Measure Zeta Potential Using Dynamic Light Scattering
SEP 5, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zeta Potential Measurement Background and Objectives
Zeta potential measurement has evolved significantly over the past several decades, becoming a critical parameter in various scientific and industrial applications. The concept of zeta potential was first introduced in the early 20th century as scientists began exploring colloidal systems and the electrical double layer theory. Initially, measurement techniques were rudimentary and limited to basic electrophoresis observations.
The development of more sophisticated measurement technologies accelerated in the 1970s and 1980s with the advent of laser-based techniques, particularly Dynamic Light Scattering (DLS). This technological advancement marked a pivotal moment in zeta potential analysis, enabling more precise and reproducible measurements of particle surface charge characteristics in colloidal suspensions.
Today, zeta potential measurement using DLS represents a cornerstone technique in numerous fields including pharmaceuticals, materials science, water treatment, food processing, and nanotechnology. The fundamental principle leverages the relationship between particle mobility in an electric field and the electrical potential at the slipping plane of the particle-liquid interface.
The current technological trajectory shows increasing integration of zeta potential measurements with complementary analytical techniques, enhanced automation, and improved algorithms for data interpretation. Recent innovations focus on expanding measurement capabilities to more complex media, higher concentration samples, and non-aqueous systems, addressing previous limitations of traditional DLS-based approaches.
The primary objectives of modern zeta potential measurement using DLS include achieving higher measurement accuracy across diverse sample types, reducing sample volume requirements, extending the measurable concentration range, and developing more robust interpretation models that account for non-ideal behaviors in complex colloidal systems.
Additionally, there is growing emphasis on standardization of measurement protocols to ensure reproducibility and comparability of results across different laboratories and instrument platforms. This standardization effort is particularly important as zeta potential data increasingly informs critical decisions in product development, quality control, and regulatory compliance.
From a technological forecasting perspective, the field is moving toward more sophisticated multi-parameter analysis systems that simultaneously measure zeta potential alongside other colloidal properties, providing a more comprehensive characterization of interfacial phenomena. The integration of artificial intelligence and machine learning approaches for data analysis represents another significant trend, potentially enabling more nuanced interpretation of complex electrophoretic mobility data.
Understanding the historical context and evolutionary path of zeta potential measurement technologies provides essential insights for identifying emerging opportunities and addressing persistent challenges in this field. The continued refinement of DLS-based zeta potential measurement techniques remains crucial for advancing numerous scientific disciplines and industrial applications.
The development of more sophisticated measurement technologies accelerated in the 1970s and 1980s with the advent of laser-based techniques, particularly Dynamic Light Scattering (DLS). This technological advancement marked a pivotal moment in zeta potential analysis, enabling more precise and reproducible measurements of particle surface charge characteristics in colloidal suspensions.
Today, zeta potential measurement using DLS represents a cornerstone technique in numerous fields including pharmaceuticals, materials science, water treatment, food processing, and nanotechnology. The fundamental principle leverages the relationship between particle mobility in an electric field and the electrical potential at the slipping plane of the particle-liquid interface.
The current technological trajectory shows increasing integration of zeta potential measurements with complementary analytical techniques, enhanced automation, and improved algorithms for data interpretation. Recent innovations focus on expanding measurement capabilities to more complex media, higher concentration samples, and non-aqueous systems, addressing previous limitations of traditional DLS-based approaches.
The primary objectives of modern zeta potential measurement using DLS include achieving higher measurement accuracy across diverse sample types, reducing sample volume requirements, extending the measurable concentration range, and developing more robust interpretation models that account for non-ideal behaviors in complex colloidal systems.
Additionally, there is growing emphasis on standardization of measurement protocols to ensure reproducibility and comparability of results across different laboratories and instrument platforms. This standardization effort is particularly important as zeta potential data increasingly informs critical decisions in product development, quality control, and regulatory compliance.
From a technological forecasting perspective, the field is moving toward more sophisticated multi-parameter analysis systems that simultaneously measure zeta potential alongside other colloidal properties, providing a more comprehensive characterization of interfacial phenomena. The integration of artificial intelligence and machine learning approaches for data analysis represents another significant trend, potentially enabling more nuanced interpretation of complex electrophoretic mobility data.
Understanding the historical context and evolutionary path of zeta potential measurement technologies provides essential insights for identifying emerging opportunities and addressing persistent challenges in this field. The continued refinement of DLS-based zeta potential measurement techniques remains crucial for advancing numerous scientific disciplines and industrial applications.
Market Applications and Demand Analysis for DLS-Based Zeta Potential
The global market for zeta potential measurement technologies has witnessed substantial growth in recent years, driven primarily by increasing applications across pharmaceutical, biotechnology, and materials science sectors. Dynamic Light Scattering (DLS) based zeta potential measurement systems have emerged as a preferred analytical technique due to their non-invasive nature, rapid analysis capabilities, and minimal sample preparation requirements.
In the pharmaceutical industry, demand for DLS-based zeta potential measurements has grown at approximately 8% annually over the past five years. This growth is primarily attributed to the rising development of complex drug delivery systems, including liposomes, nanoparticles, and protein-based therapeutics. The stability assessment of these formulations is critical for ensuring product efficacy and shelf-life, creating sustained demand for precise zeta potential measurement technologies.
