Autoclave Sterilization Cycles: Validating New Load Types
SEP 2, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Autoclave Sterilization Technology Background and Objectives
Autoclave sterilization technology has evolved significantly since its inception in the late 19th century when Charles Chamberland developed the first pressure steam sterilizer. This technology leverages the principles of moist heat under pressure to eliminate microorganisms, including bacterial spores, which are highly resistant to other sterilization methods. The fundamental mechanism involves exposing materials to saturated steam at temperatures typically ranging from 121°C to 134°C under pressure of 15-30 psi for specified time periods, ensuring complete microbial inactivation through protein denaturation and coagulation.
Over the decades, autoclave technology has progressed from basic manual systems to sophisticated computer-controlled units with precise parameter monitoring capabilities. Modern autoclaves incorporate advanced features such as air removal systems, rapid cooling mechanisms, and validation software that ensures cycle reproducibility and documentation compliance with regulatory standards including FDA, ISO, and EN requirements.
The current technological landscape presents diverse autoclave types, including gravity displacement, pre-vacuum, and steam-flush pressure-pulse systems, each designed to address specific sterilization challenges across various industries. Healthcare facilities predominantly utilize pre-vacuum autoclaves for their enhanced air removal capabilities, while pharmaceutical and biotechnology sectors often employ more specialized configurations for their unique product requirements.
The validation of new load types represents a critical frontier in autoclave technology development. As materials, packaging configurations, and product densities continue to diversify, traditional validation protocols may no longer adequately address the sterilization challenges presented by novel load compositions. This necessitates the development of more sophisticated validation methodologies that can accurately assess sterility assurance levels across heterogeneous load configurations.
The primary objective of current research in autoclave sterilization technology is to establish robust, scientifically sound validation protocols for new load types that ensure consistent sterility while optimizing cycle efficiency. This includes developing mathematical models that can predict heat penetration and microbial inactivation kinetics in complex loads, implementing real-time monitoring systems that provide spatial temperature mapping throughout the load, and creating adaptive control algorithms that can modify cycle parameters based on load characteristics.
Additional technological goals include reducing environmental impact through water and energy conservation, minimizing material degradation during sterilization processes, and developing integration capabilities with Industry 4.0 systems for enhanced process control and documentation. These advancements aim to address the growing demands for higher throughput, improved quality assurance, and reduced operational costs while maintaining absolute sterility assurance.
Over the decades, autoclave technology has progressed from basic manual systems to sophisticated computer-controlled units with precise parameter monitoring capabilities. Modern autoclaves incorporate advanced features such as air removal systems, rapid cooling mechanisms, and validation software that ensures cycle reproducibility and documentation compliance with regulatory standards including FDA, ISO, and EN requirements.
The current technological landscape presents diverse autoclave types, including gravity displacement, pre-vacuum, and steam-flush pressure-pulse systems, each designed to address specific sterilization challenges across various industries. Healthcare facilities predominantly utilize pre-vacuum autoclaves for their enhanced air removal capabilities, while pharmaceutical and biotechnology sectors often employ more specialized configurations for their unique product requirements.
The validation of new load types represents a critical frontier in autoclave technology development. As materials, packaging configurations, and product densities continue to diversify, traditional validation protocols may no longer adequately address the sterilization challenges presented by novel load compositions. This necessitates the development of more sophisticated validation methodologies that can accurately assess sterility assurance levels across heterogeneous load configurations.
The primary objective of current research in autoclave sterilization technology is to establish robust, scientifically sound validation protocols for new load types that ensure consistent sterility while optimizing cycle efficiency. This includes developing mathematical models that can predict heat penetration and microbial inactivation kinetics in complex loads, implementing real-time monitoring systems that provide spatial temperature mapping throughout the load, and creating adaptive control algorithms that can modify cycle parameters based on load characteristics.
Additional technological goals include reducing environmental impact through water and energy conservation, minimizing material degradation during sterilization processes, and developing integration capabilities with Industry 4.0 systems for enhanced process control and documentation. These advancements aim to address the growing demands for higher throughput, improved quality assurance, and reduced operational costs while maintaining absolute sterility assurance.
Market Demand Analysis for Validated Sterilization Processes
The global market for validated sterilization processes has experienced significant growth in recent years, driven primarily by increasing regulatory requirements and heightened awareness of infection control across healthcare, pharmaceutical, and food processing industries. The autoclave sterilization segment, particularly for new load type validation, represents a critical component of this expanding market.
