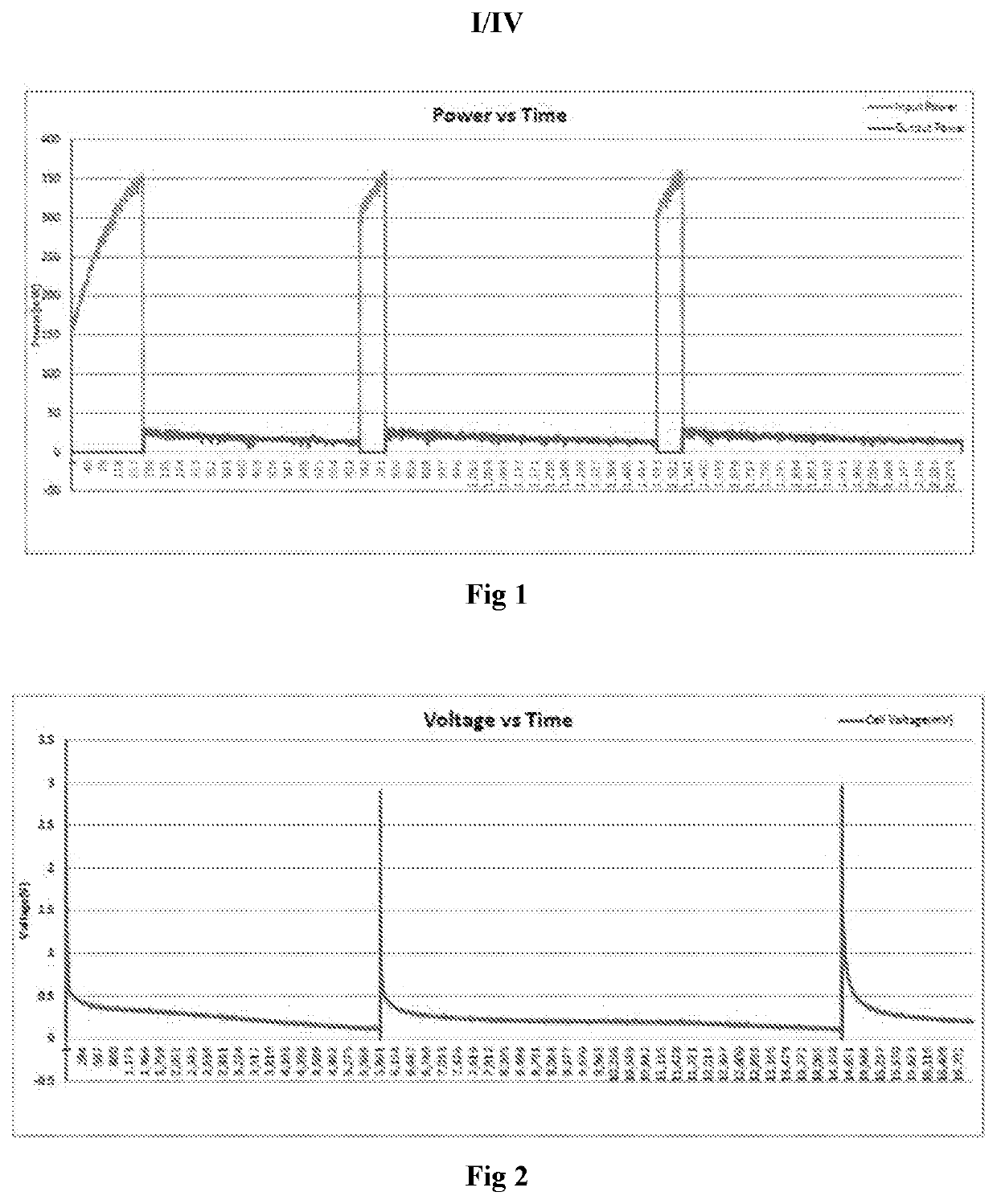
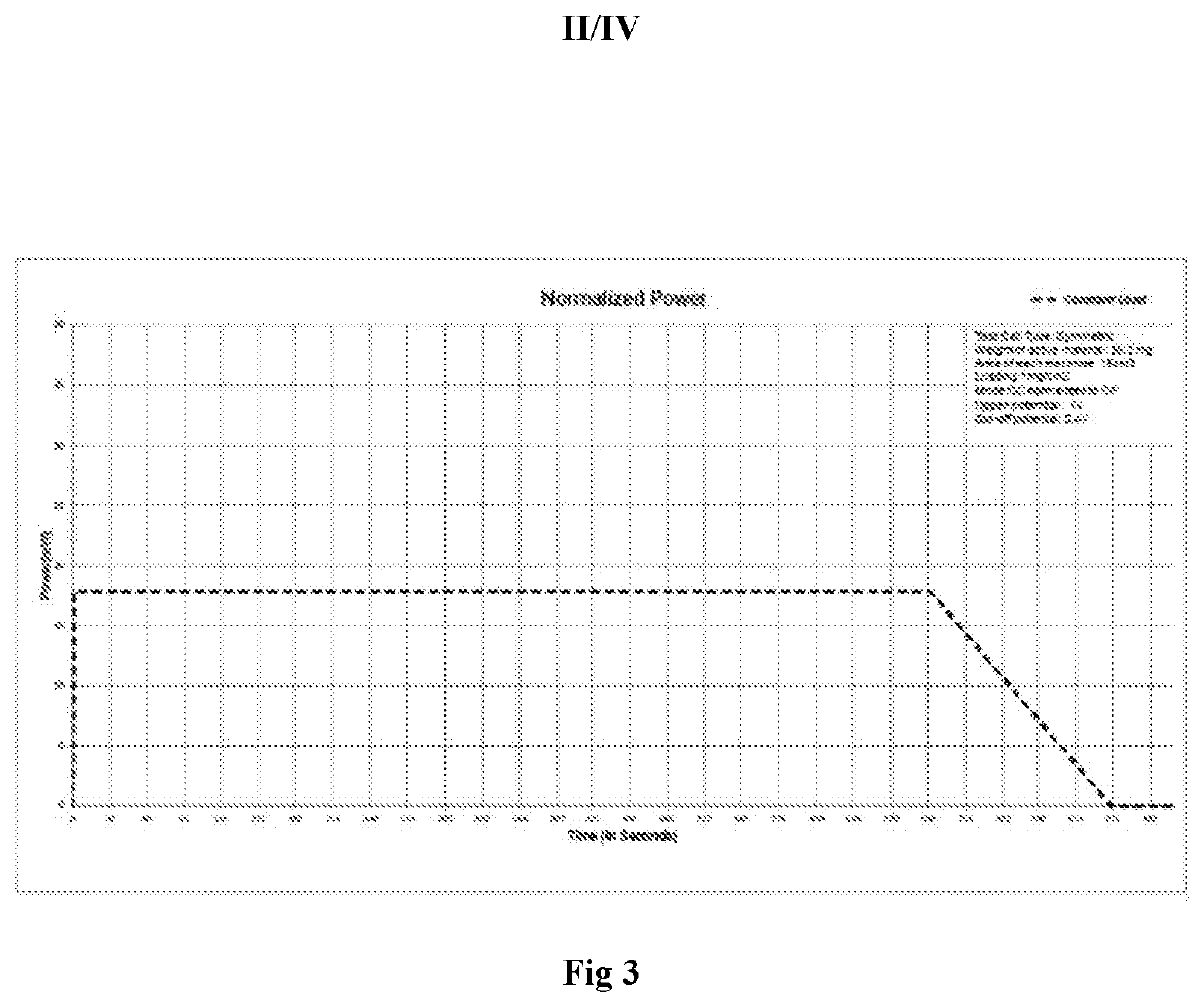
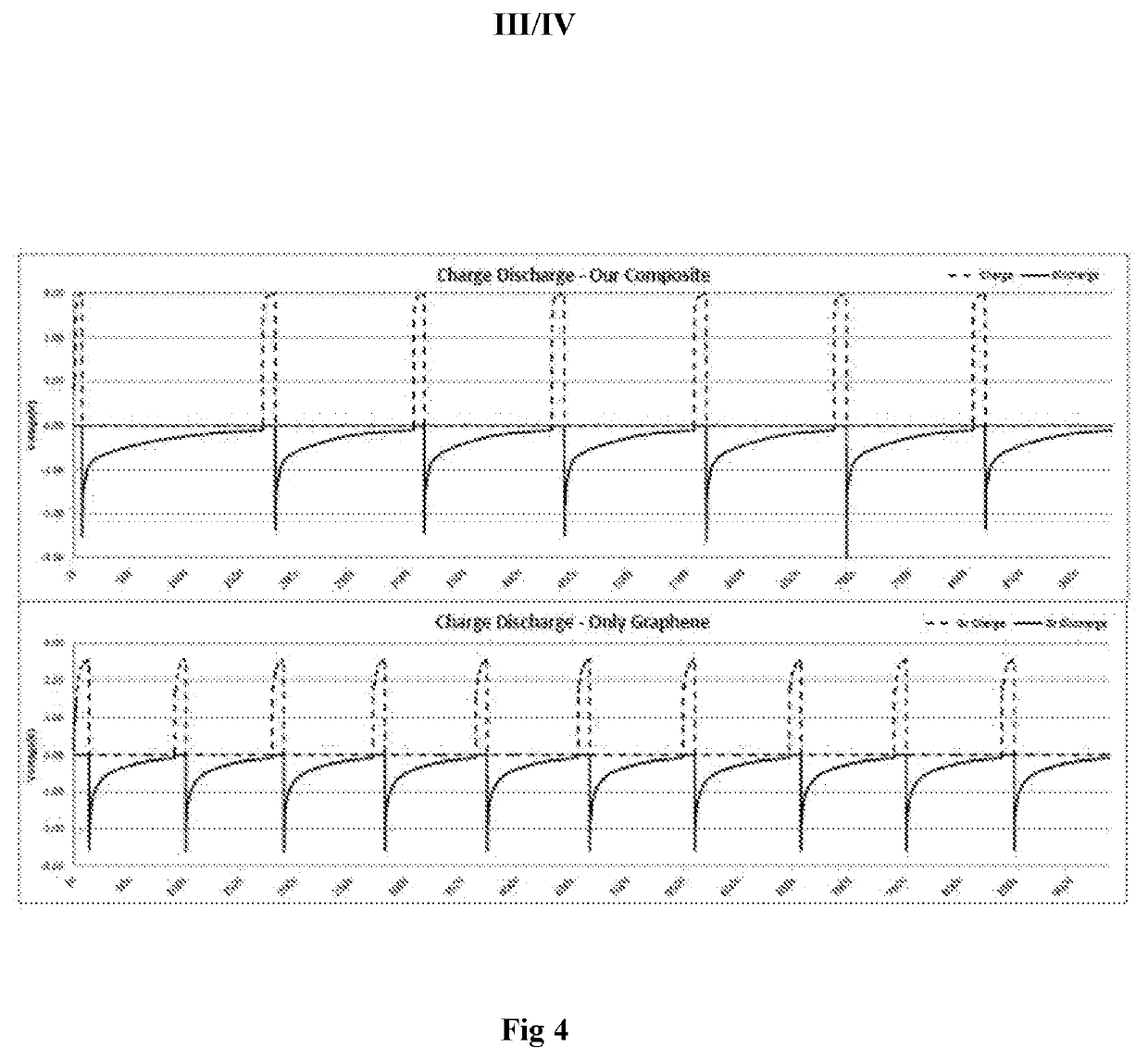
Lithium Hydroxide Vs Graphene: Conductivity Differences
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Hydroxide and Graphene Conductivity Background
Lithium hydroxide (LiOH) and graphene represent two distinct materials with significant implications for electrical conductivity applications across various industries. The exploration of their conductive properties has evolved considerably over the past decades, driven by increasing demands for high-performance energy storage systems and electronic components.
Lithium hydroxide, primarily known as a critical component in lithium-ion battery production, has been studied since the 1970s for its ionic conductivity properties. Its historical development traces back to early lithium battery research, where scientists discovered its role in facilitating lithium ion transport. The compound exhibits moderate ionic conductivity, which increases significantly when in solution or when incorporated into composite materials.
Graphene, by contrast, represents a revolutionary material first isolated in 2004 by Andre Geim and Konstantin Novoselov, who later received the Nobel Prize for this breakthrough. As a single layer of carbon atoms arranged in a two-dimensional honeycomb lattice, graphene demonstrates exceptional electronic conductivity properties that surpass most known materials, including copper and silver.