The biotechnology sector represents another significant market segment, particularly in the development of gene therapies and vaccines. The COVID-19 pandemic accelerated this trend, with manufacturers of lipid nanoparticle-based mRNA vaccines heavily relying on zeta potential measurements for quality control and stability assessment. Market analysis indicates this segment has expanded by nearly 12% since 2020.
Academic and research institutions constitute approximately 30% of the total market share, utilizing DLS-based zeta potential measurements for fundamental research in colloid science, material development, and biological systems characterization. Government funding for nanotechnology research has further bolstered this market segment.
Environmental monitoring applications represent an emerging market with significant growth potential. Water treatment facilities, environmental regulatory bodies, and mining operations increasingly employ zeta potential measurements to monitor colloidal stability in various aqueous systems. This segment is projected to grow at 10% annually over the next five years.
Geographically, North America and Europe currently dominate the market with a combined share of 65%, though Asia-Pacific regions, particularly China, Japan, and India, are experiencing the fastest growth rates. This regional expansion is driven by increasing investments in pharmaceutical manufacturing capabilities and research infrastructure.
The global market value for DLS-based zeta potential measurement instruments was estimated at $320 million in 2022, with projections suggesting growth to $450 million by 2027. Key market drivers include increasing regulatory requirements for nanomaterial characterization, growing adoption of nanotechnology across industries, and continuous technological advancements improving measurement accuracy and reproducibility.
Customer demand increasingly focuses on integrated systems that combine zeta potential measurements with other characterization techniques, such as particle size analysis and molecular weight determination, offering comprehensive sample analysis capabilities in a single instrument.
In the pharmaceutical industry, demand for DLS-based zeta potential measurements has grown at approximately 8% annually over the past five years. This growth is primarily attributed to the rising development of complex drug delivery systems, including liposomes, nanoparticles, and protein-based therapeutics. The stability assessment of these formulations is critical for ensuring product efficacy and shelf-life, creating sustained demand for precise zeta potential measurement technologies.
The biotechnology sector represents another significant market segment, particularly in the development of gene therapies and vaccines. The COVID-19 pandemic accelerated this trend, with manufacturers of lipid nanoparticle-based mRNA vaccines heavily relying on zeta potential measurements for quality control and stability assessment. Market analysis indicates this segment has expanded by nearly 12% since 2020.
Academic and research institutions constitute approximately 30% of the total market share, utilizing DLS-based zeta potential measurements for fundamental research in colloid science, material development, and biological systems characterization. Government funding for nanotechnology research has further bolstered this market segment.
Environmental monitoring applications represent an emerging market with significant growth potential. Water treatment facilities, environmental regulatory bodies, and mining operations increasingly employ zeta potential measurements to monitor colloidal stability in various aqueous systems. This segment is projected to grow at 10% annually over the next five years.
Geographically, North America and Europe currently dominate the market with a combined share of 65%, though Asia-Pacific regions, particularly China, Japan, and India, are experiencing the fastest growth rates. This regional expansion is driven by increasing investments in pharmaceutical manufacturing capabilities and research infrastructure.
The global market value for DLS-based zeta potential measurement instruments was estimated at $320 million in 2022, with projections suggesting growth to $450 million by 2027. Key market drivers include increasing regulatory requirements for nanomaterial characterization, growing adoption of nanotechnology across industries, and continuous technological advancements improving measurement accuracy and reproducibility.
Customer demand increasingly focuses on integrated systems that combine zeta potential measurements with other characterization techniques, such as particle size analysis and molecular weight determination, offering comprehensive sample analysis capabilities in a single instrument.
Current Challenges in Zeta Potential Measurement Technologies
Despite significant advancements in zeta potential measurement using Dynamic Light Scattering (DLS), several technical challenges continue to impede accurate and reliable measurements. One of the primary limitations is the sensitivity to sample concentration. At high concentrations, multiple scattering effects can distort measurements, while extremely dilute samples may not generate sufficient signal for accurate analysis, creating a narrow operational window that restricts application versatility.
Sample stability presents another significant challenge, as particles must remain suspended and maintain consistent properties throughout the measurement period. Sedimentation, aggregation, or degradation during analysis can lead to time-dependent variations in results, particularly problematic for biological samples or unstable colloidal systems that change rapidly under measurement conditions.
The presence of large particles or contaminants disproportionately affects DLS measurements due to the relationship between scattering intensity and particle size (proportional to the sixth power of diameter). Even trace amounts of dust or aggregates can dominate signals and mask the zeta potential of the primary particles of interest, necessitating meticulous sample preparation protocols.
Conductivity and ionic strength of the dispersion medium critically influence zeta potential measurements. High ionic strength compresses the electrical double layer, potentially leading to underestimation of zeta potential values. Conversely, extremely low conductivity samples may generate excessive joule heating during measurement, causing thermal gradients that distort results.
Temperature control represents another technical hurdle, as even minor temperature fluctuations can alter particle Brownian motion, viscosity of the medium, and electrical conductivity—all parameters that directly impact zeta potential calculations. Most commercial instruments struggle to maintain precise temperature control during the application of electric fields, which generate heat.
