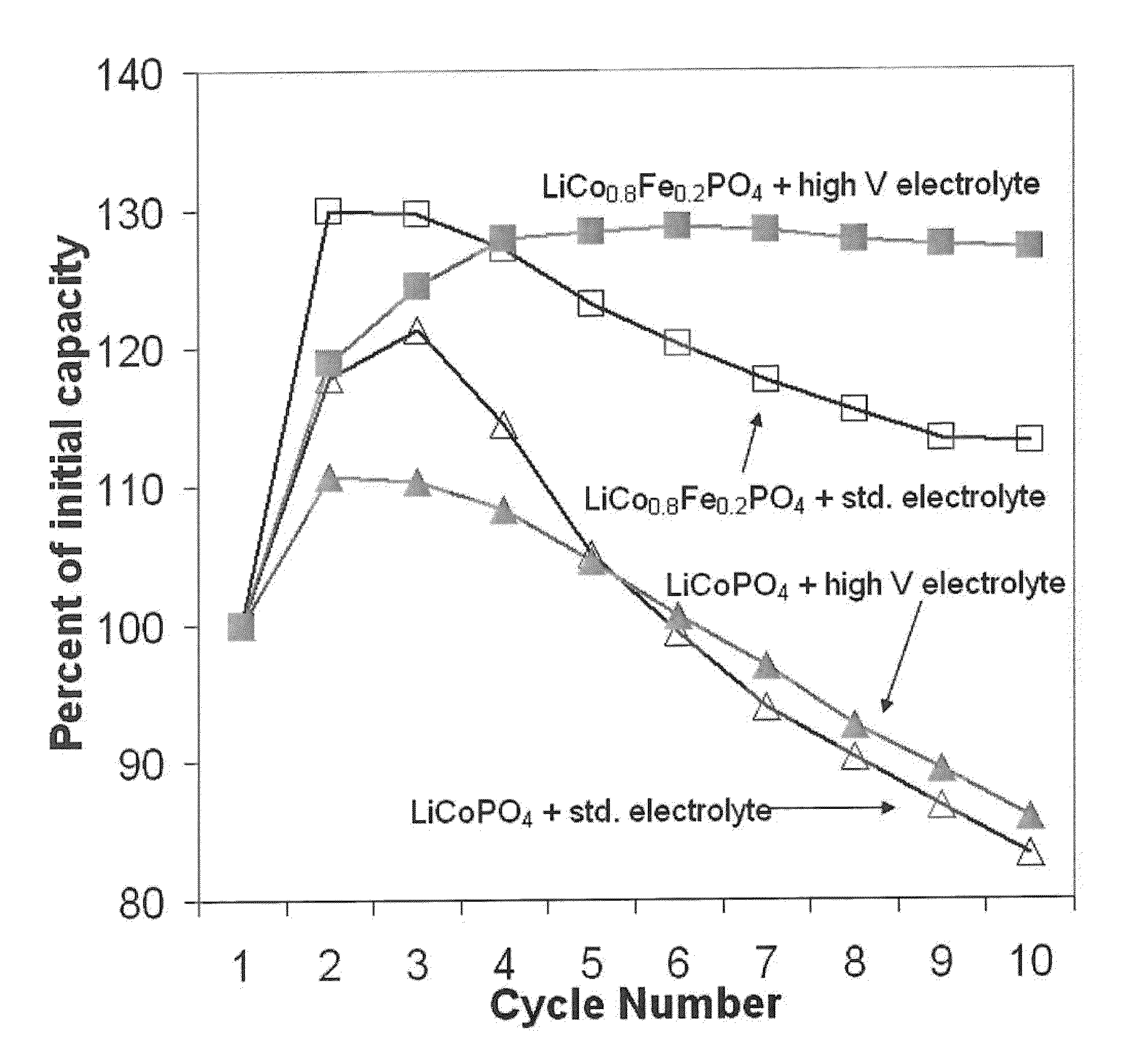
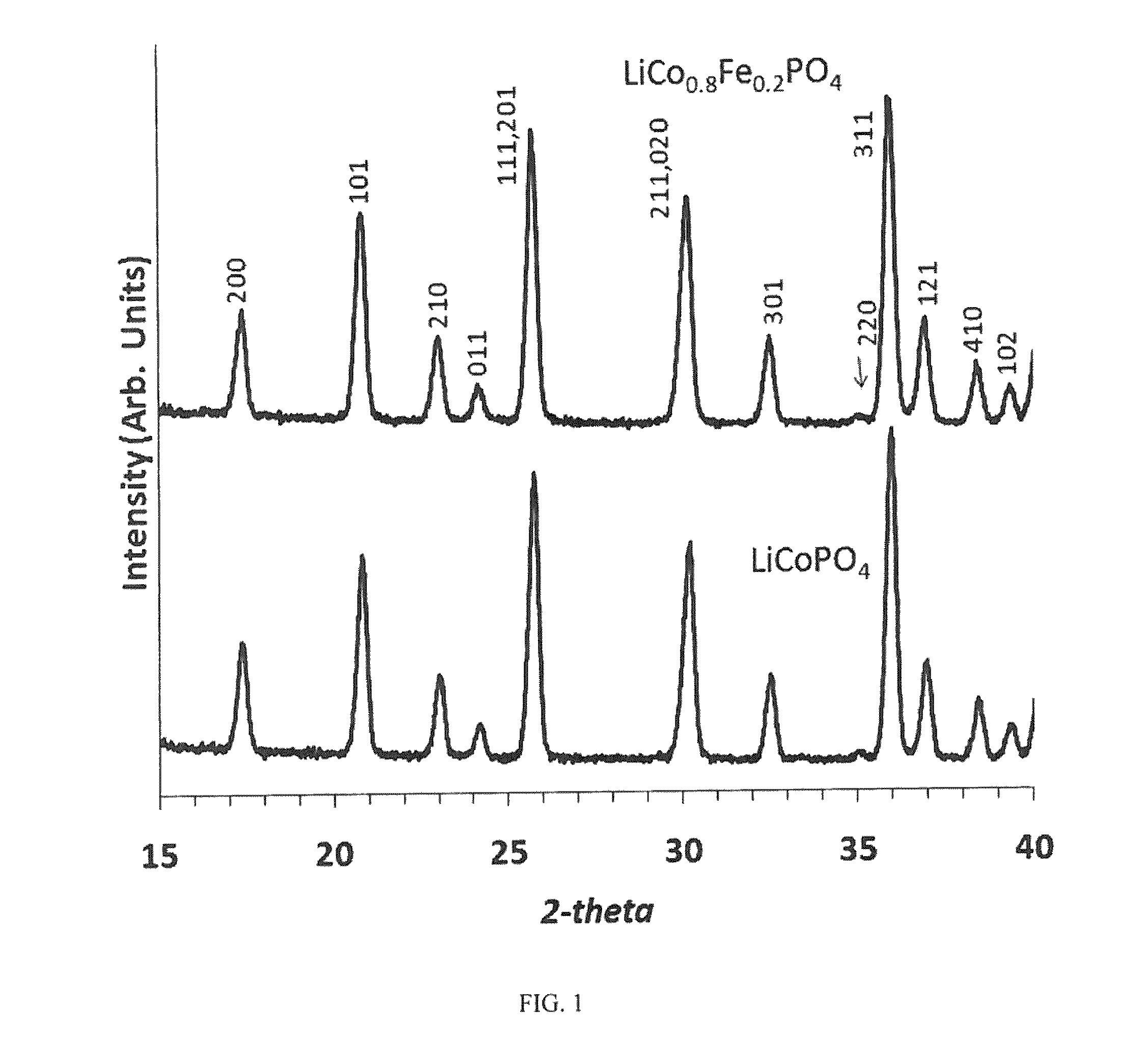
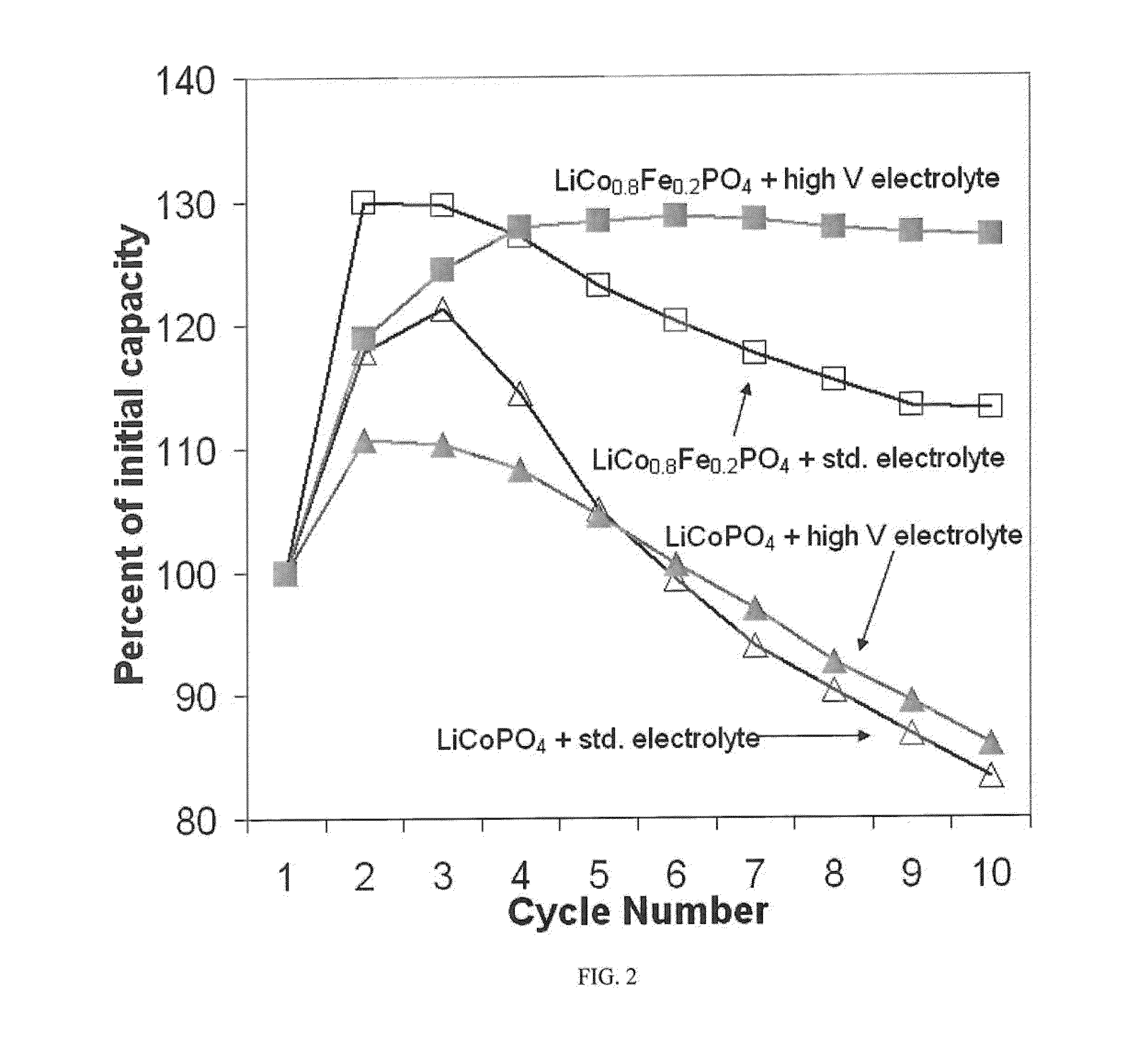
Optimize Lithium Phosphate Electrodes to Increase Cycle Life
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
LFP Battery Technology Evolution and Objectives
Lithium Iron Phosphate (LFP) battery technology has evolved significantly since its initial development in the late 1990s. The journey began when researchers at the University of Texas identified LiFePO4 as a potential cathode material, offering a safer alternative to traditional lithium-ion batteries. This discovery marked the beginning of a technological evolution that has continued to accelerate, particularly in the last decade, as demand for safer, more sustainable energy storage solutions has grown exponentially.
The early 2000s saw the first commercial applications of LFP batteries, primarily in power tools and early electric vehicles. However, these early iterations faced significant challenges, including lower energy density compared to competing lithium-ion chemistries and manufacturing complexities that limited widespread adoption. The technology breakthrough came around 2005-2007 when nano-scale engineering and carbon coating techniques were developed, substantially improving the electronic conductivity of LFP materials.
By 2010-2015, LFP batteries had established a foothold in the market, particularly in applications where safety and longevity were paramount concerns. The technology's inherent thermal stability, resistance to thermal runaway, and longer cycle life made it increasingly attractive for grid storage applications and certain electric vehicle segments. During this period, manufacturing processes were refined, leading to significant cost reductions and improved performance metrics.