The fundamental difference in conductivity mechanisms between these materials stems from their atomic structures. Lithium hydroxide conducts electricity primarily through ionic transport, where lithium ions carry the charge. This process is relatively slower and more dependent on temperature and concentration gradients. Graphene, however, exhibits ballistic electronic conductivity, where electrons move with minimal scattering, resulting in near-superconductor-like performance at room temperature.
Research trends indicate growing interest in hybrid materials that combine the properties of both substances. The integration of lithium hydroxide into graphene-based structures has shown promising results for enhancing battery performance, particularly in terms of charge/discharge rates and cycle stability.
The technological evolution in this field has been marked by significant milestones, including the development of graphene production methods beyond mechanical exfoliation, such as chemical vapor deposition (CVD) and epitaxial growth. Similarly, advances in lithium hydroxide synthesis have focused on producing higher purity grades specifically designed for battery applications.
Current research directions are exploring the fundamental physics of electron transport in these materials, with particular emphasis on understanding how structural modifications and defects affect conductivity. Computational modeling and simulation have become essential tools in predicting and optimizing the conductive properties of both materials and their composites.
Lithium hydroxide, primarily known as a critical component in lithium-ion battery production, has been studied since the 1970s for its ionic conductivity properties. Its historical development traces back to early lithium battery research, where scientists discovered its role in facilitating lithium ion transport. The compound exhibits moderate ionic conductivity, which increases significantly when in solution or when incorporated into composite materials.
Graphene, by contrast, represents a revolutionary material first isolated in 2004 by Andre Geim and Konstantin Novoselov, who later received the Nobel Prize for this breakthrough. As a single layer of carbon atoms arranged in a two-dimensional honeycomb lattice, graphene demonstrates exceptional electronic conductivity properties that surpass most known materials, including copper and silver.
The fundamental difference in conductivity mechanisms between these materials stems from their atomic structures. Lithium hydroxide conducts electricity primarily through ionic transport, where lithium ions carry the charge. This process is relatively slower and more dependent on temperature and concentration gradients. Graphene, however, exhibits ballistic electronic conductivity, where electrons move with minimal scattering, resulting in near-superconductor-like performance at room temperature.
Research trends indicate growing interest in hybrid materials that combine the properties of both substances. The integration of lithium hydroxide into graphene-based structures has shown promising results for enhancing battery performance, particularly in terms of charge/discharge rates and cycle stability.
