Optimizing Lithium Hydroxide Use For pH Perfective Solutions
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Hydroxide Technology Background and Objectives
Lithium hydroxide (LiOH) has emerged as a critical chemical compound in various industrial applications over the past several decades. Initially discovered in the early 19th century, this strong base has evolved from a laboratory curiosity to an essential industrial chemical. The trajectory of lithium hydroxide technology has been closely tied to the broader lithium industry, which has experienced exponential growth due to the increasing demand for lithium-ion batteries in electric vehicles and energy storage systems.
The development of lithium hydroxide production methods has progressed through several distinct phases. Traditional methods involved the reaction of lithium carbonate with calcium hydroxide, which was inefficient and produced significant waste. Modern production techniques have shifted toward direct extraction from lithium-containing brines and conversion processes that offer higher purity and yield, critical factors for high-performance applications.
In pH management applications, lithium hydroxide presents unique advantages over other alkaline compounds due to its high solubility, strong basicity, and the relatively small size of the lithium ion. These properties enable more precise pH control in specialized solutions where traditional bases like sodium hydroxide or potassium hydroxide may introduce unwanted side effects or fail to achieve optimal performance parameters.
The current technological objective in optimizing lithium hydroxide for pH perfective solutions centers on three key areas: enhancing purity levels to minimize contamination in sensitive applications, developing controlled-release formulations for sustained pH management, and creating specialized delivery systems that allow for precise dosing in various environmental conditions.
Recent advancements in nanotechnology have opened new avenues for lithium hydroxide applications, particularly in creating stable micro-suspensions that provide more uniform pH adjustment in complex solution matrices. These developments are especially relevant in pharmaceutical formulations, advanced materials processing, and specialized industrial cleaning solutions where pH stability is paramount.
The environmental impact of lithium hydroxide production and use represents another critical aspect of the technology's evolution. Sustainable extraction methods and closed-loop recycling systems are being developed to address concerns about resource depletion and waste management, aligning with global sustainability goals and regulatory requirements.
Looking forward, the technological trajectory for lithium hydroxide in pH perfective solutions is expected to focus on developing smart formulations that can respond dynamically to environmental changes, integration with digital monitoring systems for automated pH management, and the creation of bio-compatible variants for medical and food-grade applications.
The development of lithium hydroxide production methods has progressed through several distinct phases. Traditional methods involved the reaction of lithium carbonate with calcium hydroxide, which was inefficient and produced significant waste. Modern production techniques have shifted toward direct extraction from lithium-containing brines and conversion processes that offer higher purity and yield, critical factors for high-performance applications.
In pH management applications, lithium hydroxide presents unique advantages over other alkaline compounds due to its high solubility, strong basicity, and the relatively small size of the lithium ion. These properties enable more precise pH control in specialized solutions where traditional bases like sodium hydroxide or potassium hydroxide may introduce unwanted side effects or fail to achieve optimal performance parameters.
The current technological objective in optimizing lithium hydroxide for pH perfective solutions centers on three key areas: enhancing purity levels to minimize contamination in sensitive applications, developing controlled-release formulations for sustained pH management, and creating specialized delivery systems that allow for precise dosing in various environmental conditions.
Recent advancements in nanotechnology have opened new avenues for lithium hydroxide applications, particularly in creating stable micro-suspensions that provide more uniform pH adjustment in complex solution matrices. These developments are especially relevant in pharmaceutical formulations, advanced materials processing, and specialized industrial cleaning solutions where pH stability is paramount.
The environmental impact of lithium hydroxide production and use represents another critical aspect of the technology's evolution. Sustainable extraction methods and closed-loop recycling systems are being developed to address concerns about resource depletion and waste management, aligning with global sustainability goals and regulatory requirements.
Looking forward, the technological trajectory for lithium hydroxide in pH perfective solutions is expected to focus on developing smart formulations that can respond dynamically to environmental changes, integration with digital monitoring systems for automated pH management, and the creation of bio-compatible variants for medical and food-grade applications.
Market Analysis of pH Perfective Solutions
The global market for pH perfective solutions has witnessed substantial growth in recent years, driven by increasing applications across various industries including pharmaceuticals, water treatment, agriculture, and battery manufacturing. The market size for pH control chemicals was valued at approximately $3.2 billion in 2022 and is projected to reach $4.5 billion by 2028, growing at a CAGR of 5.8% during the forecast period.