Complex sample compositions, particularly those containing non-spherical particles, polydisperse systems, or particles with heterogeneous surface properties, challenge the fundamental assumptions of the mathematical models used in DLS analysis. Current algorithms often assume spherical, homogeneous particles, leading to interpretation errors for many real-world samples.
Electrode degradation and polarization effects during repeated measurements can alter the applied electric field, introducing systematic errors in long-term studies or when analyzing corrosive samples. The electrodes may release ions that interact with the sample, potentially altering surface charge characteristics of the particles being measured.
Finally, data interpretation remains challenging, as converting electrophoretic mobility to zeta potential relies on theoretical models with inherent assumptions. The commonly used Smoluchowski and Hückel approximations have limited applicability ranges, and selecting the appropriate model for specific sample types requires considerable expertise, often leading to inconsistent reporting across different laboratories and research groups.
Sample stability presents another significant challenge, as particles must remain suspended and maintain consistent properties throughout the measurement period. Sedimentation, aggregation, or degradation during analysis can lead to time-dependent variations in results, particularly problematic for biological samples or unstable colloidal systems that change rapidly under measurement conditions.
The presence of large particles or contaminants disproportionately affects DLS measurements due to the relationship between scattering intensity and particle size (proportional to the sixth power of diameter). Even trace amounts of dust or aggregates can dominate signals and mask the zeta potential of the primary particles of interest, necessitating meticulous sample preparation protocols.
Conductivity and ionic strength of the dispersion medium critically influence zeta potential measurements. High ionic strength compresses the electrical double layer, potentially leading to underestimation of zeta potential values. Conversely, extremely low conductivity samples may generate excessive joule heating during measurement, causing thermal gradients that distort results.
Temperature control represents another technical hurdle, as even minor temperature fluctuations can alter particle Brownian motion, viscosity of the medium, and electrical conductivity—all parameters that directly impact zeta potential calculations. Most commercial instruments struggle to maintain precise temperature control during the application of electric fields, which generate heat.
Complex sample compositions, particularly those containing non-spherical particles, polydisperse systems, or particles with heterogeneous surface properties, challenge the fundamental assumptions of the mathematical models used in DLS analysis. Current algorithms often assume spherical, homogeneous particles, leading to interpretation errors for many real-world samples.
Electrode degradation and polarization effects during repeated measurements can alter the applied electric field, introducing systematic errors in long-term studies or when analyzing corrosive samples. The electrodes may release ions that interact with the sample, potentially altering surface charge characteristics of the particles being measured.
Finally, data interpretation remains challenging, as converting electrophoretic mobility to zeta potential relies on theoretical models with inherent assumptions. The commonly used Smoluchowski and Hückel approximations have limited applicability ranges, and selecting the appropriate model for specific sample types requires considerable expertise, often leading to inconsistent reporting across different laboratories and research groups.
Existing DLS Methodologies for Zeta Potential Determination
01 Measurement techniques for zeta potential using dynamic light scattering
Dynamic light scattering (DLS) is used to measure zeta potential by analyzing the electrophoretic mobility of particles in a liquid medium. This technique involves applying an electric field to the sample and measuring the velocity of particles using scattered light. The zeta potential is calculated from the measured electrophoretic mobility using established equations. This approach provides valuable information about the stability of colloidal systems and surface charge characteristics of particles.- Measurement techniques for zeta potential using dynamic light scattering: Dynamic light scattering (DLS) is used in conjunction with electrophoretic light scattering to measure zeta potential of particles in suspension. This technique analyzes the velocity of particles under an electric field to determine their surface charge characteristics. The measurement provides information about the stability of colloidal systems and the electrostatic interactions between particles, which is crucial for understanding dispersion behavior in various applications.
- Instrumentation and apparatus for DLS zeta potential analysis: Specialized instruments have been developed for measuring zeta potential through dynamic light scattering. These systems typically include laser light sources, detectors, sample cells with electrodes, and signal processing components. Modern instruments often integrate multiple measurement capabilities, allowing for simultaneous determination of particle size distribution and zeta potential. Advanced designs focus on improving measurement accuracy, reducing sample volume requirements, and enhancing automation capabilities.
- Applications of DLS zeta potential in pharmaceutical and biomedical fields: Zeta potential measurements using dynamic light scattering are widely applied in pharmaceutical and biomedical research to characterize drug delivery systems, evaluate protein stability, and assess biocompatibility of materials. The technique helps in formulation development by monitoring the surface properties of nanoparticles, liposomes, and other drug carriers. It also provides valuable information about the interaction between biomolecules and surfaces, which is essential for developing effective therapeutic agents and medical devices.
- Environmental and industrial applications of DLS zeta potential analysis: Dynamic light scattering zeta potential measurements are utilized in environmental monitoring and industrial processes to assess colloidal stability in water treatment, mineral processing, and ceramic manufacturing. The technique helps in optimizing flocculation processes, evaluating the effectiveness of dispersants, and monitoring the behavior of particles in complex media. It provides insights into the surface properties of environmental colloids, which influence their transport, aggregation, and interaction with contaminants.
