Comparative Analysis of Batch vs Continuous Cell-free Systems
OCT 13, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Cell-free Systems Background and Objectives
Cell-free systems represent a revolutionary approach in biotechnology, emerging from the pioneering work of Nirenberg and Matthaei in the 1960s who utilized cell extracts to decipher the genetic code. These systems have evolved significantly over the past six decades, transitioning from analytical tools to sophisticated biomanufacturing platforms capable of producing complex proteins, metabolites, and other biomolecules without the constraints of cellular viability.
The fundamental principle of cell-free systems involves extracting the cellular machinery necessary for transcription and translation from living cells while eliminating cellular components that might interfere with desired biochemical processes. This creates a controlled environment where biological reactions can be manipulated with unprecedented precision, offering advantages in reaction optimization, toxic product synthesis, and rapid prototyping that traditional cell-based systems cannot match.
Recent technological advancements have dramatically enhanced the efficiency and applicability of cell-free systems, particularly in the development of both batch and continuous processing methodologies. Batch systems, characterized by finite reaction times and resource availability, have been the historical standard but face limitations in sustained productivity. Continuous systems, which feature ongoing replenishment of substrates and removal of products, represent an emerging paradigm with potential for extended reaction durations and improved yields.
The primary objective of this technical research report is to conduct a comprehensive comparative analysis of batch versus continuous cell-free systems, evaluating their respective strengths, limitations, and optimal application scenarios. We aim to identify the critical technological parameters that influence system performance, including energy regeneration mechanisms, extract preparation methods, reaction vessel design, and process control strategies.
Furthermore, this analysis seeks to establish a clear technological trajectory for cell-free systems, mapping the evolutionary path from traditional batch processes to advanced continuous operations. By examining current technological barriers and emerging solutions, we intend to forecast the future development landscape and identify promising research directions that could lead to transformative breakthroughs in biomanufacturing efficiency and scalability.
The ultimate goal is to provide strategic insights that can guide research and development investments, helping to position our organization at the forefront of this rapidly evolving field. By understanding the comparative advantages of batch and continuous cell-free systems, we can develop targeted approaches to overcome existing limitations and capitalize on emerging opportunities in pharmaceutical production, sustainable chemical manufacturing, and synthetic biology applications.
The fundamental principle of cell-free systems involves extracting the cellular machinery necessary for transcription and translation from living cells while eliminating cellular components that might interfere with desired biochemical processes. This creates a controlled environment where biological reactions can be manipulated with unprecedented precision, offering advantages in reaction optimization, toxic product synthesis, and rapid prototyping that traditional cell-based systems cannot match.
Recent technological advancements have dramatically enhanced the efficiency and applicability of cell-free systems, particularly in the development of both batch and continuous processing methodologies. Batch systems, characterized by finite reaction times and resource availability, have been the historical standard but face limitations in sustained productivity. Continuous systems, which feature ongoing replenishment of substrates and removal of products, represent an emerging paradigm with potential for extended reaction durations and improved yields.
The primary objective of this technical research report is to conduct a comprehensive comparative analysis of batch versus continuous cell-free systems, evaluating their respective strengths, limitations, and optimal application scenarios. We aim to identify the critical technological parameters that influence system performance, including energy regeneration mechanisms, extract preparation methods, reaction vessel design, and process control strategies.
Furthermore, this analysis seeks to establish a clear technological trajectory for cell-free systems, mapping the evolutionary path from traditional batch processes to advanced continuous operations. By examining current technological barriers and emerging solutions, we intend to forecast the future development landscape and identify promising research directions that could lead to transformative breakthroughs in biomanufacturing efficiency and scalability.
The ultimate goal is to provide strategic insights that can guide research and development investments, helping to position our organization at the forefront of this rapidly evolving field. By understanding the comparative advantages of batch and continuous cell-free systems, we can develop targeted approaches to overcome existing limitations and capitalize on emerging opportunities in pharmaceutical production, sustainable chemical manufacturing, and synthetic biology applications.