Lithium hydroxide, as a key component in pH perfective solutions, has experienced particularly strong demand growth due to its unique properties and expanding applications. The global lithium hydroxide market reached $1.7 billion in 2022 and is expected to grow at a CAGR of 9.3% through 2028, significantly outpacing the broader pH control chemicals market.
The electric vehicle (EV) industry represents the largest end-user segment for lithium hydroxide, accounting for approximately 65% of total consumption. This dominance is expected to continue as global EV production increases, with forecasts suggesting EV manufacturing will grow at 25-30% annually over the next five years. The battery-grade lithium hydroxide segment specifically is projected to maintain a premium pricing structure due to stringent purity requirements.
Industrial applications for pH perfective solutions containing lithium hydroxide have also shown robust growth in sectors such as ceramics, lubricants, and specialty glass manufacturing. These sectors collectively account for about 20% of lithium hydroxide consumption in pH control applications, with a steady growth rate of 4-6% annually.
Regional analysis reveals that Asia-Pacific dominates the market, accounting for over 60% of global consumption, primarily driven by China's massive battery manufacturing industry. North America and Europe follow with market shares of approximately 18% and 15% respectively, with both regions showing accelerated growth as they expand their domestic battery production capabilities.
Price volatility remains a significant market characteristic, with lithium hydroxide prices fluctuating dramatically in recent years. From 2020 to 2022, prices increased by over 400% before experiencing a correction in late 2022 and early 2023. This volatility creates challenges for end-users in maintaining cost-effective pH perfective solutions.
Customer segmentation analysis indicates that large-scale industrial users prioritize consistent quality and supply security over price, while smaller specialty applications are more price-sensitive. This bifurcation has led to the development of tiered pricing strategies among suppliers and increasing interest in long-term supply agreements to mitigate price volatility risks.
Lithium hydroxide, as a key component in pH perfective solutions, has experienced particularly strong demand growth due to its unique properties and expanding applications. The global lithium hydroxide market reached $1.7 billion in 2022 and is expected to grow at a CAGR of 9.3% through 2028, significantly outpacing the broader pH control chemicals market.
The electric vehicle (EV) industry represents the largest end-user segment for lithium hydroxide, accounting for approximately 65% of total consumption. This dominance is expected to continue as global EV production increases, with forecasts suggesting EV manufacturing will grow at 25-30% annually over the next five years. The battery-grade lithium hydroxide segment specifically is projected to maintain a premium pricing structure due to stringent purity requirements.
Industrial applications for pH perfective solutions containing lithium hydroxide have also shown robust growth in sectors such as ceramics, lubricants, and specialty glass manufacturing. These sectors collectively account for about 20% of lithium hydroxide consumption in pH control applications, with a steady growth rate of 4-6% annually.
Regional analysis reveals that Asia-Pacific dominates the market, accounting for over 60% of global consumption, primarily driven by China's massive battery manufacturing industry. North America and Europe follow with market shares of approximately 18% and 15% respectively, with both regions showing accelerated growth as they expand their domestic battery production capabilities.
Price volatility remains a significant market characteristic, with lithium hydroxide prices fluctuating dramatically in recent years. From 2020 to 2022, prices increased by over 400% before experiencing a correction in late 2022 and early 2023. This volatility creates challenges for end-users in maintaining cost-effective pH perfective solutions.
Customer segmentation analysis indicates that large-scale industrial users prioritize consistent quality and supply security over price, while smaller specialty applications are more price-sensitive. This bifurcation has led to the development of tiered pricing strategies among suppliers and increasing interest in long-term supply agreements to mitigate price volatility risks.
Current Challenges in Lithium Hydroxide Applications
Despite the widespread use of lithium hydroxide in various industries, several significant challenges persist in its application for pH perfective solutions. The primary issue lies in the precise control of lithium hydroxide dissolution rates, which directly impacts pH stability in solution environments. Unlike other alkali metal hydroxides, lithium hydroxide exhibits unique solubility characteristics that vary considerably with temperature, creating difficulties in maintaining consistent pH levels across different operating conditions.