Recent years (2016-2023) have witnessed remarkable advancements in LFP electrode optimization. Innovations in particle morphology control, electrolyte formulations, and electrode architecture have pushed the boundaries of what was previously thought possible with this chemistry. The cell-to-pack (CTP) technology introduced around 2019-2020 has been particularly transformative, allowing LFP batteries to achieve energy densities that make them competitive with other lithium-ion variants in more applications.
The current technological trajectory aims to address several key objectives. Primary among these is extending cycle life beyond the current benchmark of 2,000-3,000 cycles to potentially 5,000-6,000 cycles for mainstream applications. This would significantly reduce the lifetime cost of battery systems and enable new use cases where frequent cycling is required. Researchers are also focused on improving rate capability, particularly fast-charging performance, which has traditionally been a limitation of LFP chemistry.
Another critical objective is enhancing low-temperature performance, as LFP batteries typically suffer from reduced capacity and power output in cold conditions. Parallel efforts are underway to increase energy density through advanced electrode designs and optimized cell architectures, with the goal of reaching 200-220 Wh/kg at the cell level while maintaining LFP's inherent safety advantages and cost benefits.
The early 2000s saw the first commercial applications of LFP batteries, primarily in power tools and early electric vehicles. However, these early iterations faced significant challenges, including lower energy density compared to competing lithium-ion chemistries and manufacturing complexities that limited widespread adoption. The technology breakthrough came around 2005-2007 when nano-scale engineering and carbon coating techniques were developed, substantially improving the electronic conductivity of LFP materials.
By 2010-2015, LFP batteries had established a foothold in the market, particularly in applications where safety and longevity were paramount concerns. The technology's inherent thermal stability, resistance to thermal runaway, and longer cycle life made it increasingly attractive for grid storage applications and certain electric vehicle segments. During this period, manufacturing processes were refined, leading to significant cost reductions and improved performance metrics.
Recent years (2016-2023) have witnessed remarkable advancements in LFP electrode optimization. Innovations in particle morphology control, electrolyte formulations, and electrode architecture have pushed the boundaries of what was previously thought possible with this chemistry. The cell-to-pack (CTP) technology introduced around 2019-2020 has been particularly transformative, allowing LFP batteries to achieve energy densities that make them competitive with other lithium-ion variants in more applications.