Market Applications and Demand Analysis
The cell-free protein synthesis (CFPS) market has witnessed significant growth in recent years, driven by increasing demand for rapid protein production across pharmaceutical, biotechnology, and research sectors. The global CFPS market was valued at approximately 250 million USD in 2022 and is projected to grow at a compound annual growth rate of 8.5% through 2030, reflecting the expanding applications and technological advancements in this field.
Batch and continuous cell-free systems serve distinct market segments with varying requirements. The batch CFPS market is primarily dominated by research institutions and small-scale biopharmaceutical companies that require flexibility and lower initial investment. This segment values the simplicity and accessibility of batch systems for protein characterization, enzyme engineering, and preliminary drug development studies.
Continuous cell-free systems are gaining traction in industrial biotechnology and large-scale pharmaceutical manufacturing where sustained production and higher yields are paramount. The continuous format market is experiencing faster growth (estimated at 10-12% annually) compared to batch systems (6-7% growth), indicating a gradual shift toward continuous processing technologies across the biomanufacturing landscape.
Therapeutic protein production represents the largest application segment for both systems, accounting for approximately 40% of the total market share. The increasing prevalence of chronic diseases and growing demand for personalized medicine have significantly boosted the need for efficient protein synthesis platforms. Diagnostic applications follow closely, comprising about 30% of the market, with point-of-care diagnostics emerging as a particularly promising area for continuous cell-free systems.
Regional analysis reveals North America as the dominant market (35% share), followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to witness the highest growth rate due to increasing investments in biotechnology infrastructure and research capabilities in countries like China, Japan, and South Korea.
End-user preferences indicate that pharmaceutical companies prioritize scalability and regulatory compliance, making continuous systems increasingly attractive despite higher initial costs. Academic and research institutions, conversely, favor batch systems for their experimental flexibility and lower entry barriers. The contract manufacturing organization (CMO) segment represents the fastest-growing end-user category, with a strong preference for hybrid systems that combine elements of both batch and continuous processing.
Market challenges include high production costs, technical complexity in system optimization, and regulatory uncertainties surrounding novel manufacturing technologies. These factors currently limit wider adoption, particularly among smaller market players and in emerging economies.
Batch and continuous cell-free systems serve distinct market segments with varying requirements. The batch CFPS market is primarily dominated by research institutions and small-scale biopharmaceutical companies that require flexibility and lower initial investment. This segment values the simplicity and accessibility of batch systems for protein characterization, enzyme engineering, and preliminary drug development studies.
Continuous cell-free systems are gaining traction in industrial biotechnology and large-scale pharmaceutical manufacturing where sustained production and higher yields are paramount. The continuous format market is experiencing faster growth (estimated at 10-12% annually) compared to batch systems (6-7% growth), indicating a gradual shift toward continuous processing technologies across the biomanufacturing landscape.
Therapeutic protein production represents the largest application segment for both systems, accounting for approximately 40% of the total market share. The increasing prevalence of chronic diseases and growing demand for personalized medicine have significantly boosted the need for efficient protein synthesis platforms. Diagnostic applications follow closely, comprising about 30% of the market, with point-of-care diagnostics emerging as a particularly promising area for continuous cell-free systems.
Regional analysis reveals North America as the dominant market (35% share), followed by Europe (30%) and Asia-Pacific (25%). However, the Asia-Pacific region is expected to witness the highest growth rate due to increasing investments in biotechnology infrastructure and research capabilities in countries like China, Japan, and South Korea.
End-user preferences indicate that pharmaceutical companies prioritize scalability and regulatory compliance, making continuous systems increasingly attractive despite higher initial costs. Academic and research institutions, conversely, favor batch systems for their experimental flexibility and lower entry barriers. The contract manufacturing organization (CMO) segment represents the fastest-growing end-user category, with a strong preference for hybrid systems that combine elements of both batch and continuous processing.
Market challenges include high production costs, technical complexity in system optimization, and regulatory uncertainties surrounding novel manufacturing technologies. These factors currently limit wider adoption, particularly among smaller market players and in emerging economies.
Current Challenges in Batch and Continuous Cell-free Systems
Despite significant advancements in cell-free systems, both batch and continuous formats face substantial technical challenges that limit their widespread industrial application. Batch cell-free systems suffer from limited reaction longevity, typically operating effectively for only 4-12 hours before productivity declines significantly. This limitation stems from the gradual depletion of energy resources, accumulation of inhibitory byproducts, and degradation of essential components such as enzymes and templates.