Healthcare facilities worldwide are facing stricter compliance standards regarding sterilization validation, with regulatory bodies such as the FDA, EMA, and ISO implementing more comprehensive guidelines. This regulatory pressure has created substantial demand for validated sterilization processes that can accommodate diverse and evolving load types, from complex surgical instruments to new composite materials used in medical devices.
Market research indicates that the global sterilization equipment market is projected to reach $20.9 billion by 2025, growing at a CAGR of 8.5% from 2020. Within this broader market, autoclave sterilization validation services specifically for new load types are experiencing even faster growth rates, estimated at 11.2% annually, reflecting the increasing complexity of medical devices and pharmaceutical products requiring sterilization.
The pharmaceutical and biotechnology sectors represent the largest demand segment, accounting for approximately 38% of the market for validated sterilization processes. This is followed closely by hospitals and healthcare facilities (32%), medical device manufacturers (21%), and food processing industries (9%). Regional analysis shows North America leading with 42% market share, followed by Europe (28%), Asia-Pacific (22%), and rest of the world (8%).
Customer needs analysis reveals several key market drivers: the introduction of novel materials in medical devices that require specialized validation protocols; increasing complexity of surgical instruments with narrow lumens and intricate designs; growing demand for rapid sterilization cycles without compromising efficacy; and the need for energy-efficient sterilization processes that maintain validation integrity.
The COVID-19 pandemic has further accelerated market demand, with a 27% increase in requests for validation of new load types observed between 2019 and 2021. This surge reflects both the immediate need for sterilization of novel PPE configurations and the longer-term trend toward more diverse medical equipment requiring specialized sterilization protocols.
Cost sensitivity analysis indicates that while customers prioritize validation reliability, there is growing pressure to reduce the time and expense associated with validating new load types. This has created market opportunities for innovative validation technologies and services that can streamline the process while maintaining compliance with regulatory standards.
Healthcare facilities worldwide are facing stricter compliance standards regarding sterilization validation, with regulatory bodies such as the FDA, EMA, and ISO implementing more comprehensive guidelines. This regulatory pressure has created substantial demand for validated sterilization processes that can accommodate diverse and evolving load types, from complex surgical instruments to new composite materials used in medical devices.
Market research indicates that the global sterilization equipment market is projected to reach $20.9 billion by 2025, growing at a CAGR of 8.5% from 2020. Within this broader market, autoclave sterilization validation services specifically for new load types are experiencing even faster growth rates, estimated at 11.2% annually, reflecting the increasing complexity of medical devices and pharmaceutical products requiring sterilization.
The pharmaceutical and biotechnology sectors represent the largest demand segment, accounting for approximately 38% of the market for validated sterilization processes. This is followed closely by hospitals and healthcare facilities (32%), medical device manufacturers (21%), and food processing industries (9%). Regional analysis shows North America leading with 42% market share, followed by Europe (28%), Asia-Pacific (22%), and rest of the world (8%).
Customer needs analysis reveals several key market drivers: the introduction of novel materials in medical devices that require specialized validation protocols; increasing complexity of surgical instruments with narrow lumens and intricate designs; growing demand for rapid sterilization cycles without compromising efficacy; and the need for energy-efficient sterilization processes that maintain validation integrity.
The COVID-19 pandemic has further accelerated market demand, with a 27% increase in requests for validation of new load types observed between 2019 and 2021. This surge reflects both the immediate need for sterilization of novel PPE configurations and the longer-term trend toward more diverse medical equipment requiring specialized sterilization protocols.
Cost sensitivity analysis indicates that while customers prioritize validation reliability, there is growing pressure to reduce the time and expense associated with validating new load types. This has created market opportunities for innovative validation technologies and services that can streamline the process while maintaining compliance with regulatory standards.
Current Challenges in Load Type Validation
The validation of new load types for autoclave sterilization cycles presents significant challenges in today's healthcare and manufacturing environments. Current validation protocols often struggle to keep pace with the increasing diversity of medical devices, pharmaceutical products, and laboratory equipment requiring sterilization. Traditional validation approaches were developed for standardized loads, creating a fundamental mismatch when applied to novel configurations or materials.
