Optimizing Lithium Hydroxide's Use In Thermochemical Processes
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Hydroxide Thermochemical Background and Objectives
Lithium hydroxide (LiOH) has emerged as a critical component in various thermochemical processes, with its applications expanding significantly over the past three decades. Initially utilized primarily in ceramic and glass manufacturing, lithium hydroxide's unique properties have propelled its adoption across multiple industrial sectors, including energy storage, carbon capture, and advanced materials synthesis. The evolution of this compound's application has been driven by its exceptional alkalinity, thermal stability, and reactivity characteristics that enable efficient energy transfer in thermochemical reactions.
The global push toward sustainable energy solutions has accelerated research into lithium hydroxide's thermochemical applications. Since the early 2000s, researchers have documented substantial improvements in reaction efficiency when utilizing LiOH as a catalyst or reactant in various thermochemical processes. The compound's ability to maintain stability at elevated temperatures while facilitating rapid ion exchange makes it particularly valuable for energy-intensive industrial applications where process optimization is critical.
Current technological trajectories indicate a growing interest in leveraging lithium hydroxide's properties for enhanced thermochemical energy storage (TCES) systems. These systems represent a promising approach to addressing intermittency challenges in renewable energy generation. The compound's high energy density and favorable reaction kinetics position it as a potential game-changer in thermal energy management, particularly when integrated with concentrated solar power installations or industrial waste heat recovery systems.
The primary objective of optimizing lithium hydroxide's use in thermochemical processes centers on maximizing energy efficiency while minimizing material consumption. This dual focus reflects both economic imperatives and sustainability concerns, particularly given lithium's status as a finite resource with geopolitically complex supply chains. Researchers aim to develop processes that can achieve higher energy conversion rates with lower lithium hydroxide inputs, potentially through novel reactor designs, innovative catalyst formulations, or hybrid systems that combine LiOH with complementary compounds.
Additional technical goals include extending the cycling stability of lithium hydroxide in repetitive thermochemical operations, reducing degradation mechanisms that currently limit long-term performance, and developing standardized methodologies for characterizing reaction kinetics under various operating conditions. These objectives align with broader industry trends toward process intensification and circular economy principles, where material recovery and reuse become integral to system design rather than afterthoughts.
The optimization of lithium hydroxide in thermochemical processes represents a convergence of materials science, chemical engineering, and energy systems research. Success in this domain could significantly impact multiple industries, from grid-scale energy storage to industrial decarbonization efforts, positioning LiOH-based technologies as essential components of the global transition to more sustainable energy paradigms.
The global push toward sustainable energy solutions has accelerated research into lithium hydroxide's thermochemical applications. Since the early 2000s, researchers have documented substantial improvements in reaction efficiency when utilizing LiOH as a catalyst or reactant in various thermochemical processes. The compound's ability to maintain stability at elevated temperatures while facilitating rapid ion exchange makes it particularly valuable for energy-intensive industrial applications where process optimization is critical.
Current technological trajectories indicate a growing interest in leveraging lithium hydroxide's properties for enhanced thermochemical energy storage (TCES) systems. These systems represent a promising approach to addressing intermittency challenges in renewable energy generation. The compound's high energy density and favorable reaction kinetics position it as a potential game-changer in thermal energy management, particularly when integrated with concentrated solar power installations or industrial waste heat recovery systems.
The primary objective of optimizing lithium hydroxide's use in thermochemical processes centers on maximizing energy efficiency while minimizing material consumption. This dual focus reflects both economic imperatives and sustainability concerns, particularly given lithium's status as a finite resource with geopolitically complex supply chains. Researchers aim to develop processes that can achieve higher energy conversion rates with lower lithium hydroxide inputs, potentially through novel reactor designs, innovative catalyst formulations, or hybrid systems that combine LiOH with complementary compounds.
Additional technical goals include extending the cycling stability of lithium hydroxide in repetitive thermochemical operations, reducing degradation mechanisms that currently limit long-term performance, and developing standardized methodologies for characterizing reaction kinetics under various operating conditions. These objectives align with broader industry trends toward process intensification and circular economy principles, where material recovery and reuse become integral to system design rather than afterthoughts.