Resource management represents another critical challenge in batch systems. The finite supply of nucleotides, amino acids, and energy molecules necessitates precise initial loading, which often results in either resource wastage or premature reaction termination. Additionally, batch systems exhibit poor scalability characteristics, with performance frequently diminishing when reaction volumes increase beyond laboratory scale.
Continuous cell-free systems, while addressing some batch limitations through constant nutrient supply and byproduct removal, introduce their own set of challenges. The engineering complexity of maintaining precise flow rates, preventing membrane fouling, and ensuring homogeneous mixing significantly increases operational complexity and equipment requirements. These systems demand sophisticated control mechanisms to maintain optimal reaction conditions over extended periods.
Membrane-based continuous systems face particular difficulties with selective permeability. Designing membranes that effectively retain macromolecular machinery while allowing small molecule exchange remains technically challenging. Membrane fouling and protein adsorption progressively reduce system efficiency during extended operation, necessitating periodic replacement or cleaning protocols.
Both system types struggle with protein expression consistency and reproducibility. Batch systems show significant batch-to-batch variation due to extract preparation inconsistencies and sensitivity to minor procedural differences. Continuous systems face additional variability from flow rate fluctuations and gradual changes in membrane characteristics over time.
Cost considerations present substantial barriers to industrial adoption. High-quality cell extracts, nucleotides, and energy systems contribute to prohibitive operational expenses, particularly for large-scale applications. Continuous systems incur additional costs related to specialized equipment, monitoring systems, and increased operational complexity.
Regulatory challenges further complicate commercial implementation, particularly for pharmaceutical applications. The complex and variable nature of cell-free systems creates difficulties in establishing consistent quality control parameters and validation protocols that satisfy regulatory requirements. This regulatory uncertainty has slowed adoption in highly regulated industries despite the technical potential.
Resource management represents another critical challenge in batch systems. The finite supply of nucleotides, amino acids, and energy molecules necessitates precise initial loading, which often results in either resource wastage or premature reaction termination. Additionally, batch systems exhibit poor scalability characteristics, with performance frequently diminishing when reaction volumes increase beyond laboratory scale.
Continuous cell-free systems, while addressing some batch limitations through constant nutrient supply and byproduct removal, introduce their own set of challenges. The engineering complexity of maintaining precise flow rates, preventing membrane fouling, and ensuring homogeneous mixing significantly increases operational complexity and equipment requirements. These systems demand sophisticated control mechanisms to maintain optimal reaction conditions over extended periods.
Membrane-based continuous systems face particular difficulties with selective permeability. Designing membranes that effectively retain macromolecular machinery while allowing small molecule exchange remains technically challenging. Membrane fouling and protein adsorption progressively reduce system efficiency during extended operation, necessitating periodic replacement or cleaning protocols.
Both system types struggle with protein expression consistency and reproducibility. Batch systems show significant batch-to-batch variation due to extract preparation inconsistencies and sensitivity to minor procedural differences. Continuous systems face additional variability from flow rate fluctuations and gradual changes in membrane characteristics over time.
Cost considerations present substantial barriers to industrial adoption. High-quality cell extracts, nucleotides, and energy systems contribute to prohibitive operational expenses, particularly for large-scale applications. Continuous systems incur additional costs related to specialized equipment, monitoring systems, and increased operational complexity.
Regulatory challenges further complicate commercial implementation, particularly for pharmaceutical applications. The complex and variable nature of cell-free systems creates difficulties in establishing consistent quality control parameters and validation protocols that satisfy regulatory requirements. This regulatory uncertainty has slowed adoption in highly regulated industries despite the technical potential.
