Lithium Hydroxide Performance: High-Pressure Reaction Studies
AUG 28, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Hydroxide Technology Background and Objectives
Lithium hydroxide (LiOH) has emerged as a critical material in modern energy storage systems, particularly in the production of high-performance lithium-ion batteries. The evolution of this technology can be traced back to the early 1990s when lithium-ion batteries first entered commercial markets. Since then, the demand for higher-grade lithium hydroxide has grown exponentially, driven primarily by the electric vehicle revolution and renewable energy storage requirements.
The technological trajectory of lithium hydroxide production has undergone significant transformation over the past three decades. Traditional methods relied on the conversion of lithium carbonate through a lime-soda process, which was both energy-intensive and yielded products with varying purity levels. The industry has progressively moved toward more efficient extraction and conversion techniques, with high-pressure reaction systems representing one of the most promising recent developments.
High-pressure reaction studies for lithium hydroxide production aim to address several critical challenges in the current manufacturing landscape. These studies investigate reaction kinetics, yield optimization, and purity enhancement under elevated pressure conditions. The fundamental objective is to develop processes that can produce battery-grade lithium hydroxide (≥99.5% purity) with minimal energy consumption and environmental impact.
Current research indicates that high-pressure environments can significantly alter reaction pathways, potentially enabling more efficient conversion of lithium-containing precursors to lithium hydroxide. Preliminary studies suggest that operating at pressures between 15-30 MPa can reduce reaction times by up to 40% while simultaneously improving product consistency and reducing impurity levels, particularly sodium and calcium contaminants that adversely affect battery performance.
The technological objectives of high-pressure lithium hydroxide research extend beyond mere process optimization. They encompass the development of scalable, continuous production systems that can meet the projected tenfold increase in global demand expected by 2030. Additionally, these studies aim to establish reaction parameters that minimize water consumption—a significant concern given that traditional methods require approximately 15 tons of water per ton of lithium hydroxide produced.
Another critical goal is to understand the fundamental thermodynamics and reaction mechanisms under high-pressure conditions, which remain inadequately characterized despite their industrial relevance. This knowledge gap has hindered the development of predictive models necessary for process scale-up and optimization. By systematically investigating pressure-dependent phenomena, researchers aim to establish comprehensive reaction models that can guide industrial implementation.
The ultimate technological objective is to establish high-pressure reaction technology as a commercially viable alternative to conventional lithium hydroxide production methods, offering superior economics, product quality, and environmental performance to meet the growing demands of the clean energy transition.
The technological trajectory of lithium hydroxide production has undergone significant transformation over the past three decades. Traditional methods relied on the conversion of lithium carbonate through a lime-soda process, which was both energy-intensive and yielded products with varying purity levels. The industry has progressively moved toward more efficient extraction and conversion techniques, with high-pressure reaction systems representing one of the most promising recent developments.
High-pressure reaction studies for lithium hydroxide production aim to address several critical challenges in the current manufacturing landscape. These studies investigate reaction kinetics, yield optimization, and purity enhancement under elevated pressure conditions. The fundamental objective is to develop processes that can produce battery-grade lithium hydroxide (≥99.5% purity) with minimal energy consumption and environmental impact.
Current research indicates that high-pressure environments can significantly alter reaction pathways, potentially enabling more efficient conversion of lithium-containing precursors to lithium hydroxide. Preliminary studies suggest that operating at pressures between 15-30 MPa can reduce reaction times by up to 40% while simultaneously improving product consistency and reducing impurity levels, particularly sodium and calcium contaminants that adversely affect battery performance.
The technological objectives of high-pressure lithium hydroxide research extend beyond mere process optimization. They encompass the development of scalable, continuous production systems that can meet the projected tenfold increase in global demand expected by 2030. Additionally, these studies aim to establish reaction parameters that minimize water consumption—a significant concern given that traditional methods require approximately 15 tons of water per ton of lithium hydroxide produced.
Another critical goal is to understand the fundamental thermodynamics and reaction mechanisms under high-pressure conditions, which remain inadequately characterized despite their industrial relevance. This knowledge gap has hindered the development of predictive models necessary for process scale-up and optimization. By systematically investigating pressure-dependent phenomena, researchers aim to establish comprehensive reaction models that can guide industrial implementation.