The optimization of lithium hydroxide in thermochemical processes represents a convergence of materials science, chemical engineering, and energy systems research. Success in this domain could significantly impact multiple industries, from grid-scale energy storage to industrial decarbonization efforts, positioning LiOH-based technologies as essential components of the global transition to more sustainable energy paradigms.
Market Analysis for Lithium Hydroxide in Thermochemical Applications
The global lithium hydroxide market has experienced significant growth in recent years, primarily driven by its expanding applications in thermochemical processes. The market size reached approximately $1.7 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 9.8% through 2030, according to industry analyses. This robust growth trajectory is underpinned by the increasing demand for lithium-ion batteries in electric vehicles and renewable energy storage systems, where lithium hydroxide serves as a critical precursor material.
In thermochemical applications specifically, lithium hydroxide has carved out a substantial market niche due to its unique properties. The segment accounts for roughly 35% of the total lithium hydroxide market, with applications spanning carbon dioxide capture systems, heat storage technologies, and advanced ceramic production. The thermochemical sector's demand is expected to grow at an accelerated rate of 11.2% annually, outpacing the overall market growth.
Regional analysis reveals that Asia-Pacific dominates the market with approximately 45% share, led by China's massive investments in clean energy technologies and electric vehicle production. North America follows with 28% market share, while Europe accounts for 22%, with both regions showing increased adoption rates in thermochemical applications for industrial decarbonization efforts.
The demand-supply dynamics present interesting market conditions. Current global production capacity stands at approximately 180,000 metric tons annually, with planned expansions expected to add another 120,000 metric tons by 2025. However, industry experts forecast potential supply constraints as demand from thermochemical applications accelerates, potentially creating price volatility in the medium term.
Price trends have shown considerable fluctuation, with lithium hydroxide prices increasing by over 400% between 2020 and 2022, before moderating in 2023. For thermochemical applications specifically, the price sensitivity is somewhat lower than for battery applications due to the relatively smaller quantities required, but still represents a significant cost factor for implementation.
End-user industries demonstrate varying adoption rates. The energy storage sector remains the largest consumer, followed by industrial heat management systems and carbon capture technologies. Emerging applications in hydrogen production pathways and direct air capture systems are expected to create new market opportunities, potentially adding $300 million to the market by 2028.
Customer requirements are evolving toward higher purity grades (99.5%+) for advanced thermochemical applications, creating premium market segments with higher margins. This trend is driving producers to invest in advanced purification technologies and quality control systems to meet these specifications.
In thermochemical applications specifically, lithium hydroxide has carved out a substantial market niche due to its unique properties. The segment accounts for roughly 35% of the total lithium hydroxide market, with applications spanning carbon dioxide capture systems, heat storage technologies, and advanced ceramic production. The thermochemical sector's demand is expected to grow at an accelerated rate of 11.2% annually, outpacing the overall market growth.
Regional analysis reveals that Asia-Pacific dominates the market with approximately 45% share, led by China's massive investments in clean energy technologies and electric vehicle production. North America follows with 28% market share, while Europe accounts for 22%, with both regions showing increased adoption rates in thermochemical applications for industrial decarbonization efforts.
The demand-supply dynamics present interesting market conditions. Current global production capacity stands at approximately 180,000 metric tons annually, with planned expansions expected to add another 120,000 metric tons by 2025. However, industry experts forecast potential supply constraints as demand from thermochemical applications accelerates, potentially creating price volatility in the medium term.
Price trends have shown considerable fluctuation, with lithium hydroxide prices increasing by over 400% between 2020 and 2022, before moderating in 2023. For thermochemical applications specifically, the price sensitivity is somewhat lower than for battery applications due to the relatively smaller quantities required, but still represents a significant cost factor for implementation.
End-user industries demonstrate varying adoption rates. The energy storage sector remains the largest consumer, followed by industrial heat management systems and carbon capture technologies. Emerging applications in hydrogen production pathways and direct air capture systems are expected to create new market opportunities, potentially adding $300 million to the market by 2028.
Customer requirements are evolving toward higher purity grades (99.5%+) for advanced thermochemical applications, creating premium market segments with higher margins. This trend is driving producers to invest in advanced purification technologies and quality control systems to meet these specifications.