- Advanced methodologies and improvements in DLS zeta potential measurement: Recent advancements in dynamic light scattering zeta potential measurement include improved algorithms for data analysis, methods for measuring in concentrated suspensions, and techniques for non-aqueous systems. Innovations focus on extending the measurement range, enhancing sensitivity for small particles or low mobility samples, and reducing interference from multiple scattering effects. These methodological improvements enable more accurate characterization of complex colloidal systems and expand the applicability of the technique to challenging sample types.
02 Instrumentation and apparatus for DLS zeta potential analysis
Specialized instruments have been developed for measuring zeta potential via dynamic light scattering. These systems typically include laser light sources, detectors, sample cells with electrodes, and signal processing components. Modern instruments often integrate multiple measurement capabilities, allowing for simultaneous determination of particle size and zeta potential. Advanced designs incorporate temperature control, automated sample handling, and sophisticated data analysis software to enhance measurement accuracy and reproducibility.Expand Specific Solutions03 Applications of DLS zeta potential in pharmaceutical and biomedical fields
Zeta potential measurements using dynamic light scattering are widely applied in pharmaceutical and biomedical research to characterize drug delivery systems, evaluate protein stability, and assess biocompatibility of materials. This technique helps in understanding the interaction between biological molecules and nanoparticles, predicting the stability of drug formulations, and optimizing the design of therapeutic agents. The surface charge information provided by zeta potential analysis is crucial for developing effective drug delivery systems and understanding cellular uptake mechanisms.Expand Specific Solutions04 Environmental and industrial applications of DLS zeta potential
Dynamic light scattering zeta potential measurements are utilized in environmental monitoring and industrial processes to analyze colloidal stability in water treatment, assess soil and sediment properties, and optimize manufacturing processes. This technique helps in understanding the behavior of particles in natural water systems, evaluating the effectiveness of flocculants, and controlling the stability of industrial suspensions. The zeta potential data provides insights into the surface properties of particles in various environmental matrices and industrial formulations.Expand Specific Solutions05 Advanced methodologies and improvements in DLS zeta potential measurement
Recent advancements in dynamic light scattering zeta potential measurement include improved algorithms for data analysis, enhanced optical systems for better signal detection, and novel sample preparation techniques. These developments have led to increased sensitivity, better resolution for complex samples, and the ability to measure zeta potential in concentrated suspensions. Innovations also include non-invasive measurement approaches, real-time monitoring capabilities, and integration with complementary analytical techniques for comprehensive particle characterization.Expand Specific Solutions
Leading Manufacturers and Research Institutions in DLS Technology
The zeta potential measurement using dynamic light scattering market is currently in a growth phase, with increasing applications in pharmaceutical, material science, and environmental sectors. The global market size is estimated to be around $300-400 million, expanding at approximately 6-8% CAGR. Technologically, the field has reached moderate maturity with established measurement principles, but continues to evolve with innovations in accuracy and application-specific solutions. The competitive landscape is dominated by specialized analytical instrument manufacturers like Malvern Panalytical, Horiba, and Anton Paar, who offer comprehensive solutions, while emerging players such as Particle Metrix and Izon Science focus on niche applications. Academic institutions including Carnegie Mellon University and Max Planck Society contribute significantly to advancing fundamental research in this field.
Malvern Panalytical Ltd.
Technical Solution: Malvern Panalytical has developed advanced Zetasizer series instruments that utilize Dynamic Light Scattering (DLS) technology combined with electrophoretic light scattering to measure zeta potential. Their systems employ a patented M3-PALS (Mixed Mode Measurement-Phase Analysis Light Scattering) technique that allows for accurate measurements across a wide range of sample concentrations. The technology applies an electric field to the sample, causing charged particles to move, while simultaneously measuring their velocity using phase analysis light scattering. This approach enables determination of electrophoretic mobility which is then converted to zeta potential using established theoretical models such as the Smoluchowski or Hückel equations. Their latest systems incorporate automated titration capabilities for pH and concentration-dependent studies.
Strengths: Industry-leading precision with reported accuracy within ±2mV for standard samples; comprehensive software with automated quality control features; wide measurement range (3.8nm to 100μm). Weaknesses: Higher cost compared to competitors; complex operation requiring trained personnel; measurements can be affected by sample polydispersity.
Otsuka Electronics Co., Ltd.
Technical Solution: Otsuka Electronics has developed the ELSZ-2000 series that employs laser Doppler electrophoresis combined with DLS for zeta potential measurements. Their technology utilizes a unique optical configuration with a 532nm laser source and incorporates a reference beam mixing technique to enhance signal quality. The system features a specialized cell design with gold-plated electrodes to minimize contamination and ensure measurement stability. Otsuka's approach includes automatic laser alignment and focus adjustment to optimize measurement conditions for each sample. Their proprietary signal processing algorithms employ fast Fourier transform methods to analyze the frequency spectrum of scattered light, enabling accurate determination of electrophoretic mobility even for complex samples. The technology also incorporates automatic measurement sequence capabilities for temperature-dependent studies and titration experiments.
Strengths: Excellent stability for long-duration measurements; high sensitivity for low-mobility particles; robust performance with high-salt concentration samples. Weaknesses: Less market presence in Western countries limiting support availability; more complex user interface compared to leading competitors; fewer application-specific protocols available.