The ultimate technological objective is to establish high-pressure reaction technology as a commercially viable alternative to conventional lithium hydroxide production methods, offering superior economics, product quality, and environmental performance to meet the growing demands of the clean energy transition.
Market Analysis for High-Pressure Lithium Hydroxide Applications
The global market for high-pressure lithium hydroxide applications has witnessed significant growth in recent years, primarily driven by the expanding electric vehicle (EV) industry and renewable energy storage systems. The demand for high-purity lithium hydroxide produced through high-pressure reaction methods has increased substantially, with the global market value reaching $3.2 billion in 2022 and projected to grow at a CAGR of 14.7% through 2030.
The EV battery sector represents the largest application segment, accounting for approximately 65% of the high-pressure lithium hydroxide market. This dominance stems from the superior performance characteristics of lithium hydroxide produced under high-pressure conditions, including enhanced purity levels, consistent particle size distribution, and improved electrochemical properties that directly translate to better battery performance metrics.
Regional analysis indicates that Asia-Pacific currently leads the market with a 48% share, driven by China's aggressive EV manufacturing ecosystem and battery production capabilities. North America follows with 27% market share, while Europe accounts for 21% of the global consumption. The remaining 4% is distributed across other regions, with emerging markets showing accelerated adoption rates.
Market segmentation by application reveals diversification beyond EV batteries. Industrial lubricants represent 12% of the market, ceramics and glass manufacturing account for 8%, and specialized chemical processes comprise 7%. The remaining 8% encompasses various niche applications including aerospace materials, pharmaceutical processing, and advanced polymer production.
Price analysis shows that high-pressure synthesized lithium hydroxide commands a premium of 15-22% over conventionally produced alternatives, reflecting its superior quality attributes and performance benefits in high-demand applications. This price differential has remained relatively stable despite overall lithium compound price volatility, indicating strong value recognition among end-users.
Supply chain dynamics present both opportunities and challenges. Currently, five major producers control approximately 73% of the high-pressure lithium hydroxide production capacity globally. This concentration has created potential supply vulnerabilities, particularly as demand continues to outpace capacity expansion. Industry forecasts suggest that current production capacity will need to triple by 2028 to meet projected demand.
Customer analysis reveals shifting preferences toward higher performance specifications, with 78% of industrial buyers prioritizing consistent quality over price considerations. This trend particularly benefits high-pressure reaction methodologies that deliver more predictable product characteristics compared to traditional production techniques.
Market barriers include significant capital requirements for high-pressure reaction equipment, technical expertise limitations, and regulatory compliance complexities related to high-pressure operations. These factors have contributed to the relatively concentrated market structure and present challenges for new entrants.
The EV battery sector represents the largest application segment, accounting for approximately 65% of the high-pressure lithium hydroxide market. This dominance stems from the superior performance characteristics of lithium hydroxide produced under high-pressure conditions, including enhanced purity levels, consistent particle size distribution, and improved electrochemical properties that directly translate to better battery performance metrics.
Regional analysis indicates that Asia-Pacific currently leads the market with a 48% share, driven by China's aggressive EV manufacturing ecosystem and battery production capabilities. North America follows with 27% market share, while Europe accounts for 21% of the global consumption. The remaining 4% is distributed across other regions, with emerging markets showing accelerated adoption rates.
Market segmentation by application reveals diversification beyond EV batteries. Industrial lubricants represent 12% of the market, ceramics and glass manufacturing account for 8%, and specialized chemical processes comprise 7%. The remaining 8% encompasses various niche applications including aerospace materials, pharmaceutical processing, and advanced polymer production.
Price analysis shows that high-pressure synthesized lithium hydroxide commands a premium of 15-22% over conventionally produced alternatives, reflecting its superior quality attributes and performance benefits in high-demand applications. This price differential has remained relatively stable despite overall lithium compound price volatility, indicating strong value recognition among end-users.
Supply chain dynamics present both opportunities and challenges. Currently, five major producers control approximately 73% of the high-pressure lithium hydroxide production capacity globally. This concentration has created potential supply vulnerabilities, particularly as demand continues to outpace capacity expansion. Industry forecasts suggest that current production capacity will need to triple by 2028 to meet projected demand.