Comparative Technical Solutions: Batch vs Continuous Methods
01 Cell-free protein synthesis systems
Cell-free protein synthesis systems allow for the production of proteins outside of living cells. These systems typically contain all the necessary components for transcription and translation, including ribosomes, enzymes, and nucleic acids. They offer advantages such as rapid protein production, the ability to produce toxic proteins, and simplified purification processes. These systems can be derived from various organisms including bacteria, yeast, and mammalian cells, and can be optimized for specific applications in biotechnology and pharmaceutical research.- Cell-free protein synthesis systems: Cell-free protein synthesis systems allow for the production of proteins outside of living cells. These systems typically contain all the necessary components for transcription and translation, including ribosomes, enzymes, nucleotides, and amino acids. They offer advantages such as rapid protein production, the ability to produce toxic proteins, and simplified purification processes. These systems can be derived from various organisms including bacteria, yeast, and mammalian cells, each with specific applications in biotechnology and pharmaceutical research.
- Cell-free diagnostics and biosensors: Cell-free systems are being utilized for diagnostic applications and biosensor development. These systems can detect specific biomarkers, pathogens, or environmental contaminants without requiring intact cells. The technology enables rapid, sensitive, and portable detection methods that can be deployed in resource-limited settings. Cell-free diagnostic platforms often incorporate synthetic gene circuits or CRISPR-based detection systems that produce visible signals in response to target molecules, offering potential solutions for point-of-care testing and environmental monitoring.
- Cell-free metabolic engineering: Cell-free metabolic engineering involves the design and manipulation of biochemical pathways outside of living cells to produce valuable compounds. This approach allows researchers to bypass cellular constraints such as toxicity, growth requirements, and competing pathways. By reconstituting metabolic pathways in vitro, scientists can optimize reaction conditions, rapidly test pathway designs, and produce chemicals that would be challenging to synthesize in living organisms. Applications include the production of biofuels, pharmaceuticals, fine chemicals, and natural products.
