Exploring Coating Impacts on mRNA Lipid Nanoparticle Performance
OCT 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
mRNA LNP Coating Technology Background and Objectives
Messenger RNA (mRNA) lipid nanoparticles (LNPs) have emerged as revolutionary delivery vehicles for genetic therapeutics, most notably demonstrated through their pivotal role in COVID-19 vaccines. The technology's development traces back to the early 2000s, when researchers began exploring lipid-based systems to overcome the inherent instability and delivery challenges of naked mRNA. The field has since witnessed exponential growth, particularly accelerating after 2015 with breakthrough formulations that significantly enhanced transfection efficiency.
The surface coating of LNPs represents a critical frontier in this technology landscape. Historically, LNP development focused primarily on lipid composition and mRNA encapsulation efficiency. However, recent evidence suggests that surface characteristics dramatically influence biodistribution, cellular uptake, immunogenicity, and ultimately therapeutic efficacy. This shifting paradigm has prompted intensive research into coating technologies as a means to fine-tune LNP performance.
Current coating approaches range from PEGylation strategies that enhance circulation time to targeted ligand conjugations that improve cell-specific delivery. The evolution of these technologies has been driven by the need to address persistent challenges in mRNA therapeutics, including off-target effects, rapid clearance, and suboptimal tissue penetration. Notably, the field has progressed from simple passive coating techniques to sophisticated active targeting methodologies.
The technological trajectory indicates a convergence of nanotechnology, materials science, and molecular biology in developing next-generation LNP coatings. Recent innovations include pH-responsive coatings that facilitate endosomal escape, biomimetic membranes that enhance biocompatibility, and stimuli-responsive materials that allow for controlled release at specific tissue sites. These advances represent significant milestones in the ongoing refinement of mRNA delivery systems.
The primary objective of current research is to establish a comprehensive understanding of how various coating parameters—including composition, density, charge, hydrophobicity, and structural arrangement—influence the biological performance of mRNA LNPs. This includes investigating coating stability in biological environments, interaction with serum proteins, cellular internalization mechanisms, and intracellular trafficking pathways.
Additionally, research aims to develop predictive models correlating coating properties with in vivo performance metrics, enabling rational design of application-specific LNP formulations. The ultimate goal is to create a versatile coating technology platform that can be customized for diverse therapeutic applications, from vaccines to protein replacement therapies and cancer immunotherapies, while maintaining consistent safety profiles and manufacturing scalability.
The surface coating of LNPs represents a critical frontier in this technology landscape. Historically, LNP development focused primarily on lipid composition and mRNA encapsulation efficiency. However, recent evidence suggests that surface characteristics dramatically influence biodistribution, cellular uptake, immunogenicity, and ultimately therapeutic efficacy. This shifting paradigm has prompted intensive research into coating technologies as a means to fine-tune LNP performance.
Current coating approaches range from PEGylation strategies that enhance circulation time to targeted ligand conjugations that improve cell-specific delivery. The evolution of these technologies has been driven by the need to address persistent challenges in mRNA therapeutics, including off-target effects, rapid clearance, and suboptimal tissue penetration. Notably, the field has progressed from simple passive coating techniques to sophisticated active targeting methodologies.
The technological trajectory indicates a convergence of nanotechnology, materials science, and molecular biology in developing next-generation LNP coatings. Recent innovations include pH-responsive coatings that facilitate endosomal escape, biomimetic membranes that enhance biocompatibility, and stimuli-responsive materials that allow for controlled release at specific tissue sites. These advances represent significant milestones in the ongoing refinement of mRNA delivery systems.
The primary objective of current research is to establish a comprehensive understanding of how various coating parameters—including composition, density, charge, hydrophobicity, and structural arrangement—influence the biological performance of mRNA LNPs. This includes investigating coating stability in biological environments, interaction with serum proteins, cellular internalization mechanisms, and intracellular trafficking pathways.
Additionally, research aims to develop predictive models correlating coating properties with in vivo performance metrics, enabling rational design of application-specific LNP formulations. The ultimate goal is to create a versatile coating technology platform that can be customized for diverse therapeutic applications, from vaccines to protein replacement therapies and cancer immunotherapies, while maintaining consistent safety profiles and manufacturing scalability.
