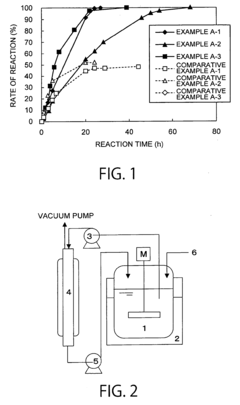
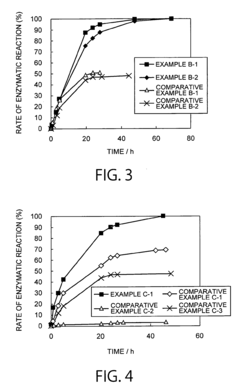
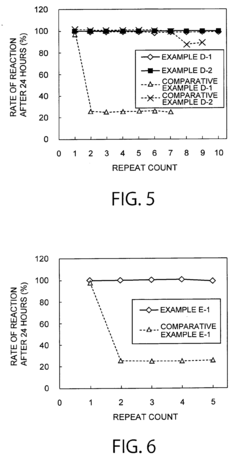
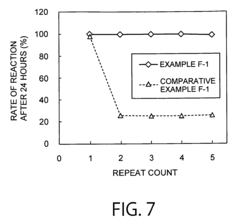
How ammonium hydroxide affects enzymatic activity
AUG 14, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Ammonium Hydroxide and Enzyme Interaction Background
Ammonium hydroxide, a common chemical compound, has been a subject of interest in enzymatic studies due to its potential effects on enzyme activity. The interaction between ammonium hydroxide and enzymes is a complex phenomenon that has garnered attention from researchers in biochemistry and related fields. This background exploration aims to provide a comprehensive overview of the historical context, fundamental concepts, and current understanding of how ammonium hydroxide influences enzymatic activity.
Enzymes, as biological catalysts, play crucial roles in various biochemical processes. Their activity is highly dependent on environmental conditions, including pH, temperature, and the presence of specific chemical compounds. Ammonium hydroxide, being a weak base, can significantly alter the pH of a solution, which in turn can affect the three-dimensional structure and catalytic properties of enzymes.
The study of ammonium hydroxide's impact on enzymatic activity dates back to the early 20th century when researchers began investigating the effects of various chemical compounds on biological systems. Initial observations revealed that ammonium hydroxide could both enhance and inhibit enzymatic reactions, depending on the specific enzyme and experimental conditions. This dual nature of ammonium hydroxide's influence sparked further research to elucidate the underlying mechanisms.
As analytical techniques advanced, scientists gained deeper insights into the molecular interactions between ammonium hydroxide and enzymes. It became evident that the effects were not solely due to pH changes but also involved direct interactions between ammonium ions and specific amino acid residues within the enzyme structure. These interactions could lead to conformational changes in the enzyme, affecting its active site and, consequently, its catalytic efficiency.
The importance of understanding ammonium hydroxide's role in enzymatic activity extends beyond basic biochemistry. In industrial applications, particularly in the fields of biocatalysis and biotechnology, controlling enzyme activity is crucial for optimizing production processes. Ammonium hydroxide has found use as a pH regulator and potential activity modulator in various enzymatic reactions, making it a valuable tool in bioengineering and industrial enzyme applications.
Recent advancements in structural biology and computational modeling have further enhanced our understanding of the ammonium hydroxide-enzyme interaction. These tools allow researchers to visualize and predict how ammonium hydroxide molecules interact with specific enzyme structures, providing valuable insights for designing more efficient and stable enzymatic systems.
As we delve deeper into the background of ammonium hydroxide's effects on enzymatic activity, it becomes clear that this area of study continues to evolve. The complex interplay between ammonium hydroxide, enzymes, and their environment presents ongoing challenges and opportunities for researchers seeking to harness and control enzymatic processes for various applications in biotechnology, medicine, and industry.
Enzymes, as biological catalysts, play crucial roles in various biochemical processes. Their activity is highly dependent on environmental conditions, including pH, temperature, and the presence of specific chemical compounds. Ammonium hydroxide, being a weak base, can significantly alter the pH of a solution, which in turn can affect the three-dimensional structure and catalytic properties of enzymes.