Customer analysis reveals shifting preferences toward higher performance specifications, with 78% of industrial buyers prioritizing consistent quality over price considerations. This trend particularly benefits high-pressure reaction methodologies that deliver more predictable product characteristics compared to traditional production techniques.
Market barriers include significant capital requirements for high-pressure reaction equipment, technical expertise limitations, and regulatory compliance complexities related to high-pressure operations. These factors have contributed to the relatively concentrated market structure and present challenges for new entrants.
Current Technical Challenges in High-Pressure Reaction Systems
High-pressure reaction systems for lithium hydroxide production face several significant technical challenges that impede optimal performance and efficiency. The primary challenge lies in maintaining precise pressure control throughout the reaction process. Current pressure regulation systems often struggle with rapid fluctuations during exothermic stages of lithium hydroxide formation, leading to inconsistent product quality and reduced yield rates. These fluctuations can cause uneven particle size distribution, which directly impacts the electrochemical performance of the final product in battery applications.
Material degradation presents another substantial obstacle in high-pressure environments. Reactor vessels and components experience accelerated corrosion when exposed to the highly alkaline conditions necessary for lithium hydroxide synthesis. This corrosion not only compromises system integrity but also introduces metal ion contamination into the final product, potentially degrading battery performance when the lithium hydroxide is incorporated into cathode materials.
Energy efficiency remains suboptimal in existing high-pressure systems. The significant energy input required to maintain elevated pressures, often exceeding 20 MPa, contributes to high operational costs and carbon footprints. Current heat recovery systems capture only 60-70% of thermal energy, leaving substantial room for improvement in overall process efficiency.
Scaling challenges persist when transitioning from laboratory to industrial production scales. Phenomena that are manageable at smaller scales, such as heat distribution and mixing efficiency, become increasingly problematic in larger reactors. This scaling difficulty has created a bottleneck in the commercial implementation of promising laboratory-developed high-pressure lithium hydroxide synthesis methods.
Safety concerns represent a critical challenge, with high-pressure systems requiring sophisticated monitoring and emergency response mechanisms. Current pressure relief systems occasionally experience delayed response times, creating potential hazards during unexpected pressure surges. The integration of real-time monitoring with automated safety protocols remains inadequate in many existing systems.
Process monitoring technology presents limitations in harsh reaction environments. Conventional sensors often demonstrate reduced accuracy and shortened lifespans when exposed to the combination of high pressure, elevated temperatures, and caustic chemical environments present in lithium hydroxide production. This monitoring deficiency hampers process optimization and quality control efforts.
Batch-to-batch consistency remains difficult to achieve, with variations in reaction kinetics under high-pressure conditions leading to product heterogeneity. These variations become particularly problematic when lithium hydroxide is destined for high-performance battery applications where impurity profiles and crystal structure must meet increasingly stringent specifications.
Material degradation presents another substantial obstacle in high-pressure environments. Reactor vessels and components experience accelerated corrosion when exposed to the highly alkaline conditions necessary for lithium hydroxide synthesis. This corrosion not only compromises system integrity but also introduces metal ion contamination into the final product, potentially degrading battery performance when the lithium hydroxide is incorporated into cathode materials.
Energy efficiency remains suboptimal in existing high-pressure systems. The significant energy input required to maintain elevated pressures, often exceeding 20 MPa, contributes to high operational costs and carbon footprints. Current heat recovery systems capture only 60-70% of thermal energy, leaving substantial room for improvement in overall process efficiency.
Scaling challenges persist when transitioning from laboratory to industrial production scales. Phenomena that are manageable at smaller scales, such as heat distribution and mixing efficiency, become increasingly problematic in larger reactors. This scaling difficulty has created a bottleneck in the commercial implementation of promising laboratory-developed high-pressure lithium hydroxide synthesis methods.
Safety concerns represent a critical challenge, with high-pressure systems requiring sophisticated monitoring and emergency response mechanisms. Current pressure relief systems occasionally experience delayed response times, creating potential hazards during unexpected pressure surges. The integration of real-time monitoring with automated safety protocols remains inadequate in many existing systems.
Process monitoring technology presents limitations in harsh reaction environments. Conventional sensors often demonstrate reduced accuracy and shortened lifespans when exposed to the combination of high pressure, elevated temperatures, and caustic chemical environments present in lithium hydroxide production. This monitoring deficiency hampers process optimization and quality control efforts.