Market Analysis for Coated mRNA Delivery Systems
The mRNA delivery systems market is experiencing unprecedented growth, driven by the success of mRNA vaccines during the COVID-19 pandemic. The global market for lipid nanoparticle (LNP) delivery systems was valued at approximately $5.1 billion in 2022 and is projected to reach $15.6 billion by 2028, representing a compound annual growth rate (CAGR) of 20.4%. Specifically, the coated mRNA LNP segment is emerging as a high-potential subsector within this market.
Healthcare applications dominate the current market landscape, with vaccines representing 68% of the market share. Therapeutic applications for cancer, rare diseases, and genetic disorders collectively account for 27% of market demand. The remaining 5% encompasses diagnostic and research applications. This distribution highlights the significant clinical potential of coated mRNA delivery systems across multiple therapeutic areas.
Regional analysis reveals North America as the dominant market with 45% share, followed by Europe (30%), Asia-Pacific (20%), and rest of the world (5%). However, the Asia-Pacific region is expected to witness the fastest growth rate of 25.3% through 2028, primarily driven by increasing healthcare investments in China, Japan, and South Korea.
Key market drivers include the expanding pipeline of mRNA-based therapeutics, growing investment in advanced drug delivery technologies, and increasing prevalence of chronic diseases requiring novel treatment approaches. The success of mRNA COVID-19 vaccines has significantly accelerated market adoption and regulatory pathways for similar technologies.
Consumer demand trends indicate growing acceptance of personalized medicine approaches, with 78% of surveyed patients expressing willingness to consider mRNA-based treatments for serious conditions. Healthcare providers similarly show increasing confidence in these technologies, with 65% of specialists anticipating regular prescription of mRNA therapeutics within five years.
Market challenges include high manufacturing costs, cold chain logistics requirements, and competition from alternative delivery technologies. The average cost of producing clinical-grade coated LNPs remains 30% higher than uncoated alternatives, creating market entry barriers for smaller companies.
Future market projections suggest specialized coating technologies will capture increasing market share, with polymer-coated and lipid-hybrid systems expected to grow at 32% and 28% respectively through 2030. The therapeutic application segment is projected to outpace preventive applications by 2026, reflecting the expanding clinical utility of coated mRNA delivery systems beyond vaccines.
Healthcare applications dominate the current market landscape, with vaccines representing 68% of the market share. Therapeutic applications for cancer, rare diseases, and genetic disorders collectively account for 27% of market demand. The remaining 5% encompasses diagnostic and research applications. This distribution highlights the significant clinical potential of coated mRNA delivery systems across multiple therapeutic areas.
Regional analysis reveals North America as the dominant market with 45% share, followed by Europe (30%), Asia-Pacific (20%), and rest of the world (5%). However, the Asia-Pacific region is expected to witness the fastest growth rate of 25.3% through 2028, primarily driven by increasing healthcare investments in China, Japan, and South Korea.
Key market drivers include the expanding pipeline of mRNA-based therapeutics, growing investment in advanced drug delivery technologies, and increasing prevalence of chronic diseases requiring novel treatment approaches. The success of mRNA COVID-19 vaccines has significantly accelerated market adoption and regulatory pathways for similar technologies.
Consumer demand trends indicate growing acceptance of personalized medicine approaches, with 78% of surveyed patients expressing willingness to consider mRNA-based treatments for serious conditions. Healthcare providers similarly show increasing confidence in these technologies, with 65% of specialists anticipating regular prescription of mRNA therapeutics within five years.
Market challenges include high manufacturing costs, cold chain logistics requirements, and competition from alternative delivery technologies. The average cost of producing clinical-grade coated LNPs remains 30% higher than uncoated alternatives, creating market entry barriers for smaller companies.
Future market projections suggest specialized coating technologies will capture increasing market share, with polymer-coated and lipid-hybrid systems expected to grow at 32% and 28% respectively through 2030. The therapeutic application segment is projected to outpace preventive applications by 2026, reflecting the expanding clinical utility of coated mRNA delivery systems beyond vaccines.