Key Technical Innovations in Electrophoretic Light Scattering
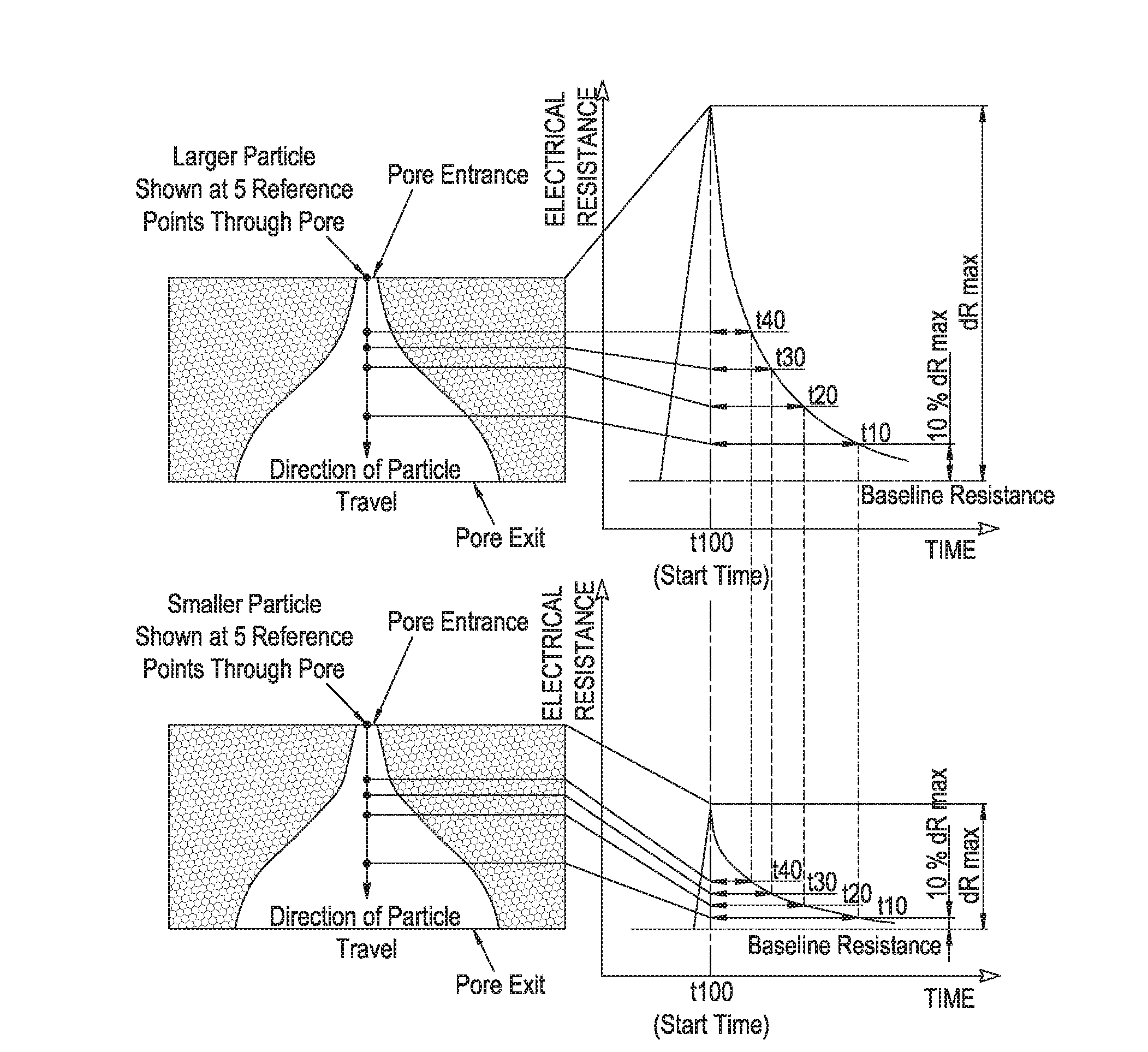
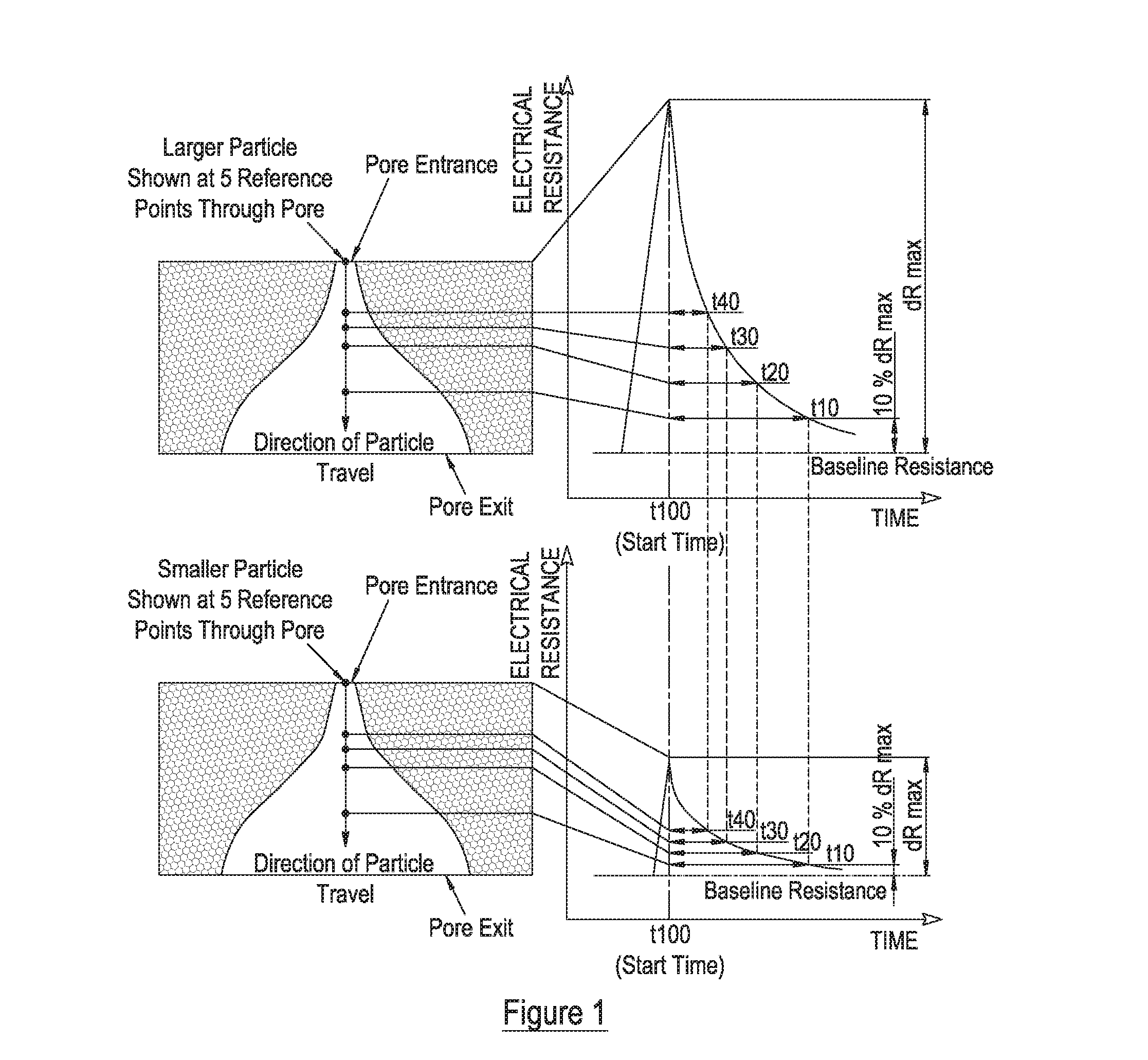
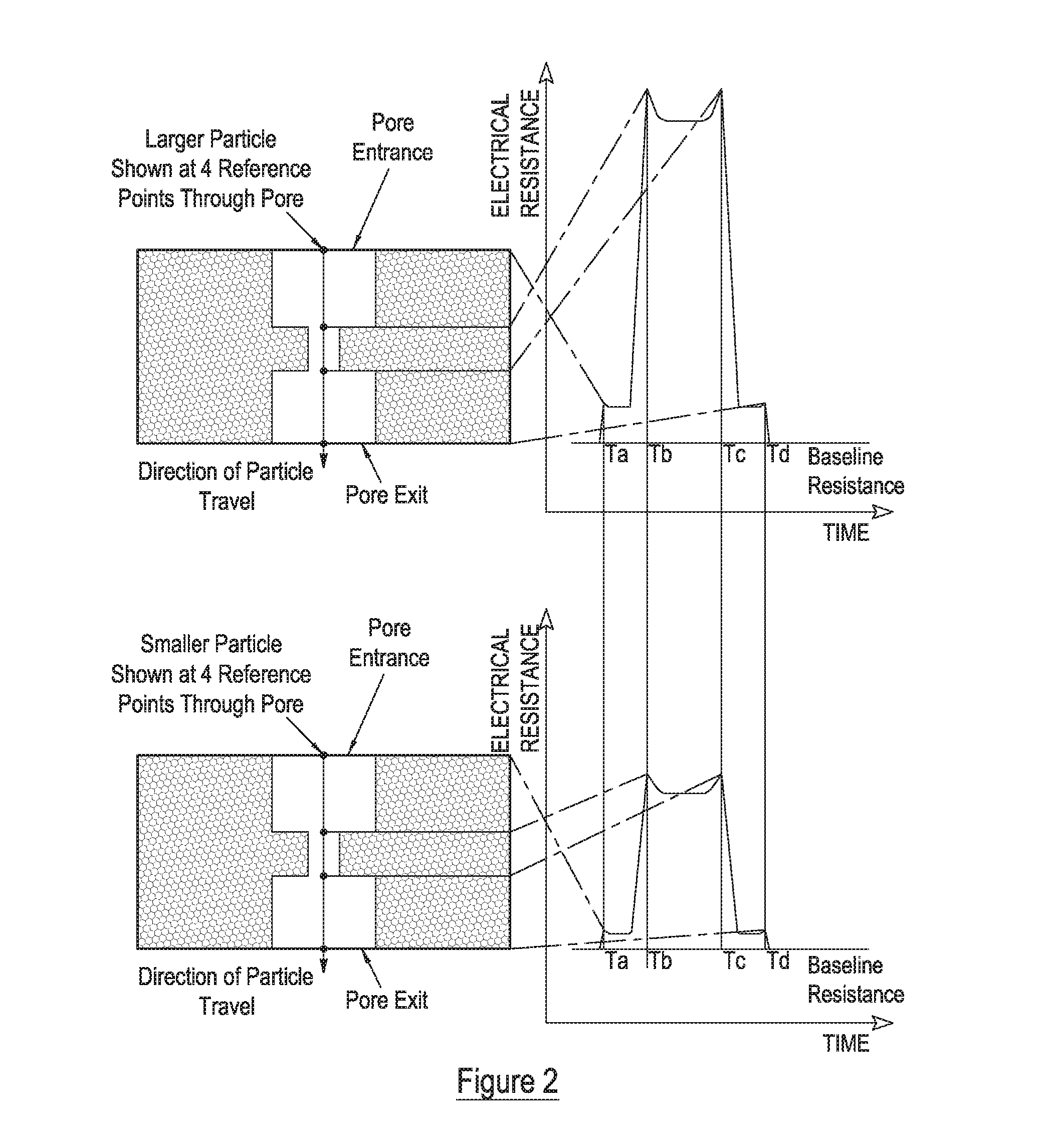
Measurement of particle charge
PatentActiveUS20160223492A1
Innovation
- A method that measures the surface charge of individual particles using a resistive pulse sensor, allowing for particle-by-particle analysis without requiring precise geometry knowledge, by resolving velocity components into convection, electrophoresis, and electro-osmosis, and using calibration particles to derive zeta potential, with adjustments for signal quality and pore zeta potential changes.
Method of obtaining stable suspensions of heterocrystals of titanium dioxide or particles of silicon dioxide and stable suspensions obtained by this method for initiation of active form of oxygen in body at use in medical forms
PatentPendingUS20240358746A1
Innovation
- A method involving aggressive acid treatment to modulate the electrostatic properties of the nanoparticle surfaces, followed by thermal treatment and cavitation homogenization to create stable suspensions with a high percentage of electronically-excited oxygen on the surface, ensuring long-term stability and catalytic activity.
Sample Preparation Techniques and Optimization
Sample preparation is a critical determinant of accurate zeta potential measurements using dynamic light scattering (DLS). The quality and reliability of results depend significantly on how samples are prepared and optimized prior to analysis. Proper preparation techniques must address several key factors including particle concentration, dispersion medium properties, and sample homogeneity.