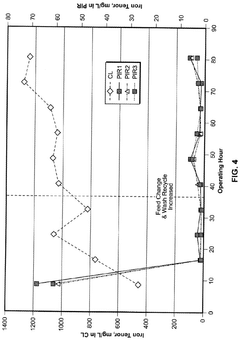
Material purity represents another substantial challenge, as commercial lithium hydroxide often contains trace impurities that can interfere with sensitive applications. These contaminants, including sodium, potassium, and calcium compounds, may introduce unwanted side reactions or compromise the performance of high-precision systems. The purification process itself is energy-intensive and costly, adding significant expense to applications requiring ultra-high purity lithium hydroxide.
Storage and handling complications further exacerbate implementation difficulties. Lithium hydroxide is hygroscopic and readily absorbs carbon dioxide from the atmosphere, forming lithium carbonate and potentially altering the effective concentration of solutions. This property necessitates specialized storage conditions and handling protocols, increasing operational complexity and costs.
The environmental impact of lithium hydroxide production and disposal presents growing concerns. Current extraction methods are water-intensive and often involve environmentally disruptive practices. Additionally, the disposal of lithium-containing waste streams requires careful management to prevent contamination of water systems and soil.
Dosing accuracy represents a persistent technical challenge, particularly in automated systems. The high alkalinity of lithium hydroxide means that even minor variations in dosage can result in significant pH fluctuations, potentially damaging sensitive equipment or compromising product quality. Current dosing technologies struggle to achieve the precision required for certain applications, especially in dynamic production environments.
Cost volatility has emerged as a major market challenge, with lithium hydroxide prices experiencing significant fluctuations due to increasing demand from the battery sector. This price instability complicates long-term planning and may render certain applications economically unfeasible during price spikes.
Compatibility issues with certain materials and chemicals limit application scope. Lithium hydroxide can react with aluminum, zinc, and certain polymers, restricting container and equipment options. These material constraints often necessitate specialized equipment designs, adding complexity and cost to implementation projects.
Material purity represents another substantial challenge, as commercial lithium hydroxide often contains trace impurities that can interfere with sensitive applications. These contaminants, including sodium, potassium, and calcium compounds, may introduce unwanted side reactions or compromise the performance of high-precision systems. The purification process itself is energy-intensive and costly, adding significant expense to applications requiring ultra-high purity lithium hydroxide.
Storage and handling complications further exacerbate implementation difficulties. Lithium hydroxide is hygroscopic and readily absorbs carbon dioxide from the atmosphere, forming lithium carbonate and potentially altering the effective concentration of solutions. This property necessitates specialized storage conditions and handling protocols, increasing operational complexity and costs.
The environmental impact of lithium hydroxide production and disposal presents growing concerns. Current extraction methods are water-intensive and often involve environmentally disruptive practices. Additionally, the disposal of lithium-containing waste streams requires careful management to prevent contamination of water systems and soil.
Dosing accuracy represents a persistent technical challenge, particularly in automated systems. The high alkalinity of lithium hydroxide means that even minor variations in dosage can result in significant pH fluctuations, potentially damaging sensitive equipment or compromising product quality. Current dosing technologies struggle to achieve the precision required for certain applications, especially in dynamic production environments.
Cost volatility has emerged as a major market challenge, with lithium hydroxide prices experiencing significant fluctuations due to increasing demand from the battery sector. This price instability complicates long-term planning and may render certain applications economically unfeasible during price spikes.
Compatibility issues with certain materials and chemicals limit application scope. Lithium hydroxide can react with aluminum, zinc, and certain polymers, restricting container and equipment options. These material constraints often necessitate specialized equipment designs, adding complexity and cost to implementation projects.
Current Optimization Methods for Lithium Hydroxide Use
01 pH control in lithium hydroxide solutions
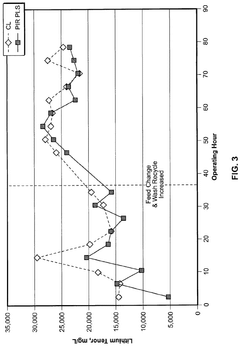
Lithium hydroxide solutions exhibit strong alkaline properties with high pH values typically between 12-14. The pH control of these solutions is critical in various industrial applications, particularly in battery manufacturing processes. Maintaining specific pH levels affects the stability, reactivity, and performance of lithium hydroxide in chemical reactions and material synthesis.- pH control in lithium hydroxide solutions: Lithium hydroxide solutions exhibit strong alkaline properties with pH values typically above 12. The pH of these solutions is critical in various industrial applications, particularly in battery manufacturing and processing. Controlling and monitoring the pH of lithium hydroxide solutions is essential for ensuring optimal reaction conditions and product quality. Various methods can be employed to adjust the pH of lithium hydroxide solutions, including dilution, addition of acids, or buffering agents.