One primary challenge is the thermal mapping of complex load geometries. Modern medical devices frequently incorporate intricate designs with multiple materials and small crevices, creating potential "cold spots" where sterilization may be compromised. These areas are difficult to identify and monitor during validation, requiring advanced thermal mapping techniques that many facilities lack the capability to implement effectively.
Material compatibility issues further complicate validation efforts. New polymers, composites, and specialized coatings used in medical devices may respond unpredictably to autoclave conditions. Some materials experience accelerated degradation, while others may release compounds that interfere with sterilization efficacy or create safety concerns. Current validation methodologies often fail to adequately assess these material-specific responses.
The increasing prevalence of mixed-load sterilization cycles presents another significant hurdle. Healthcare facilities seeking operational efficiency often combine different device types in a single cycle, creating complex validation scenarios where air removal, steam penetration, and heat distribution must be verified across heterogeneous loads. Standard validation protocols typically assume homogeneous load characteristics, leading to potential gaps in sterilization assurance.
Regulatory expectations add another layer of complexity. Different global regulatory bodies maintain varying requirements for load validation, creating compliance challenges for organizations operating internationally. The FDA, EU MDR, and other regulatory frameworks have increasingly stringent expectations for validation documentation, particularly for novel load configurations, without providing standardized methodologies.
Resource constraints further exacerbate these challenges. Comprehensive load validation requires specialized equipment, technical expertise, and significant time investment. Many organizations lack dedicated validation specialists or sufficient budget allocation for thorough validation studies, leading to compromised protocols or excessive reliance on third-party testing services.
Data interpretation presents a final significant challenge. The statistical analysis of validation results for new load types often involves complex datasets with multiple variables. Determining appropriate acceptance criteria and confidence levels for novel configurations requires sophisticated statistical approaches that many validation teams are not equipped to implement, potentially leading to either overly conservative or insufficiently rigorous validation conclusions.
One primary challenge is the thermal mapping of complex load geometries. Modern medical devices frequently incorporate intricate designs with multiple materials and small crevices, creating potential "cold spots" where sterilization may be compromised. These areas are difficult to identify and monitor during validation, requiring advanced thermal mapping techniques that many facilities lack the capability to implement effectively.
Material compatibility issues further complicate validation efforts. New polymers, composites, and specialized coatings used in medical devices may respond unpredictably to autoclave conditions. Some materials experience accelerated degradation, while others may release compounds that interfere with sterilization efficacy or create safety concerns. Current validation methodologies often fail to adequately assess these material-specific responses.
The increasing prevalence of mixed-load sterilization cycles presents another significant hurdle. Healthcare facilities seeking operational efficiency often combine different device types in a single cycle, creating complex validation scenarios where air removal, steam penetration, and heat distribution must be verified across heterogeneous loads. Standard validation protocols typically assume homogeneous load characteristics, leading to potential gaps in sterilization assurance.
Regulatory expectations add another layer of complexity. Different global regulatory bodies maintain varying requirements for load validation, creating compliance challenges for organizations operating internationally. The FDA, EU MDR, and other regulatory frameworks have increasingly stringent expectations for validation documentation, particularly for novel load configurations, without providing standardized methodologies.
Resource constraints further exacerbate these challenges. Comprehensive load validation requires specialized equipment, technical expertise, and significant time investment. Many organizations lack dedicated validation specialists or sufficient budget allocation for thorough validation studies, leading to compromised protocols or excessive reliance on third-party testing services.
Data interpretation presents a final significant challenge. The statistical analysis of validation results for new load types often involves complex datasets with multiple variables. Determining appropriate acceptance criteria and confidence levels for novel configurations requires sophisticated statistical approaches that many validation teams are not equipped to implement, potentially leading to either overly conservative or insufficiently rigorous validation conclusions.
Current Validation Methodologies for New Load Types
01 Validation methods for autoclave sterilization cycles
Various methods are employed to validate autoclave sterilization cycles, ensuring that the sterilization process meets required standards. These methods include biological indicators, chemical indicators, and physical measurements to verify that the necessary conditions for sterilization (temperature, pressure, time) are achieved throughout the load. Validation protocols typically involve placement of indicators at multiple locations within the autoclave chamber, particularly in hard-to-reach areas or cold spots, to confirm uniform sterilization.- Validation methods for autoclave sterilization cycles: Various methods are employed to validate autoclave sterilization cycles, including biological indicators, chemical indicators, and physical measurements. These validation methods ensure that the sterilization process meets predetermined specifications and consistently delivers the required sterility assurance level. The validation process typically involves installation qualification, operational qualification, and performance qualification to verify that the autoclave operates correctly and achieves effective sterilization.