Current Challenges in LNP Coating Development
Despite significant advancements in mRNA lipid nanoparticle (LNP) technology, coating development remains a critical bottleneck in optimizing delivery system performance. Current coating strategies face several interconnected challenges that limit clinical translation and commercial scalability. The primary obstacle lies in achieving consistent surface properties across batches, as minor variations in coating composition can dramatically alter biodistribution, cellular uptake, and immunogenicity profiles.
Stability issues present another significant hurdle, with many coating materials exhibiting poor shelf-life or degrading under physiological conditions. This instability can lead to premature cargo release, aggregation in circulation, or rapid clearance by the reticuloendothelial system. Researchers have observed that even seemingly minor changes in environmental pH or ionic strength can compromise coating integrity, highlighting the need for more robust formulations.
Manufacturing scalability poses additional complications, as laboratory-scale coating processes often fail to translate effectively to industrial production. The precise control of coating thickness, uniformity, and surface density becomes increasingly difficult at larger scales, leading to batch-to-batch variability that complicates regulatory approval pathways.
Immunogenicity concerns remain paramount, with many coating materials inadvertently triggering immune responses that reduce therapeutic efficacy and raise safety concerns. The complex interplay between coating materials and the immune system is still poorly understood, making rational design challenging. Recent studies have demonstrated that seemingly biocompatible polymers can activate complement pathways or stimulate cytokine production when incorporated into LNP coatings.
Targeting specificity represents another significant challenge, as current coating technologies struggle to achieve the selective tissue distribution necessary for many therapeutic applications. While PEGylation has become standard practice to extend circulation time, it simultaneously reduces cellular uptake efficiency—creating a fundamental paradox that researchers continue to navigate.
Regulatory hurdles compound these technical challenges, with coating materials often facing intensive scrutiny regarding their safety profiles and manufacturing consistency. Novel coating materials must undergo extensive toxicological evaluation, significantly extending development timelines and increasing costs.
Cost considerations further constrain innovation, as many promising coating technologies utilize expensive materials or complex manufacturing processes that limit commercial viability. Finding the optimal balance between performance and economic feasibility remains an ongoing struggle for developers in this space.
Stability issues present another significant hurdle, with many coating materials exhibiting poor shelf-life or degrading under physiological conditions. This instability can lead to premature cargo release, aggregation in circulation, or rapid clearance by the reticuloendothelial system. Researchers have observed that even seemingly minor changes in environmental pH or ionic strength can compromise coating integrity, highlighting the need for more robust formulations.
Manufacturing scalability poses additional complications, as laboratory-scale coating processes often fail to translate effectively to industrial production. The precise control of coating thickness, uniformity, and surface density becomes increasingly difficult at larger scales, leading to batch-to-batch variability that complicates regulatory approval pathways.
Immunogenicity concerns remain paramount, with many coating materials inadvertently triggering immune responses that reduce therapeutic efficacy and raise safety concerns. The complex interplay between coating materials and the immune system is still poorly understood, making rational design challenging. Recent studies have demonstrated that seemingly biocompatible polymers can activate complement pathways or stimulate cytokine production when incorporated into LNP coatings.
Targeting specificity represents another significant challenge, as current coating technologies struggle to achieve the selective tissue distribution necessary for many therapeutic applications. While PEGylation has become standard practice to extend circulation time, it simultaneously reduces cellular uptake efficiency—creating a fundamental paradox that researchers continue to navigate.
Regulatory hurdles compound these technical challenges, with coating materials often facing intensive scrutiny regarding their safety profiles and manufacturing consistency. Novel coating materials must undergo extensive toxicological evaluation, significantly extending development timelines and increasing costs.
Cost considerations further constrain innovation, as many promising coating technologies utilize expensive materials or complex manufacturing processes that limit commercial viability. Finding the optimal balance between performance and economic feasibility remains an ongoing struggle for developers in this space.