Batch-to-batch consistency remains difficult to achieve, with variations in reaction kinetics under high-pressure conditions leading to product heterogeneity. These variations become particularly problematic when lithium hydroxide is destined for high-performance battery applications where impurity profiles and crystal structure must meet increasingly stringent specifications.
Current High-Pressure Reaction Methodologies
01 Lithium hydroxide production methods
Various methods for producing lithium hydroxide with improved performance characteristics. These methods include extraction from lithium-containing minerals, conversion from lithium carbonate, and novel processing techniques that yield high-purity lithium hydroxide suitable for battery applications. The production processes focus on efficiency, yield optimization, and reducing impurities to enhance the overall performance of the final product.- Lithium hydroxide production methods: Various methods for producing lithium hydroxide with improved performance characteristics. These methods include extraction from lithium-containing minerals, conversion from lithium carbonate, and direct production processes that yield high-purity lithium hydroxide suitable for battery applications. The production techniques focus on efficiency, purity levels, and scalability to meet industrial demands.
- Battery applications and performance enhancement: Lithium hydroxide is a critical component in high-performance lithium-ion batteries, particularly for electric vehicles and energy storage systems. Its use leads to improved battery capacity, longer cycle life, and enhanced thermal stability. The high purity of lithium hydroxide contributes to better electrochemical performance and safety characteristics in advanced battery systems.
- Purification and quality control of lithium hydroxide: Techniques for purifying lithium hydroxide to meet stringent quality requirements for high-tech applications. These include removal of impurities such as sodium, calcium, and magnesium ions, crystallization processes, and advanced filtration methods. Quality control measures ensure consistent performance characteristics such as particle size distribution, moisture content, and chemical purity.
- Environmental and sustainable processing: Environmentally friendly approaches to lithium hydroxide production and processing that reduce carbon footprint and minimize waste. These include closed-loop systems, water recycling, energy-efficient processes, and reduced use of harmful chemicals. Sustainable methods focus on responsible resource management while maintaining or improving the performance characteristics of the final lithium hydroxide product.
- Material properties and characterization: Analysis and characterization of lithium hydroxide's physical and chemical properties that affect its performance in various applications. These include studies on crystal structure, thermal stability, reactivity, solubility, and hygroscopic nature. Advanced analytical techniques are employed to understand how these properties influence the material's behavior in batteries, ceramics, lubricants, and other industrial applications.
02 Battery applications and performance enhancement
Lithium hydroxide is a critical component in high-performance lithium-ion batteries, particularly for electric vehicles and energy storage systems. Its use improves battery capacity, charging efficiency, and cycle life. The performance of lithium hydroxide in batteries is influenced by its purity, particle size, and morphology, with higher-grade materials enabling better electrochemical properties and thermal stability in battery systems.Expand Specific Solutions03 Purification and quality control techniques
Advanced purification methods for lithium hydroxide that enhance its performance in various applications. These techniques include crystallization, filtration, ion exchange, and other separation processes designed to remove impurities such as sodium, calcium, and magnesium ions. Quality control measures ensure consistent particle size distribution, moisture content, and chemical composition, which are critical for optimal performance in end-use applications.Expand Specific Solutions04 Environmental and sustainability aspects
Environmentally friendly approaches to lithium hydroxide production and utilization that improve sustainability while maintaining performance. These include green extraction methods, recycling of lithium from spent batteries, reduced water consumption in processing, and lower carbon footprint manufacturing techniques. Sustainable practices in lithium hydroxide production contribute to improved environmental performance throughout the lithium value chain.Expand Specific Solutions05 Industrial applications beyond batteries
Lithium hydroxide performance in non-battery applications such as lubricating greases, air purification systems, ceramic production, and glass manufacturing. In these applications, lithium hydroxide provides benefits including improved thermal stability, enhanced mechanical properties, and superior chemical resistance. The performance characteristics required for these diverse applications differ from battery-grade requirements, focusing on specific properties relevant to each industrial use case.Expand Specific Solutions
Key Industry Players in Lithium Hydroxide Production
The lithium hydroxide performance market is currently in a growth phase, with increasing demand driven by electric vehicle battery applications. The market size is expanding rapidly, projected to reach significant scale by 2030 as major players invest in production capacity. Technologically, high-pressure reaction studies represent an advancing frontier with varying maturity levels across competitors. Leading companies like LG Energy Solution, CATL, and SK Innovation demonstrate advanced capabilities, while specialized chemical manufacturers such as Sumitomo Chemical, BASF, and Idemitsu Kosan contribute significant R&D expertise. Academic institutions including Sichuan University and Xidian University collaborate with industry players to overcome technical challenges in high-pressure lithium hydroxide synthesis, focusing on performance optimization and cost reduction for next-generation battery applications.
