Comparative study of lepidolite and petalite in lithium market trends
AUG 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Source Overview
Lithium, a critical component in modern energy storage technologies, is sourced primarily from two types of deposits: brine and hard rock. Among hard rock sources, lepidolite and petalite are two significant lithium-bearing minerals that have gained attention in recent years due to the growing demand for lithium in various industries, particularly in the production of lithium-ion batteries.
Lepidolite, a lilac-gray or rose-colored phyllosilicate mineral, is a member of the mica group. It typically contains between 3.5% and 7.7% lithium oxide by weight. Lepidolite deposits are found in various locations worldwide, including Portugal, Brazil, and parts of Africa. The mineral's distinctive color and relatively high lithium content make it an attractive source for lithium extraction.
Petalite, on the other hand, is a lithium aluminum silicate mineral that appears as colorless to pale pink crystals. It contains approximately 2.0% to 4.7% lithium oxide by weight. Significant petalite deposits have been identified in Zimbabwe, Canada, and Australia. While petalite has a lower lithium content compared to lepidolite, it often occurs in larger, more concentrated deposits, which can make extraction more economically viable in certain circumstances.
Both minerals play crucial roles in diversifying the global lithium supply chain, which has traditionally been dominated by brine sources in South America. The increasing interest in hard rock lithium sources, including lepidolite and petalite, is driven by several factors. These include the need for supply chain resilience, the potential for faster production ramp-up compared to brine operations, and the ability to produce high-purity lithium compounds suitable for battery-grade applications.
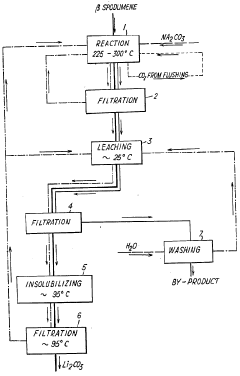
The extraction processes for lepidolite and petalite differ from those used in brine operations. Hard rock lithium extraction typically involves mining, crushing, and processing the ore through various metallurgical techniques. These may include roasting, leaching, and chemical conversion to produce lithium carbonate or lithium hydroxide. While these processes can be more energy-intensive than brine extraction, they often offer advantages in terms of production speed and the ability to operate in diverse geographical locations.
As the global demand for lithium continues to surge, driven primarily by the electric vehicle and renewable energy storage sectors, the importance of lepidolite and petalite as lithium sources is likely to grow. This trend is prompting increased exploration activities and technological innovations aimed at improving the efficiency and sustainability of hard rock lithium extraction methods.
Lepidolite, a lilac-gray or rose-colored phyllosilicate mineral, is a member of the mica group. It typically contains between 3.5% and 7.7% lithium oxide by weight. Lepidolite deposits are found in various locations worldwide, including Portugal, Brazil, and parts of Africa. The mineral's distinctive color and relatively high lithium content make it an attractive source for lithium extraction.
Petalite, on the other hand, is a lithium aluminum silicate mineral that appears as colorless to pale pink crystals. It contains approximately 2.0% to 4.7% lithium oxide by weight. Significant petalite deposits have been identified in Zimbabwe, Canada, and Australia. While petalite has a lower lithium content compared to lepidolite, it often occurs in larger, more concentrated deposits, which can make extraction more economically viable in certain circumstances.
Both minerals play crucial roles in diversifying the global lithium supply chain, which has traditionally been dominated by brine sources in South America. The increasing interest in hard rock lithium sources, including lepidolite and petalite, is driven by several factors. These include the need for supply chain resilience, the potential for faster production ramp-up compared to brine operations, and the ability to produce high-purity lithium compounds suitable for battery-grade applications.
The extraction processes for lepidolite and petalite differ from those used in brine operations. Hard rock lithium extraction typically involves mining, crushing, and processing the ore through various metallurgical techniques. These may include roasting, leaching, and chemical conversion to produce lithium carbonate or lithium hydroxide. While these processes can be more energy-intensive than brine extraction, they often offer advantages in terms of production speed and the ability to operate in diverse geographical locations.