For colloidal systems, optimal particle concentration typically ranges between 0.001% and 0.1% by volume, depending on the material's optical properties. Concentrations that are too high can lead to multiple scattering effects, while excessively dilute samples may produce insufficient signal strength. Systematic dilution studies are recommended to determine the ideal concentration range for specific sample types.
The dispersion medium's ionic strength and pH significantly influence zeta potential measurements. Buffer solutions should be carefully selected to maintain stable pH conditions throughout the measurement process. For aqueous systems, the ionic strength should typically be kept below 100 mM to avoid electrode polarization effects. When preparing samples, high-purity water (resistivity >18.2 MΩ·cm) is essential to prevent contamination that could alter surface charge characteristics.
Temperature control during sample preparation is equally important, as zeta potential values exhibit temperature dependence. Samples should be equilibrated at the measurement temperature (typically 25°C) for at least 15 minutes before analysis. This ensures thermal equilibrium and stable electrokinetic properties throughout the measurement period.
Filtration or centrifugation steps are often necessary to remove large particulates or aggregates that can interfere with DLS measurements. For submicron particles, filtration through 0.2-0.45 μm membrane filters is recommended, while for larger particles, gentle centrifugation at low g-forces may be more appropriate. These procedures should be validated to ensure they do not alter the sample's intrinsic properties.
