish robust methodologies that guarantee consistent and reliable data across diverse experimental conditions. This includes developing standardized protocols for sample preparation, measurement parameters, and data analysis that minimize variability and enhance reproducibility. Additionally, there is a growing emphasis on creating reference materials and calibration standards specifically designed for DLS applications.
Another critical goal is to improve the resolution of DLS for polydisperse samples, where multiple particle populations with different sizes coexist. Traditional DLS algorithms often struggle with accurately resolving complex mixtures, leading to inconsistent results when analyzing heterogeneous systems. Advanced mathematical models and machine learning approaches are being explored to overcome these limitations.
The integration of DLS with automated sample handling systems represents another important objective, aimed at reducing human error and increasing throughput in industrial and research settings. Such integration requires sophisticated software interfaces and quality control mechanisms to maintain data consistency across large sample sets.
Ultimately, the field is moving toward establishing universal standards for DLS measurements that would enable direct comparison of results obtained from different instruments and laboratories. This standardization effort is crucial for applications in regulated industries such as pharmaceuticals, where consistent particle characterization is essential for product development and quality control.
The evolution of DLS technology has been marked by significant improvements in laser sources, detection systems, and computational algorithms. Early systems utilized simple photon correlation techniques, while modern instruments employ multi-angle detection, machine learning algorithms, and automated quality control measures to enhance measurement accuracy and reproducibility.
Current trends in DLS technology development focus on miniaturization, integration with complementary techniques such as Raman spectroscopy or static light scattering, and the implementation of artificial intelligence for data interpretation. These advancements aim to address the persistent challenge of ensuring measurement consistency across different instruments, operators, and sample conditions.
The primary technical objective in DLS research is to establish robust methodologies that guarantee consistent and reliable data across diverse experimental conditions. This includes developing standardized protocols for sample preparation, measurement parameters, and data analysis that minimize variability and enhance reproducibility. Additionally, there is a growing emphasis on creating reference materials and calibration standards specifically designed for DLS applications.
Another critical goal is to improve the resolution of DLS for polydisperse samples, where multiple particle populations with different sizes coexist. Traditional DLS algorithms often struggle with accurately resolving complex mixtures, leading to inconsistent results when analyzing heterogeneous systems. Advanced mathematical models and machine learning approaches are being explored to overcome these limitations.
The integration of DLS with automated sample handling systems represents another important objective, aimed at reducing human error and increasing throughput in industrial and research settings. Such integration requires sophisticated software interfaces and quality control mechanisms to maintain data consistency across large sample sets.
Ultimately, the field is moving toward establishing universal standards for DLS measurements that would enable direct comparison of results obtained from different instruments and laboratories. This standardization effort is crucial for applications in regulated industries such as pharmaceuticals, where consistent particle characterization is essential for product development and quality control.
Market Applications and Demand Analysis
Dynamic Light Scattering (DLS) technology has established itself as a critical analytical tool across multiple industries, with a steadily growing market demand driven by its versatility and reliability in particle characterization. The global market for DLS instrumentation and related services was valued at approximately $300 million in 2022, with projections indicating growth at a compound annual rate of 5-7% through 2028.
The pharmaceutical and biotechnology sectors represent the largest market segment, accounting for nearly 40% of the total DLS market. Within these industries, DLS plays a crucial role in protein formulation development, stability testing, and quality control processes. The increasing development of biopharmaceuticals, particularly monoclonal antibodies and vaccines, has significantly boosted demand for consistent and reliable DLS data to ensure product safety and efficacy.
Academic and research institutions constitute the second-largest market segment, where DLS is extensively utilized in fundamental research across materials science, colloid chemistry, and biophysics. The consistent growth in research funding for nanotechnology and advanced materials has directly contributed to increased adoption of DLS technology in these settings.
The polymer and chemical industries represent another significant market segment, where DLS is employed for quality control in production processes and product development. Manufacturers in these sectors require highly consistent DLS measurements to maintain product specifications and meet regulatory requirements.