As the global demand for lithium continues to surge, driven primarily by the electric vehicle and renewable energy storage sectors, the importance of lepidolite and petalite as lithium sources is likely to grow. This trend is prompting increased exploration activities and technological innovations aimed at improving the efficiency and sustainability of hard rock lithium extraction methods.
Market Demand Analysis
The lithium market has experienced significant growth in recent years, driven primarily by the increasing demand for lithium-ion batteries in electric vehicles and energy storage systems. This surge in demand has led to a heightened interest in various lithium sources, with lepidolite and petalite emerging as important alternatives to traditional brine and spodumene deposits.
Market analysis indicates that the global lithium market is expected to continue its upward trajectory, with projections suggesting a compound annual growth rate (CAGR) of over 10% in the coming years. This growth is largely attributed to the rapid adoption of electric vehicles worldwide, as governments and consumers alike push for more sustainable transportation options.
Lepidolite and petalite, both lithium-bearing minerals, have garnered increased attention from industry players due to their potential to diversify lithium supply chains. Lepidolite, a lithium-rich mica mineral, has seen growing interest due to its relatively high lithium content and the development of new extraction technologies. Petalite, a lithium aluminum silicate, has also gained traction as a viable lithium source, particularly in regions where traditional lithium resources are scarce.
The demand for these minerals is closely tied to the overall lithium market trends. As battery manufacturers seek to secure stable and diverse lithium supplies, both lepidolite and petalite are becoming increasingly attractive options. This shift is partly driven by concerns over the concentration of lithium production in a few key regions and the desire to reduce dependency on any single source.
In terms of market segmentation, the automotive sector remains the primary driver of lithium demand, accounting for a substantial portion of the market. However, other sectors such as consumer electronics and grid energy storage are also contributing to the growing demand for lithium, and by extension, for lepidolite and petalite.
Geographically, the demand for these minerals is not uniformly distributed. Regions with established battery manufacturing capabilities, such as Asia-Pacific, are showing particularly strong interest in diversifying their lithium sources. Meanwhile, countries with significant lepidolite or petalite deposits are exploring opportunities to develop these resources to meet the growing global demand.
The market dynamics for lepidolite and petalite are also influenced by factors such as extraction costs, processing technologies, and environmental considerations. As the lithium industry continues to evolve, there is a growing emphasis on sustainable and environmentally friendly extraction methods, which could further impact the market potential of these minerals.
In conclusion, the market demand for lepidolite and petalite is closely intertwined with the broader lithium market trends. As the global push for electrification continues, these minerals are likely to play an increasingly important role in meeting the growing demand for lithium, offering potential alternatives to traditional sources and contributing to a more diverse and resilient lithium supply chain.
Market analysis indicates that the global lithium market is expected to continue its upward trajectory, with projections suggesting a compound annual growth rate (CAGR) of over 10% in the coming years. This growth is largely attributed to the rapid adoption of electric vehicles worldwide, as governments and consumers alike push for more sustainable transportation options.
Lepidolite and petalite, both lithium-bearing minerals, have garnered increased attention from industry players due to their potential to diversify lithium supply chains. Lepidolite, a lithium-rich mica mineral, has seen growing interest due to its relatively high lithium content and the development of new extraction technologies. Petalite, a lithium aluminum silicate, has also gained traction as a viable lithium source, particularly in regions where traditional lithium resources are scarce.
The demand for these minerals is closely tied to the overall lithium market trends. As battery manufacturers seek to secure stable and diverse lithium supplies, both lepidolite and petalite are becoming increasingly attractive options. This shift is partly driven by concerns over the concentration of lithium production in a few key regions and the desire to reduce dependency on any single source.
In terms of market segmentation, the automotive sector remains the primary driver of lithium demand, accounting for a substantial portion of the market. However, other sectors such as consumer electronics and grid energy storage are also contributing to the growing demand for lithium, and by extension, for lepidolite and petalite.