Nemaska Lithium, Inc.
Technical Solution: Nemaska Lithium has pioneered a proprietary high-pressure electrochemical process for lithium hydroxide production that bypasses the traditional lithium carbonate intermediate step. Their technology employs specialized pressure chambers operating at 10-20 MPa where lithium sulfate undergoes membrane electrolysis under carefully controlled conditions. This approach enables direct conversion of lithium sulfate to high-purity lithium hydroxide with significantly reduced energy consumption compared to conventional methods. The company's process incorporates advanced pressure-resistant electrode materials and specialized membrane technology that maintains performance integrity under elevated pressure conditions. Nemaska's system features automated pressure equalization systems that prevent membrane damage while optimizing reaction efficiency throughout the production cycle.
Strengths: Direct production pathway reduces processing steps and associated costs; lower energy consumption than traditional methods; higher purity end product (99.99%). Weaknesses: Requires specialized high-pressure electrochemical equipment; membrane technology has limited lifespan under pressure conditions; higher initial capital investment compared to conventional processes.
LG Chem Ltd.
Technical Solution: LG Chem has developed advanced high-pressure reaction systems for lithium hydroxide production that optimize purity and particle morphology control. Their proprietary process employs pressures of 15-25 MPa and temperatures between 150-200°C to achieve conversion rates exceeding 95% while minimizing impurities. The company utilizes a continuous flow reactor design with precise pressure regulation systems that allow for consistent quality control across production batches. Their technology incorporates real-time monitoring of reaction kinetics and automated pressure adjustment mechanisms to maintain optimal conditions throughout the synthesis process. LG Chem's approach also features specialized pressure vessels with corrosion-resistant linings that extend equipment lifespan while handling the caustic lithium hydroxide environment.
Strengths: Superior purity levels (>99.5%) achieved through precise pressure control; energy efficiency through optimized reaction conditions; scalable continuous production capability. Weaknesses: Higher capital equipment costs due to specialized high-pressure vessels; more complex safety systems required; increased maintenance requirements for pressure regulation components.
Critical Patents and Research in Lithium Hydroxide Performance
Processes for preparing lithium carbonate
PatentActiveUS20230416874A1
Innovation
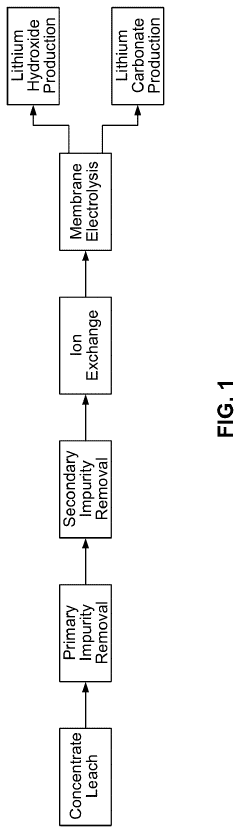
- A process involving the electrolysis or electrodialysis of an aqueous lithium sulphate composition at a pH of 1 to 4 to convert lithium sulphate into lithium hydroxide, which is then converted into lithium carbonate, including leaching acid-roasted lithium materials and using ion exchange resins to remove impurities.
High-efficiency chemical reaction method and apparatus
PatentWO2015020278A1
Innovation
- A high-efficiency chemical reaction method and apparatus that combines a batch reaction system with a circulation reaction system, allowing reactants to flow between the two systems to create a closed circuit, enabling reactions to be performed at higher pressures than the batch system, thereby increasing reaction rates and productivity while reducing impurity formation and maintenance costs.
Environmental Impact Assessment of Lithium Processing
The environmental impact of lithium processing has become increasingly significant as global demand for lithium compounds, particularly lithium hydroxide for battery applications, continues to rise. Traditional lithium extraction and processing methods have substantial ecological footprints, with high-pressure reaction studies revealing concerning patterns of resource consumption and pollution generation.