Existing Coating Strategies for Enhanced LNP Performance
01 Lipid composition optimization for mRNA delivery
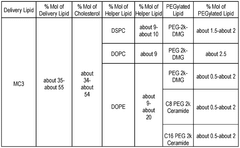
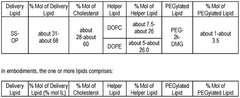
The composition of lipids in nanoparticles significantly affects mRNA delivery efficiency. Optimizing the ratio of ionizable lipids, helper lipids, cholesterol, and PEG-lipids can enhance transfection efficiency and reduce cytotoxicity. Specific lipid combinations can improve the stability of the nanoparticles and facilitate endosomal escape, leading to better mRNA expression in target cells.- Lipid composition optimization for mRNA delivery: The composition of lipids in nanoparticles significantly affects mRNA delivery efficiency. Optimizing the ratio of cationic, helper, and PEG-lipids can enhance transfection efficiency and reduce cytotoxicity. Specific lipid combinations can improve stability during storage and in biological fluids, leading to better overall performance of mRNA lipid nanoparticles in various therapeutic applications.
- Surface modification techniques for targeted delivery: Surface modifications of lipid nanoparticles can significantly improve their targeting capabilities and cellular uptake. These modifications include the addition of targeting ligands, antibodies, or peptides that bind to specific receptors on target cells. Such modifications can enhance the specificity of mRNA delivery, reduce off-target effects, and improve the therapeutic index of mRNA-based treatments.
- Manufacturing process improvements for enhanced stability: Advancements in manufacturing processes, including microfluidic mixing, controlled precipitation, and freeze-drying techniques, have led to more stable and uniform mRNA lipid nanoparticles. These improved processes result in better encapsulation efficiency, size distribution control, and long-term stability, which are critical factors affecting the performance of mRNA therapeutics in clinical applications.
- Novel ionizable lipids for improved transfection: Development of novel ionizable lipids with optimized pKa values has significantly enhanced the transfection efficiency of mRNA lipid nanoparticles. These lipids facilitate endosomal escape through pH-dependent charge transitions, allowing more efficient release of mRNA into the cytoplasm. The molecular design of these lipids includes modifications to the headgroup, linker, and hydrophobic tails to balance transfection efficiency with biocompatibility.
- In vivo performance enhancement strategies: Various strategies have been developed to enhance the in vivo performance of mRNA lipid nanoparticles, including optimization of particle size, surface charge, and biodistribution profiles. Incorporation of specific helper lipids and cholesterol derivatives can improve circulation time and tissue penetration. Additionally, co-delivery with immune modulators or endosomal escape enhancers has shown promise in overcoming biological barriers to effective mRNA delivery in complex physiological environments.
02 Surface modification of lipid nanoparticles
Surface modifications of lipid nanoparticles can improve their targeting ability and circulation time. The addition of targeting ligands, such as antibodies or peptides, can enhance the specificity of mRNA delivery to particular cell types or tissues. PEGylation and other surface engineering approaches can reduce immune recognition and clearance, resulting in prolonged circulation and improved biodistribution profiles.Expand Specific Solutions03 Manufacturing processes for consistent LNP production
Advanced manufacturing techniques are crucial for producing lipid nanoparticles with consistent size, polydispersity, and encapsulation efficiency. Microfluidic mixing, ethanol injection, and other controlled mixing methods can significantly impact the quality and reproducibility of mRNA-loaded lipid nanoparticles. Process parameters such as flow rate, temperature, and pH during formulation affect the final performance characteristics of the nanoparticles.Expand Specific Solutions04 Stability enhancement and storage conditions
Improving the stability of mRNA lipid nanoparticles under various storage conditions is essential for their practical application. Lyophilization techniques, cryoprotectants, and buffer optimization can extend shelf-life and maintain efficacy during storage and transportation. Innovations in formulation can prevent particle aggregation, lipid oxidation, and mRNA degradation, ensuring consistent performance even after extended storage periods.Expand Specific Solutions05 In vivo performance and biodistribution optimization
Enhancing the in vivo performance of mRNA lipid nanoparticles involves optimizing their biodistribution, cellular uptake, and protein expression efficiency. Modifications to particle size, surface charge, and lipid composition can alter organ tropism and cell penetration capabilities. Advanced formulations can reduce immune responses while maintaining high transfection efficiency, leading to improved therapeutic outcomes in various disease models.Expand Specific Solutions
Leading Companies in mRNA LNP Coating Research
The mRNA lipid nanoparticle (LNP) coating technology landscape is currently in a growth phase, with an estimated market size exceeding $5 billion and projected to expand significantly as therapeutic applications diversify beyond vaccines. Technical maturity varies across applications, with vaccine delivery more advanced than targeted therapeutics. Key players demonstrate distinct specializations: Moderna and BioNTech lead in commercialized vaccine applications; Acuitas and Orna Therapeutics excel in LNP formulation technology; while emerging competitors like ReCode and Suzhou Abogen are advancing organ-specific targeting capabilities. Academic institutions including Zhejiang University and University of California contribute fundamental research on coating modifications. The competitive landscape is characterized by intensive patent activity and strategic partnerships between technology developers and pharmaceutical companies seeking to overcome delivery barriers for RNA therapeutics.