The technological evolution in this field has been marked by significant milestones, including the development of graphene production methods beyond mechanical exfoliation, such as chemical vapor deposition (CVD) and epitaxial growth. Similarly, advances in lithium hydroxide synthesis have focused on producing higher purity grades specifically designed for battery applications.
Current research directions are exploring the fundamental physics of electron transport in these materials, with particular emphasis on understanding how structural modifications and defects affect conductivity. Computational modeling and simulation have become essential tools in predicting and optimizing the conductive properties of both materials and their composites.
Market Applications and Demand Analysis
The market for advanced conductive materials is experiencing significant growth, driven by the expanding electronics, energy storage, and renewable energy sectors. When comparing lithium hydroxide and graphene in terms of conductivity applications, distinct market segments and demand patterns emerge based on their unique properties.
Lithium hydroxide primarily serves the lithium-ion battery market, which is projected to reach $182 billion by 2030, growing at a CAGR of approximately 18%. The electric vehicle industry represents the largest demand driver, with forecasts suggesting EV production will exceed 25 million units annually by 2030. Major automotive manufacturers have committed over $300 billion to electrification strategies, directly impacting lithium hydroxide demand. While lithium hydroxide itself is not valued for conductivity, it serves as a critical precursor for cathode materials that determine battery performance.
Graphene, conversely, targets high-performance electronics and next-generation energy applications due to its exceptional conductivity. The global graphene market, valued at $87 million in 2022, is expected to grow at 38% CAGR through 2030. Demand is particularly strong in semiconductor manufacturing, flexible electronics, and supercapacitors where graphene's conductivity advantages over traditional materials translate to substantial performance improvements.
Regional demand patterns show interesting divergence. Asia Pacific dominates lithium hydroxide consumption with China accounting for 75% of global processing capacity. For graphene, while Asia leads in production volume, North America and Europe show stronger demand growth rates in high-value applications requiring superior conductivity.
Industry surveys indicate that manufacturers are increasingly evaluating conductivity-performance tradeoffs against cost considerations. Lithium hydroxide-based solutions currently command greater market share due to established supply chains and manufacturing processes, despite graphene's superior conductivity properties. The price differential remains significant, with battery-grade lithium hydroxide trading at $15,000-$20,000 per ton while high-quality graphene can cost $100-$200 per gram.
Emerging applications in wearable electronics, medical devices, and aerospace components are creating new demand vectors for highly conductive materials. Market research indicates these sectors value graphene's conductivity advantages more highly than traditional industries, potentially accelerating adoption despite higher costs. Conversely, grid-scale energy storage applications remain firmly in lithium hydroxide's domain due to cost sensitivity and established manufacturing infrastructure.
Consumer electronics manufacturers report increasing interest in graphene-enhanced components, with 62% of surveyed companies exploring graphene integration for next-generation products where conductivity improvements directly enhance user experience or enable new functionalities.
Lithium hydroxide primarily serves the lithium-ion battery market, which is projected to reach $182 billion by 2030, growing at a CAGR of approximately 18%. The electric vehicle industry represents the largest demand driver, with forecasts suggesting EV production will exceed 25 million units annually by 2030. Major automotive manufacturers have committed over $300 billion to electrification strategies, directly impacting lithium hydroxide demand. While lithium hydroxide itself is not valued for conductivity, it serves as a critical precursor for cathode materials that determine battery performance.
Graphene, conversely, targets high-performance electronics and next-generation energy applications due to its exceptional conductivity. The global graphene market, valued at $87 million in 2022, is expected to grow at 38% CAGR through 2030. Demand is particularly strong in semiconductor manufacturing, flexible electronics, and supercapacitors where graphene's conductivity advantages over traditional materials translate to substantial performance improvements.
Regional demand patterns show interesting divergence. Asia Pacific dominates lithium hydroxide consumption with China accounting for 75% of global processing capacity. For graphene, while Asia leads in production volume, North America and Europe show stronger demand growth rates in high-value applications requiring superior conductivity.
Industry surveys indicate that manufacturers are increasingly evaluating conductivity-performance tradeoffs against cost considerations. Lithium hydroxide-based solutions currently command greater market share due to established supply chains and manufacturing processes, despite graphene's superior conductivity properties. The price differential remains significant, with battery-grade lithium hydroxide trading at $15,000-$20,000 per ton while high-quality graphene can cost $100-$200 per gram.
Emerging applications in wearable electronics, medical devices, and aerospace components are creating new demand vectors for highly conductive materials. Market research indicates these sectors value graphene's conductivity advantages more highly than traditional industries, potentially accelerating adoption despite higher costs. Conversely, grid-scale energy storage applications remain firmly in lithium hydroxide's domain due to cost sensitivity and established manufacturing infrastructure.
Consumer electronics manufacturers report increasing interest in graphene-enhanced components, with 62% of surveyed companies exploring graphene integration for next-generation products where conductivity improvements directly enhance user experience or enable new functionalities.