Geographically, the demand for these minerals is not uniformly distributed. Regions with established battery manufacturing capabilities, such as Asia-Pacific, are showing particularly strong interest in diversifying their lithium sources. Meanwhile, countries with significant lepidolite or petalite deposits are exploring opportunities to develop these resources to meet the growing global demand.
The market dynamics for lepidolite and petalite are also influenced by factors such as extraction costs, processing technologies, and environmental considerations. As the lithium industry continues to evolve, there is a growing emphasis on sustainable and environmentally friendly extraction methods, which could further impact the market potential of these minerals.
In conclusion, the market demand for lepidolite and petalite is closely intertwined with the broader lithium market trends. As the global push for electrification continues, these minerals are likely to play an increasingly important role in meeting the growing demand for lithium, offering potential alternatives to traditional sources and contributing to a more diverse and resilient lithium supply chain.
Lepidolite vs Petalite
Lepidolite and petalite are two significant lithium-bearing minerals that play crucial roles in the global lithium market. Both minerals have distinct characteristics that influence their extraction processes, market demand, and overall economic viability.
Lepidolite, a lithium-rich mica mineral, typically contains 1.5% to 4% lithium oxide. It is often found in pegmatite deposits and can be identified by its purple to pink coloration. Lepidolite's complex crystal structure makes lithium extraction more challenging compared to other lithium sources. However, recent technological advancements have improved the efficiency of lepidolite processing, making it an increasingly attractive option for lithium production.
Petalite, on the other hand, is a lithium aluminum silicate mineral with a lithium oxide content ranging from 2% to 4.5%. It is characterized by its white to grayish appearance and is also commonly found in pegmatite deposits. Petalite has a simpler crystal structure compared to lepidolite, which generally allows for easier lithium extraction.
In terms of market trends, both minerals have seen increased interest due to the growing demand for lithium in various industries, particularly in the electric vehicle and energy storage sectors. However, their market positions differ based on several factors. Lepidolite has gained traction in recent years due to its relatively abundant deposits and the development of more efficient extraction technologies. This has led to increased exploration and investment in lepidolite projects worldwide.
Petalite, while less abundant than lepidolite, has maintained a strong position in the market due to its higher lithium content and easier processing. It is particularly favored in the glass and ceramics industries, where its low iron content is advantageous. The petalite market has seen steady growth, albeit at a slower pace compared to lepidolite.
The choice between lepidolite and petalite for lithium production often depends on factors such as deposit size, extraction costs, and end-use applications. Lepidolite projects typically require larger-scale operations to be economically viable, while petalite can be profitable even in smaller deposits. This difference has influenced investment patterns and project development strategies in the lithium mining sector.
As the lithium market continues to evolve, both lepidolite and petalite are expected to play important roles in meeting the growing global demand for lithium. Ongoing research and development efforts are focused on improving extraction efficiencies and reducing production costs for both minerals, which could further impact their relative market positions in the future.
Lepidolite, a lithium-rich mica mineral, typically contains 1.5% to 4% lithium oxide. It is often found in pegmatite deposits and can be identified by its purple to pink coloration. Lepidolite's complex crystal structure makes lithium extraction more challenging compared to other lithium sources. However, recent technological advancements have improved the efficiency of lepidolite processing, making it an increasingly attractive option for lithium production.
Petalite, on the other hand, is a lithium aluminum silicate mineral with a lithium oxide content ranging from 2% to 4.5%. It is characterized by its white to grayish appearance and is also commonly found in pegmatite deposits. Petalite has a simpler crystal structure compared to lepidolite, which generally allows for easier lithium extraction.
In terms of market trends, both minerals have seen increased interest due to the growing demand for lithium in various industries, particularly in the electric vehicle and energy storage sectors. However, their market positions differ based on several factors. Lepidolite has gained traction in recent years due to its relatively abundant deposits and the development of more efficient extraction technologies. This has led to increased exploration and investment in lepidolite projects worldwide.