- Monitoring and control systems for sterilization cycles: Advanced monitoring and control systems are essential for autoclave sterilization cycle validation. These systems include sensors for measuring critical parameters such as temperature, pressure, and time throughout the sterilization process. Real-time data collection and analysis allow for immediate detection of deviations from the validated cycle parameters. Automated control systems can adjust process variables to maintain optimal sterilization conditions and generate comprehensive documentation for regulatory compliance.
- Sterilization cycle parameters and optimization: Effective autoclave sterilization requires careful optimization of cycle parameters including temperature, pressure, exposure time, and steam quality. The validation process establishes the acceptable ranges for these parameters to ensure consistent sterilization efficacy. Different materials and load configurations may require specific cycle parameters to achieve proper sterilization. Optimization studies help determine the most efficient combination of parameters that consistently achieve the required sterility assurance level while minimizing cycle time and utility consumption.
- Load configuration and placement validation: The arrangement and configuration of items within an autoclave significantly impact sterilization effectiveness. Validation protocols must include testing of various load configurations to ensure steam penetration and heat distribution throughout the load. Critical factors include spacing between items, orientation of packaging, and total mass of the load. Temperature mapping studies using multiple sensors placed throughout the load help identify cold spots where sterilization might be compromised. Validation must demonstrate that all items in the load consistently reach and maintain the required sterilization conditions.
- Documentation and regulatory compliance for sterilization validation: Comprehensive documentation is crucial for autoclave sterilization cycle validation to meet regulatory requirements. This includes detailed protocols, validation reports, standard operating procedures, and routine monitoring records. The documentation must demonstrate that the sterilization process has been thoroughly validated and consistently produces sterile products. Regular revalidation is necessary when changes occur to equipment, processes, or load configurations. Proper documentation provides evidence of compliance with standards such as ISO 17665 and regulatory requirements from agencies like FDA and EMA.
02 Monitoring systems for autoclave cycle parameters
Advanced monitoring systems are used to track critical parameters during autoclave sterilization cycles. These systems continuously record temperature, pressure, time, and steam quality throughout the sterilization process. Real-time monitoring allows for immediate detection of cycle deviations, while data logging capabilities provide documentation for regulatory compliance. Some systems incorporate wireless sensors, digital interfaces, and automated reporting features to enhance accuracy and efficiency in validation procedures.Expand Specific Solutions03 Qualification procedures for autoclave equipment
Comprehensive qualification procedures are essential for autoclave equipment validation, typically including installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). These procedures verify that the autoclave is properly installed, operates according to specifications, and consistently delivers effective sterilization across various load configurations. Qualification protocols often include calibration of instruments, verification of control systems, and challenge tests to ensure the equipment can handle worst-case scenarios.Expand Specific Solutions04 Load configuration and packaging validation
The arrangement of items within an autoclave and their packaging significantly impact sterilization effectiveness. Validation studies must account for various load configurations, packaging materials, and container systems to ensure steam penetration and heat distribution. Testing includes validation of maximum load densities, optimal spacing arrangements, and compatibility of packaging materials with steam sterilization. Proper load configuration validation helps prevent sterilization failures caused by air pockets, inadequate steam contact, or thermal barriers.Expand Specific Solutions05 Regulatory compliance and documentation requirements
Autoclave sterilization cycle validation must adhere to specific regulatory standards and documentation requirements. This includes maintaining detailed records of validation protocols, test results, and cycle parameters for regulatory inspections. Documentation typically covers equipment calibration, maintenance records, operator training, and routine performance testing. Compliance with standards such as ISO, FDA guidelines, and pharmacopeia requirements ensures that validation procedures are recognized as adequate by regulatory authorities across different jurisdictions.Expand Specific Solutions
Key Industry Players and Equipment Manufacturers
The autoclave sterilization validation market is currently in a growth phase, with increasing demand driven by stringent regulatory requirements in healthcare and pharmaceutical industries. The global market size is estimated to exceed $2 billion, expanding at approximately 6-8% CAGR. From a technological maturity perspective, the landscape shows varying degrees of advancement. Industry leaders like STERIS (American Sterilizer Co.) and Shinva Medical Instrument demonstrate mature validation protocols, while companies such as Stryker Corp. and 3D Systems are advancing specialized load validation technologies. Emerging players like Turbett Surgical and Oneighty C Technologies are introducing innovative approaches to validation processes. Regional leaders including W&H Sterilization, Melag Medizintechnik, and Jiangyin Binjiang Medical Equipment are strengthening their positions through enhanced validation capabilities tailored to specific market requirements.