- Cell-free synthetic biology platforms: Cell-free synthetic biology platforms provide controlled environments for designing and testing genetic circuits and biological parts without the complexity of living cells. These systems enable rapid prototyping of genetic constructs, characterization of regulatory elements, and optimization of gene expression. Researchers can implement complex genetic networks and study their behavior in defined conditions. The technology accelerates the design-build-test cycle in synthetic biology and facilitates the development of novel biological functions for applications in medicine, agriculture, and industry.
- Cell-free therapeutic production systems: Cell-free systems are being developed for the production of therapeutic proteins, vaccines, and other biopharmaceuticals. These systems offer advantages over traditional cell-based manufacturing, including reduced production time, simplified purification processes, and the ability to produce proteins that would be toxic to living cells. The technology enables on-demand production of personalized medicines and rapid response to emerging pathogens. Recent advances have improved the scalability and cost-effectiveness of cell-free therapeutic production, making it increasingly viable for commercial applications.
02 Cell-free diagnostics and biosensors
Cell-free systems are utilized in diagnostic applications and biosensor development. These systems can detect specific biomarkers, pathogens, or molecules of interest without requiring intact cells. They often incorporate synthetic gene circuits or enzymatic reactions that produce detectable signals in response to target analytes. Cell-free diagnostics offer advantages such as stability at room temperature, rapid results, and potential for point-of-care applications. These technologies are particularly valuable for resource-limited settings and rapid disease detection.Expand Specific Solutions03 Cell-free metabolic engineering