Emerging applications in food science, cosmetics, and environmental monitoring are creating new market opportunities. In the food industry, DLS is increasingly used for characterizing emulsions and detecting contaminants, while cosmetic manufacturers employ it for formulation development and stability testing of complex products.
Market analysis reveals a growing demand for automated DLS systems with enhanced data consistency features. End-users across all sectors are increasingly prioritizing instruments that offer reproducible results with minimal operator intervention, reflecting the critical importance of measurement consistency in research and quality control applications.
Regional market distribution shows North America and Europe as dominant markets, collectively accounting for approximately 65% of global DLS instrument sales. However, the Asia-Pacific region, particularly China and India, is experiencing the fastest growth rate, driven by expanding pharmaceutical manufacturing capabilities and increasing research activities.
The market demand for consistency-enhancing solutions in DLS technology is particularly strong, as evidenced by recent customer surveys indicating that data reproducibility ranks among the top three purchasing considerations for new DLS instrumentation, alongside measurement sensitivity and ease of use.
The pharmaceutical and biotechnology sectors represent the largest market segment, accounting for nearly 40% of the total DLS market. Within these industries, DLS plays a crucial role in protein formulation development, stability testing, and quality control processes. The increasing development of biopharmaceuticals, particularly monoclonal antibodies and vaccines, has significantly boosted demand for consistent and reliable DLS data to ensure product safety and efficacy.
Academic and research institutions constitute the second-largest market segment, where DLS is extensively utilized in fundamental research across materials science, colloid chemistry, and biophysics. The consistent growth in research funding for nanotechnology and advanced materials has directly contributed to increased adoption of DLS technology in these settings.
The polymer and chemical industries represent another significant market segment, where DLS is employed for quality control in production processes and product development. Manufacturers in these sectors require highly consistent DLS measurements to maintain product specifications and meet regulatory requirements.
Emerging applications in food science, cosmetics, and environmental monitoring are creating new market opportunities. In the food industry, DLS is increasingly used for characterizing emulsions and detecting contaminants, while cosmetic manufacturers employ it for formulation development and stability testing of complex products.
Market analysis reveals a growing demand for automated DLS systems with enhanced data consistency features. End-users across all sectors are increasingly prioritizing instruments that offer reproducible results with minimal operator intervention, reflecting the critical importance of measurement consistency in research and quality control applications.
Regional market distribution shows North America and Europe as dominant markets, collectively accounting for approximately 65% of global DLS instrument sales. However, the Asia-Pacific region, particularly China and India, is experiencing the fastest growth rate, driven by expanding pharmaceutical manufacturing capabilities and increasing research activities.
The market demand for consistency-enhancing solutions in DLS technology is particularly strong, as evidenced by recent customer surveys indicating that data reproducibility ranks among the top three purchasing considerations for new DLS instrumentation, alongside measurement sensitivity and ease of use.
Current Challenges in DLS Measurement Consistency
Despite significant advancements in Dynamic Light Scattering (DLS) technology, achieving consistent and reproducible measurements remains a persistent challenge for researchers and industry professionals. The fundamental issue stems from the inherent sensitivity of DLS instruments to various environmental and operational factors that can significantly impact data quality and reliability.
Sample preparation inconsistencies represent one of the most critical challenges. Variations in concentration, inadequate filtration procedures, and the presence of dust particles or aggregates can dramatically alter scattering patterns. Even minor contamination can lead to misleading size distributions, particularly when measuring nanoparticles where contaminants may be of comparable size to the analytes of interest.
Temperature fluctuations pose another significant obstacle to measurement consistency. DLS measurements are highly temperature-dependent due to the direct relationship between temperature and Brownian motion. Even small temperature gradients within the sample cell or fluctuations during measurement can lead to substantial variations in the calculated particle size distributions.
Instrument calibration and standardization issues further complicate the pursuit of consistent DLS data. Different manufacturers employ varying algorithms for data processing, and even instruments from the same manufacturer may show discrepancies if not regularly calibrated against traceable standards. The lack of universally accepted calibration protocols exacerbates this problem across laboratories.