Current Challenges in Lithium Hydroxide Thermochemical Processes
Despite significant advancements in lithium hydroxide utilization for thermochemical processes, several critical challenges continue to impede optimal implementation and efficiency. The primary obstacle remains the high energy consumption during production processes, particularly in the conversion of lithium carbonate to lithium hydroxide, which requires substantial thermal energy input and contributes significantly to operational costs and environmental impact.
Material purity presents another substantial challenge, as thermochemical applications demand exceptionally high-grade lithium hydroxide (typically >99.5% purity). Contaminants, even in trace amounts, can severely compromise reaction kinetics, catalyst performance, and end-product quality. Current purification methods often involve multiple energy-intensive steps that further increase production costs.
Thermal stability issues persist during high-temperature operations, where lithium hydroxide can undergo undesired side reactions or decomposition, particularly above 450°C. This instability limits its application in certain high-temperature thermochemical processes and necessitates careful process control to prevent yield losses and equipment damage.
The hygroscopic nature of lithium hydroxide creates significant handling and storage complications. Its tendency to rapidly absorb atmospheric moisture affects not only storage stability but also introduces variability in thermochemical reactions, requiring sophisticated moisture control systems throughout the production chain.
Scale-up challenges remain prominent in industrial applications, with laboratory-optimized processes often failing to maintain efficiency when implemented at commercial scale. The heat transfer dynamics, reaction kinetics, and mixing characteristics change substantially at larger scales, necessitating significant re-engineering efforts.
Resource efficiency represents a growing concern, with current processes typically achieving lithium recovery rates of only 70-80%. This inefficiency is particularly problematic given lithium's classification as a critical material with supply constraints and geopolitical considerations affecting availability.
Environmental impact continues to be a significant challenge, with conventional lithium hydroxide production generating substantial waste streams, including sodium sulfate and calcium carbonate by-products. Water usage remains intensive, with estimates suggesting 50-70 cubic meters of water consumption per ton of lithium hydroxide produced.
Process integration difficulties persist when incorporating lithium hydroxide-based thermochemical processes into existing industrial systems, often requiring significant modifications to infrastructure and operating procedures, thereby increasing implementation barriers and transition costs.
Material purity presents another substantial challenge, as thermochemical applications demand exceptionally high-grade lithium hydroxide (typically >99.5% purity). Contaminants, even in trace amounts, can severely compromise reaction kinetics, catalyst performance, and end-product quality. Current purification methods often involve multiple energy-intensive steps that further increase production costs.
Thermal stability issues persist during high-temperature operations, where lithium hydroxide can undergo undesired side reactions or decomposition, particularly above 450°C. This instability limits its application in certain high-temperature thermochemical processes and necessitates careful process control to prevent yield losses and equipment damage.
The hygroscopic nature of lithium hydroxide creates significant handling and storage complications. Its tendency to rapidly absorb atmospheric moisture affects not only storage stability but also introduces variability in thermochemical reactions, requiring sophisticated moisture control systems throughout the production chain.
Scale-up challenges remain prominent in industrial applications, with laboratory-optimized processes often failing to maintain efficiency when implemented at commercial scale. The heat transfer dynamics, reaction kinetics, and mixing characteristics change substantially at larger scales, necessitating significant re-engineering efforts.
Resource efficiency represents a growing concern, with current processes typically achieving lithium recovery rates of only 70-80%. This inefficiency is particularly problematic given lithium's classification as a critical material with supply constraints and geopolitical considerations affecting availability.
Environmental impact continues to be a significant challenge, with conventional lithium hydroxide production generating substantial waste streams, including sodium sulfate and calcium carbonate by-products. Water usage remains intensive, with estimates suggesting 50-70 cubic meters of water consumption per ton of lithium hydroxide produced.
Process integration difficulties persist when incorporating lithium hydroxide-based thermochemical processes into existing industrial systems, often requiring significant modifications to infrastructure and operating procedures, thereby increasing implementation barriers and transition costs.