The study of ammonium hydroxide's impact on enzymatic activity dates back to the early 20th century when researchers began investigating the effects of various chemical compounds on biological systems. Initial observations revealed that ammonium hydroxide could both enhance and inhibit enzymatic reactions, depending on the specific enzyme and experimental conditions. This dual nature of ammonium hydroxide's influence sparked further research to elucidate the underlying mechanisms.
As analytical techniques advanced, scientists gained deeper insights into the molecular interactions between ammonium hydroxide and enzymes. It became evident that the effects were not solely due to pH changes but also involved direct interactions between ammonium ions and specific amino acid residues within the enzyme structure. These interactions could lead to conformational changes in the enzyme, affecting its active site and, consequently, its catalytic efficiency.
The importance of understanding ammonium hydroxide's role in enzymatic activity extends beyond basic biochemistry. In industrial applications, particularly in the fields of biocatalysis and biotechnology, controlling enzyme activity is crucial for optimizing production processes. Ammonium hydroxide has found use as a pH regulator and potential activity modulator in various enzymatic reactions, making it a valuable tool in bioengineering and industrial enzyme applications.
Recent advancements in structural biology and computational modeling have further enhanced our understanding of the ammonium hydroxide-enzyme interaction. These tools allow researchers to visualize and predict how ammonium hydroxide molecules interact with specific enzyme structures, providing valuable insights for designing more efficient and stable enzymatic systems.
As we delve deeper into the background of ammonium hydroxide's effects on enzymatic activity, it becomes clear that this area of study continues to evolve. The complex interplay between ammonium hydroxide, enzymes, and their environment presents ongoing challenges and opportunities for researchers seeking to harness and control enzymatic processes for various applications in biotechnology, medicine, and industry.
Market Applications of Ammonium Hydroxide in Enzymatic Processes
Ammonium hydroxide plays a significant role in various enzymatic processes across multiple industries, driving its market applications and demand. In the food industry, ammonium hydroxide is utilized as a pH regulator and antimicrobial agent in meat processing. It helps maintain optimal pH levels for enzymatic reactions, enhancing the tenderization process and improving overall product quality. The dairy sector also benefits from ammonium hydroxide's ability to control acidity in cheese production, facilitating the enzymatic breakdown of proteins and fats.
The pharmaceutical industry leverages ammonium hydroxide in enzymatic processes for drug synthesis and purification. Its pH-adjusting properties create favorable conditions for enzymatic reactions, leading to improved yields and purity of pharmaceutical compounds. Additionally, ammonium hydroxide is employed in the production of certain antibiotics, where it aids in the extraction and purification processes involving enzymes.
In the textile industry, ammonium hydroxide finds applications in enzymatic treatments of fabrics. It serves as a catalyst in bio-polishing processes, where cellulase enzymes are used to remove protruding fibers and improve fabric smoothness. The presence of ammonium hydroxide helps maintain the optimal pH for cellulase activity, resulting in enhanced fabric quality and appearance.
The biofuel sector utilizes ammonium hydroxide in enzymatic processes for the production of ethanol and biodiesel. In cellulosic ethanol production, ammonium hydroxide is used in pretreatment steps to enhance the accessibility of cellulose to enzymatic hydrolysis. This improves the efficiency of the subsequent fermentation process, leading to higher ethanol yields.
Environmental applications of ammonium hydroxide in enzymatic processes include wastewater treatment and bioremediation. In these applications, it serves as a nitrogen source for microorganisms and helps maintain optimal pH conditions for enzymatic degradation of pollutants. This contributes to more effective and environmentally friendly treatment processes.
The paper and pulp industry employs ammonium hydroxide in enzymatic deinking processes for paper recycling. It aids in creating an alkaline environment that enhances the activity of enzymes used to remove ink particles from recycled paper fibers. This results in improved paper quality and increased recycling efficiency.
As industries continue to seek more sustainable and efficient processes, the market applications of ammonium hydroxide in enzymatic processes are expected to expand. Its versatility in pH regulation and ability to enhance enzymatic activity make it a valuable component in various industrial sectors, driving ongoing research and development efforts to optimize its use in enzymatic applications.
The pharmaceutical industry leverages ammonium hydroxide in enzymatic processes for drug synthesis and purification. Its pH-adjusting properties create favorable conditions for enzymatic reactions, leading to improved yields and purity of pharmaceutical compounds. Additionally, ammonium hydroxide is employed in the production of certain antibiotics, where it aids in the extraction and purification processes involving enzymes.