Ultrasonic treatment represents another valuable technique for dispersing agglomerates, particularly for nanomaterials that tend to form clusters. However, sonication parameters (power, duration, mode) must be carefully optimized, as excessive sonication can alter surface properties or cause particle fragmentation. A typical approach involves short sonication bursts (30-60 seconds) with cooling intervals to prevent sample heating.
For complex biological samples or polymer dispersions, additional stabilizing agents such as surfactants or steric stabilizers may be necessary. These additives should be selected carefully to ensure they do not interfere with the measurement while providing sufficient colloidal stability. The concentration of such additives should be kept at the minimum effective level to avoid introducing artifacts in the zeta potential determination.
For colloidal systems, optimal particle concentration typically ranges between 0.001% and 0.1% by volume, depending on the material's optical properties. Concentrations that are too high can lead to multiple scattering effects, while excessively dilute samples may produce insufficient signal strength. Systematic dilution studies are recommended to determine the ideal concentration range for specific sample types.
The dispersion medium's ionic strength and pH significantly influence zeta potential measurements. Buffer solutions should be carefully selected to maintain stable pH conditions throughout the measurement process. For aqueous systems, the ionic strength should typically be kept below 100 mM to avoid electrode polarization effects. When preparing samples, high-purity water (resistivity >18.2 MΩ·cm) is essential to prevent contamination that could alter surface charge characteristics.