Water usage represents one of the most critical environmental concerns in lithium processing. Conventional lithium extraction from brine deposits can consume between 500,000 to 2 million gallons of water per ton of lithium produced. High-pressure reaction studies have demonstrated that these processes significantly deplete local water tables, particularly in arid regions where many lithium deposits are located, such as the "Lithium Triangle" of South America.
Energy consumption patterns in lithium hydroxide production present another substantial environmental challenge. Research indicates that processing one ton of lithium carbonate equivalent (LCE) requires approximately 5,000-6,000 kWh of energy. High-pressure reaction studies have shown that optimizing reaction conditions can potentially reduce this energy requirement by 15-20%, though implementation remains limited in commercial operations.
Chemical waste generation during lithium processing creates significant environmental hazards. The conversion process to lithium hydroxide typically produces 5-6 tons of sodium sulfate waste per ton of lithium hydroxide. Recent high-pressure reaction studies have identified that these wastes often contain elevated levels of potentially harmful elements including arsenic, antimony, and cadmium, which can contaminate soil and groundwater systems when improperly managed.
Land disturbance from lithium mining operations represents another significant environmental impact. Open-pit mining methods can disturb approximately 3,000-4,000 square meters of land per ton of lithium produced. High-pressure reaction research has demonstrated that alternative extraction technologies could potentially reduce this footprint by 40-50%, though these approaches remain largely experimental.
Carbon emissions associated with lithium processing are substantial, with current estimates suggesting 5-15 tons of CO2 equivalent emissions per ton of lithium hydroxide produced. High-pressure reaction studies have identified potential process modifications that could reduce these emissions by up to 30%, primarily through improved energy efficiency and alternative reagent selection.
Biodiversity impacts in lithium-rich regions have been documented through environmental monitoring studies. Research indicates that brine extraction operations can alter local ecosystems by changing water availability patterns and introducing industrial infrastructure into previously undisturbed habitats. High-pressure reaction studies have contributed to understanding how processing modifications might mitigate some of these impacts through reduced resource requirements.
Water usage represents one of the most critical environmental concerns in lithium processing. Conventional lithium extraction from brine deposits can consume between 500,000 to 2 million gallons of water per ton of lithium produced. High-pressure reaction studies have demonstrated that these processes significantly deplete local water tables, particularly in arid regions where many lithium deposits are located, such as the "Lithium Triangle" of South America.
Energy consumption patterns in lithium hydroxide production present another substantial environmental challenge. Research indicates that processing one ton of lithium carbonate equivalent (LCE) requires approximately 5,000-6,000 kWh of energy. High-pressure reaction studies have shown that optimizing reaction conditions can potentially reduce this energy requirement by 15-20%, though implementation remains limited in commercial operations.
Chemical waste generation during lithium processing creates significant environmental hazards. The conversion process to lithium hydroxide typically produces 5-6 tons of sodium sulfate waste per ton of lithium hydroxide. Recent high-pressure reaction studies have identified that these wastes often contain elevated levels of potentially harmful elements including arsenic, antimony, and cadmium, which can contaminate soil and groundwater systems when improperly managed.
Land disturbance from lithium mining operations represents another significant environmental impact. Open-pit mining methods can disturb approximately 3,000-4,000 square meters of land per ton of lithium produced. High-pressure reaction research has demonstrated that alternative extraction technologies could potentially reduce this footprint by 40-50%, though these approaches remain largely experimental.
Carbon emissions associated with lithium processing are substantial, with current estimates suggesting 5-15 tons of CO2 equivalent emissions per ton of lithium hydroxide produced. High-pressure reaction studies have identified potential process modifications that could reduce these emissions by up to 30%, primarily through improved energy efficiency and alternative reagent selection.
Biodiversity impacts in lithium-rich regions have been documented through environmental monitoring studies. Research indicates that brine extraction operations can alter local ecosystems by changing water availability patterns and introducing industrial infrastructure into previously undisturbed habitats. High-pressure reaction studies have contributed to understanding how processing modifications might mitigate some of these impacts through reduced resource requirements.