Current Technical Challenges in Conductivity Research
The conductivity research field faces significant challenges when comparing lithium hydroxide and graphene materials. One primary obstacle is the development of standardized measurement protocols for accurately assessing conductivity across different material states. Current methodologies often produce inconsistent results due to variations in sample preparation, environmental conditions, and measurement techniques, making direct comparisons between these materials problematic.
Temperature dependency presents another critical challenge, as both materials exhibit different conductivity behaviors across temperature ranges. While graphene maintains relatively stable conductivity across wider temperature spans, lithium hydroxide shows more dramatic fluctuations, particularly in high-temperature environments. This variability complicates the design of applications requiring consistent performance across diverse operating conditions.
Surface modification effects on conductivity remain poorly understood. Research indicates that even minor surface alterations can significantly impact the conductivity properties of both materials, but quantifying these effects systematically has proven difficult. The scientific community lacks comprehensive models that can predict how specific surface treatments affect conductivity parameters in real-world applications.
Scale-up challenges persist in transitioning from laboratory measurements to industrial applications. Laboratory-scale conductivity measurements often fail to translate accurately to larger production environments, creating significant barriers to commercialization. This discrepancy is particularly pronounced with graphene, where production methods significantly influence conductivity properties.
Interface conductivity between these materials and other components in composite systems represents another frontier challenge. The conductivity behavior at material interfaces often differs substantially from bulk properties, yet current research lacks sophisticated tools to characterize these interface phenomena accurately.
Computational modeling limitations hinder progress in this field. Existing models struggle to accurately simulate the complex quantum and molecular interactions that determine conductivity properties, particularly for graphene's unique two-dimensional structure. The computational resources required for accurate modeling remain prohibitively expensive for many research institutions.
Environmental stability of conductivity properties presents ongoing challenges, with both materials showing sensitivity to humidity, oxidation, and other environmental factors. Graphene's conductivity can degrade through oxidation or contamination, while lithium hydroxide's properties are highly sensitive to moisture exposure, complicating long-term performance predictions in real-world applications.
Temperature dependency presents another critical challenge, as both materials exhibit different conductivity behaviors across temperature ranges. While graphene maintains relatively stable conductivity across wider temperature spans, lithium hydroxide shows more dramatic fluctuations, particularly in high-temperature environments. This variability complicates the design of applications requiring consistent performance across diverse operating conditions.
Surface modification effects on conductivity remain poorly understood. Research indicates that even minor surface alterations can significantly impact the conductivity properties of both materials, but quantifying these effects systematically has proven difficult. The scientific community lacks comprehensive models that can predict how specific surface treatments affect conductivity parameters in real-world applications.
Scale-up challenges persist in transitioning from laboratory measurements to industrial applications. Laboratory-scale conductivity measurements often fail to translate accurately to larger production environments, creating significant barriers to commercialization. This discrepancy is particularly pronounced with graphene, where production methods significantly influence conductivity properties.
Interface conductivity between these materials and other components in composite systems represents another frontier challenge. The conductivity behavior at material interfaces often differs substantially from bulk properties, yet current research lacks sophisticated tools to characterize these interface phenomena accurately.
Computational modeling limitations hinder progress in this field. Existing models struggle to accurately simulate the complex quantum and molecular interactions that determine conductivity properties, particularly for graphene's unique two-dimensional structure. The computational resources required for accurate modeling remain prohibitively expensive for many research institutions.
Environmental stability of conductivity properties presents ongoing challenges, with both materials showing sensitivity to humidity, oxidation, and other environmental factors. Graphene's conductivity can degrade through oxidation or contamination, while lithium hydroxide's properties are highly sensitive to moisture exposure, complicating long-term performance predictions in real-world applications.
Comparative Analysis of Conductivity Mechanisms
01 Lithium hydroxide treatment of graphene for enhanced conductivity
Lithium hydroxide can be used to treat graphene materials to enhance their electrical conductivity. This treatment process involves the interaction of lithium hydroxide with graphene sheets, which can modify the surface properties and electronic structure of graphene. The treatment helps in removing impurities and defects from graphene, resulting in improved electron mobility and conductivity. This approach is particularly useful for applications requiring high-performance conductive materials.- Lithium hydroxide treatment of graphene for enhanced conductivity: Lithium hydroxide can be used to treat graphene materials to enhance their electrical conductivity. This treatment process involves the interaction of lithium hydroxide with graphene sheets, which can modify the surface properties and electronic structure of graphene. The treatment helps in removing impurities and defects from graphene, resulting in improved electron mobility and conductivity. This approach is particularly useful for applications requiring high-performance conductive materials.
- Graphene-lithium hydroxide composites for battery applications: Composites of graphene and lithium hydroxide can be formulated to enhance the performance of lithium-ion batteries. These composites serve as efficient electrode materials with improved conductivity and electrochemical properties. The incorporation of lithium hydroxide into graphene-based structures facilitates lithium-ion transport and enhances the overall battery efficiency. These materials demonstrate superior cycling stability and higher capacity compared to conventional electrode materials.