Data interpretation and analysis present additional challenges. The conversion of correlation functions to particle size distributions involves complex mathematical models and assumptions that may not be appropriate for all sample types. Polydisperse samples are particularly problematic, as the presence of multiple particle populations can lead to ambiguous results that are highly dependent on the specific analysis algorithms employed.
Operator variability introduces another layer of inconsistency. Differences in sample handling techniques, parameter selection, and data processing choices can lead to significant variations in results even when using identical instruments and samples. This human factor is often underestimated but can substantially impact measurement reproducibility.
Inter-laboratory comparisons have repeatedly demonstrated concerning levels of variability in DLS measurements of identical samples. Studies have shown differences as high as 20-30% in reported hydrodynamic diameters between different laboratories, highlighting the magnitude of the consistency challenge facing the field.
The increasing application of DLS in regulated environments, such as pharmaceutical quality control and nanomaterial characterization for regulatory submissions, has elevated the importance of addressing these consistency challenges. Without robust solutions, the reliability of DLS as a analytical technique for critical applications remains compromised.
Sample preparation inconsistencies represent one of the most critical challenges. Variations in concentration, inadequate filtration procedures, and the presence of dust particles or aggregates can dramatically alter scattering patterns. Even minor contamination can lead to misleading size distributions, particularly when measuring nanoparticles where contaminants may be of comparable size to the analytes of interest.
Temperature fluctuations pose another significant obstacle to measurement consistency. DLS measurements are highly temperature-dependent due to the direct relationship between temperature and Brownian motion. Even small temperature gradients within the sample cell or fluctuations during measurement can lead to substantial variations in the calculated particle size distributions.
Instrument calibration and standardization issues further complicate the pursuit of consistent DLS data. Different manufacturers employ varying algorithms for data processing, and even instruments from the same manufacturer may show discrepancies if not regularly calibrated against traceable standards. The lack of universally accepted calibration protocols exacerbates this problem across laboratories.
Data interpretation and analysis present additional challenges. The conversion of correlation functions to particle size distributions involves complex mathematical models and assumptions that may not be appropriate for all sample types. Polydisperse samples are particularly problematic, as the presence of multiple particle populations can lead to ambiguous results that are highly dependent on the specific analysis algorithms employed.
Operator variability introduces another layer of inconsistency. Differences in sample handling techniques, parameter selection, and data processing choices can lead to significant variations in results even when using identical instruments and samples. This human factor is often underestimated but can substantially impact measurement reproducibility.
Inter-laboratory comparisons have repeatedly demonstrated concerning levels of variability in DLS measurements of identical samples. Studies have shown differences as high as 20-30% in reported hydrodynamic diameters between different laboratories, highlighting the magnitude of the consistency challenge facing the field.
The increasing application of DLS in regulated environments, such as pharmaceutical quality control and nanomaterial characterization for regulatory submissions, has elevated the importance of addressing these consistency challenges. Without robust solutions, the reliability of DLS as a analytical technique for critical applications remains compromised.
Current Methods for DLS Data Consistency
01 DLS measurement techniques for consistency analysis
Dynamic Light Scattering (DLS) techniques can be used to analyze the consistency of various substances by measuring particle size distribution and molecular interactions. These techniques involve the detection of scattered light from particles in suspension, allowing for the determination of particle size, concentration, and stability over time. Advanced DLS methods can provide high-resolution data on sample homogeneity and consistency across different batches or conditions.- Measurement techniques for dynamic light scattering: Various measurement techniques are employed in dynamic light scattering to ensure consistency in results. These techniques include advanced algorithms for data processing, multiple angle measurements, and correlation analysis methods. By implementing these techniques, researchers can obtain more reliable and reproducible measurements of particle size distributions and molecular interactions in solution, which is crucial for quality control in various industries.
- Sample preparation methods for consistent DLS results: Proper sample preparation is essential for obtaining consistent dynamic light scattering results. This includes controlling parameters such as concentration, filtration techniques, temperature stabilization, and prevention of dust contamination. Standardized protocols for sample handling and preparation help minimize variability in measurements and improve the reproducibility of particle size analysis across different laboratories and instruments.