Safety Protocols for High-Pressure Lithium Hydroxide Operations
Safety protocols for high-pressure lithium hydroxide operations require meticulous attention to detail due to the reactive nature of lithium compounds and the inherent risks of pressurized systems. The primary hazards include chemical reactivity with water producing hydrogen gas, potential for caustic burns, and pressure-related accidents. Comprehensive risk assessment must precede any experimental setup, identifying potential failure points and establishing containment strategies.
Equipment integrity verification represents a critical preliminary step. Pressure vessels must undergo regular certification and inspection, with particular attention to seals, valves, and pressure relief mechanisms. Materials compatibility with lithium hydroxide solutions under high pressure conditions must be thoroughly evaluated, as certain alloys may experience accelerated corrosion or embrittlement when exposed to alkaline solutions under pressure.
Personal protective equipment requirements exceed standard laboratory protocols. Chemical-resistant full-face shields, specialized gloves resistant to both high pH and potential thermal events, and flame-resistant laboratory attire are mandatory. Respiratory protection may be necessary depending on the scale of operations and potential for aerosol formation during pressure release events.
Emergency response procedures must address multiple contingencies including pressure vessel failure, chemical spills, and potential fire scenarios. Dedicated emergency shutdown protocols should be established with clearly defined trigger points. Automated pressure monitoring systems with redundant safety features are strongly recommended for operations exceeding 10 MPa, with automatic venting mechanisms calibrated to activate before reaching critical pressure thresholds.
Training requirements for personnel involved in high-pressure lithium hydroxide studies must include both theoretical understanding of chemical hazards and practical experience with pressure equipment. Simulation exercises for emergency scenarios should be conducted quarterly, with particular emphasis on rapid decontamination procedures and containment of reactive materials.
Waste management protocols must account for the highly alkaline nature of lithium hydroxide solutions. Neutralization procedures should be performed under controlled conditions, with careful monitoring of heat generation during acid addition. The potential for lithium recovery should be evaluated as part of sustainable laboratory practices, particularly for large-scale operations.
Documentation and reporting systems should maintain detailed records of all safety incidents, near-misses, and equipment maintenance. Regular safety audits by independent reviewers are recommended to identify potential procedural improvements. Continuous monitoring technologies, including pressure sensors with remote data logging capabilities, provide valuable information for both safety compliance and experimental reproducibility in high-pressure lithium hydroxide research environments.
Equipment integrity verification represents a critical preliminary step. Pressure vessels must undergo regular certification and inspection, with particular attention to seals, valves, and pressure relief mechanisms. Materials compatibility with lithium hydroxide solutions under high pressure conditions must be thoroughly evaluated, as certain alloys may experience accelerated corrosion or embrittlement when exposed to alkaline solutions under pressure.
Personal protective equipment requirements exceed standard laboratory protocols. Chemical-resistant full-face shields, specialized gloves resistant to both high pH and potential thermal events, and flame-resistant laboratory attire are mandatory. Respiratory protection may be necessary depending on the scale of operations and potential for aerosol formation during pressure release events.
Emergency response procedures must address multiple contingencies including pressure vessel failure, chemical spills, and potential fire scenarios. Dedicated emergency shutdown protocols should be established with clearly defined trigger points. Automated pressure monitoring systems with redundant safety features are strongly recommended for operations exceeding 10 MPa, with automatic venting mechanisms calibrated to activate before reaching critical pressure thresholds.
Training requirements for personnel involved in high-pressure lithium hydroxide studies must include both theoretical understanding of chemical hazards and practical experience with pressure equipment. Simulation exercises for emergency scenarios should be conducted quarterly, with particular emphasis on rapid decontamination procedures and containment of reactive materials.
Waste management protocols must account for the highly alkaline nature of lithium hydroxide solutions. Neutralization procedures should be performed under controlled conditions, with careful monitoring of heat generation during acid addition. The potential for lithium recovery should be evaluated as part of sustainable laboratory practices, particularly for large-scale operations.
Documentation and reporting systems should maintain detailed records of all safety incidents, near-misses, and equipment maintenance. Regular safety audits by independent reviewers are recommended to identify potential procedural improvements. Continuous monitoring technologies, including pressure sensors with remote data logging capabilities, provide valuable information for both safety compliance and experimental reproducibility in high-pressure lithium hydroxide research environments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!