- Doping graphene with lithium compounds for conductivity modification: Doping graphene with lithium compounds, including lithium hydroxide, can effectively modify its electronic properties and conductivity. The doping process introduces lithium ions into the graphene structure, which alters the charge carrier concentration and mobility. This modification can be controlled to achieve specific conductivity levels for various applications. The doping approach offers a versatile method to tailor graphene's electrical properties for specific technological requirements.
- Synthesis methods for lithium hydroxide-graphene materials: Various synthesis methods have been developed to create lithium hydroxide-graphene materials with enhanced conductivity. These methods include hydrothermal synthesis, chemical vapor deposition, solution-based processing, and electrochemical techniques. Each synthesis approach offers different advantages in terms of controlling the interaction between lithium hydroxide and graphene, which directly affects the conductivity of the resulting material. The synthesis parameters can be optimized to achieve specific conductivity characteristics for targeted applications.
- Applications of lithium hydroxide-treated graphene in energy storage devices: Lithium hydroxide-treated graphene materials find extensive applications in energy storage devices due to their enhanced conductivity. These materials are particularly valuable in supercapacitors, lithium-ion batteries, and other electrochemical energy storage systems. The improved conductivity leads to faster charge/discharge rates, higher energy density, and better overall performance of these devices. The combination of lithium hydroxide and graphene creates advanced materials that address key challenges in modern energy storage technologies.
02 Graphene-lithium hydroxide composites for battery applications
Composites of graphene and lithium hydroxide can be formulated to enhance the performance of lithium-ion batteries. These composites serve as efficient electrode materials with improved conductivity and electrochemical properties. The incorporation of lithium hydroxide into graphene-based structures facilitates lithium-ion transport and enhances the overall battery efficiency. These materials demonstrate higher capacity, better cycling stability, and improved rate capability compared to conventional electrode materials.Expand Specific Solutions03 Doping graphene with lithium compounds for conductivity modification
Doping graphene with lithium compounds, including lithium hydroxide, can effectively modify its electronic properties and conductivity. The doping process introduces lithium ions into the graphene structure, which can alter the charge carrier concentration and mobility. This modification allows for tailoring the conductivity of graphene for specific applications. The controlled doping of graphene with lithium compounds enables the development of materials with customized electronic properties.Expand Specific Solutions04 Synthesis methods for lithium hydroxide-graphene materials
Various synthesis methods have been developed for producing lithium hydroxide-graphene materials with enhanced conductivity. These methods include hydrothermal synthesis, chemical vapor deposition, solution-based processing, and electrochemical techniques. The synthesis parameters significantly influence the structure, morphology, and conductivity of the resulting materials. Optimized synthesis procedures can yield graphene materials with uniform lithium hydroxide distribution and superior conductive properties.Expand Specific Solutions05 Applications of lithium hydroxide-treated graphene in energy storage and electronics
Lithium hydroxide-treated graphene materials find extensive applications in energy storage devices and electronic components due to their enhanced conductivity. These materials are utilized in supercapacitors, lithium-ion batteries, solar cells, and flexible electronics. The improved conductivity of lithium hydroxide-treated graphene contributes to better device performance, including faster charging rates, higher energy density, and extended cycle life. The versatility of these materials makes them valuable for next-generation energy and electronic applications.Expand Specific Solutions
Key Industry Players and Research Institutions
The lithium hydroxide vs graphene conductivity market is in a growth phase, with increasing demand driven by energy storage applications. The global market size for advanced battery materials is expanding rapidly, projected to reach significant volumes by 2030. Technologically, graphene solutions are still emerging while lithium hydroxide has established commercial applications. Leading companies like LG Chem, Toray Industries, and Toshiba are advancing lithium-based technologies, while graphene research is spearheaded by The University of Manchester, International Iberian Nanotechnology Laboratory, and Morion NanoTech. Semiconductor Energy Laboratory and NEC Corp are developing hybrid solutions combining both materials' properties. Companies like Honeycomb Battery and Hefei Guoxuan are commercializing these technologies for energy storage applications, indicating growing market maturity.
The University of Manchester
Technical Solution: The University of Manchester has pioneered groundbreaking research in graphene conductivity since Andre Geim and Konstantin Novoselov first isolated the material in 2004. Their technical approach focuses on developing high-quality graphene sheets with exceptional electrical conductivity (approximately 10^6 S/m) through mechanical exfoliation and chemical vapor deposition (CVD) methods. The university has established comprehensive characterization techniques to measure graphene's conductivity under various conditions, demonstrating that single-layer graphene exhibits ballistic transport of electrons with minimal scattering, allowing electrons to travel submicrometer distances without scattering. Their research has confirmed graphene's superior room-temperature electron mobility (exceeding 200,000 cm²/V·s), significantly outperforming lithium hydroxide's ionic conductivity. The university has also developed functionalization methods to tune graphene's electronic properties while maintaining its high conductivity, enabling applications in flexible electronics, sensors, and energy storage devices.
Strengths: World-leading expertise in graphene research with access to advanced characterization facilities; pioneered fundamental understanding of graphene's electronic properties; established scalable production methods. Weaknesses: Some graphene production techniques remain costly for mass production; challenges in maintaining consistent conductivity across large-area graphene sheets; integration with existing technologies requires further development.