Cell-free metabolic engineering involves the design and manipulation of biochemical pathways outside of living cells. This approach allows researchers to bypass cellular constraints and directly control reaction conditions for the production of valuable compounds. By reconstituting metabolic pathways in vitro, researchers can optimize enzyme concentrations, cofactor regeneration, and substrate availability. These systems are particularly useful for producing chemicals that might be toxic to living cells or require reaction conditions incompatible with cellular life, enabling more efficient bioproduction processes.Expand Specific Solutions04 Cell-free synthetic biology platforms
Cell-free synthetic biology platforms provide controlled environments for designing and testing genetic circuits and biological parts. These systems allow for rapid prototyping of genetic constructs without the need for time-consuming cell transformation and cultivation steps. They enable the study of complex biological processes in simplified contexts and facilitate the development of novel biological functions. Cell-free platforms can be customized with specific components to achieve desired outcomes and are valuable tools for education, research, and biotechnological applications.Expand Specific Solutions05 Cell-free therapeutic production systems
Cell-free systems are employed for the production of therapeutic proteins, vaccines, and other biopharmaceuticals. These systems offer advantages such as rapid production timelines, reduced risk of contamination, and the ability to produce complex or modified proteins. They can be optimized for high-yield production of specific therapeutic molecules and can incorporate non-natural amino acids or post-translational modifications. Cell-free therapeutic production systems are particularly valuable for personalized medicine applications and rapid response to emerging health threats.Expand Specific Solutions
Key Innovations in Cell-free Expression Systems
Intensified Perfusion Production Method
PatentInactiveUS20080206819A1
Innovation
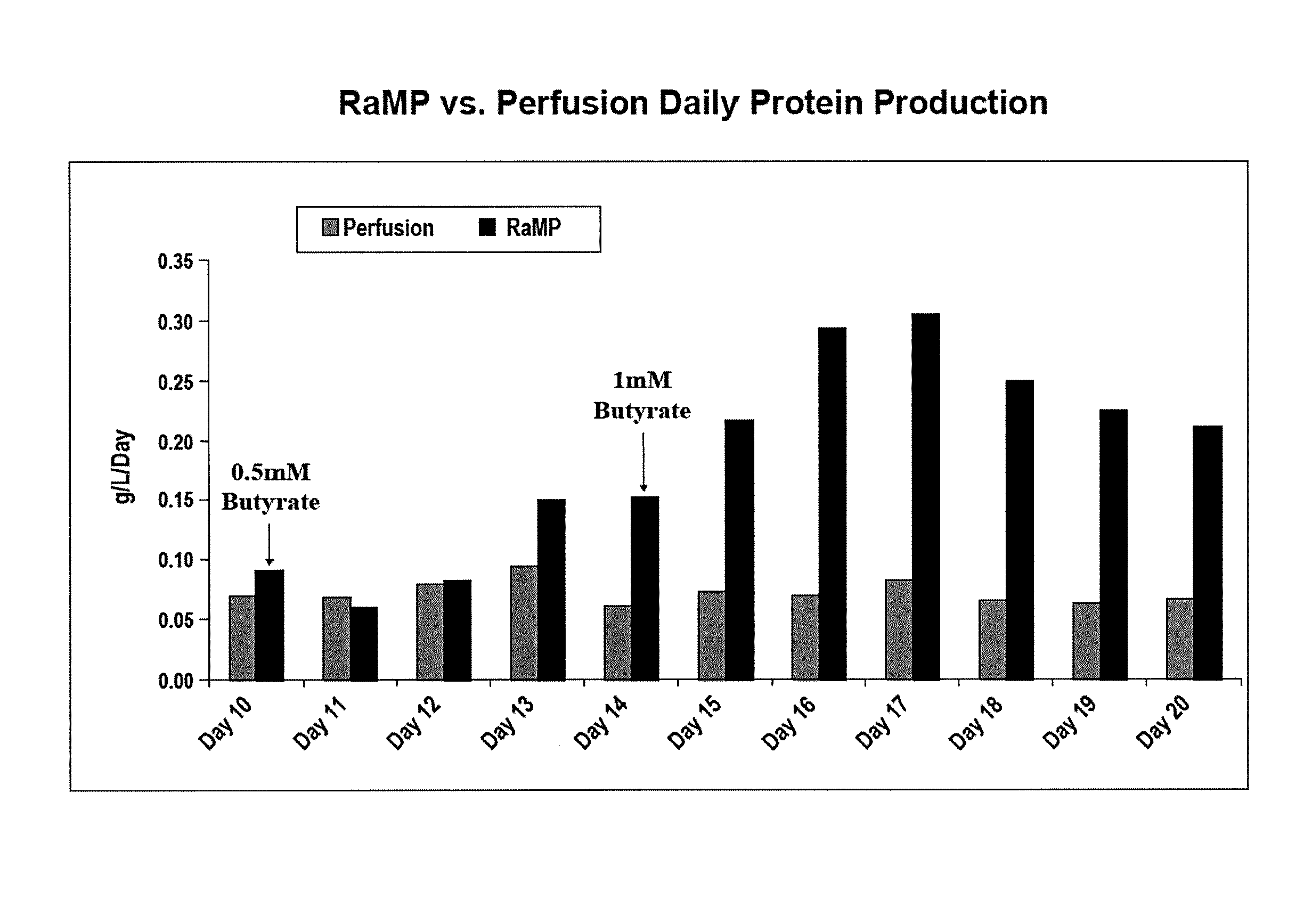
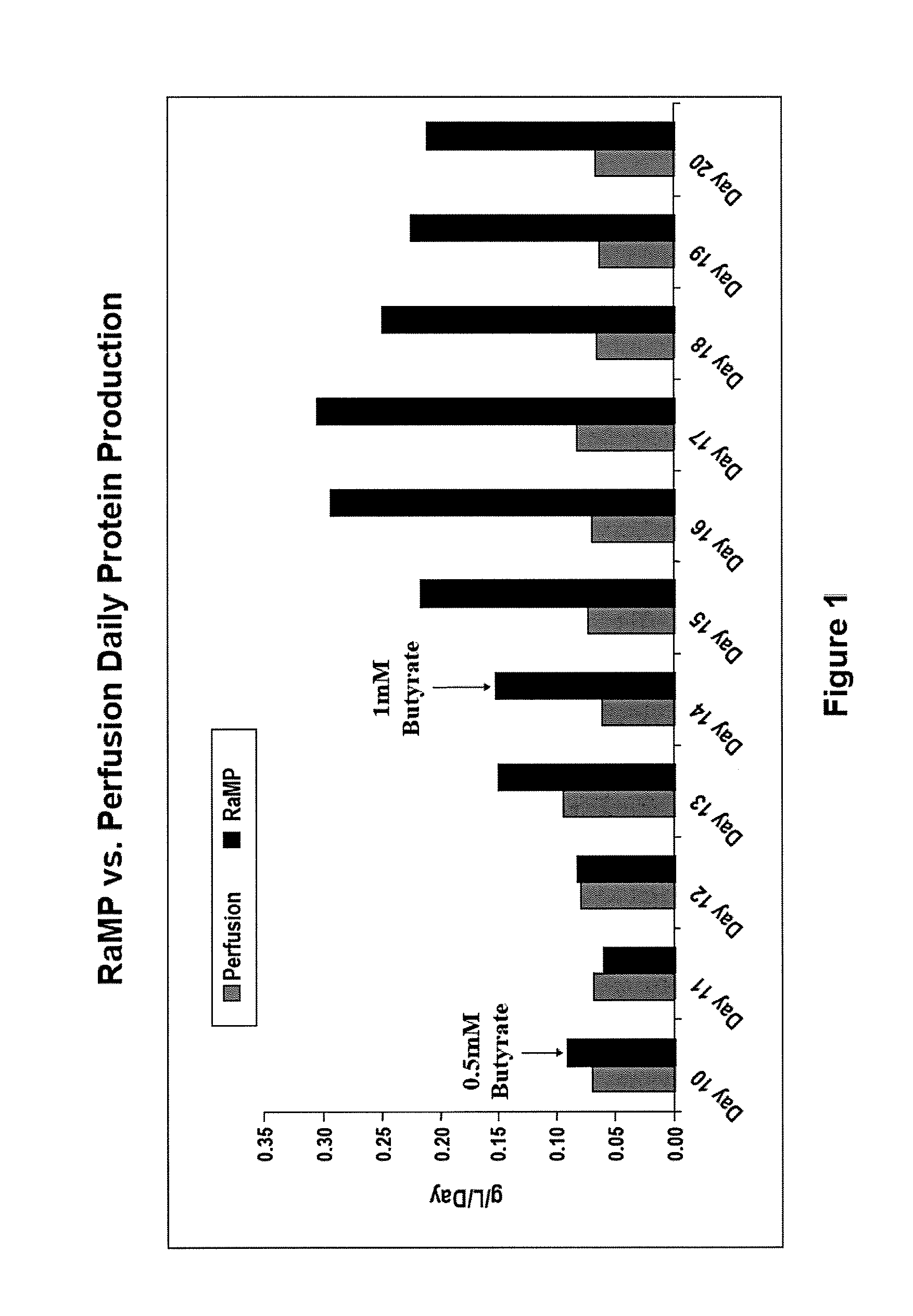
- A novel process for perfusion cell culture that maintains high cell viability and density by gradually introducing induction agents, such as sodium butyrate, into the culture medium, allowing for extended production periods without significant cell death, and utilizing a pre-sterilized disposable bioreactor with agitation and filtration to concentrate the protein of interest.