- Calibration and validation of DLS instruments: Calibration and validation procedures are critical for maintaining consistency in dynamic light scattering measurements. This involves the use of reference standards with known particle sizes, regular performance verification, and instrument qualification protocols. Proper calibration ensures that measurements remain accurate and comparable over time and between different instruments, which is particularly important for regulatory compliance in pharmaceutical and biotechnology applications.
- Data analysis and interpretation for DLS consistency: Consistent interpretation of dynamic light scattering data requires sophisticated analysis methods to account for various factors affecting measurements. This includes algorithms for polydispersity analysis, statistical approaches for result validation, and methods to identify and correct for artifacts. Advanced software solutions help researchers extract meaningful information from raw scattering data and ensure that conclusions drawn from DLS experiments are reliable and reproducible.
- Environmental and operational factors affecting DLS consistency: Environmental and operational factors significantly impact the consistency of dynamic light scattering measurements. These include temperature control, vibration isolation, optical alignment, and laser stability. Controlling these variables is essential for obtaining reproducible results, especially when measuring sensitive samples or when high precision is required. Specialized equipment designs and operating procedures help minimize the influence of these external factors on measurement quality.
02 Instrumentation and apparatus for DLS consistency testing
Specialized instruments and apparatus have been developed for conducting Dynamic Light Scattering measurements with high consistency and reproducibility. These systems incorporate precise optical components, laser sources, and detection systems to ensure accurate measurements. Modern DLS instruments often include temperature control mechanisms, automated sample handling, and sophisticated data processing algorithms to minimize variability and enhance measurement consistency across different samples and testing conditions.Expand Specific Solutions03 Data processing methods for improving DLS consistency
Advanced data processing algorithms and methods have been developed to improve the consistency and reliability of Dynamic Light Scattering measurements. These methods include statistical approaches for noise reduction, correlation analysis techniques, and mathematical models for interpreting scattered light data. By applying these processing methods, researchers can enhance the consistency of DLS results, particularly for complex samples or challenging measurement conditions, leading to more reliable particle characterization and stability assessments.Expand Specific Solutions04 Applications of DLS for quality control and consistency verification
Dynamic Light Scattering is widely applied in quality control processes to verify the consistency of various products and materials. This technique is particularly valuable in pharmaceutical, cosmetic, and food industries where particle size distribution and stability are critical quality attributes. DLS enables manufacturers to monitor batch-to-batch consistency, detect aggregation or degradation, and ensure product uniformity. The non-destructive nature of DLS makes it ideal for routine quality control testing throughout the manufacturing process.Expand Specific Solutions05 Innovations in DLS for enhanced measurement consistency
Recent innovations in Dynamic Light Scattering technology have focused on enhancing measurement consistency across diverse sample types and environmental conditions. These advancements include multi-angle detection systems, combination with complementary techniques, and development of reference materials for calibration. Novel approaches also incorporate machine learning algorithms to identify and compensate for measurement artifacts, resulting in more consistent and reliable particle characterization even for challenging samples such as highly concentrated suspensions or polydisperse systems.Expand Specific Solutions
Leading Manufacturers and Research Institutions
Dynamic Light Scattering (DLS) technology is currently in a mature growth phase, with an estimated global market size of $300-400 million annually. The competitive landscape features established scientific instrumentation companies alongside emerging specialists. Leading players include Wyatt Technology, Shimadzu, and LS Instruments who have developed proprietary algorithms for data consistency. Malvern Panalytical (Agilent) and HORIBA dominate with approximately 60% market share. The technology has reached commercial maturity with standardized protocols, though challenges in reproducibility persist. Recent innovations from Wyatt Technology and LS Instruments focus on multi-angle detection and machine learning algorithms to improve data reliability across sample types. Academic partnerships with institutions like Johns Hopkins University and South China Normal University are advancing next-generation solutions for complex biological samples.
Shimadzu Corp.