Morion NanoTech
Technical Solution: Morion NanoTech has developed a proprietary technology platform focused on comparing and enhancing the conductivity properties of both lithium hydroxide and graphene materials. Their technical approach involves creating hybrid composite materials that leverage the ionic conductivity of lithium hydroxide (typically 10^-2 to 10^-4 S/cm) with the electronic conductivity of graphene (approximately 10^6 S/m). The company employs advanced nano-engineering techniques to create structured interfaces between these materials, optimizing electron and ion transport pathways. Their research has demonstrated that while lithium hydroxide conducts primarily through ion movement in aqueous solutions, graphene's conductivity relies on delocalized pi-electrons across its 2D lattice. Morion's hybrid materials incorporate lithium hydroxide's ionic properties into graphene-based matrices, creating materials with tailored conductivity profiles for specific applications. Their technology enables precise control of conductivity parameters through structural modifications at the nanoscale, allowing for customization based on application requirements.
Strengths: Innovative approach combining benefits of both materials; customizable conductivity properties for specific applications; potential for breakthrough performance in energy storage devices. Weaknesses: Relatively new technology with limited commercial deployment; challenges in scaling production while maintaining quality; higher cost compared to traditional materials.
Critical Patents and Research Breakthroughs
High capacitance composites
PatentActiveUS20200343054A1
Innovation
- A composite comprising graphene at 65-95% concentration and graphitic carbon nitride coated on mesoporous carbon (mc@g-C3N4) with a binder, combined with a Deep Eutectic Solvent electrolyte, is used to create high energy density EDLCs with improved surface area and stability, enabling fast charge and discharge rates.
A method for improving cycle stability of cathode material for lithium-ion battery
PatentActiveZA202108331A
Innovation
- A method involving the alternating coating of lithium iron phosphate and graphene layers on the surface of cathode materials, with a preferred structure of two lithium iron phosphate layers and one graphene layer, to enhance conductivity and stability, utilizing graphene's high carrier mobility to disperse overpotential.
Environmental Impact and Sustainability Considerations
The environmental footprint of lithium hydroxide and graphene production represents a critical consideration in their industrial application, particularly when evaluating their conductivity properties. Lithium hydroxide extraction primarily relies on mining operations that cause significant land disruption, water consumption, and potential contamination of local ecosystems. The conventional production methods require approximately 500,000 gallons of water per ton of lithium extracted, creating substantial pressure on water resources in often arid mining regions.
Graphene manufacturing, while not dependent on mining, presents different environmental challenges. Chemical vapor deposition (CVD) methods for high-quality graphene production consume considerable energy and often utilize hazardous chemicals like methane and hydrogen. However, recent advancements in graphene production from waste carbon sources offer promising sustainability improvements, potentially reducing carbon footprint by up to 70% compared to traditional methods.
Life cycle assessments reveal that lithium hydroxide's environmental impact extends beyond production to include transportation and processing emissions. The carbon footprint of battery-grade lithium hydroxide is estimated at 5-15 kg CO2 equivalent per kilogram produced, varying significantly based on production method and energy sources. Graphene's carbon footprint, while initially higher during production (approximately 20-30 kg CO2 equivalent per kilogram), may offer lifetime environmental benefits through enhanced conductivity efficiency in applications.
Recycling capabilities represent another sustainability dimension in this comparison. Current lithium recycling technologies recover only 50-60% of lithium from end-of-life products, though emerging hydrometallurgical processes show promise for improving this rate. Graphene presents greater recycling challenges due to its integration into composite materials, with current recovery rates below 30% in most applications.
Water pollution risks differ significantly between these materials. Lithium extraction can release toxic chemicals including hydrochloric acid and petroleum compounds into groundwater systems. Graphene production generally poses lower water contamination risks, though nanoparticle release during manufacturing remains a concern requiring further research on potential aquatic ecosystem impacts.
Looking forward, sustainability innovations are emerging for both materials. Closed-loop lithium extraction systems have demonstrated 90% reductions in water usage, while direct lithium extraction technologies from geothermal brines offer significantly reduced environmental footprints. For graphene, biomass-derived production methods and solvent-free exfoliation techniques represent promising pathways toward more environmentally responsible manufacturing processes with potentially comparable conductivity properties.
Graphene manufacturing, while not dependent on mining, presents different environmental challenges. Chemical vapor deposition (CVD) methods for high-quality graphene production consume considerable energy and often utilize hazardous chemicals like methane and hydrogen. However, recent advancements in graphene production from waste carbon sources offer promising sustainability improvements, potentially reducing carbon footprint by up to 70% compared to traditional methods.
Life cycle assessments reveal that lithium hydroxide's environmental impact extends beyond production to include transportation and processing emissions. The carbon footprint of battery-grade lithium hydroxide is estimated at 5-15 kg CO2 equivalent per kilogram produced, varying significantly based on production method and energy sources. Graphene's carbon footprint, while initially higher during production (approximately 20-30 kg CO2 equivalent per kilogram), may offer lifetime environmental benefits through enhanced conductivity efficiency in applications.