In the textile industry, ammonium hydroxide finds applications in enzymatic treatments of fabrics. It serves as a catalyst in bio-polishing processes, where cellulase enzymes are used to remove protruding fibers and improve fabric smoothness. The presence of ammonium hydroxide helps maintain the optimal pH for cellulase activity, resulting in enhanced fabric quality and appearance.
The biofuel sector utilizes ammonium hydroxide in enzymatic processes for the production of ethanol and biodiesel. In cellulosic ethanol production, ammonium hydroxide is used in pretreatment steps to enhance the accessibility of cellulose to enzymatic hydrolysis. This improves the efficiency of the subsequent fermentation process, leading to higher ethanol yields.
Environmental applications of ammonium hydroxide in enzymatic processes include wastewater treatment and bioremediation. In these applications, it serves as a nitrogen source for microorganisms and helps maintain optimal pH conditions for enzymatic degradation of pollutants. This contributes to more effective and environmentally friendly treatment processes.
The paper and pulp industry employs ammonium hydroxide in enzymatic deinking processes for paper recycling. It aids in creating an alkaline environment that enhances the activity of enzymes used to remove ink particles from recycled paper fibers. This results in improved paper quality and increased recycling efficiency.
As industries continue to seek more sustainable and efficient processes, the market applications of ammonium hydroxide in enzymatic processes are expected to expand. Its versatility in pH regulation and ability to enhance enzymatic activity make it a valuable component in various industrial sectors, driving ongoing research and development efforts to optimize its use in enzymatic applications.
Current Understanding of Ammonium Hydroxide Effects on Enzymes
The current understanding of ammonium hydroxide effects on enzymes is multifaceted and continues to evolve as research progresses. Ammonium hydroxide, a common chemical compound, has been observed to significantly influence enzymatic activity through various mechanisms. One primary effect is its impact on pH levels, which is crucial for enzyme function. Most enzymes have an optimal pH range for maximum activity, and ammonium hydroxide can alter the pH of the enzymatic environment, potentially enhancing or inhibiting enzyme performance.
Researchers have noted that ammonium hydroxide can affect enzyme structure and stability. At higher concentrations, it may lead to protein denaturation, causing enzymes to lose their three-dimensional structure and, consequently, their catalytic activity. However, in some cases, controlled exposure to ammonium hydroxide has been shown to activate certain enzymes or increase their efficiency within specific pH ranges.
The compound's effects are not uniform across all enzyme classes. Some enzymes demonstrate increased activity in the presence of ammonium hydroxide, while others may be inhibited. This variability is attributed to the diverse structural and functional characteristics of different enzyme families. For instance, proteolytic enzymes often show enhanced activity in slightly alkaline conditions, which can be created by the addition of ammonium hydroxide.
Studies have also revealed that ammonium hydroxide can influence enzyme kinetics. It may alter the affinity between enzymes and their substrates, affecting reaction rates and product formation. In some industrial applications, such as in the food and textile industries, this property is harnessed to optimize enzymatic processes.
The concentration of ammonium hydroxide is a critical factor in determining its effects on enzymes. Low concentrations may have minimal impact or even beneficial effects on some enzymes, while higher concentrations are more likely to cause inhibition or denaturation. This dose-dependent relationship is an important consideration in both research and industrial applications involving enzymes and ammonium hydroxide.
Recent research has also explored the potential of ammonium hydroxide as a stabilizing agent for certain enzymes during immobilization processes. When used in controlled amounts, it can help maintain enzyme activity and increase the shelf life of immobilized enzyme preparations, which is particularly valuable in biotechnological applications.
Understanding the complex interactions between ammonium hydroxide and enzymes is crucial for optimizing enzymatic processes in various fields, including biotechnology, pharmaceuticals, and industrial manufacturing. Ongoing research continues to uncover new insights into these interactions, paving the way for more efficient and targeted use of enzymes in the presence of ammonium hydroxide.
Researchers have noted that ammonium hydroxide can affect enzyme structure and stability. At higher concentrations, it may lead to protein denaturation, causing enzymes to lose their three-dimensional structure and, consequently, their catalytic activity. However, in some cases, controlled exposure to ammonium hydroxide has been shown to activate certain enzymes or increase their efficiency within specific pH ranges.