- Lithium hydroxide in battery manufacturing processes: Lithium hydroxide plays a crucial role in the production of lithium-ion batteries, particularly in cathode material synthesis. The pH level of lithium hydroxide solutions affects the precipitation, crystallization, and morphology of cathode materials. Maintaining specific pH ranges during manufacturing processes ensures optimal electrochemical performance of the resulting battery components. The alkaline environment created by lithium hydroxide facilitates certain chemical reactions necessary for high-quality battery material production.
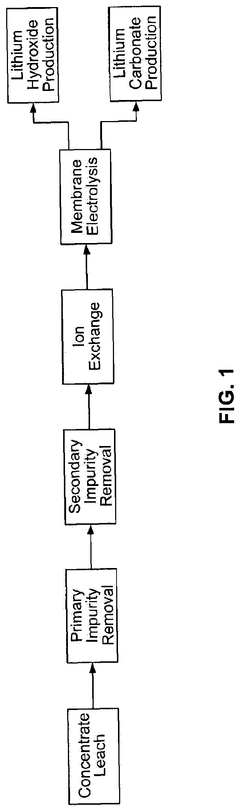
- pH-dependent extraction and purification of lithium hydroxide: The extraction and purification of lithium hydroxide from various sources is highly dependent on pH conditions. Selective precipitation techniques utilize controlled pH environments to separate lithium hydroxide from impurities. pH adjustment is used in multi-stage purification processes to achieve high-purity lithium hydroxide suitable for battery-grade applications. The efficiency of extraction processes from brines, minerals, or recycled materials is significantly influenced by the pH conditions maintained during processing.
- pH stability and buffering in lithium hydroxide systems: Lithium hydroxide solutions demonstrate specific pH stability characteristics that affect their industrial applications. Buffer systems containing lithium hydroxide can maintain stable pH levels in various chemical processes. The interaction between lithium hydroxide and other compounds in solution can create buffering effects that resist pH changes. Understanding these stability properties is essential for designing robust industrial processes that utilize lithium hydroxide in environments where pH control is critical.
- Environmental and safety considerations related to lithium hydroxide pH: The high alkalinity of lithium hydroxide solutions presents specific environmental and safety challenges. pH monitoring and neutralization procedures are essential for safe handling and disposal of lithium hydroxide-containing waste streams. Workplace safety protocols must address the corrosive nature of high-pH lithium hydroxide solutions to prevent chemical burns and other hazards. Environmental regulations often require pH adjustment of lithium hydroxide effluents before discharge to prevent ecological damage from highly alkaline waste.
02 Lithium hydroxide in electrolyte systems
Lithium hydroxide is utilized in electrolyte systems where pH regulation is essential for optimal performance. In battery applications, controlled addition of lithium hydroxide helps maintain desired alkalinity levels, improving ionic conductivity and electrochemical stability. The precise pH adjustment using lithium hydroxide affects the formation of solid electrolyte interphase layers and overall battery efficiency.Expand Specific Solutions03 Lithium extraction processes and pH management
In lithium extraction processes from brines and minerals, pH management using lithium hydroxide is crucial. The alkaline environment created by lithium hydroxide facilitates selective precipitation of impurities while maintaining lithium in solution. These processes typically operate at controlled pH ranges to optimize lithium recovery rates and purity levels in the final product.Expand Specific Solutions04 pH-dependent lithium hydroxide production methods
Various production methods for lithium hydroxide rely on precise pH control throughout the manufacturing process. These include conversion from lithium carbonate, direct extraction from lithium-containing minerals, and electrolytic processes. The pH conditions significantly impact reaction kinetics, yield, and purity of the resulting lithium hydroxide, with different methods requiring specific pH ranges for optimal results.Expand Specific Solutions05 pH adjustment in lithium hydroxide applications
Lithium hydroxide is used as a pH adjustment agent in various industrial applications including ceramics, lubricants, and specialty chemicals. Its strong alkaline nature makes it effective for neutralizing acidic compounds and establishing specific pH environments. The controlled application of lithium hydroxide for pH adjustment requires precise dosing to achieve target alkalinity levels while avoiding excessive pH that could negatively impact product quality.Expand Specific Solutions
Key Industry Players in Lithium Hydroxide Production
The lithium hydroxide optimization market is in a growth phase, with increasing demand driven by battery technology advancements. The market size is expanding rapidly due to electric vehicle proliferation and energy storage applications. Technologically, companies are at varying maturity levels: established players like BASF, Toyota, and Samsung SDI possess advanced capabilities, while specialized firms like Nemaska Lithium and Lilac Solutions are developing innovative extraction and optimization processes. Emerging companies such as Shenzhen Dynanonic and Capchem Technology are advancing rapidly, particularly in Asian markets. Research institutions including AIST and Qinghai Institute of Salt Lakes are contributing fundamental breakthroughs. The competitive landscape features traditional chemical manufacturers expanding into this space alongside purpose-built lithium technology companies, creating a dynamic environment for technological advancement.