Shinva Medical Instrument Co., Ltd.
Technical Solution: Shinva Medical has pioneered an integrated validation approach for new autoclave load types that combines traditional biological validation with advanced computational modeling. Their system employs a three-phase validation protocol: theoretical modeling, laboratory testing, and production verification. For new load types, Shinva's technology creates digital twins of the load configuration and simulates heat penetration and steam distribution using computational fluid dynamics. This is followed by physical validation using their proprietary multi-point temperature monitoring system with up to 48 wireless sensors that can be placed throughout complex loads. Their validation technology incorporates real-time data analytics that automatically identifies cold spots and calculates F0 values throughout the sterilization cycle. Shinva's approach is particularly effective for validating complex medical device assemblies and mixed loads, with their software providing automated documentation for regulatory compliance.
Strengths: Comprehensive integration of digital simulation with physical testing reduces validation time by up to 40%; wireless sensor technology allows for validation of geometrically complex loads without disrupting steam flow. Weaknesses: Their computational models require extensive calibration for new materials; the system has higher initial implementation costs compared to traditional validation methods.
Stryker Corp.
Technical Solution: Stryker has developed an advanced validation system specifically designed for complex surgical instrument sets and implantable devices. Their approach utilizes a combination of physical and chemical indicators alongside biological validation methods. Stryker's proprietary "SureCycle" technology employs load-specific algorithms that adjust sterilization parameters based on the thermal characteristics of different materials within the load. The system features high-precision temperature and pressure sensors that create detailed 3D thermal maps of the load during the sterilization cycle. For new load types, Stryker employs a progressive challenge testing methodology where increasingly difficult-to-sterilize configurations are validated to establish safety margins. Their validation protocol includes specialized biological indicators placed at predetermined cold spots, with fractional cycle testing to determine the minimum time required for complete sterilization. The system also incorporates material compatibility testing to ensure that sterilization cycles do not compromise the functionality or longevity of sensitive instruments.
Strengths: Specialized expertise in validating complex surgical instrument sets; advanced material compatibility database that prevents damage to sensitive components; rapid validation protocols that reduce time-to-market for new medical devices. Weaknesses: System is primarily optimized for medical devices rather than general purpose loads; higher cost structure compared to more generic validation approaches.
Critical Parameters and Scientific Principles
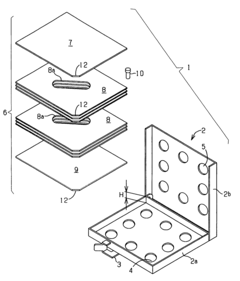
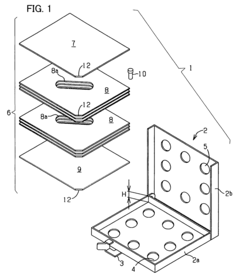
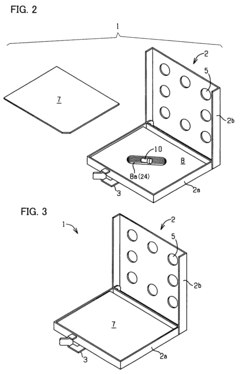
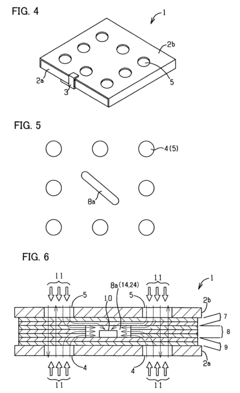
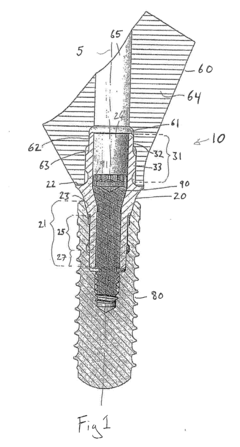
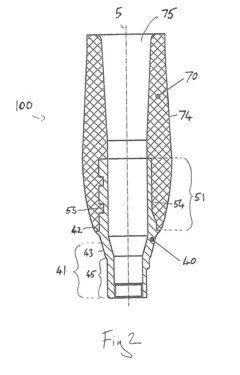
Process challenge device for a high-pressure steam sterilizer and sheet for a challenge device
PatentInactiveUS8333933B2
Innovation
- A process challenge device comprising steam permeable bodies and a holder, where the steam permeable bodies form a cavity with openings that communicate with each other, allowing steam to reach an indicator, which changes appearance upon exposure to a predetermined temperature, and can be reused without damaging the components, with the indicator being easily removable and user-selectable.