Temperature control during sample preparation is equally important, as zeta potential values exhibit temperature dependence. Samples should be equilibrated at the measurement temperature (typically 25°C) for at least 15 minutes before analysis. This ensures thermal equilibrium and stable electrokinetic properties throughout the measurement period.
Filtration or centrifugation steps are often necessary to remove large particulates or aggregates that can interfere with DLS measurements. For submicron particles, filtration through 0.2-0.45 μm membrane filters is recommended, while for larger particles, gentle centrifugation at low g-forces may be more appropriate. These procedures should be validated to ensure they do not alter the sample's intrinsic properties.
Ultrasonic treatment represents another valuable technique for dispersing agglomerates, particularly for nanomaterials that tend to form clusters. However, sonication parameters (power, duration, mode) must be carefully optimized, as excessive sonication can alter surface properties or cause particle fragmentation. A typical approach involves short sonication bursts (30-60 seconds) with cooling intervals to prevent sample heating.
For complex biological samples or polymer dispersions, additional stabilizing agents such as surfactants or steric stabilizers may be necessary. These additives should be selected carefully to ensure they do not interfere with the measurement while providing sufficient colloidal stability. The concentration of such additives should be kept at the minimum effective level to avoid introducing artifacts in the zeta potential determination.
Data Interpretation and Quality Control Standards
The interpretation of zeta potential data obtained through Dynamic Light Scattering (DLS) requires rigorous analytical frameworks and quality control measures to ensure reliability and reproducibility. Raw electrophoretic mobility measurements must undergo systematic conversion to zeta potential values using appropriate theoretical models such as the Smoluchowski or Hückel equations, with selection based on particle size, shape, and suspension medium characteristics.
Quality control in zeta potential measurements begins with instrument calibration using certified reference materials with known zeta potential values. Regular verification using these standards ensures measurement accuracy and system stability over time. Measurement precision should be established through statistical analysis of replicate measurements, with coefficient of variation typically maintained below 10% for reliable data.
Environmental factors significantly impact data quality and must be strictly controlled. Temperature fluctuations can alter particle Brownian motion and electrophoretic mobility, necessitating temperature stabilization within ±0.5°C during measurements. Sample conductivity should be monitored as excessive ionic strength can compress the electrical double layer and affect zeta potential readings. pH monitoring is equally critical as it directly influences surface charge density and subsequent zeta potential values.
Data filtering protocols must be implemented to identify and exclude anomalous measurements. Signal-to-noise ratio thresholds (typically >5:1) should be established, and count rate stability monitored throughout measurement cycles. Phase plots from correlogram analysis provide valuable quality indicators, with smooth, monotonic decay patterns suggesting reliable data acquisition.
Multi-angle measurements enhance data robustness by confirming consistency across different scattering angles. Discrepancies between angles may indicate sample polydispersity or the presence of large aggregates that could skew results. Time-dependent measurements are essential for systems with potential temporal instability, allowing detection of dynamic changes in colloidal stability.
Reporting standards should include comprehensive documentation of all measurement parameters: temperature, pH, conductivity, diluent composition, sample concentration, equilibration time, applied field strength, and number of measurement runs. Statistical parameters including mean, median, mode, and standard deviation should accompany all zeta potential values, providing complete characterization of distribution profiles and measurement uncertainty.
Cross-validation with complementary techniques such as electrophoretic light scattering or microelectrophoresis strengthens data reliability, particularly for complex or novel sample types where established protocols may be insufficient.
Quality control in zeta potential measurements begins with instrument calibration using certified reference materials with known zeta potential values. Regular verification using these standards ensures measurement accuracy and system stability over time. Measurement precision should be established through statistical analysis of replicate measurements, with coefficient of variation typically maintained below 10% for reliable data.
Environmental factors significantly impact data quality and must be strictly controlled. Temperature fluctuations can alter particle Brownian motion and electrophoretic mobility, necessitating temperature stabilization within ±0.5°C during measurements. Sample conductivity should be monitored as excessive ionic strength can compress the electrical double layer and affect zeta potential readings. pH monitoring is equally critical as it directly influences surface charge density and subsequent zeta potential values.
Data filtering protocols must be implemented to identify and exclude anomalous measurements. Signal-to-noise ratio thresholds (typically >5:1) should be established, and count rate stability monitored throughout measurement cycles. Phase plots from correlogram analysis provide valuable quality indicators, with smooth, monotonic decay patterns suggesting reliable data acquisition.
Multi-angle measurements enhance data robustness by confirming consistency across different scattering angles. Discrepancies between angles may indicate sample polydispersity or the presence of large aggregates that could skew results. Time-dependent measurements are essential for systems with potential temporal instability, allowing detection of dynamic changes in colloidal stability.
Reporting standards should include comprehensive documentation of all measurement parameters: temperature, pH, conductivity, diluent composition, sample concentration, equilibration time, applied field strength, and number of measurement runs. Statistical parameters including mean, median, mode, and standard deviation should accompany all zeta potential values, providing complete characterization of distribution profiles and measurement uncertainty.
Cross-validation with complementary techniques such as electrophoretic light scattering or microelectrophoresis strengthens data reliability, particularly for complex or novel sample types where established protocols may be insufficient.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!