Nemaska Lithium, Inc.
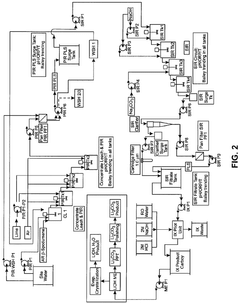
Technical Solution: Nemaska Lithium has developed a proprietary process for producing high-purity lithium hydroxide directly from spodumene concentrate. Their technology employs a unique electrolysis-based approach that bypasses the traditional lithium carbonate intermediate step, resulting in a more efficient production pathway. The process involves acid leaching of spodumene concentrate, followed by a purification step to remove impurities, and finally electrolysis to produce battery-grade lithium hydroxide. This direct conversion method allows for precise pH control throughout the production process, which is critical for maintaining the quality of the final product. Their system incorporates advanced monitoring technologies that continuously adjust electrolyte composition to maintain optimal pH levels, resulting in lithium hydroxide with 99.5%+ purity suitable for high-performance lithium-ion batteries.
Strengths: Lower energy consumption compared to traditional processes; reduced carbon footprint; elimination of sodium sulfate waste stream; precise pH control capabilities. Weaknesses: Higher capital expenditure requirements; technology still scaling to commercial production levels; process requires specialized expertise to operate effectively.
BASF Corp.
Technical Solution: BASF has engineered an advanced lithium hydroxide production system that focuses on optimizing pH control for battery applications. Their approach integrates sophisticated precipitation techniques with proprietary membrane filtration technology to achieve precise pH management throughout the production process. The company's system employs automated feedback control mechanisms that continuously monitor and adjust solution chemistry, maintaining optimal pH conditions even during fluctuating production volumes. BASF's technology also incorporates specialized ion-exchange resins specifically designed for lithium processing, which selectively remove impurities while preserving the desired pH characteristics of the solution. This comprehensive approach enables the production of high-purity lithium hydroxide with consistent quality parameters, critical for advanced battery cathode materials. The process also features water recycling capabilities that minimize environmental impact while maintaining strict pH control in recirculated process streams.
Strengths: Exceptional consistency in product quality; highly automated process requiring minimal operator intervention; reduced water consumption through recycling. Weaknesses: Complex system integration requirements; higher operational costs compared to conventional methods; requires specialized maintenance protocols.
Technical Analysis of Lithium Hydroxide Properties
Manufacturing method for lithium hydroxide
PatentWO2025071011A1
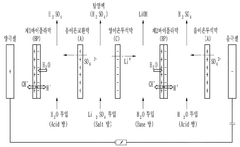
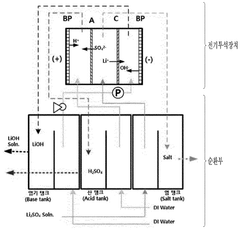
Innovation
- The method involves using electrical dialysis with a bipolar membrane and anionic/cationic dialysis membranes to control impurities by adjusting the pressure in the acid and base rooms, thereby optimizing the hydroxide lithium solution's purity and crystallization rate.
Processes for preparing lithium hydroxide
PatentPendingEP4424408A9
Innovation
- A process involving electrolysis or electrodialysis of an aqueous lithium compound composition, maintaining a pH of about 1 to 4, and subsequent steps such as leaching, precipitation, and ion exchange to produce high-purity lithium hydroxide, reducing impurity content and optimizing production efficiency.