Method for the production of proteins and peptides
PatentInactiveUS20130017576A1
Innovation
- A method using a cell-free lysate from eukaryotic cells with functional microsomal vesicles, where nucleic acid coding for antibody fragments with signal sequences is added, allowing cotranslational translocation into microsomal vesicles, followed by detergent-mediated release of the synthesized proteins, eliminating the need for chemical pre-treatment and chaperones, and enabling production in a completely cell-free medium.
Scalability and Process Engineering Considerations
Scaling cell-free systems from laboratory to industrial applications presents distinct challenges for both batch and continuous processes. Batch systems, while simpler to implement initially, face significant hurdles when scaled up. The primary limitation stems from the finite reaction lifetime (typically 4-12 hours) due to resource depletion and byproduct accumulation. This necessitates larger reaction vessels and increased reagent quantities, leading to exponential cost increases without proportional productivity gains. Additionally, heat and mass transfer become problematic in larger batch reactors, potentially causing temperature gradients and uneven reaction conditions that compromise product quality.
Continuous cell-free systems offer more promising scalability pathways through steady-state operations. By continuously supplying fresh reagents and removing byproducts, these systems can maintain productivity for extended periods—some experimental setups have demonstrated stable operation for over 100 hours. This extended operational window significantly improves volumetric productivity and resource utilization efficiency. The continuous format also enables more precise process control, with real-time monitoring and adjustment capabilities that enhance product consistency.
Engineering considerations for batch scale-up focus on reactor design optimization, including improved mixing strategies and temperature control systems. Computational fluid dynamics modeling has proven valuable in predicting and mitigating potential scale-up issues. For batch processes, single-use technologies have emerged as a flexible solution that reduces cleaning validation requirements and cross-contamination risks.
Continuous systems require more sophisticated engineering approaches, including membrane technology development for selective component retention and removal. Hollow fiber reactors and microfluidic platforms have shown particular promise, allowing for compartmentalization that mimics cellular organization. Process intensification strategies, such as immobilized enzyme systems and multi-stage reaction cascades, further enhance continuous operation efficiency.
Economic analyses indicate that while continuous systems have higher initial capital expenditure requirements, their superior operational efficiency often results in lower cost-per-unit of product over extended production campaigns. This advantage becomes particularly pronounced for high-value biopharmaceuticals and enzymes where product quality consistency commands premium pricing.
Recent advances in process analytical technology (PAT) have significantly improved monitoring capabilities for both formats, though continuous systems benefit more substantially from real-time quality control implementation. The integration of automation and machine learning algorithms for process optimization represents the frontier of cell-free system engineering, potentially enabling autonomous operation with minimal human intervention.
Continuous cell-free systems offer more promising scalability pathways through steady-state operations. By continuously supplying fresh reagents and removing byproducts, these systems can maintain productivity for extended periods—some experimental setups have demonstrated stable operation for over 100 hours. This extended operational window significantly improves volumetric productivity and resource utilization efficiency. The continuous format also enables more precise process control, with real-time monitoring and adjustment capabilities that enhance product consistency.
Engineering considerations for batch scale-up focus on reactor design optimization, including improved mixing strategies and temperature control systems. Computational fluid dynamics modeling has proven valuable in predicting and mitigating potential scale-up issues. For batch processes, single-use technologies have emerged as a flexible solution that reduces cleaning validation requirements and cross-contamination risks.
Continuous systems require more sophisticated engineering approaches, including membrane technology development for selective component retention and removal. Hollow fiber reactors and microfluidic platforms have shown particular promise, allowing for compartmentalization that mimics cellular organization. Process intensification strategies, such as immobilized enzyme systems and multi-stage reaction cascades, further enhance continuous operation efficiency.
Economic analyses indicate that while continuous systems have higher initial capital expenditure requirements, their superior operational efficiency often results in lower cost-per-unit of product over extended production campaigns. This advantage becomes particularly pronounced for high-value biopharmaceuticals and enzymes where product quality consistency commands premium pricing.