The current technological trajectory aims to address several key objectives. Primary among these is extending cycle life beyond the current benchmark of 2,000-3,000 cycles to potentially 5,000-6,000 cycles for mainstream applications. This would significantly reduce the lifetime cost of battery systems and enable new use cases where frequent cycling is required. Researchers are also focused on improving rate capability, particularly fast-charging performance, which has traditionally been a limitation of LFP chemistry.
Another critical objective is enhancing low-temperature performance, as LFP batteries typically suffer from reduced capacity and power output in cold conditions. Parallel efforts are underway to increase energy density through advanced electrode designs and optimized cell architectures, with the goal of reaching 200-220 Wh/kg at the cell level while maintaining LFP's inherent safety advantages and cost benefits.
Market Analysis for Long-Cycle Battery Applications
The long-cycle battery market is experiencing unprecedented growth driven by increasing demand across multiple sectors. Electric vehicles (EVs) represent the largest and fastest-growing segment, with global EV sales projected to reach 14 million units in 2023, up from 10.5 million in 2022. This market is expected to maintain a compound annual growth rate (CAGR) of approximately 25% through 2030, creating substantial demand for batteries with extended cycle life capabilities.
Energy storage systems (ESS) constitute another significant market segment, particularly for grid-scale applications and renewable energy integration. This sector is growing at a CAGR of 20-22%, with particular emphasis on batteries that can withstand 3,000+ charge cycles while maintaining high capacity retention. The economic viability of these systems depends heavily on battery longevity, making cycle life optimization a critical factor in market adoption.
Industrial applications, including material handling equipment, backup power systems, and telecommunications infrastructure, represent a stable but growing market segment with specific requirements for long-cycle batteries. These applications typically demand 2,000-5,000 cycle capabilities under varied operational conditions, creating a specialized market niche estimated at $12 billion globally.
Consumer electronics manufacturers are increasingly prioritizing device longevity as a competitive advantage, driving demand for batteries that can maintain performance over 1,000+ cycles. This trend is particularly evident in premium laptop, smartphone, and wearable device segments where manufacturers are willing to pay premium prices for advanced battery technologies.
Geographically, Asia-Pacific dominates the long-cycle battery market with China leading manufacturing capacity, followed by South Korea and Japan. However, significant investments in North America and Europe are reshaping the global landscape, with new gigafactories specifically designed for advanced lithium phosphate electrode production.
Price sensitivity varies significantly across application segments. While EV manufacturers remain highly cost-conscious (targeting below $100/kWh), grid storage operators demonstrate greater willingness to pay premium prices for proven cycle life advantages. The industrial segment occupies a middle ground, balancing initial cost against total cost of ownership calculations.
Market forecasts indicate that batteries with optimized lithium phosphate electrodes capable of achieving 3,000+ cycles while maintaining 80% capacity retention could command a 15-20% price premium in current markets. This premium is expected to normalize as production scales, but early market entrants with demonstrable cycle life advantages stand to capture significant market share and establish technology leadership positions.
Energy storage systems (ESS) constitute another significant market segment, particularly for grid-scale applications and renewable energy integration. This sector is growing at a CAGR of 20-22%, with particular emphasis on batteries that can withstand 3,000+ charge cycles while maintaining high capacity retention. The economic viability of these systems depends heavily on battery longevity, making cycle life optimization a critical factor in market adoption.
Industrial applications, including material handling equipment, backup power systems, and telecommunications infrastructure, represent a stable but growing market segment with specific requirements for long-cycle batteries. These applications typically demand 2,000-5,000 cycle capabilities under varied operational conditions, creating a specialized market niche estimated at $12 billion globally.
Consumer electronics manufacturers are increasingly prioritizing device longevity as a competitive advantage, driving demand for batteries that can maintain performance over 1,000+ cycles. This trend is particularly evident in premium laptop, smartphone, and wearable device segments where manufacturers are willing to pay premium prices for advanced battery technologies.
Geographically, Asia-Pacific dominates the long-cycle battery market with China leading manufacturing capacity, followed by South Korea and Japan. However, significant investments in North America and Europe are reshaping the global landscape, with new gigafactories specifically designed for advanced lithium phosphate electrode production.
Price sensitivity varies significantly across application segments. While EV manufacturers remain highly cost-conscious (targeting below $100/kWh), grid storage operators demonstrate greater willingness to pay premium prices for proven cycle life advantages. The industrial segment occupies a middle ground, balancing initial cost against total cost of ownership calculations.
Market forecasts indicate that batteries with optimized lithium phosphate electrodes capable of achieving 3,000+ cycles while maintaining 80% capacity retention could command a 15-20% price premium in current markets. This premium is expected to normalize as production scales, but early market entrants with demonstrable cycle life advantages stand to capture significant market share and establish technology leadership positions.