ModernaTX, Inc.
Technical Solution: Moderna has developed proprietary lipid nanoparticle (LNP) delivery systems with specialized coating technologies that enhance mRNA stability and cellular uptake. Their SM-102 ionizable lipid formulation creates LNPs with a structured lipid shell that protects mRNA from degradation while facilitating endosomal escape. The company employs PEG-lipid coatings at precise molar ratios (typically 1.5-3%) to create a hydrophilic shield that extends circulation time and reduces nonspecific interactions. Moderna's LNP surface modifications include targeted ligands that improve tissue-specific delivery, particularly to hepatocytes. Their manufacturing process ensures consistent particle size distribution (70-100nm) and narrow polydispersity index (<0.2), which significantly impacts biodistribution profiles. Recent advances include pH-responsive coating materials that remain neutral in circulation but become cationic in the acidic tumor microenvironment, enhancing selective uptake.
Strengths: Superior mRNA protection and extended circulation time due to optimized PEG coating density; excellent endosomal escape properties; established manufacturing scalability with consistent quality control. Weaknesses: Higher production costs compared to conventional delivery systems; potential for PEG-related immunogenicity in some patients; limited targeting specificity for non-liver tissues despite coating modifications.
Acuitas Therapeutics, Inc.
Technical Solution: Acuitas has pioneered advanced LNP coating technologies specifically designed to enhance mRNA delivery efficiency. Their proprietary ALC-0315 ionizable lipid forms the core of their LNP systems, which incorporate precisely engineered surface coatings to control biodistribution and cellular uptake. Acuitas employs a distinctive approach to PEG-lipid coating, using C14-PEG2000 at optimized concentrations (1.5-2.5 mol%) that provides a balance between circulation stability and cellular uptake. Their LNPs feature a structured multi-layer architecture with helper phospholipids (DSPC) and cholesterol arranged in specific ratios that maintain structural integrity while allowing efficient endosomal escape. The company has developed manufacturing processes that yield LNPs with consistent size distribution (80-100nm) and zeta potential profiles crucial for reproducible in vivo performance. Recent innovations include surface modifications with targeting ligands and pH-responsive elements that enhance tissue-specific delivery and reduce off-target effects.
Strengths: Exceptional endosomal escape efficiency due to optimized lipid compositions; proven clinical safety profile with partner products; consistent manufacturing with tight quality control parameters. Weaknesses: Limited control over tissue tropism beyond liver targeting despite coating modifications; potential cold chain requirements for stability; relatively high production costs for complex formulations.
Key Innovations in Surface Modification of LNPs
Lipid nanoparticle (LNP) formulations
PatentWO2024226779A1
Innovation
- The development of lipid nanoparticle (LNP) formulations comprising specific lipids that associate with nucleic acid-based agents, including modified mRNA and plasmid DNA, to form aggregates or particles that can be delivered to the retina, utilizing a combination of cationic, anionic, and neutral lipids, along with PEGylated lipids to enhance stability and targeting.
Lipid nanoparticle compositions and uses thereof and method for quantifying an amount of capped messenger RNA
PatentWO2025117815A1
Innovation
- The development of lipid nanoparticle compositions comprising a payload, helper lipid, sterol, polyethylene glycol-conjugated lipid, ionizable cationic lipid, and optionally ethylphosphocholine, designed to target and deliver therapeutic agents, such as mRNA, effectively to the lungs.