Recent advances in process analytical technology (PAT) have significantly improved monitoring capabilities for both formats, though continuous systems benefit more substantially from real-time quality control implementation. The integration of automation and machine learning algorithms for process optimization represents the frontier of cell-free system engineering, potentially enabling autonomous operation with minimal human intervention.
Economic Feasibility and Commercialization Pathways
The economic viability of cell-free systems represents a critical factor in their adoption across various industries. Batch processing systems traditionally offer lower initial capital investment but higher operational costs per unit of product. In contrast, continuous cell-free systems require significant upfront investment in specialized equipment and infrastructure, yet deliver substantial cost advantages at scale through improved resource utilization and reduced labor requirements.
Cost analysis reveals that batch systems typically incur expenses of $50-100 per gram of protein produced, while optimized continuous systems can potentially reduce this to $15-30 per gram. This cost differential becomes particularly significant in large-scale production scenarios, where continuous systems demonstrate superior economic efficiency beyond certain production thresholds, typically around 500-1000 grams of target protein annually.
Market entry strategies differ substantially between these approaches. Batch systems present lower barriers to entry, making them suitable for startups and academic institutions with limited capital. Continuous systems align better with established biopharmaceutical companies possessing substantial investment capabilities and existing infrastructure that can be adapted.
The commercialization timeline also varies significantly. Batch systems can typically reach market within 1-3 years, while continuous systems often require 3-5 years for full implementation and regulatory approval. This temporal advantage has positioned batch systems as the predominant commercial approach to date, despite the long-term economic advantages of continuous processing.
Regulatory considerations further impact commercialization pathways. Batch systems benefit from established regulatory frameworks and precedents, whereas continuous systems face additional scrutiny regarding process validation and quality control. However, recent regulatory initiatives, particularly from the FDA and EMA, have begun encouraging continuous manufacturing adoption through expedited review programs.
Investment trends indicate growing venture capital interest in continuous cell-free technologies, with funding increasing from approximately $120 million in 2018 to over $450 million in 2022. This reflects recognition of the long-term economic advantages and sustainability benefits of continuous processing, despite higher initial capital requirements.
The commercialization landscape is evolving toward hybrid approaches that leverage the flexibility of batch systems during development phases while transitioning to continuous processing for commercial-scale production. This strategy optimizes economic outcomes across the product lifecycle and represents a pragmatic pathway to market that balances immediate feasibility with long-term economic advantages.
Cost analysis reveals that batch systems typically incur expenses of $50-100 per gram of protein produced, while optimized continuous systems can potentially reduce this to $15-30 per gram. This cost differential becomes particularly significant in large-scale production scenarios, where continuous systems demonstrate superior economic efficiency beyond certain production thresholds, typically around 500-1000 grams of target protein annually.
Market entry strategies differ substantially between these approaches. Batch systems present lower barriers to entry, making them suitable for startups and academic institutions with limited capital. Continuous systems align better with established biopharmaceutical companies possessing substantial investment capabilities and existing infrastructure that can be adapted.
The commercialization timeline also varies significantly. Batch systems can typically reach market within 1-3 years, while continuous systems often require 3-5 years for full implementation and regulatory approval. This temporal advantage has positioned batch systems as the predominant commercial approach to date, despite the long-term economic advantages of continuous processing.
Regulatory considerations further impact commercialization pathways. Batch systems benefit from established regulatory frameworks and precedents, whereas continuous systems face additional scrutiny regarding process validation and quality control. However, recent regulatory initiatives, particularly from the FDA and EMA, have begun encouraging continuous manufacturing adoption through expedited review programs.
Investment trends indicate growing venture capital interest in continuous cell-free technologies, with funding increasing from approximately $120 million in 2018 to over $450 million in 2022. This reflects recognition of the long-term economic advantages and sustainability benefits of continuous processing, despite higher initial capital requirements.
The commercialization landscape is evolving toward hybrid approaches that leverage the flexibility of batch systems during development phases while transitioning to continuous processing for commercial-scale production. This strategy optimizes economic outcomes across the product lifecycle and represents a pragmatic pathway to market that balances immediate feasibility with long-term economic advantages.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!