Petalite, while less abundant than lepidolite, has maintained a strong position in the market due to its higher lithium content and easier processing. It is particularly favored in the glass and ceramics industries, where its low iron content is advantageous. The petalite market has seen steady growth, albeit at a slower pace compared to lepidolite.
The choice between lepidolite and petalite for lithium production often depends on factors such as deposit size, extraction costs, and end-use applications. Lepidolite projects typically require larger-scale operations to be economically viable, while petalite can be profitable even in smaller deposits. This difference has influenced investment patterns and project development strategies in the lithium mining sector.
As the lithium market continues to evolve, both lepidolite and petalite are expected to play important roles in meeting the growing global demand for lithium. Ongoing research and development efforts are focused on improving extraction efficiencies and reducing production costs for both minerals, which could further impact their relative market positions in the future.
Current Extraction Methods
01 Lithium extraction from lepidolite and petalite
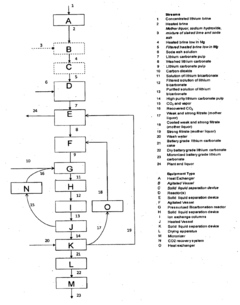
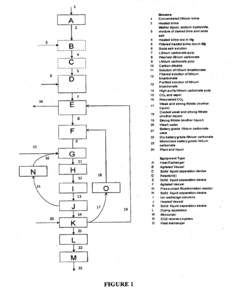
Processes for extracting lithium from lepidolite and petalite ores. These methods typically involve crushing, roasting, leaching, and purification steps to obtain lithium compounds. The extraction techniques aim to improve efficiency and yield in lithium production from these mineral sources.- Lithium extraction from lepidolite and petalite: Processes for extracting lithium from lepidolite and petalite ores. These methods typically involve crushing, roasting, leaching, and purification steps to obtain lithium compounds. The extraction techniques aim to improve efficiency and yield of lithium recovery from these mineral sources.
- Ceramic and glass applications: Utilization of lepidolite and petalite in ceramic and glass manufacturing. These minerals are used as fluxes and raw materials in the production of various ceramic and glass products, contributing to improved properties such as thermal shock resistance and reduced thermal expansion.
- Battery and energy storage applications: Use of lepidolite and petalite as sources of lithium for battery production, particularly in the manufacturing of lithium-ion batteries. These minerals are processed to obtain lithium compounds suitable for use in energy storage devices, contributing to the development of advanced battery technologies.
- Environmental remediation and waste treatment: Application of lepidolite and petalite in environmental remediation processes. These minerals are used in the treatment of contaminated water and soil, leveraging their ion-exchange properties to remove pollutants and heavy metals from the environment.
- Composite materials and coatings: Incorporation of lepidolite and petalite in the development of composite materials and coatings. These minerals are used to enhance properties such as thermal insulation, fire resistance, and durability in various industrial and construction applications.
02 Ceramic and glass applications
Utilization of lepidolite and petalite in ceramic and glass manufacturing. These minerals are used as fluxes and raw materials in the production of various ceramic products and specialized glasses. Their unique properties contribute to improved thermal shock resistance and reduced thermal expansion in the final products.Expand Specific Solutions03 Battery and energy storage applications
Development of battery and energy storage technologies using lepidolite and petalite as lithium sources. These minerals are processed to produce lithium compounds used in the manufacture of lithium-ion batteries and other energy storage devices, contributing to advancements in renewable energy and electric vehicle industries.Expand Specific Solutions04 Environmental remediation and waste treatment
Application of lepidolite and petalite in environmental remediation and waste treatment processes. These minerals are used in the development of adsorbents and ion exchange materials for removing contaminants from water and soil, as well as in the treatment of industrial waste streams.Expand Specific Solutions05 Jewelry and ornamental uses
Utilization of lepidolite and petalite in jewelry making and ornamental applications. These minerals are valued for their aesthetic properties, including color and luster, and are used in the creation of gemstones, beads, and decorative objects. Techniques for cutting, polishing, and enhancing these minerals for ornamental purposes are developed.Expand Specific Solutions
Key Industry Players
The comparative study of lepidolite and petalite in lithium market trends reveals a competitive landscape shaped by increasing demand for lithium in battery technologies. The industry is in a growth phase, with the global lithium market expected to expand significantly in the coming years. While both lepidolite and petalite are viable lithium sources, their technological maturity differs. Companies like Novalith Technologies and Yichun Yinli New Energy are advancing lepidolite processing, while others such as Redwood Materials and BASF are focusing on broader lithium extraction and recycling technologies. Research institutions like Central South University and Korea Institute of Geoscience & Mineral Resources are contributing to the development of more efficient extraction methods for both minerals.