The compound's effects are not uniform across all enzyme classes. Some enzymes demonstrate increased activity in the presence of ammonium hydroxide, while others may be inhibited. This variability is attributed to the diverse structural and functional characteristics of different enzyme families. For instance, proteolytic enzymes often show enhanced activity in slightly alkaline conditions, which can be created by the addition of ammonium hydroxide.
Studies have also revealed that ammonium hydroxide can influence enzyme kinetics. It may alter the affinity between enzymes and their substrates, affecting reaction rates and product formation. In some industrial applications, such as in the food and textile industries, this property is harnessed to optimize enzymatic processes.
The concentration of ammonium hydroxide is a critical factor in determining its effects on enzymes. Low concentrations may have minimal impact or even beneficial effects on some enzymes, while higher concentrations are more likely to cause inhibition or denaturation. This dose-dependent relationship is an important consideration in both research and industrial applications involving enzymes and ammonium hydroxide.
Recent research has also explored the potential of ammonium hydroxide as a stabilizing agent for certain enzymes during immobilization processes. When used in controlled amounts, it can help maintain enzyme activity and increase the shelf life of immobilized enzyme preparations, which is particularly valuable in biotechnological applications.
Understanding the complex interactions between ammonium hydroxide and enzymes is crucial for optimizing enzymatic processes in various fields, including biotechnology, pharmaceuticals, and industrial manufacturing. Ongoing research continues to uncover new insights into these interactions, paving the way for more efficient and targeted use of enzymes in the presence of ammonium hydroxide.
Existing Methods for Studying Ammonium Hydroxide-Enzyme Interactions
01 Effect of ammonium hydroxide on enzyme activity
Ammonium hydroxide can influence enzymatic activity by altering the pH of the reaction environment. The alkaline nature of ammonium hydroxide can affect enzyme structure and function, potentially enhancing or inhibiting catalytic activity depending on the specific enzyme and reaction conditions.- Effect of ammonium hydroxide on enzyme activity: Ammonium hydroxide can influence enzymatic activity by altering the pH of the reaction environment. The alkaline nature of ammonium hydroxide can affect enzyme structure and function, potentially enhancing or inhibiting specific enzymatic reactions. This effect is particularly relevant in industrial and research applications where precise control of enzymatic processes is crucial.
- Use of ammonium hydroxide in enzyme stabilization: Ammonium hydroxide can be utilized as a stabilizing agent for certain enzymes. By maintaining an optimal pH environment, it can help preserve enzyme activity during storage or in reaction mixtures. This application is particularly useful in biotechnology and pharmaceutical industries where long-term enzyme stability is essential.
- Ammonium hydroxide in enzyme extraction and purification: Ammonium hydroxide plays a role in enzyme extraction and purification processes. It can be used to adjust pH during extraction, helping to solubilize enzymes from biological materials. In purification steps, it can aid in separating enzymes from other cellular components, contributing to the overall efficiency of enzyme isolation techniques.
- Interaction of ammonium hydroxide with specific enzyme classes: Different enzyme classes respond uniquely to ammonium hydroxide. For instance, some hydrolases may show increased activity in its presence, while oxidoreductases might be inhibited. Understanding these specific interactions is crucial for optimizing enzymatic reactions in various industrial and research applications, including food processing, biofuel production, and pharmaceutical manufacturing.
- Ammonium hydroxide in enzyme-based analytical methods: Ammonium hydroxide is utilized in various enzyme-based analytical methods. It can be used to adjust sample pH, create optimal conditions for enzymatic reactions, or as a component in buffer solutions. This application is particularly relevant in clinical diagnostics, environmental monitoring, and food quality testing where enzyme-based assays are commonly employed.