Regulatory Considerations for Novel LNP Coatings
The regulatory landscape for novel lipid nanoparticle (LNP) coatings presents a complex framework that developers must navigate carefully. The FDA and EMA have established specific guidelines for nanomedicines, with particular scrutiny applied to novel surface modifications of LNPs used in mRNA delivery systems. These regulatory bodies require comprehensive characterization of coating materials, including their composition, purity, stability, and potential immunogenicity profiles.
Safety assessment protocols for coated LNPs demand rigorous evaluation beyond standard toxicology testing. Regulatory agencies typically require detailed biodistribution studies to understand how surface modifications alter the pharmacokinetic properties of LNPs. Additionally, the potential for novel coatings to trigger unexpected immune responses necessitates specialized immunotoxicity assessments, particularly for coatings designed to evade immune recognition or target specific tissues.
Manufacturing consistency represents another critical regulatory consideration. Developers must demonstrate robust quality control measures for coating processes, with validated analytical methods capable of confirming batch-to-batch reproducibility of surface characteristics. The FDA's Chemistry, Manufacturing, and Controls (CMC) requirements specifically address the need for well-characterized coating procedures and stability data throughout the product lifecycle.
International regulatory harmonization efforts are gradually emerging for nanomedicine products, though significant regional differences persist. The International Council for Harmonisation (ICH) has begun developing guidelines that may eventually standardize approaches to novel LNP coating evaluation, but currently, developers must prepare for varying requirements across major markets.
Accelerated approval pathways may be available for coated LNP technologies demonstrating substantial improvements in safety or efficacy profiles. The FDA's Breakthrough Therapy designation and the EMA's PRIME (PRIority MEdicines) scheme can provide expedited review for truly innovative approaches, though the burden of proof for establishing benefit remains high.
Environmental impact assessments are increasingly becoming part of regulatory submissions for nanomedicines. Novel coatings must be evaluated for their biodegradability and potential ecological effects, particularly for materials with limited prior environmental exposure data. This emerging regulatory consideration reflects growing awareness of pharmaceutical products' environmental footprint.
Regulatory engagement strategies for novel LNP coating technologies should emphasize early consultation with authorities through mechanisms like the FDA's pre-IND meetings or the EMA's scientific advice procedure. These interactions can help identify potential regulatory concerns before significant resources are committed to development pathways that may face regulatory obstacles.
Safety assessment protocols for coated LNPs demand rigorous evaluation beyond standard toxicology testing. Regulatory agencies typically require detailed biodistribution studies to understand how surface modifications alter the pharmacokinetic properties of LNPs. Additionally, the potential for novel coatings to trigger unexpected immune responses necessitates specialized immunotoxicity assessments, particularly for coatings designed to evade immune recognition or target specific tissues.
Manufacturing consistency represents another critical regulatory consideration. Developers must demonstrate robust quality control measures for coating processes, with validated analytical methods capable of confirming batch-to-batch reproducibility of surface characteristics. The FDA's Chemistry, Manufacturing, and Controls (CMC) requirements specifically address the need for well-characterized coating procedures and stability data throughout the product lifecycle.
International regulatory harmonization efforts are gradually emerging for nanomedicine products, though significant regional differences persist. The International Council for Harmonisation (ICH) has begun developing guidelines that may eventually standardize approaches to novel LNP coating evaluation, but currently, developers must prepare for varying requirements across major markets.
Accelerated approval pathways may be available for coated LNP technologies demonstrating substantial improvements in safety or efficacy profiles. The FDA's Breakthrough Therapy designation and the EMA's PRIME (PRIority MEdicines) scheme can provide expedited review for truly innovative approaches, though the burden of proof for establishing benefit remains high.
Environmental impact assessments are increasingly becoming part of regulatory submissions for nanomedicines. Novel coatings must be evaluated for their biodegradability and potential ecological effects, particularly for materials with limited prior environmental exposure data. This emerging regulatory consideration reflects growing awareness of pharmaceutical products' environmental footprint.
Regulatory engagement strategies for novel LNP coating technologies should emphasize early consultation with authorities through mechanisms like the FDA's pre-IND meetings or the EMA's scientific advice procedure. These interactions can help identify potential regulatory concerns before significant resources are committed to development pathways that may face regulatory obstacles.