Novalith Technologies Pty Ltd.
Technical Solution: Novalith Technologies has developed a novel lithium extraction process that can be applied to both lepidolite and petalite ores. Their technology, called LiCAL™, uses carbon dioxide as a reagent to selectively leach lithium from the ore[7]. This process significantly reduces the environmental impact compared to traditional acid-based extraction methods. The LiCAL™ technology can produce battery-grade lithium chemicals directly from the ore, potentially streamlining the supply chain[8]. Novalith's approach is designed to be more cost-effective and environmentally sustainable, which could influence market preferences between lepidolite and petalite sources based on their amenability to this process[9].
Strengths: Low environmental impact, cost-effective, direct production of battery-grade lithium. Weaknesses: Relatively new technology, may require further optimization for different ore types.
BASF Corp.
Technical Solution: BASF has developed a proprietary process for lithium extraction from both lepidolite and petalite ores, focusing on improving efficiency and reducing environmental impact. Their technology utilizes a combination of mechanical and chemical processes to extract lithium from these ores[4]. The process involves crushing and grinding the ore, followed by a series of chemical treatments to selectively extract lithium. BASF's method is designed to maximize lithium recovery while minimizing water usage and chemical consumption[5]. Additionally, the company has invested in research to develop more efficient lithium-ion battery materials, which could influence the demand for lithium from various sources, including lepidolite and petalite[6].
Strengths: Efficient extraction process, reduced environmental footprint, integrated approach from extraction to battery materials. Weaknesses: May require significant capital investment for large-scale implementation, process efficiency may vary depending on ore quality.
Innovative Extraction Tech
Process for producing lithium carbonate from concentrated lithium brine
PatentInactiveUS20180320247A1
Innovation
- A method involving the precipitation of impure lithium carbonate with soda ash, followed by a lithium bicarbonation step, and subsequent separation of impurities using ion selective mediums to remove boron and other impurities, allowing for the precipitation of high-purity lithium carbonate without the need for extensive boron removal processes.
Production of lithium salts
PatentInactiveGB1002410A
Innovation
- A process involving the hydrothermal reaction of calcined lithium-bearing alumino silicates with aqueous sodium bicarbonate or sodium sesquicarbonate under controlled conditions to produce a slurry containing lithium carbonate and sodium aluminosilicate, followed by straightforward recovery procedures, allowing for the production of lithium carbonate and other salts with minimal impurities and reduced chemical consumption.
Environmental Impact
The environmental impact of lithium extraction from lepidolite and petalite sources is a critical consideration in the comparative study of these minerals in lithium market trends. Both minerals present unique challenges and opportunities in terms of their environmental footprint throughout the mining and processing stages.
Lepidolite mining typically involves open-pit extraction methods, which can lead to significant land disturbance and habitat destruction. The process often requires the removal of large amounts of overburden, potentially affecting local ecosystems and biodiversity. However, lepidolite deposits are often found in areas with existing mining infrastructure, which can minimize the need for new development and associated environmental disruption.