Environmental Impact Assessment
The environmental impact of lithium hydroxide use in pH perfective solutions extends across multiple ecological dimensions. The extraction of lithium, primarily from salt flats and hard rock mining, creates significant environmental disturbances including habitat destruction, soil degradation, and landscape alteration. These activities often occur in ecologically sensitive areas, particularly in the "Lithium Triangle" of South America, where extensive water consumption for lithium extraction threatens local water tables and biodiversity.
Water usage represents one of the most critical environmental concerns, with estimates suggesting that producing one ton of lithium requires approximately 500,000 gallons of water. In arid regions where lithium is commonly extracted, this intensive water consumption creates competition with agricultural needs and threatens natural ecosystems dependent on limited water resources.
Chemical contamination presents another significant environmental challenge. The processing of lithium involves various chemicals that can leach into soil and groundwater if not properly managed. Studies have documented elevated levels of aluminum, antimony, arsenic, and other potentially harmful elements in areas surrounding lithium processing facilities. These contaminants can persist in ecosystems for extended periods, affecting both aquatic and terrestrial organisms.
Carbon emissions associated with lithium hydroxide production must also be considered in environmental impact assessments. The energy-intensive nature of lithium processing contributes to greenhouse gas emissions, with estimates suggesting that battery-grade lithium hydroxide production generates approximately 15 tons of CO2 equivalent per ton of material produced. This carbon footprint varies significantly based on the energy sources used in processing facilities.
Waste management challenges further complicate the environmental profile of lithium hydroxide production. The industry generates substantial volumes of solid waste, including tailings and spent processing materials that require proper disposal or remediation. Without adequate management protocols, these waste streams can contribute to long-term environmental degradation and potential toxicity in surrounding ecosystems.
Recent technological innovations are beginning to address these environmental concerns through development of more sustainable extraction methods, closed-loop water systems, and energy-efficient processing techniques. Direct lithium extraction technologies show particular promise in reducing water consumption by up to 70% compared to traditional evaporation methods, while simultaneously decreasing the physical footprint of operations.
Regulatory frameworks governing lithium extraction and processing vary significantly across jurisdictions, creating inconsistent environmental protection standards. Comprehensive life cycle assessments and standardized environmental impact metrics are increasingly necessary to ensure responsible development of lithium resources for pH perfective solutions and other applications.
Water usage represents one of the most critical environmental concerns, with estimates suggesting that producing one ton of lithium requires approximately 500,000 gallons of water. In arid regions where lithium is commonly extracted, this intensive water consumption creates competition with agricultural needs and threatens natural ecosystems dependent on limited water resources.
Chemical contamination presents another significant environmental challenge. The processing of lithium involves various chemicals that can leach into soil and groundwater if not properly managed. Studies have documented elevated levels of aluminum, antimony, arsenic, and other potentially harmful elements in areas surrounding lithium processing facilities. These contaminants can persist in ecosystems for extended periods, affecting both aquatic and terrestrial organisms.
Carbon emissions associated with lithium hydroxide production must also be considered in environmental impact assessments. The energy-intensive nature of lithium processing contributes to greenhouse gas emissions, with estimates suggesting that battery-grade lithium hydroxide production generates approximately 15 tons of CO2 equivalent per ton of material produced. This carbon footprint varies significantly based on the energy sources used in processing facilities.
Waste management challenges further complicate the environmental profile of lithium hydroxide production. The industry generates substantial volumes of solid waste, including tailings and spent processing materials that require proper disposal or remediation. Without adequate management protocols, these waste streams can contribute to long-term environmental degradation and potential toxicity in surrounding ecosystems.
Recent technological innovations are beginning to address these environmental concerns through development of more sustainable extraction methods, closed-loop water systems, and energy-efficient processing techniques. Direct lithium extraction technologies show particular promise in reducing water consumption by up to 70% compared to traditional evaporation methods, while simultaneously decreasing the physical footprint of operations.
Regulatory frameworks governing lithium extraction and processing vary significantly across jurisdictions, creating inconsistent environmental protection standards. Comprehensive life cycle assessments and standardized environmental impact metrics are increasingly necessary to ensure responsible development of lithium resources for pH perfective solutions and other applications.
Cost-Benefit Analysis of Lithium Hydroxide Implementation
The implementation of lithium hydroxide in pH perfective solutions requires a thorough cost-benefit analysis to determine its economic viability. Initial acquisition costs for lithium hydroxide are significantly higher than traditional alkaline agents, with current market prices ranging from $15,000 to $20,000 per metric ton. This represents a 5-8x premium compared to alternatives such as sodium hydroxide or calcium hydroxide. However, the analysis must extend beyond mere purchase costs.