Sterilisation method of dental components comprising polymethyl methacrylate
PatentInactiveEP2698171A1
Innovation
- Steam sterilization of thermoset PMMA dental components at temperatures between 121°C and 134°C for up to 30 minutes, which surprisingly does not degrade the material and maintains its mechanical and biocompatibility properties.
Regulatory Compliance and Standards
Autoclave sterilization processes are governed by a comprehensive framework of regulatory standards that ensure safety, efficacy, and consistency across healthcare and industrial applications. The FDA's Quality System Regulation (21 CFR Part 820) establishes fundamental requirements for medical device manufacturers, mandating validation of sterilization processes for each new load configuration. This regulation is complemented by ISO 17665-1:2006, which provides specific guidelines for the development, validation, and routine control of moist heat sterilization processes.
For healthcare facilities, compliance with standards set by the Association for the Advancement of Medical Instrumentation (AAMI) is essential, particularly AAMI ST79 which offers detailed guidance on steam sterilization validation. The European Medical Device Regulation (MDR 2017/745) imposes additional requirements for products marketed in European territories, emphasizing thorough documentation of validation procedures.
When validating new load types, organizations must adhere to a three-phase validation approach as outlined in ISO 17665-2: installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). This structured methodology ensures that sterilization equipment functions correctly, operates within specified parameters, and consistently achieves sterility across various load configurations.
Documentation requirements are particularly stringent, with regulatory bodies requiring comprehensive records of validation protocols, acceptance criteria, test results, and deviation management. These records must demonstrate that the sterilization process consistently achieves a sterility assurance level (SAL) of 10^-6 for critical medical devices, meaning a probability of not more than one viable microorganism in one million sterilized items.
Regulatory compliance also extends to personnel qualifications and training. Standards require that validation activities be performed by individuals with appropriate education, training, and experience in sterilization technologies. This requirement ensures that validation studies are designed and executed with proper scientific rigor.
Recent regulatory trends indicate increasing scrutiny of validation practices for complex load configurations. Regulatory bodies are placing greater emphasis on worst-case scenario testing and the use of biological indicators positioned at locations determined through temperature mapping studies. Additionally, there is growing recognition of the need for specialized approaches when validating mixed loads containing materials with different thermal properties and geometries.
Non-compliance with these regulatory standards can result in significant consequences, including product recalls, regulatory actions, and potential liability issues. Therefore, organizations must maintain robust quality management systems that ensure ongoing compliance with evolving regulatory requirements for autoclave sterilization validation.
For healthcare facilities, compliance with standards set by the Association for the Advancement of Medical Instrumentation (AAMI) is essential, particularly AAMI ST79 which offers detailed guidance on steam sterilization validation. The European Medical Device Regulation (MDR 2017/745) imposes additional requirements for products marketed in European territories, emphasizing thorough documentation of validation procedures.
When validating new load types, organizations must adhere to a three-phase validation approach as outlined in ISO 17665-2: installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). This structured methodology ensures that sterilization equipment functions correctly, operates within specified parameters, and consistently achieves sterility across various load configurations.
Documentation requirements are particularly stringent, with regulatory bodies requiring comprehensive records of validation protocols, acceptance criteria, test results, and deviation management. These records must demonstrate that the sterilization process consistently achieves a sterility assurance level (SAL) of 10^-6 for critical medical devices, meaning a probability of not more than one viable microorganism in one million sterilized items.
Regulatory compliance also extends to personnel qualifications and training. Standards require that validation activities be performed by individuals with appropriate education, training, and experience in sterilization technologies. This requirement ensures that validation studies are designed and executed with proper scientific rigor.