In contrast, petalite mining may have a smaller initial footprint due to the mineral's higher lithium content, potentially requiring less material to be extracted for equivalent lithium yields. This could translate to reduced land disturbance and lower volumes of waste rock. Nevertheless, petalite deposits may be located in more remote or environmentally sensitive areas, necessitating careful consideration of ecosystem impacts and mitigation strategies.
Water usage and management are crucial factors in the environmental assessment of both minerals. Lepidolite processing typically requires significant water resources for mineral separation and chemical treatment. This can strain local water supplies, particularly in arid regions where many lithium deposits are found. Petalite processing, while also water-intensive, may offer opportunities for more efficient water use due to its simpler mineral structure and potentially lower impurity content.
Energy consumption and greenhouse gas emissions are important considerations in the comparative environmental impact analysis. Lepidolite processing often involves energy-intensive roasting steps to make the lithium more accessible for extraction. This can result in higher carbon emissions compared to petalite processing, which may require less thermal treatment. However, the overall energy balance depends on factors such as deposit location, processing technology, and energy source mix.
Waste management presents distinct challenges for both minerals. Lepidolite processing generates significant quantities of waste material, including potentially hazardous chemical byproducts. Proper disposal and potential valorization of these wastes are critical for minimizing environmental impact. Petalite processing may produce less waste volume but still requires careful management of tailings and other byproducts to prevent soil and water contamination.
Rehabilitation and closure planning are essential aspects of the environmental impact assessment for both lepidolite and petalite mining operations. The long-term ecological restoration of mined areas, including soil remediation and revegetation, must be considered in the overall environmental footprint of lithium extraction from these sources. The feasibility and cost of rehabilitation may vary between lepidolite and petalite sites, depending on factors such as local climate, soil conditions, and the extent of land disturbance.
Lepidolite mining typically involves open-pit extraction methods, which can lead to significant land disturbance and habitat destruction. The process often requires the removal of large amounts of overburden, potentially affecting local ecosystems and biodiversity. However, lepidolite deposits are often found in areas with existing mining infrastructure, which can minimize the need for new development and associated environmental disruption.
In contrast, petalite mining may have a smaller initial footprint due to the mineral's higher lithium content, potentially requiring less material to be extracted for equivalent lithium yields. This could translate to reduced land disturbance and lower volumes of waste rock. Nevertheless, petalite deposits may be located in more remote or environmentally sensitive areas, necessitating careful consideration of ecosystem impacts and mitigation strategies.
Water usage and management are crucial factors in the environmental assessment of both minerals. Lepidolite processing typically requires significant water resources for mineral separation and chemical treatment. This can strain local water supplies, particularly in arid regions where many lithium deposits are found. Petalite processing, while also water-intensive, may offer opportunities for more efficient water use due to its simpler mineral structure and potentially lower impurity content.
Energy consumption and greenhouse gas emissions are important considerations in the comparative environmental impact analysis. Lepidolite processing often involves energy-intensive roasting steps to make the lithium more accessible for extraction. This can result in higher carbon emissions compared to petalite processing, which may require less thermal treatment. However, the overall energy balance depends on factors such as deposit location, processing technology, and energy source mix.
Waste management presents distinct challenges for both minerals. Lepidolite processing generates significant quantities of waste material, including potentially hazardous chemical byproducts. Proper disposal and potential valorization of these wastes are critical for minimizing environmental impact. Petalite processing may produce less waste volume but still requires careful management of tailings and other byproducts to prevent soil and water contamination.
Rehabilitation and closure planning are essential aspects of the environmental impact assessment for both lepidolite and petalite mining operations. The long-term ecological restoration of mined areas, including soil remediation and revegetation, must be considered in the overall environmental footprint of lithium extraction from these sources. The feasibility and cost of rehabilitation may vary between lepidolite and petalite sites, depending on factors such as local climate, soil conditions, and the extent of land disturbance.