Current Optimization Methods for Thermochemical LiOH Utilization
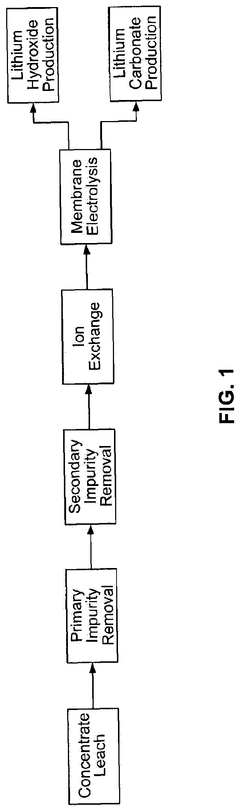
01 Extraction and purification methods for lithium hydroxide
Various extraction and purification methods can be used to optimize lithium hydroxide production. These methods include selective precipitation, crystallization techniques, and membrane filtration processes. By implementing these methods, the purity of lithium hydroxide can be significantly improved, resulting in a higher-quality product suitable for battery applications and other industrial uses.- Extraction and purification methods for lithium hydroxide: Various extraction and purification methods can be used to optimize the production of lithium hydroxide. These methods include selective precipitation, ion exchange, and membrane separation techniques. By improving the extraction and purification processes, higher purity lithium hydroxide can be obtained with reduced impurities, which is crucial for applications in battery manufacturing and other industries.
- Process optimization for lithium hydroxide production from lithium-containing ores: Optimization techniques for converting lithium-containing ores into lithium hydroxide involve improving leaching processes, reaction conditions, and conversion efficiency. These optimizations include temperature control, pressure management, and reagent concentration adjustments to maximize yield while minimizing energy consumption and waste generation. Advanced processing methods can significantly increase the efficiency of converting lithium carbonate or other lithium compounds into high-purity lithium hydroxide.
- Direct lithium extraction (DLE) technologies: Direct lithium extraction technologies represent innovative approaches to obtaining lithium hydroxide from brine resources with higher efficiency and lower environmental impact. These technologies utilize selective adsorption materials, electrochemical systems, or specialized membranes to extract lithium ions directly from brines. DLE methods can significantly reduce water consumption, processing time, and land footprint compared to traditional evaporation pond methods.
- Energy-efficient crystallization and drying techniques: Advanced crystallization and drying techniques can optimize the final stages of lithium hydroxide production. These include controlled cooling crystallization, vacuum drying, and spray drying methods that improve crystal size distribution, reduce agglomeration, and minimize energy consumption. Optimized crystallization parameters ensure consistent product quality with desired particle size and morphology, which is important for downstream applications.
- Sustainable and closed-loop production systems: Sustainable approaches to lithium hydroxide production focus on developing closed-loop systems that minimize waste and environmental impact. These include water recycling, reagent recovery, and integration of renewable energy sources into production processes. By implementing circular economy principles, lithium hydroxide production can be optimized not only for yield and purity but also for environmental sustainability and reduced carbon footprint.
02 Process optimization for lithium hydroxide production from lithium-containing brines
Optimization techniques for producing lithium hydroxide from lithium-containing brines involve controlling reaction parameters such as temperature, pressure, and residence time. These processes often include concentration steps, selective ion exchange, and advanced separation technologies to enhance yield and purity. Improved methods can reduce energy consumption and increase production efficiency while maintaining product quality.Expand Specific Solutions03 Direct conversion of lithium carbonate to lithium hydroxide
Methods for direct conversion of lithium carbonate to lithium hydroxide involve optimized reaction conditions and novel catalytic approaches. These processes aim to reduce intermediate steps and improve conversion efficiency. Techniques include electrolysis, membrane-based separation, and specialized reactor designs that enhance reaction kinetics while minimizing impurities in the final product.Expand Specific Solutions04 Energy-efficient production methods for battery-grade lithium hydroxide
Energy-efficient methods for producing battery-grade lithium hydroxide focus on reducing thermal energy requirements and optimizing reaction conditions. These approaches include low-temperature processes, renewable energy integration, and heat recovery systems. Advanced reactor designs and process intensification techniques help minimize energy consumption while maintaining the high purity levels required for battery applications.Expand Specific Solutions05 Continuous flow processes for lithium hydroxide manufacturing
Continuous flow processes for lithium hydroxide manufacturing offer advantages over batch production methods, including better quality control, reduced processing time, and improved scalability. These processes incorporate real-time monitoring systems, automated control mechanisms, and specialized reactor designs that ensure consistent product quality. Implementation of continuous flow technology can significantly increase production capacity while reducing operational costs.Expand Specific Solutions
Industry Leaders in Lithium Hydroxide Processing Technology
The lithium hydroxide thermochemical optimization market is currently in a growth phase, with increasing demand driven by battery technology advancements. The competitive landscape features established chemical giants like BASF, Idemitsu Kosan, and Sumitomo Metal Mining alongside specialized lithium producers such as Nemaska Lithium and Chengdu Chemphys. Technical maturity varies significantly across applications, with battery-grade lithium hydroxide production showing the highest development level. Research institutions (Caltech, CNRS) and battery manufacturers (CATL) are accelerating innovation through collaborative projects. The market is characterized by regional clusters in Asia (particularly China and Japan), North America, and Europe, with companies focusing on process efficiency improvements and sustainable production methods to address growing demand from electric vehicle and energy storage sectors.