Current LFP Electrode Limitations and Technical Barriers
Lithium iron phosphate (LFP) electrodes, while offering advantages in safety, cost, and environmental impact, face significant limitations that hinder their widespread adoption in high-performance energy storage applications. The primary technical barrier is their relatively low energy density compared to other lithium-ion chemistries, with theoretical capacity limited to approximately 170 mAh/g. This inherent limitation stems from the fundamental crystal structure and electrochemical properties of the LiFePO4 material.
A critical challenge in LFP electrode optimization is the poor electronic conductivity, typically around 10^-9 S/cm, which is several orders of magnitude lower than other cathode materials. This poor conductivity leads to increased internal resistance, voltage hysteresis, and reduced rate capability, particularly at high discharge rates or low temperatures. The issue is further exacerbated during cycling, as the formation of insulating phases can progressively degrade electrode performance.
The lithium-ion diffusion kinetics in LFP electrodes present another significant barrier. The one-dimensional diffusion channels in the olivine structure are easily blocked by defects or structural distortions, creating "bottlenecks" that impede ion transport. This characteristic manifests as capacity fade during cycling, especially at higher C-rates, limiting the practical application of LFP in fast-charging scenarios.
Electrode manufacturing inconsistencies contribute substantially to performance variability. Current industrial processes struggle with achieving uniform particle size distribution, consistent carbon coating, and homogeneous conductive additive dispersion. These manufacturing challenges result in electrodes with non-uniform current distribution, localized hotspots, and accelerated degradation in certain regions.
The electrode-electrolyte interface stability represents another critical barrier. While LFP is generally considered more stable than other cathode materials, prolonged cycling still leads to interfacial degradation. The formation and growth of solid electrolyte interphase (SEI) layers consume active lithium and increase cell impedance over time. Current electrolyte formulations are not optimized specifically for LFP chemistry, leading to suboptimal interface properties.
Mechanical stress during cycling constitutes a significant degradation mechanism. The volume changes during lithiation/delithiation, though smaller than in some other electrode materials, still cause particle cracking and electrode delamination over extended cycling. This mechanical degradation is particularly pronounced at elevated temperatures or during fast charging conditions, leading to accelerated capacity fade and reduced cycle life.
Addressing these technical barriers requires a multidisciplinary approach combining materials science, electrochemistry, and advanced manufacturing techniques to develop next-generation LFP electrodes with enhanced performance characteristics and extended cycle life.
A critical challenge in LFP electrode optimization is the poor electronic conductivity, typically around 10^-9 S/cm, which is several orders of magnitude lower than other cathode materials. This poor conductivity leads to increased internal resistance, voltage hysteresis, and reduced rate capability, particularly at high discharge rates or low temperatures. The issue is further exacerbated during cycling, as the formation of insulating phases can progressively degrade electrode performance.
The lithium-ion diffusion kinetics in LFP electrodes present another significant barrier. The one-dimensional diffusion channels in the olivine structure are easily blocked by defects or structural distortions, creating "bottlenecks" that impede ion transport. This characteristic manifests as capacity fade during cycling, especially at higher C-rates, limiting the practical application of LFP in fast-charging scenarios.
Electrode manufacturing inconsistencies contribute substantially to performance variability. Current industrial processes struggle with achieving uniform particle size distribution, consistent carbon coating, and homogeneous conductive additive dispersion. These manufacturing challenges result in electrodes with non-uniform current distribution, localized hotspots, and accelerated degradation in certain regions.
The electrode-electrolyte interface stability represents another critical barrier. While LFP is generally considered more stable than other cathode materials, prolonged cycling still leads to interfacial degradation. The formation and growth of solid electrolyte interphase (SEI) layers consume active lithium and increase cell impedance over time. Current electrolyte formulations are not optimized specifically for LFP chemistry, leading to suboptimal interface properties.
Mechanical stress during cycling constitutes a significant degradation mechanism. The volume changes during lithiation/delithiation, though smaller than in some other electrode materials, still cause particle cracking and electrode delamination over extended cycling. This mechanical degradation is particularly pronounced at elevated temperatures or during fast charging conditions, leading to accelerated capacity fade and reduced cycle life.
Addressing these technical barriers requires a multidisciplinary approach combining materials science, electrochemistry, and advanced manufacturing techniques to develop next-generation LFP electrodes with enhanced performance characteristics and extended cycle life.
Current Approaches to LFP Electrode Optimization
01 Electrode composition modifications for improved cycle life
Modifying the composition of lithium phosphate electrodes can significantly enhance their cycle life. This includes incorporating specific dopants, optimizing particle size distribution, and using composite structures. These modifications help to stabilize the electrode structure during charging and discharging cycles, reduce capacity fading, and improve overall electrochemical performance. The enhanced structural stability leads to better retention of capacity over numerous cycles.- Electrode composition modifications for improved cycle life: Various modifications to lithium phosphate electrode compositions can significantly enhance cycle life. These include doping with elements like manganese, nickel, or cobalt, creating composite structures with carbon materials, and optimizing particle size distribution. These modifications improve structural stability, electronic conductivity, and resistance to degradation during repeated charge-discharge cycles, resulting in batteries with extended operational lifespans.