Geopolitical Factors
The geopolitical landscape plays a crucial role in shaping the lithium market trends, particularly when comparing lepidolite and petalite as lithium sources. These two minerals are found in different geographical locations, which significantly impacts their accessibility and strategic importance in the global lithium supply chain.
Lepidolite deposits are predominantly located in countries such as Portugal, Brazil, and Zimbabwe, while petalite is more commonly found in Canada, Australia, and Namibia. This distribution of resources has led to varying levels of geopolitical influence and competition among nations seeking to secure their lithium supplies.
The concentration of lepidolite in certain regions has prompted countries like Portugal to position themselves as key players in the European lithium market. This strategic advantage has implications for the European Union's efforts to reduce dependency on external lithium sources and develop a more self-sufficient supply chain for electric vehicle batteries.
Conversely, the presence of petalite in countries like Canada and Australia has strengthened their positions as reliable lithium suppliers to major markets such as the United States and Asia. These geopolitical alignments have influenced trade agreements and partnerships in the lithium industry, affecting the market dynamics for both minerals.
The ongoing trade tensions between major economies, particularly the United States and China, have also impacted the lithium market. As China is a significant processor of lithium minerals, including both lepidolite and petalite, any trade restrictions or tariffs can have ripple effects on the global supply chain and pricing of lithium products derived from these minerals.
Furthermore, government policies and regulations regarding mining and environmental protection in different countries affect the extraction and processing of lepidolite and petalite. For instance, stricter environmental regulations in some petalite-rich regions may favor the development of lepidolite resources in countries with more lenient policies, potentially shifting the balance between these two lithium sources in the market.
The geopolitical factors also extend to the realm of technology transfer and intellectual property. Countries with advanced processing technologies for either lepidolite or petalite may gain a competitive edge, influencing international collaborations and investments in the lithium sector.
In conclusion, the geopolitical factors surrounding lepidolite and petalite significantly influence their respective roles in the lithium market. The geographical distribution of these minerals, coupled with international relations, trade policies, and regulatory environments, creates a complex landscape that shapes the comparative advantages and challenges of utilizing lepidolite versus petalite in the evolving global lithium industry.
Lepidolite deposits are predominantly located in countries such as Portugal, Brazil, and Zimbabwe, while petalite is more commonly found in Canada, Australia, and Namibia. This distribution of resources has led to varying levels of geopolitical influence and competition among nations seeking to secure their lithium supplies.
The concentration of lepidolite in certain regions has prompted countries like Portugal to position themselves as key players in the European lithium market. This strategic advantage has implications for the European Union's efforts to reduce dependency on external lithium sources and develop a more self-sufficient supply chain for electric vehicle batteries.
Conversely, the presence of petalite in countries like Canada and Australia has strengthened their positions as reliable lithium suppliers to major markets such as the United States and Asia. These geopolitical alignments have influenced trade agreements and partnerships in the lithium industry, affecting the market dynamics for both minerals.
The ongoing trade tensions between major economies, particularly the United States and China, have also impacted the lithium market. As China is a significant processor of lithium minerals, including both lepidolite and petalite, any trade restrictions or tariffs can have ripple effects on the global supply chain and pricing of lithium products derived from these minerals.
Furthermore, government policies and regulations regarding mining and environmental protection in different countries affect the extraction and processing of lepidolite and petalite. For instance, stricter environmental regulations in some petalite-rich regions may favor the development of lepidolite resources in countries with more lenient policies, potentially shifting the balance between these two lithium sources in the market.
The geopolitical factors also extend to the realm of technology transfer and intellectual property. Countries with advanced processing technologies for either lepidolite or petalite may gain a competitive edge, influencing international collaborations and investments in the lithium sector.
In conclusion, the geopolitical factors surrounding lepidolite and petalite significantly influence their respective roles in the lithium market. The geographical distribution of these minerals, coupled with international relations, trade policies, and regulatory environments, creates a complex landscape that shapes the comparative advantages and challenges of utilizing lepidolite versus petalite in the evolving global lithium industry.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!