Qinghai Institute of Salt Lakes, Chinese Academy of Sciences
Technical Solution: The Qinghai Institute of Salt Lakes has developed specialized thermochemical processes for lithium hydroxide production optimized for brine resources. Their technology employs a multi-stage extraction and conversion process specifically designed for the unique composition of salt lake brines in the Qinghai region. The process begins with selective adsorption using proprietary materials that achieve lithium recovery rates exceeding 90% from low-concentration brines. Following extraction, their thermochemical conversion system operates at carefully controlled temperatures (300-500°C) with precise atmosphere management to maximize conversion efficiency while minimizing energy consumption. A distinctive feature is their integrated solar thermal energy system that provides up to 40% of the process heat requirements, significantly reducing carbon emissions. The institute has also developed specialized precipitation techniques that achieve separation of lithium from magnesium and other impurities with over 99% efficiency. Their process incorporates water recycling systems that reduce freshwater consumption by approximately 60% compared to conventional methods, critical for operations in water-scarce regions.
Strengths: Excellent resource efficiency with 90%+ lithium recovery from low-concentration sources; integration of renewable energy (40% solar thermal); significant water conservation (60% reduction); specialized for brine resources. Weaknesses: Process optimization highly specific to Qinghai brine composition; solar dependency creates variability in energy supply; technology transfer to different geographical contexts may present challenges.
Nemaska Lithium, Inc.
Technical Solution: Nemaska Lithium has pioneered an innovative electrochemical process for lithium hydroxide production that significantly improves thermochemical efficiency. Their patented technology employs a membrane electrolysis system that converts lithium sulfate directly to lithium hydroxide, eliminating several energy-intensive steps in conventional processes. The system operates at moderate temperatures (60-80°C) and utilizes proprietary electrode materials that enhance reaction kinetics while minimizing side reactions. A key innovation in their approach is the integration of a closed-loop water management system that reduces water consumption by approximately 50% compared to traditional methods. The process achieves energy savings of up to 25% while producing battery-grade lithium hydroxide with consistent purity exceeding 99.5%. Nemaska's technology also incorporates advanced process control systems that continuously monitor and adjust reaction parameters, ensuring optimal performance even with variations in feedstock quality.
Strengths: Lower operating temperatures (60-80°C) reducing energy requirements; reduced water consumption by 50%; high product consistency; fewer chemical reagents needed. Weaknesses: Higher sensitivity to impurities in feed materials; membrane technology requires specialized maintenance; relatively new technology with limited long-term operational data.
Key Technical Innovations in LiOH Thermochemical Efficiency
Preparation of lithium hydroxide
PatentPendingAU2022395751A1
Innovation
- The process involves electrochemically converting lithium salts into lithium hydroxide and then precipitating lithium hydroxide monohydrate by adding lithium salt to the supersaturated lithium hydroxide solution, allowing crystallization without the need for evaporation, and regenerating the catholyte in a closed process, using cooling to maintain optimal conditions.