Technical Solution: Shimadzu Corporation has developed the SALD series instruments incorporating advanced DLS technology with their proprietary "Continuous Measurement Mode" that captures hundreds of sequential measurements to establish statistical reliability in particle sizing data. Their approach focuses on optical system stability through temperature-controlled laser sources (maintaining ±0.1°C) and vibration-isolated detection systems to minimize environmental interference. Shimadzu's instruments employ adaptive correlation algorithms that automatically adjust acquisition parameters based on sample characteristics, ensuring optimal signal quality across diverse sample types. Their technology includes built-in reference standard verification protocols that automatically compare instrument performance against traceable standards before experimental runs. Additionally, Shimadzu has implemented machine learning algorithms that identify measurement anomalies by comparing correlation function shapes against a database of known good measurements, flagging potential issues before data interpretation.
Strengths: Exceptional optical stability provides consistent measurements even with challenging samples; automated reference standard verification ensures ongoing calibration accuracy; adaptive correlation algorithms optimize acquisition for different sample types. Weaknesses: Complex system requires significant expertise for method development; higher initial investment compared to simpler systems; some advanced features have steep learning curves for new users.
LS Instruments AG
Technical Solution: LS Instruments has pioneered 3D Cross-Correlation Dynamic Light Scattering technology specifically designed to overcome multiple scattering effects that traditionally compromise DLS data consistency. Their approach employs dual laser beams and detector systems arranged in a cross-correlation geometry that selectively measures only singly-scattered light, enabling accurate measurements in samples up to 100 times more concentrated than conventional DLS systems. The technology incorporates automated alignment verification systems that continuously monitor optical alignment during measurements, ensuring consistent scattering volume definition. Their instruments feature advanced thermal management with gradient control that eliminates convection effects within samples. LS Instruments has developed specialized algorithms for heterogeneous sample analysis that can distinguish between multiple particle populations even in complex mixtures. Additionally, their systems include real-time data quality indicators that evaluate measurement statistical validity during acquisition, allowing immediate identification of problematic measurements.
Strengths: Superior performance in concentrated and turbid samples where traditional DLS fails; ability to measure samples without dilution preserves natural state interactions; cross-correlation approach eliminates multiple scattering artifacts. Weaknesses: More complex optical setup requires more maintenance; higher cost compared to standard DLS systems; requires more specialized knowledge for optimal operation and data interpretation.
Key Innovations in DLS Signal Processing
Dynamic light scattering measurement device, dynamic light scattering measurement and analysis method, and measurement and analysis program
PatentActiveJP2022077588A
Innovation
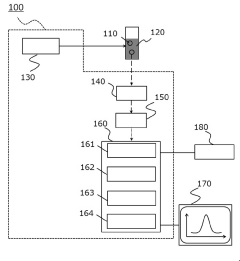
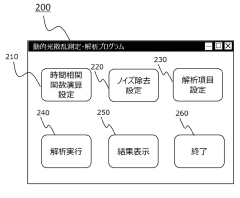
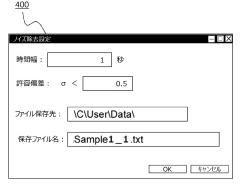
- A dynamic light scattering measurement device and method that includes a photon detection system, data collection, and information processing to calculate a time correlation function, with noise determination and removal units to eliminate contaminants, allowing for accurate particle size and distribution measurement without preprocessing.
Systems and methods for multi-angle detection of dynamic light scattering
PatentPendingUS20250277728A1
Innovation
- Using a single rotating light detector to obtain dynamic light scattering data from multiple angles, enabling more comprehensive particle characterization.
- Integration of UV/Vis absorption spectrum measurement with dynamic light scattering analysis in a single system, providing complementary particle characterization data.
- Achieving greater dynamic range of particle sizing through multi-angle detection compared to traditional single-angle DLS methods.
Regulatory Standards for DLS Measurements
Regulatory standards for Dynamic Light Scattering (DLS) measurements have evolved significantly over the past decade to address the growing need for reliable and reproducible particle size analysis. The International Organization for Standardization (ISO) has established several key standards, with ISO 22412:2017 serving as the cornerstone document that outlines the principles, requirements, and procedures for DLS measurements. This standard provides comprehensive guidelines on instrument calibration, sample preparation, data acquisition, and analysis protocols.