- Coating technologies for lithium phosphate electrodes: Protective coatings applied to lithium phosphate electrode materials can substantially improve cycle life performance. These coatings, which may include carbon layers, metal oxides, or polymer films, act as barriers against electrolyte degradation reactions and help maintain structural integrity during cycling. Advanced coating techniques ensure uniform coverage and optimal thickness, preventing capacity fade and extending battery lifespan.
- Electrolyte formulations compatible with lithium phosphate: Specialized electrolyte formulations designed for lithium phosphate electrodes can significantly enhance cycle life. These formulations may include additives that form stable solid-electrolyte interfaces, solvents with improved stability at high voltages, and lithium salts that minimize side reactions. The optimized electrolyte chemistry reduces degradation mechanisms at the electrode-electrolyte interface, leading to more durable battery performance.
- Manufacturing processes affecting cycle stability: Advanced manufacturing techniques for lithium phosphate electrodes directly impact cycle life performance. Processes such as controlled synthesis methods, precise temperature treatments, and innovative electrode fabrication techniques can optimize crystal structure, particle morphology, and electrode porosity. These manufacturing improvements lead to more homogeneous materials with fewer defects, resulting in batteries with superior cycling stability.
- Battery management strategies for extending cycle life: Intelligent battery management systems and charging protocols can maximize the cycle life of lithium phosphate electrodes. These strategies include optimized charging rates, voltage limits tailored to lithium phosphate chemistry, temperature management systems, and algorithms that prevent harmful operating conditions. By controlling the operational parameters within ideal ranges, these approaches minimize stress factors that contribute to electrode degradation over time.
02 Surface coating and treatment techniques
Various surface coating and treatment techniques can be applied to lithium phosphate electrodes to extend their cycle life. These include carbon coating, metal oxide coatings, polymer coatings, and surface functionalization. Such treatments create protective layers that prevent direct contact between the electrode material and the electrolyte, reducing unwanted side reactions and electrolyte decomposition. This results in improved structural integrity during cycling and enhanced electrochemical stability.Expand Specific Solutions03 Electrolyte formulation and additives
The formulation of electrolytes and incorporation of specific additives play a crucial role in extending the cycle life of lithium phosphate electrodes. Optimized electrolyte compositions with functional additives can form stable solid electrolyte interphase (SEI) layers, prevent electrode dissolution, and mitigate side reactions. These formulations help maintain electrode integrity during repeated cycling and improve the overall performance and longevity of lithium phosphate-based battery systems.Expand Specific Solutions04 Advanced manufacturing processes
Advanced manufacturing processes significantly impact the cycle life of lithium phosphate electrodes. Techniques such as controlled synthesis methods, precise temperature regulation during production, optimized sintering processes, and innovative electrode assembly methods can enhance the structural integrity and electrochemical performance of these electrodes. These manufacturing innovations result in more uniform particle distribution, better interfacial contacts, and reduced defects, all contributing to extended cycle life.Expand Specific Solutions05 Nanostructured and composite electrode designs
Nanostructured and composite designs for lithium phosphate electrodes offer significant improvements in cycle life. These include core-shell structures, hierarchical architectures, 3D electrode frameworks, and carbon-based composites. Such designs provide enhanced electron transport pathways, accommodate volume changes during cycling, and offer larger electrode-electrolyte contact areas. The improved structural stability and electrochemical kinetics result in better capacity retention and extended cycle life for lithium phosphate-based battery systems.Expand Specific Solutions
Leading Companies in LFP Battery Technology
The lithium phosphate electrode optimization market is in a growth phase, with increasing demand driven by the electric vehicle and energy storage sectors. The market size is expanding rapidly, projected to reach significant value as battery technology becomes central to renewable energy adoption. Technologically, the field is advancing from early-stage development toward commercial maturity, with key players demonstrating varying levels of innovation. Companies like Contemporary Amperex Technology (CATL), LG Energy Solution, and Samsung SDI lead with established manufacturing capabilities, while specialized firms such as StoreDot and Sion Power focus on breakthrough technologies. Research institutions including National Taiwan University and CNRS contribute fundamental advancements. The competitive landscape features both traditional battery manufacturers and newer entrants developing next-generation solutions to address the critical challenge of extending battery cycle life.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed an advanced lithium iron phosphate (LFP) electrode optimization technology called "Cell-to-Pack" (CTP) that significantly improves cycle life. Their approach involves nano-engineering of LFP particles with carbon coating and doping with elements like manganese and zinc to enhance conductivity. CATL's electrodes feature precisely controlled particle size distribution (1-5μm) and morphology to maximize active material utilization. They've implemented a gradient porosity structure that facilitates better lithium-ion diffusion while maintaining structural integrity during cycling. Their proprietary electrolyte additives form a more stable solid electrolyte interphase (SEI) layer, reducing parasitic reactions that typically degrade cycle life. CATL has reported achieving over 6,000 cycles at 80% capacity retention, representing a substantial improvement over conventional LFP electrodes.