Processes for preparing lithium hydroxide
PatentPendingEP4424408A9
Innovation
- A process involving electrolysis or electrodialysis of an aqueous lithium compound composition, maintaining a pH of about 1 to 4, and subsequent steps such as leaching, precipitation, and ion exchange to produce high-purity lithium hydroxide, reducing impurity content and optimizing production efficiency.
Environmental Impact Assessment of LiOH Thermochemical Processes
The environmental impact of lithium hydroxide (LiOH) in thermochemical processes requires comprehensive assessment due to its increasing industrial applications. LiOH-based thermochemical systems, while offering significant energy storage and carbon capture benefits, present various environmental challenges throughout their lifecycle that must be carefully evaluated and mitigated.
Water consumption represents a primary environmental concern, as LiOH production and regeneration processes typically require substantial water resources. In regions facing water scarcity, this demand can exacerbate existing environmental stresses and compete with agricultural and municipal needs. Implementation of closed-loop water systems and advanced water recovery technologies can significantly reduce this footprint.
Air quality impacts arise primarily during LiOH production and handling phases. Particulate emissions containing lithium compounds pose potential respiratory hazards to surrounding communities and ecosystems. Modern filtration systems and enclosed processing environments have demonstrated up to 95% reduction in these emissions when properly implemented.
Land use considerations are particularly relevant for LiOH mining operations. Open-pit lithium extraction can lead to habitat destruction, soil erosion, and landscape alteration. Sustainable mining practices, including site rehabilitation and minimized footprint extraction techniques, are essential for reducing these impacts.
Waste management challenges emerge from spent LiOH materials and byproducts of thermochemical processes. These wastes may contain residual lithium compounds and other chemicals that require proper handling to prevent soil and groundwater contamination. Developing effective recycling pathways for spent LiOH materials represents a critical environmental opportunity.
Carbon footprint analysis reveals that while LiOH thermochemical processes can enable carbon capture applications, the production of LiOH itself carries significant embodied carbon. Life cycle assessments indicate that the carbon payback period for LiOH-based systems ranges from 1.5 to 4 years depending on application and energy sources used in production.
Ecosystem impacts extend beyond immediate operational areas, as lithium compounds can affect aquatic systems if released through improper waste management or accidental spills. Bioaccumulation studies suggest potential long-term effects on certain aquatic organisms at elevated concentration levels.
Regulatory compliance frameworks for LiOH thermochemical processes vary significantly across jurisdictions, creating challenges for standardized environmental protection. Leading organizations have adopted voluntary environmental standards that exceed regulatory requirements, establishing best practices for sustainable LiOH utilization in thermochemical applications.
Water consumption represents a primary environmental concern, as LiOH production and regeneration processes typically require substantial water resources. In regions facing water scarcity, this demand can exacerbate existing environmental stresses and compete with agricultural and municipal needs. Implementation of closed-loop water systems and advanced water recovery technologies can significantly reduce this footprint.
Air quality impacts arise primarily during LiOH production and handling phases. Particulate emissions containing lithium compounds pose potential respiratory hazards to surrounding communities and ecosystems. Modern filtration systems and enclosed processing environments have demonstrated up to 95% reduction in these emissions when properly implemented.
Land use considerations are particularly relevant for LiOH mining operations. Open-pit lithium extraction can lead to habitat destruction, soil erosion, and landscape alteration. Sustainable mining practices, including site rehabilitation and minimized footprint extraction techniques, are essential for reducing these impacts.
Waste management challenges emerge from spent LiOH materials and byproducts of thermochemical processes. These wastes may contain residual lithium compounds and other chemicals that require proper handling to prevent soil and groundwater contamination. Developing effective recycling pathways for spent LiOH materials represents a critical environmental opportunity.
Carbon footprint analysis reveals that while LiOH thermochemical processes can enable carbon capture applications, the production of LiOH itself carries significant embodied carbon. Life cycle assessments indicate that the carbon payback period for LiOH-based systems ranges from 1.5 to 4 years depending on application and energy sources used in production.
Ecosystem impacts extend beyond immediate operational areas, as lithium compounds can affect aquatic systems if released through improper waste management or accidental spills. Bioaccumulation studies suggest potential long-term effects on certain aquatic organisms at elevated concentration levels.