In the pharmaceutical industry, regulatory bodies such as the FDA and EMA have incorporated DLS standards into their guidance documents for characterizing nanomaterials and colloidal systems. The FDA's "Guidance for Industry: Liposome Drug Products" specifically references DLS as a recommended technique for size determination, mandating adherence to validated protocols with defined acceptance criteria. Similarly, the EMA's "Reflection Paper on Nanotechnology-based Medicinal Products for Human Use" emphasizes the importance of standardized DLS measurements.
ASTM International has developed ASTM E2490, which provides standard guidelines for measurement of particle size distribution by DLS. This standard outlines specific requirements for instrument performance verification, including the use of certified reference materials traceable to national metrology institutes such as NIST in the US or NPL in the UK.
The United States Pharmacopeia (USP) has incorporated DLS standards in chapters <429> and <1430>, which detail the requirements for light scattering techniques in pharmaceutical applications. These chapters specify acceptance criteria for measurement repeatability, intermediate precision, and reproducibility, typically requiring coefficient of variation values below 10% for size measurements.
International Conference on Harmonisation (ICH) guidelines, particularly ICH Q2(R1) on validation of analytical procedures, provide a framework for validating DLS methods in regulated environments. These guidelines emphasize the importance of method validation parameters such as specificity, linearity, range, accuracy, precision, and robustness.
For environmental and nanomaterial safety assessments, the Organization for Economic Co-operation and Development (OECD) has published Test Guidelines that incorporate DLS measurements, specifying minimum reporting requirements and quality control procedures. These guidelines are particularly relevant for regulatory submissions related to nanomaterial characterization and risk assessment.
Compliance with these regulatory standards requires laboratories to implement robust quality management systems, participate in proficiency testing programs, and maintain comprehensive documentation of instrument qualification, method validation, and analyst training. Regular performance verification using certified reference materials is essential to demonstrate ongoing compliance with regulatory expectations.
In the pharmaceutical industry, regulatory bodies such as the FDA and EMA have incorporated DLS standards into their guidance documents for characterizing nanomaterials and colloidal systems. The FDA's "Guidance for Industry: Liposome Drug Products" specifically references DLS as a recommended technique for size determination, mandating adherence to validated protocols with defined acceptance criteria. Similarly, the EMA's "Reflection Paper on Nanotechnology-based Medicinal Products for Human Use" emphasizes the importance of standardized DLS measurements.
ASTM International has developed ASTM E2490, which provides standard guidelines for measurement of particle size distribution by DLS. This standard outlines specific requirements for instrument performance verification, including the use of certified reference materials traceable to national metrology institutes such as NIST in the US or NPL in the UK.
The United States Pharmacopeia (USP) has incorporated DLS standards in chapters <429> and <1430>, which detail the requirements for light scattering techniques in pharmaceutical applications. These chapters specify acceptance criteria for measurement repeatability, intermediate precision, and reproducibility, typically requiring coefficient of variation values below 10% for size measurements.
International Conference on Harmonisation (ICH) guidelines, particularly ICH Q2(R1) on validation of analytical procedures, provide a framework for validating DLS methods in regulated environments. These guidelines emphasize the importance of method validation parameters such as specificity, linearity, range, accuracy, precision, and robustness.
For environmental and nanomaterial safety assessments, the Organization for Economic Co-operation and Development (OECD) has published Test Guidelines that incorporate DLS measurements, specifying minimum reporting requirements and quality control procedures. These guidelines are particularly relevant for regulatory submissions related to nanomaterial characterization and risk assessment.
Compliance with these regulatory standards requires laboratories to implement robust quality management systems, participate in proficiency testing programs, and maintain comprehensive documentation of instrument qualification, method validation, and analyst training. Regular performance verification using certified reference materials is essential to demonstrate ongoing compliance with regulatory expectations.