Strengths: Industry-leading cycle life performance with excellent thermal stability and safety characteristics. Their integrated manufacturing approach ensures consistent quality control. Weaknesses: The complex nano-engineering processes increase production costs compared to standard LFP electrodes, and the technology requires specialized manufacturing equipment.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed a proprietary "SRS" (Stress Relief Structure) technology for lithium phosphate electrodes that significantly enhances cycle life. Their approach incorporates a three-dimensional conductive network within the electrode structure using carbon nanotubes and graphene derivatives, creating flexible pathways that accommodate volume changes during cycling. The company employs a dual-phase coating technique where primary LFP particles (approximately 100nm) are first coated with conductive carbon, then assembled into secondary structures with polymer binders engineered to maintain mechanical integrity over thousands of cycles. LG's electrodes feature gradient porosity designs with higher porosity near the separator interface, optimizing ion transport while maintaining structural stability. Their manufacturing process includes precise control of electrode calendering parameters to achieve optimal density (2.8-3.2 g/cm³) without compromising ion diffusion pathways.
Strengths: Superior mechanical stability during cycling with excellent rate capability and low temperature performance compared to conventional LFP electrodes. Their technology is compatible with existing manufacturing infrastructure. Weaknesses: The complex electrode architecture requires precise quality control during manufacturing, and the advanced materials increase production costs by approximately 15-20% compared to standard LFP formulations.
Key Patents and Innovations in Electrode Material Science
High voltage lithium ion positive electrode material with improved cycle life
PatentInactiveUS9114779B2
Innovation
- Development of a substituted lithiated metal phosphate material with the formula Li1-3tM2+1-t-dTt3+Dd2+PO4, where M includes Co and/or other metals, T includes Fe, Al, or Ga, and D includes additional divalent ions, to enhance cycle life and conductivity by stabilizing the triphylite structure during lithium ion transfer.
A process to enhance the cycle life of li-ion battery electrode
PatentPendingTW201626619A
Innovation
- A plasma-assisted chemical vapor deposition method is used to form a fluorocarbon polymer film on the electrode surface, enhancing its durability and conductivity.
Material Sustainability and Resource Considerations
The sustainability of lithium phosphate electrode materials represents a critical dimension in the advancement of battery technology with extended cycle life. The sourcing of raw materials for lithium iron phosphate (LFP) cathodes presents significant advantages over other lithium-ion battery chemistries. Phosphorus and iron are abundant elements in the Earth's crust, making LFP batteries less susceptible to supply chain disruptions compared to cobalt or nickel-based alternatives. This abundance translates to reduced extraction impacts and more stable pricing, supporting long-term manufacturing sustainability.
Resource efficiency in electrode manufacturing processes directly impacts cycle life optimization. Current production methods for lithium phosphate electrodes typically involve energy-intensive calcination steps at temperatures exceeding 600°C. Innovations in low-temperature synthesis routes, such as hydrothermal or sol-gel methods, can reduce energy consumption by 30-40% while potentially yielding materials with more uniform morphology that contributes to extended cycle life.
The recyclability of lithium phosphate materials presents both opportunities and challenges. Unlike cobalt-based cathodes, the lower intrinsic material value of LFP has historically limited recycling incentives. However, recent advances in direct recycling technologies have demonstrated recovery rates exceeding 90% for lithium and phosphate components, with the recovered materials showing comparable electrochemical performance to virgin materials when reintroduced into new electrodes.
Water consumption represents another critical sustainability metric in electrode manufacturing. Conventional wet processing methods require approximately 5-7 liters of water per kWh of battery capacity produced. Emerging dry processing techniques can reduce this water footprint by up to 80% while simultaneously eliminating the need for environmentally problematic N-Methyl-2-pyrrolidone (NMP) solvents, contributing to both environmental sustainability and potentially enhanced electrode performance.
Life cycle assessment (LCA) studies indicate that optimizing lithium phosphate electrodes for extended cycle life delivers compounding sustainability benefits. Each doubling of cycle life effectively halves the environmental impact per unit of energy delivered over the battery's lifetime. This relationship underscores the importance of viewing sustainability not merely as a manufacturing consideration but as intrinsically linked to performance optimization strategies.
The carbon footprint associated with lithium phosphate electrode production currently ranges from 35-50 kg CO₂-equivalent per kWh of battery capacity. Implementing best practices in sustainable manufacturing could reduce this by approximately 25%, with further reductions possible through renewable energy integration in production facilities and optimized local supply chains that minimize transportation emissions.
Resource efficiency in electrode manufacturing processes directly impacts cycle life optimization. Current production methods for lithium phosphate electrodes typically involve energy-intensive calcination steps at temperatures exceeding 600°C. Innovations in low-temperature synthesis routes, such as hydrothermal or sol-gel methods, can reduce energy consumption by 30-40% while potentially yielding materials with more uniform morphology that contributes to extended cycle life.