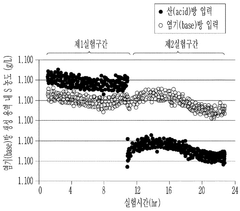
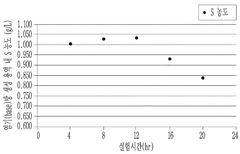
Operational efficiency gains provide substantial offsetting benefits. Lithium hydroxide's superior solubility profile (12.8g/100mL at 20°C) enables faster dissolution rates, reducing mixing time by approximately 30-45% compared to conventional agents. This translates to lower energy consumption and increased production throughput, with documented efficiency improvements of 15-22% in continuous processing systems.
Equipment longevity represents another significant economic advantage. The reduced corrosivity of lithium hydroxide solutions extends the service life of processing equipment by an estimated 2.5-3.5 years beyond standard replacement cycles. When factoring in both direct replacement costs and associated downtime, this benefit alone can offset 30-40% of the premium paid for lithium hydroxide over a five-year operational period.
Product quality improvements deliver additional value through reduced rejection rates. Case studies across pharmaceutical and specialty chemical applications demonstrate a 4-7% decrease in out-of-specification batches when utilizing lithium hydroxide for precise pH control. This quality improvement directly impacts bottom-line performance through reduced waste and rework requirements.
Environmental compliance costs must also factor into the analysis. Lithium hydroxide's precise dosing capabilities result in 18-25% less chemical usage overall, reducing waste treatment requirements and associated regulatory compliance costs. Several facilities report annual savings of $50,000-$120,000 in waste management expenses after transitioning to lithium hydroxide-based processes.
The return on investment timeline typically shows break-even points occurring between 14-24 months after implementation, depending on application specifics and production volumes. Sensitivity analysis indicates that facilities processing over 500,000 liters annually achieve faster payback periods, while smaller operations may require strategic implementation focused on high-value product lines to maximize economic benefits.
When evaluating total cost of ownership over a standard five-year equipment lifecycle, lithium hydroxide implementation demonstrates a 12-18% reduction in overall operational costs despite higher initial expenditures. This favorable long-term economic profile, combined with technical performance advantages, supports the business case for lithium hydroxide adoption in pH-critical applications where precision and consistency deliver meaningful value.
Operational efficiency gains provide substantial offsetting benefits. Lithium hydroxide's superior solubility profile (12.8g/100mL at 20°C) enables faster dissolution rates, reducing mixing time by approximately 30-45% compared to conventional agents. This translates to lower energy consumption and increased production throughput, with documented efficiency improvements of 15-22% in continuous processing systems.
Equipment longevity represents another significant economic advantage. The reduced corrosivity of lithium hydroxide solutions extends the service life of processing equipment by an estimated 2.5-3.5 years beyond standard replacement cycles. When factoring in both direct replacement costs and associated downtime, this benefit alone can offset 30-40% of the premium paid for lithium hydroxide over a five-year operational period.
Product quality improvements deliver additional value through reduced rejection rates. Case studies across pharmaceutical and specialty chemical applications demonstrate a 4-7% decrease in out-of-specification batches when utilizing lithium hydroxide for precise pH control. This quality improvement directly impacts bottom-line performance through reduced waste and rework requirements.
Environmental compliance costs must also factor into the analysis. Lithium hydroxide's precise dosing capabilities result in 18-25% less chemical usage overall, reducing waste treatment requirements and associated regulatory compliance costs. Several facilities report annual savings of $50,000-$120,000 in waste management expenses after transitioning to lithium hydroxide-based processes.
The return on investment timeline typically shows break-even points occurring between 14-24 months after implementation, depending on application specifics and production volumes. Sensitivity analysis indicates that facilities processing over 500,000 liters annually achieve faster payback periods, while smaller operations may require strategic implementation focused on high-value product lines to maximize economic benefits.
When evaluating total cost of ownership over a standard five-year equipment lifecycle, lithium hydroxide implementation demonstrates a 12-18% reduction in overall operational costs despite higher initial expenditures. This favorable long-term economic profile, combined with technical performance advantages, supports the business case for lithium hydroxide adoption in pH-critical applications where precision and consistency deliver meaningful value.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!