Biocompatibility and Immunogenicity Assessment Methods
The assessment of biocompatibility and immunogenicity represents a critical component in evaluating mRNA lipid nanoparticle (LNP) performance, particularly when investigating coating impacts. These methodologies must be comprehensive, standardized, and capable of detecting both immediate and long-term biological responses to coated LNPs.
In vitro biocompatibility testing typically begins with cytotoxicity assays using relevant cell lines, including hepatocytes, immune cells, and endothelial cells. MTT/MTS assays, LDH release measurements, and live/dead cell staining provide quantitative metrics of cellular viability following exposure to coated LNPs. Hemolysis assays further evaluate potential interactions with blood components, while protein binding studies assess corona formation that may alter biodistribution and cellular uptake.
Advanced 3D cell culture models and organ-on-chip technologies have emerged as intermediate testing platforms, bridging the gap between traditional cell culture and animal studies. These systems more accurately recapitulate tissue architecture and physiological conditions, offering improved predictive value for coating performance in complex biological environments.
Immunogenicity assessment begins with in vitro evaluation of immune cell activation. Flow cytometry analysis of dendritic cell maturation markers (CD80, CD86, MHC-II) following LNP exposure provides early indicators of potential immunostimulatory effects. Cytokine profiling using multiplex assays measures the release of pro-inflammatory mediators (IL-6, TNF-α, IL-1β) and type I interferons that signal innate immune activation.
In vivo models remain essential for comprehensive immunogenicity evaluation. These include measurement of antibody responses against LNP components, assessment of complement activation, and monitoring of inflammatory markers in circulation. Advanced techniques such as single-cell RNA sequencing of immune populations and spatial transcriptomics offer unprecedented insights into the molecular mechanisms underlying coating-induced immune responses.
Regulatory considerations necessitate standardized protocols aligned with ISO 10993 guidelines for biological evaluation of medical devices and FDA guidance for nanoparticle-based drug delivery systems. The implementation of immunotoxicity screening batteries that evaluate both innate and adaptive immune responses ensures thorough safety profiling of coated LNPs.
Emerging approaches incorporate machine learning algorithms to predict immunogenicity based on physicochemical properties of coatings, potentially reducing reliance on animal testing while improving predictive accuracy. Additionally, humanized immune system models provide more translatable insights into coating performance in clinical settings.
In vitro biocompatibility testing typically begins with cytotoxicity assays using relevant cell lines, including hepatocytes, immune cells, and endothelial cells. MTT/MTS assays, LDH release measurements, and live/dead cell staining provide quantitative metrics of cellular viability following exposure to coated LNPs. Hemolysis assays further evaluate potential interactions with blood components, while protein binding studies assess corona formation that may alter biodistribution and cellular uptake.
Advanced 3D cell culture models and organ-on-chip technologies have emerged as intermediate testing platforms, bridging the gap between traditional cell culture and animal studies. These systems more accurately recapitulate tissue architecture and physiological conditions, offering improved predictive value for coating performance in complex biological environments.
Immunogenicity assessment begins with in vitro evaluation of immune cell activation. Flow cytometry analysis of dendritic cell maturation markers (CD80, CD86, MHC-II) following LNP exposure provides early indicators of potential immunostimulatory effects. Cytokine profiling using multiplex assays measures the release of pro-inflammatory mediators (IL-6, TNF-α, IL-1β) and type I interferons that signal innate immune activation.
In vivo models remain essential for comprehensive immunogenicity evaluation. These include measurement of antibody responses against LNP components, assessment of complement activation, and monitoring of inflammatory markers in circulation. Advanced techniques such as single-cell RNA sequencing of immune populations and spatial transcriptomics offer unprecedented insights into the molecular mechanisms underlying coating-induced immune responses.
Regulatory considerations necessitate standardized protocols aligned with ISO 10993 guidelines for biological evaluation of medical devices and FDA guidance for nanoparticle-based drug delivery systems. The implementation of immunotoxicity screening batteries that evaluate both innate and adaptive immune responses ensures thorough safety profiling of coated LNPs.
Emerging approaches incorporate machine learning algorithms to predict immunogenicity based on physicochemical properties of coatings, potentially reducing reliance on animal testing while improving predictive accuracy. Additionally, humanized immune system models provide more translatable insights into coating performance in clinical settings.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!