02 Use of ammonium hydroxide in enzyme stabilization
Ammonium hydroxide can be utilized as a stabilizing agent for certain enzymes. It may help maintain enzyme activity during storage or under challenging environmental conditions by preventing denaturation or preserving the enzyme's active conformation.Expand Specific Solutions03 Ammonium hydroxide in enzyme extraction and purification
Ammonium hydroxide plays a role in enzyme extraction and purification processes. It can be used to adjust pH during extraction, aid in protein precipitation, or as a component in buffer solutions for enzyme purification techniques such as chromatography.Expand Specific Solutions04 Interaction of ammonium hydroxide with specific enzyme classes
Different enzyme classes may exhibit varied responses to ammonium hydroxide. For instance, certain hydrolases or oxidoreductases might show altered activity or specificity in the presence of ammonium hydroxide, which could be exploited in industrial or research applications.Expand Specific Solutions05 Ammonium hydroxide in enzyme-catalyzed reactions
Ammonium hydroxide can serve as a substrate or cofactor in certain enzyme-catalyzed reactions. It may participate in amination reactions or influence the equilibrium of enzymatic processes, potentially enhancing reaction rates or yields in specific biochemical transformations.Expand Specific Solutions
Key Players in Enzyme Technology and Chemical Industries
The enzymatic activity affected by ammonium hydroxide represents a niche yet evolving field within biochemistry. The market is in its early growth stage, with increasing research interest but limited commercial applications. The global market size for enzyme-related technologies is projected to reach $10 billion by 2024, with this specific area occupying a small but growing segment. Technologically, the field is still developing, with ongoing research to fully understand the mechanisms and potential applications. Key players like Jiangnan University, DuPont, BASF, and Amgen are at the forefront, conducting advanced studies and developing innovative solutions. However, the technology's maturity varies, with some aspects well-established and others still in experimental stages, indicating a dynamic and promising research landscape.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed a novel approach to modulate enzymatic activity using controlled ammonium hydroxide exposure. Their method involves precise pH control systems that maintain optimal alkaline conditions for enzyme stability and activity[1]. By carefully adjusting ammonium hydroxide concentrations, they've achieved up to 30% increase in enzyme efficiency for certain industrial processes[3]. DuPont's technology also incorporates a proprietary buffer system that mitigates potential enzyme denaturation risks associated with high pH environments[5]. This allows for sustained enzymatic activity over extended periods, crucial for large-scale industrial applications.
Strengths: Precise pH control, increased enzyme efficiency, and prolonged enzyme stability. Weaknesses: May require specialized equipment and careful monitoring, potentially increasing operational costs.
BASF Corp.
Technical Solution: BASF has engineered a sophisticated enzyme stabilization technology that leverages the effects of ammonium hydroxide on protein structure. Their approach involves encapsulating enzymes in a protective matrix that selectively allows ammonium hydroxide interaction[2]. This controlled exposure method has shown to enhance the thermostability of certain enzymes by up to 25°C[4]. BASF's technology also incorporates a gradual release mechanism for ammonium hydroxide, ensuring a consistent pH environment that promotes optimal enzyme conformation and activity[6]. The company has successfully applied this technology in various sectors, including detergents and biofuel production, reporting a 40% increase in product yield in some cases[8].
Strengths: Enhanced enzyme thermostability, broad industrial applicability, and significant yield improvements. Weaknesses: Complex formulation process and potential scalability challenges for certain applications.
Critical Research on Ammonium Hydroxide's Impact on Enzyme Kinetics
Immobilized enzyme and use thereof in continuous production
PatentPendingEP4438723A1
Innovation
- The use of a cyanuric chloride-activated amino carrier, combined with Polyethyleneimine (PEI) modification, to create a more stable and active immobilized enzyme, specifically for AmDH and Formate Dehydrogenase (FDH), which enhances catalytic activity and reusability.
Process for production of optically active amino acid or optically active amino acid amide
PatentInactiveEP2508615A1
Innovation
- The method involves using a biocatalyst selected from enzymes or microorganisms capable of stereoselectively hydrolyzing DL-tert-leucine amide, with continuous or intermittent separation of ammonia from the reaction solution using reduced pressure evaporation or adsorption on cation exchange resin or zeolite, to maintain enzyme activity and increase substrate concentration without increasing acid or enzyme amounts.
Safety and Environmental Considerations
The use of ammonium hydroxide in enzymatic processes necessitates careful consideration of safety and environmental factors. Ammonium hydroxide, a solution of ammonia in water, is corrosive and can pose significant risks to human health and the environment if not handled properly. Workers exposed to high concentrations may experience severe respiratory irritation, eye damage, and skin burns. Therefore, proper personal protective equipment, including respirators, goggles, and chemical-resistant gloves, is essential when handling this compound.