Recent regulatory trends indicate increasing scrutiny of validation practices for complex load configurations. Regulatory bodies are placing greater emphasis on worst-case scenario testing and the use of biological indicators positioned at locations determined through temperature mapping studies. Additionally, there is growing recognition of the need for specialized approaches when validating mixed loads containing materials with different thermal properties and geometries.
Non-compliance with these regulatory standards can result in significant consequences, including product recalls, regulatory actions, and potential liability issues. Therefore, organizations must maintain robust quality management systems that ensure ongoing compliance with evolving regulatory requirements for autoclave sterilization validation.
Risk Assessment and Quality Assurance
Risk assessment is a critical component in the validation process for new load types in autoclave sterilization cycles. Comprehensive risk analysis should be conducted using established methodologies such as Failure Mode and Effects Analysis (FMEA) or Hazard Analysis and Critical Control Points (HACCP). These approaches enable systematic identification of potential failure points throughout the sterilization process, from load configuration to cycle completion. For new load types, particular attention must be paid to thermal distribution risks, as novel configurations may create unexpected cold spots where sterilization efficacy could be compromised.
Quality assurance frameworks must be established with clearly defined acceptance criteria that align with regulatory standards such as ISO 17665 and AAMI ST79. These criteria should include minimum temperature thresholds, exposure time requirements, and biological indicator performance specifications tailored to the specific characteristics of the new load type. Documentation systems must capture all validation parameters, test results, and deviations to ensure traceability and compliance with regulatory expectations.
Statistical process control methods should be implemented to monitor critical process parameters during validation studies. This includes establishing appropriate sampling plans based on load heterogeneity and complexity. For highly variable load types, increased sampling frequency and distribution may be necessary to ensure statistical confidence in the validation results. Upper and lower control limits should be established for key parameters such as temperature, pressure, and humidity to facilitate ongoing monitoring.
Risk mitigation strategies should be developed for each identified risk, with particular emphasis on those classified as high-priority. These strategies might include cycle modifications, load configuration adjustments, or enhanced monitoring protocols. A formal change control process must be established to manage any modifications to validated cycles, ensuring that changes are properly assessed, documented, and verified before implementation.
Continuous quality improvement processes should be integrated into the validation framework. This includes regular review of validation data, analysis of trends, and implementation of corrective and preventive actions when necessary. For new load types, post-validation monitoring should be more intensive initially, with frequency adjusted based on accumulated performance data and risk assessment outcomes.
Personnel training represents another critical quality assurance element. Staff involved in autoclave operation and validation must receive specialized training on the unique characteristics and requirements of new load types. Competency assessments should verify understanding of critical parameters, monitoring requirements, and response protocols for cycle deviations specific to the new load configuration.
Quality assurance frameworks must be established with clearly defined acceptance criteria that align with regulatory standards such as ISO 17665 and AAMI ST79. These criteria should include minimum temperature thresholds, exposure time requirements, and biological indicator performance specifications tailored to the specific characteristics of the new load type. Documentation systems must capture all validation parameters, test results, and deviations to ensure traceability and compliance with regulatory expectations.
Statistical process control methods should be implemented to monitor critical process parameters during validation studies. This includes establishing appropriate sampling plans based on load heterogeneity and complexity. For highly variable load types, increased sampling frequency and distribution may be necessary to ensure statistical confidence in the validation results. Upper and lower control limits should be established for key parameters such as temperature, pressure, and humidity to facilitate ongoing monitoring.
Risk mitigation strategies should be developed for each identified risk, with particular emphasis on those classified as high-priority. These strategies might include cycle modifications, load configuration adjustments, or enhanced monitoring protocols. A formal change control process must be established to manage any modifications to validated cycles, ensuring that changes are properly assessed, documented, and verified before implementation.
Continuous quality improvement processes should be integrated into the validation framework. This includes regular review of validation data, analysis of trends, and implementation of corrective and preventive actions when necessary. For new load types, post-validation monitoring should be more intensive initially, with frequency adjusted based on accumulated performance data and risk assessment outcomes.
Personnel training represents another critical quality assurance element. Staff involved in autoclave operation and validation must receive specialized training on the unique characteristics and requirements of new load types. Competency assessments should verify understanding of critical parameters, monitoring requirements, and response protocols for cycle deviations specific to the new load configuration.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!