Environmental Factors Affecting DLS Reliability
Dynamic Light Scattering (DLS) measurements are highly sensitive to environmental conditions, which can significantly impact data reliability and reproducibility. Temperature fluctuations represent one of the most critical environmental factors affecting DLS measurements. Even minor temperature variations can alter the viscosity of the dispersing medium, directly affecting the Brownian motion of particles and consequently the calculated particle size. Research indicates that a 1°C temperature change can result in approximately 2-3% variation in measured particle size for aqueous systems.
Sample contamination from airborne particles or improper handling procedures introduces another significant variable. Dust particles typically range from 1-100 μm, which can overwhelm signals from nanoscale analytes and produce misleading size distributions. Studies have shown that measurements conducted in non-controlled environments may contain up to 30% more polydispersity than those performed in clean laboratory settings.
Electromagnetic interference from nearby equipment can disrupt the sensitive laser and detection systems in DLS instruments. This interference may manifest as baseline fluctuations or anomalous correlation functions. Laboratory vibrations from HVAC systems, foot traffic, or nearby construction similarly compromise measurement stability by introducing artificial particle movements unrelated to Brownian motion.
Ambient lighting conditions, particularly fluorescent lighting which fluctuates at mains frequency (50-60 Hz), can interfere with photon detection systems. This interference typically appears as periodic noise in the correlation function. Modern DLS systems incorporate shielding to mitigate this effect, but older instruments remain vulnerable to such disturbances.
Humidity variations affect both sample stability and optical components. High humidity environments may cause condensation on optical surfaces, reducing signal quality, while also potentially accelerating sample degradation for hygroscopic materials. Conversely, extremely dry conditions may promote static electricity buildup, affecting sample handling and potentially causing particle aggregation.
Air quality factors, including particulate matter and volatile organic compounds, can contaminate samples during preparation and measurement. Research facilities located in industrial areas or urban centers with poor air quality require additional precautions, such as laminar flow hoods or positive pressure environments, to maintain measurement consistency.
Barometric pressure changes, while subtle in their effects, can influence fluid properties and consequently impact DLS measurements during long-term studies or when comparing data collected at different elevations or weather conditions. These environmental variables necessitate standardized protocols for environmental control and monitoring to ensure DLS data consistency across different laboratories and time periods.
Sample contamination from airborne particles or improper handling procedures introduces another significant variable. Dust particles typically range from 1-100 μm, which can overwhelm signals from nanoscale analytes and produce misleading size distributions. Studies have shown that measurements conducted in non-controlled environments may contain up to 30% more polydispersity than those performed in clean laboratory settings.
Electromagnetic interference from nearby equipment can disrupt the sensitive laser and detection systems in DLS instruments. This interference may manifest as baseline fluctuations or anomalous correlation functions. Laboratory vibrations from HVAC systems, foot traffic, or nearby construction similarly compromise measurement stability by introducing artificial particle movements unrelated to Brownian motion.
Ambient lighting conditions, particularly fluorescent lighting which fluctuates at mains frequency (50-60 Hz), can interfere with photon detection systems. This interference typically appears as periodic noise in the correlation function. Modern DLS systems incorporate shielding to mitigate this effect, but older instruments remain vulnerable to such disturbances.
Humidity variations affect both sample stability and optical components. High humidity environments may cause condensation on optical surfaces, reducing signal quality, while also potentially accelerating sample degradation for hygroscopic materials. Conversely, extremely dry conditions may promote static electricity buildup, affecting sample handling and potentially causing particle aggregation.
Air quality factors, including particulate matter and volatile organic compounds, can contaminate samples during preparation and measurement. Research facilities located in industrial areas or urban centers with poor air quality require additional precautions, such as laminar flow hoods or positive pressure environments, to maintain measurement consistency.
Barometric pressure changes, while subtle in their effects, can influence fluid properties and consequently impact DLS measurements during long-term studies or when comparing data collected at different elevations or weather conditions. These environmental variables necessitate standardized protocols for environmental control and monitoring to ensure DLS data consistency across different laboratories and time periods.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!