Recycling capabilities represent another sustainability dimension in this comparison. Current lithium recycling technologies recover only 50-60% of lithium from end-of-life products, though emerging hydrometallurgical processes show promise for improving this rate. Graphene presents greater recycling challenges due to its integration into composite materials, with current recovery rates below 30% in most applications.
Water pollution risks differ significantly between these materials. Lithium extraction can release toxic chemicals including hydrochloric acid and petroleum compounds into groundwater systems. Graphene production generally poses lower water contamination risks, though nanoparticle release during manufacturing remains a concern requiring further research on potential aquatic ecosystem impacts.
Looking forward, sustainability innovations are emerging for both materials. Closed-loop lithium extraction systems have demonstrated 90% reductions in water usage, while direct lithium extraction technologies from geothermal brines offer significantly reduced environmental footprints. For graphene, biomass-derived production methods and solvent-free exfoliation techniques represent promising pathways toward more environmentally responsible manufacturing processes with potentially comparable conductivity properties.
Manufacturing Scalability and Cost Analysis
The manufacturing scalability of lithium hydroxide and graphene presents significant contrasts that directly impact their commercial viability in conductive applications. Lithium hydroxide benefits from established industrial production methods, with global capacity exceeding 300,000 metric tons annually. This mature manufacturing ecosystem translates to relatively stable production costs ranging from $7,000 to $15,000 per ton, depending on purity requirements and regional factors.
In contrast, graphene production remains predominantly at pilot or small-batch scale despite significant advancements since its isolation in 2004. Current global production capacity for high-quality graphene is estimated at less than 5,000 tons annually, with considerable variation in quality and consistency across manufacturers.
Cost analysis reveals that graphene production expenses remain substantially higher, with prices ranging from $50,000 to $200,000 per ton for bulk graphene and reaching millions of dollars per ton for high-purity single-layer graphene. These elevated costs stem from energy-intensive production processes, specialized equipment requirements, and rigorous quality control measures necessary to maintain conductivity properties.
The scalability gap manifests in several key dimensions. Lithium hydroxide production can be readily scaled through conventional chemical processing facilities with predictable linear cost increases. Graphene manufacturing faces non-linear scaling challenges, where quality often deteriorates as production volume increases, directly impacting conductivity performance.
Energy consumption metrics further highlight this disparity. Lithium hydroxide production requires approximately 5-8 kWh per kilogram, while graphene production methods such as chemical vapor deposition or electrochemical exfoliation demand 50-200 kWh per kilogram, significantly impacting production economics and environmental footprint.
Recent technological innovations are gradually improving graphene's manufacturing outlook. Roll-to-roll production techniques have demonstrated potential for reducing costs by 40-60%, while solution-based processing methods show promise for large-scale applications. However, these advances have yet to achieve the consistency in electrical conductivity properties required for direct competition with lithium-based solutions in many applications.
Market projections indicate that while lithium hydroxide will maintain its cost advantage through 2030, graphene manufacturing costs could decrease by 30-50% within five years as production technologies mature and economies of scale improve. This trajectory suggests a potential convergence point where graphene's superior conductivity properties might overcome its cost disadvantages in specific high-performance applications.
In contrast, graphene production remains predominantly at pilot or small-batch scale despite significant advancements since its isolation in 2004. Current global production capacity for high-quality graphene is estimated at less than 5,000 tons annually, with considerable variation in quality and consistency across manufacturers.
Cost analysis reveals that graphene production expenses remain substantially higher, with prices ranging from $50,000 to $200,000 per ton for bulk graphene and reaching millions of dollars per ton for high-purity single-layer graphene. These elevated costs stem from energy-intensive production processes, specialized equipment requirements, and rigorous quality control measures necessary to maintain conductivity properties.
The scalability gap manifests in several key dimensions. Lithium hydroxide production can be readily scaled through conventional chemical processing facilities with predictable linear cost increases. Graphene manufacturing faces non-linear scaling challenges, where quality often deteriorates as production volume increases, directly impacting conductivity performance.
Energy consumption metrics further highlight this disparity. Lithium hydroxide production requires approximately 5-8 kWh per kilogram, while graphene production methods such as chemical vapor deposition or electrochemical exfoliation demand 50-200 kWh per kilogram, significantly impacting production economics and environmental footprint.
Recent technological innovations are gradually improving graphene's manufacturing outlook. Roll-to-roll production techniques have demonstrated potential for reducing costs by 40-60%, while solution-based processing methods show promise for large-scale applications. However, these advances have yet to achieve the consistency in electrical conductivity properties required for direct competition with lithium-based solutions in many applications.
Market projections indicate that while lithium hydroxide will maintain its cost advantage through 2030, graphene manufacturing costs could decrease by 30-50% within five years as production technologies mature and economies of scale improve. This trajectory suggests a potential convergence point where graphene's superior conductivity properties might overcome its cost disadvantages in specific high-performance applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!