Adequate ventilation in laboratory and industrial settings is crucial to prevent the accumulation of ammonia vapors, which can be explosive in certain concentrations. Emergency eyewash stations and safety showers should be readily accessible in areas where ammonium hydroxide is used. Furthermore, strict protocols for storage, handling, and disposal must be implemented to prevent accidental releases.
From an environmental perspective, ammonium hydroxide can have detrimental effects on aquatic ecosystems if released into water bodies. It can cause pH changes and contribute to eutrophication, leading to algal blooms and oxygen depletion. Proper waste management and neutralization procedures are essential to mitigate these risks. Companies must comply with local and national regulations regarding the discharge of ammonia-containing wastewater.
The production and transportation of ammonium hydroxide also have environmental implications. Energy-intensive manufacturing processes contribute to greenhouse gas emissions, while transportation risks include potential spills or leaks. To address these concerns, organizations should consider implementing cleaner production technologies and optimizing logistics to reduce the environmental footprint.
In the context of enzymatic activity, the use of ammonium hydroxide as a pH regulator or in buffer solutions requires careful monitoring and control. Fluctuations in pH can not only affect enzyme performance but also potentially lead to the formation of harmful byproducts. Implementing robust process control systems and regular quality checks is crucial to ensure both safety and optimal enzymatic activity.
As sustainability becomes increasingly important, researchers and industries are exploring alternatives to ammonium hydroxide that may offer similar benefits with reduced environmental and safety risks. This includes the development of enzyme-friendly, biodegradable buffer systems and the use of less hazardous alkaline compounds. Ongoing research in green chemistry and bioengineering may provide safer and more environmentally friendly solutions for pH regulation in enzymatic processes.
Adequate ventilation in laboratory and industrial settings is crucial to prevent the accumulation of ammonia vapors, which can be explosive in certain concentrations. Emergency eyewash stations and safety showers should be readily accessible in areas where ammonium hydroxide is used. Furthermore, strict protocols for storage, handling, and disposal must be implemented to prevent accidental releases.
From an environmental perspective, ammonium hydroxide can have detrimental effects on aquatic ecosystems if released into water bodies. It can cause pH changes and contribute to eutrophication, leading to algal blooms and oxygen depletion. Proper waste management and neutralization procedures are essential to mitigate these risks. Companies must comply with local and national regulations regarding the discharge of ammonia-containing wastewater.
The production and transportation of ammonium hydroxide also have environmental implications. Energy-intensive manufacturing processes contribute to greenhouse gas emissions, while transportation risks include potential spills or leaks. To address these concerns, organizations should consider implementing cleaner production technologies and optimizing logistics to reduce the environmental footprint.
In the context of enzymatic activity, the use of ammonium hydroxide as a pH regulator or in buffer solutions requires careful monitoring and control. Fluctuations in pH can not only affect enzyme performance but also potentially lead to the formation of harmful byproducts. Implementing robust process control systems and regular quality checks is crucial to ensure both safety and optimal enzymatic activity.
As sustainability becomes increasingly important, researchers and industries are exploring alternatives to ammonium hydroxide that may offer similar benefits with reduced environmental and safety risks. This includes the development of enzyme-friendly, biodegradable buffer systems and the use of less hazardous alkaline compounds. Ongoing research in green chemistry and bioengineering may provide safer and more environmentally friendly solutions for pH regulation in enzymatic processes.
Analytical Techniques for Enzyme Activity Measurement
Analytical techniques for enzyme activity measurement are crucial for understanding how ammonium hydroxide affects enzymatic activity. These techniques provide quantitative data on enzyme kinetics, substrate conversion rates, and overall catalytic efficiency under various conditions.
Spectrophotometric assays are widely used for measuring enzyme activity in the presence of ammonium hydroxide. These methods rely on detecting changes in light absorption as substrates are converted to products. For example, the hydrolysis of p-nitrophenyl phosphate by alkaline phosphatase can be monitored by measuring the increase in absorbance at 405 nm due to the release of p-nitrophenol. This technique allows for real-time monitoring of enzyme activity and is particularly useful for studying the effects of ammonium hydroxide on reaction rates.
Fluorescence-based assays offer another sensitive approach for measuring enzyme activity. These methods utilize fluorogenic substrates that become fluorescent upon enzymatic cleavage. The intensity of fluorescence emission is directly proportional to the amount of product formed, allowing for precise quantification of enzyme activity. Fluorescence techniques are particularly advantageous when studying the effects of ammonium hydroxide on enzymes with low catalytic rates or in complex biological samples.