Regulatory compliance frameworks for LiOH thermochemical processes vary significantly across jurisdictions, creating challenges for standardized environmental protection. Leading organizations have adopted voluntary environmental standards that exceed regulatory requirements, establishing best practices for sustainable LiOH utilization in thermochemical applications.
Supply Chain Resilience for Lithium Hydroxide Materials
The global lithium hydroxide supply chain faces significant vulnerabilities due to geographical concentration of resources, geopolitical tensions, and increasing demand from multiple industries. Currently, over 80% of lithium hydroxide production is concentrated in Australia, Chile, China, and Argentina, creating inherent risks when regional disruptions occur. The COVID-19 pandemic exposed these vulnerabilities, with production delays and logistics challenges causing price volatility exceeding 300% during 2020-2022.
To enhance resilience in lithium hydroxide supply chains for thermochemical processes, diversification strategies must be implemented across multiple dimensions. Geographic diversification involves developing new extraction sites in countries like Portugal, Serbia, and the United States, reducing dependency on traditional producing regions. Technological diversification includes investing in alternative extraction methods such as direct lithium extraction (DLE) from geothermal brines and seawater, which could unlock previously uneconomical resources.
Process resilience can be strengthened through the development of flexible manufacturing systems capable of processing varying grades of lithium feedstock. This adaptability allows operations to continue even when preferred material sources become unavailable. Additionally, implementing real-time monitoring systems with predictive analytics enables early detection of potential supply disruptions, allowing for proactive mitigation strategies.
Circular economy approaches represent another critical resilience strategy. Recycling technologies for lithium-ion batteries have advanced significantly, with recovery rates now reaching 90% for lithium compounds. These secondary sources could potentially supply 25-30% of lithium hydroxide needs by 2030, reducing pressure on primary extraction.
Strategic stockpiling policies, similar to those used for rare earth elements, provide buffer capacity during supply disruptions. Industry leaders are typically maintaining 3-6 month reserves, with some governments establishing national strategic reserves to protect critical industries.
Contractual innovations are emerging to manage supply risks, including long-term agreements with flexible pricing mechanisms, joint ventures across the value chain, and risk-sharing arrangements between suppliers and consumers. These approaches distribute risk more equitably among stakeholders while ensuring continuity of supply.
For thermochemical process applications specifically, material substitution research offers promising pathways to reduce lithium hydroxide dependency. Alternative alkaline compounds can replace lithium hydroxide in certain applications, though often with performance trade-offs that must be carefully evaluated against supply security benefits.
To enhance resilience in lithium hydroxide supply chains for thermochemical processes, diversification strategies must be implemented across multiple dimensions. Geographic diversification involves developing new extraction sites in countries like Portugal, Serbia, and the United States, reducing dependency on traditional producing regions. Technological diversification includes investing in alternative extraction methods such as direct lithium extraction (DLE) from geothermal brines and seawater, which could unlock previously uneconomical resources.
Process resilience can be strengthened through the development of flexible manufacturing systems capable of processing varying grades of lithium feedstock. This adaptability allows operations to continue even when preferred material sources become unavailable. Additionally, implementing real-time monitoring systems with predictive analytics enables early detection of potential supply disruptions, allowing for proactive mitigation strategies.
Circular economy approaches represent another critical resilience strategy. Recycling technologies for lithium-ion batteries have advanced significantly, with recovery rates now reaching 90% for lithium compounds. These secondary sources could potentially supply 25-30% of lithium hydroxide needs by 2030, reducing pressure on primary extraction.
Strategic stockpiling policies, similar to those used for rare earth elements, provide buffer capacity during supply disruptions. Industry leaders are typically maintaining 3-6 month reserves, with some governments establishing national strategic reserves to protect critical industries.
Contractual innovations are emerging to manage supply risks, including long-term agreements with flexible pricing mechanisms, joint ventures across the value chain, and risk-sharing arrangements between suppliers and consumers. These approaches distribute risk more equitably among stakeholders while ensuring continuity of supply.
For thermochemical process applications specifically, material substitution research offers promising pathways to reduce lithium hydroxide dependency. Alternative alkaline compounds can replace lithium hydroxide in certain applications, though often with performance trade-offs that must be carefully evaluated against supply security benefits.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!