The recyclability of lithium phosphate materials presents both opportunities and challenges. Unlike cobalt-based cathodes, the lower intrinsic material value of LFP has historically limited recycling incentives. However, recent advances in direct recycling technologies have demonstrated recovery rates exceeding 90% for lithium and phosphate components, with the recovered materials showing comparable electrochemical performance to virgin materials when reintroduced into new electrodes.
Water consumption represents another critical sustainability metric in electrode manufacturing. Conventional wet processing methods require approximately 5-7 liters of water per kWh of battery capacity produced. Emerging dry processing techniques can reduce this water footprint by up to 80% while simultaneously eliminating the need for environmentally problematic N-Methyl-2-pyrrolidone (NMP) solvents, contributing to both environmental sustainability and potentially enhanced electrode performance.
Life cycle assessment (LCA) studies indicate that optimizing lithium phosphate electrodes for extended cycle life delivers compounding sustainability benefits. Each doubling of cycle life effectively halves the environmental impact per unit of energy delivered over the battery's lifetime. This relationship underscores the importance of viewing sustainability not merely as a manufacturing consideration but as intrinsically linked to performance optimization strategies.
The carbon footprint associated with lithium phosphate electrode production currently ranges from 35-50 kg CO₂-equivalent per kWh of battery capacity. Implementing best practices in sustainable manufacturing could reduce this by approximately 25%, with further reductions possible through renewable energy integration in production facilities and optimized local supply chains that minimize transportation emissions.
Manufacturing Scalability and Cost Analysis
The scalability of lithium phosphate electrode manufacturing processes represents a critical factor in the widespread adoption of this technology for enhanced cycle life batteries. Current manufacturing methods for lithium phosphate electrodes involve several complex steps including material synthesis, slurry preparation, coating, drying, calendering, and cell assembly. These processes require significant capital investment and specialized equipment, creating barriers to rapid scaling.
Analysis of production costs reveals that material expenses constitute approximately 60-70% of total manufacturing costs for lithium phosphate electrodes. The remaining costs are distributed among equipment depreciation, energy consumption, labor, and quality control measures. Recent innovations in continuous flow synthesis and water-based processing have demonstrated potential cost reductions of 15-25% compared to traditional batch processing methods.
Energy consumption during high-temperature calcination and extended drying processes significantly impacts both manufacturing costs and environmental footprint. Advanced manufacturing techniques utilizing microwave-assisted synthesis and rapid thermal processing have shown promising results in reducing energy requirements by up to 40% while maintaining or even improving electrode performance characteristics.
Equipment utilization efficiency presents another critical factor in manufacturing economics. Current industry standards achieve approximately 65-75% equipment utilization rates, with significant downtime for maintenance, calibration, and product changeovers. Implementation of Industry 4.0 principles with predictive maintenance and real-time quality monitoring systems has demonstrated potential to increase utilization rates to over 85% in pilot facilities.
Supply chain considerations also impact manufacturing scalability, particularly regarding the availability of high-purity raw materials. Phosphate sources with consistent quality specifications remain a potential bottleneck for large-scale production. Diversification of supply sources and development of purification technologies for lower-grade materials represent important strategies for ensuring manufacturing resilience.
Cost modeling projections indicate that with optimized manufacturing processes and economies of scale, lithium phosphate electrodes with enhanced cycle life could reach cost parity with conventional lithium-ion battery electrodes within 3-5 years. This timeline assumes continued improvements in process efficiency and material utilization rates, with particular emphasis on reducing waste generation during manufacturing.
Analysis of production costs reveals that material expenses constitute approximately 60-70% of total manufacturing costs for lithium phosphate electrodes. The remaining costs are distributed among equipment depreciation, energy consumption, labor, and quality control measures. Recent innovations in continuous flow synthesis and water-based processing have demonstrated potential cost reductions of 15-25% compared to traditional batch processing methods.
Energy consumption during high-temperature calcination and extended drying processes significantly impacts both manufacturing costs and environmental footprint. Advanced manufacturing techniques utilizing microwave-assisted synthesis and rapid thermal processing have shown promising results in reducing energy requirements by up to 40% while maintaining or even improving electrode performance characteristics.
Equipment utilization efficiency presents another critical factor in manufacturing economics. Current industry standards achieve approximately 65-75% equipment utilization rates, with significant downtime for maintenance, calibration, and product changeovers. Implementation of Industry 4.0 principles with predictive maintenance and real-time quality monitoring systems has demonstrated potential to increase utilization rates to over 85% in pilot facilities.
Supply chain considerations also impact manufacturing scalability, particularly regarding the availability of high-purity raw materials. Phosphate sources with consistent quality specifications remain a potential bottleneck for large-scale production. Diversification of supply sources and development of purification technologies for lower-grade materials represent important strategies for ensuring manufacturing resilience.
Cost modeling projections indicate that with optimized manufacturing processes and economies of scale, lithium phosphate electrodes with enhanced cycle life could reach cost parity with conventional lithium-ion battery electrodes within 3-5 years. This timeline assumes continued improvements in process efficiency and material utilization rates, with particular emphasis on reducing waste generation during manufacturing.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!