Electrochemical methods, such as amperometry and potentiometry, provide an alternative approach for measuring enzyme activity in the presence of ammonium hydroxide. These techniques detect changes in electrical current or potential resulting from enzymatic reactions. For instance, glucose oxidase activity can be measured by monitoring the production of hydrogen peroxide using an amperometric biosensor. Electrochemical methods offer high sensitivity and the ability to perform continuous measurements in real-time.
Calorimetric techniques, including isothermal titration calorimetry (ITC) and differential scanning calorimetry (DSC), measure the heat released or absorbed during enzymatic reactions. These methods provide valuable information on reaction thermodynamics and can be used to study the effects of ammonium hydroxide on enzyme stability and activity. ITC, in particular, allows for the determination of binding constants and reaction stoichiometry, offering insights into how ammonium hydroxide influences enzyme-substrate interactions.
High-performance liquid chromatography (HPLC) and mass spectrometry (MS) are powerful analytical tools for quantifying enzyme activity by directly measuring substrate depletion or product formation. These techniques offer high specificity and sensitivity, making them suitable for complex reaction mixtures. HPLC-MS can be particularly useful for studying the effects of ammonium hydroxide on enzyme-catalyzed reactions involving multiple substrates or products.
In conclusion, a combination of these analytical techniques provides a comprehensive understanding of how ammonium hydroxide affects enzymatic activity. By employing multiple methods, researchers can obtain complementary data on reaction kinetics, substrate specificity, and enzyme stability under various conditions. This multi-faceted approach ensures robust and reliable results when investigating the complex interactions between ammonium hydroxide and enzymatic systems.
Spectrophotometric assays are widely used for measuring enzyme activity in the presence of ammonium hydroxide. These methods rely on detecting changes in light absorption as substrates are converted to products. For example, the hydrolysis of p-nitrophenyl phosphate by alkaline phosphatase can be monitored by measuring the increase in absorbance at 405 nm due to the release of p-nitrophenol. This technique allows for real-time monitoring of enzyme activity and is particularly useful for studying the effects of ammonium hydroxide on reaction rates.
Fluorescence-based assays offer another sensitive approach for measuring enzyme activity. These methods utilize fluorogenic substrates that become fluorescent upon enzymatic cleavage. The intensity of fluorescence emission is directly proportional to the amount of product formed, allowing for precise quantification of enzyme activity. Fluorescence techniques are particularly advantageous when studying the effects of ammonium hydroxide on enzymes with low catalytic rates or in complex biological samples.
Electrochemical methods, such as amperometry and potentiometry, provide an alternative approach for measuring enzyme activity in the presence of ammonium hydroxide. These techniques detect changes in electrical current or potential resulting from enzymatic reactions. For instance, glucose oxidase activity can be measured by monitoring the production of hydrogen peroxide using an amperometric biosensor. Electrochemical methods offer high sensitivity and the ability to perform continuous measurements in real-time.
Calorimetric techniques, including isothermal titration calorimetry (ITC) and differential scanning calorimetry (DSC), measure the heat released or absorbed during enzymatic reactions. These methods provide valuable information on reaction thermodynamics and can be used to study the effects of ammonium hydroxide on enzyme stability and activity. ITC, in particular, allows for the determination of binding constants and reaction stoichiometry, offering insights into how ammonium hydroxide influences enzyme-substrate interactions.
High-performance liquid chromatography (HPLC) and mass spectrometry (MS) are powerful analytical tools for quantifying enzyme activity by directly measuring substrate depletion or product formation. These techniques offer high specificity and sensitivity, making them suitable for complex reaction mixtures. HPLC-MS can be particularly useful for studying the effects of ammonium hydroxide on enzyme-catalyzed reactions involving multiple substrates or products.
In conclusion, a combination of these analytical techniques provides a comprehensive understanding of how ammonium hydroxide affects enzymatic activity. By employing multiple methods, researchers can obtain complementary data on reaction kinetics, substrate specificity, and enzyme stability under various conditions. This multi-faceted approach ensures robust and reliable results when investigating the complex interactions between ammonium hydroxide and enzymatic systems.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!