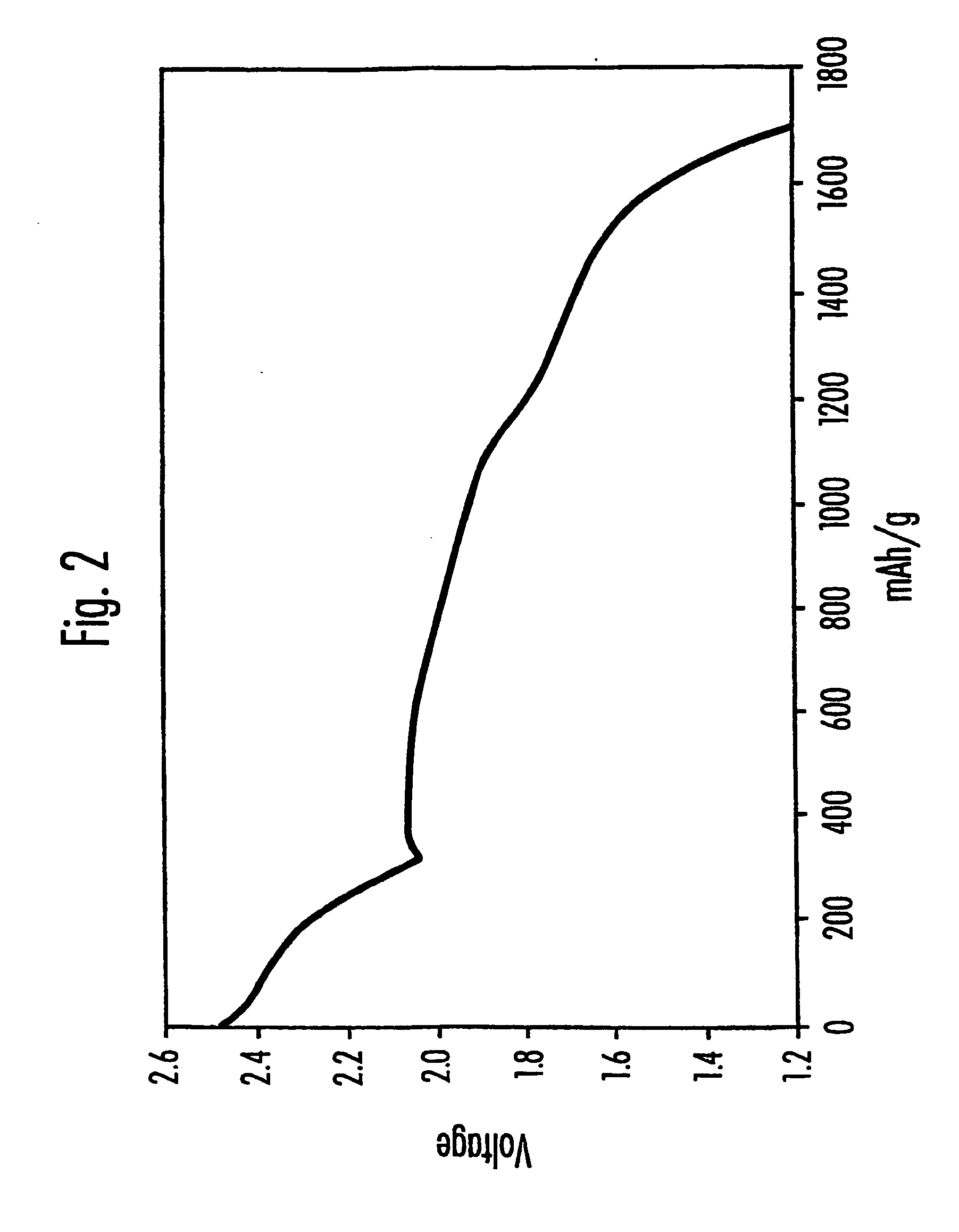
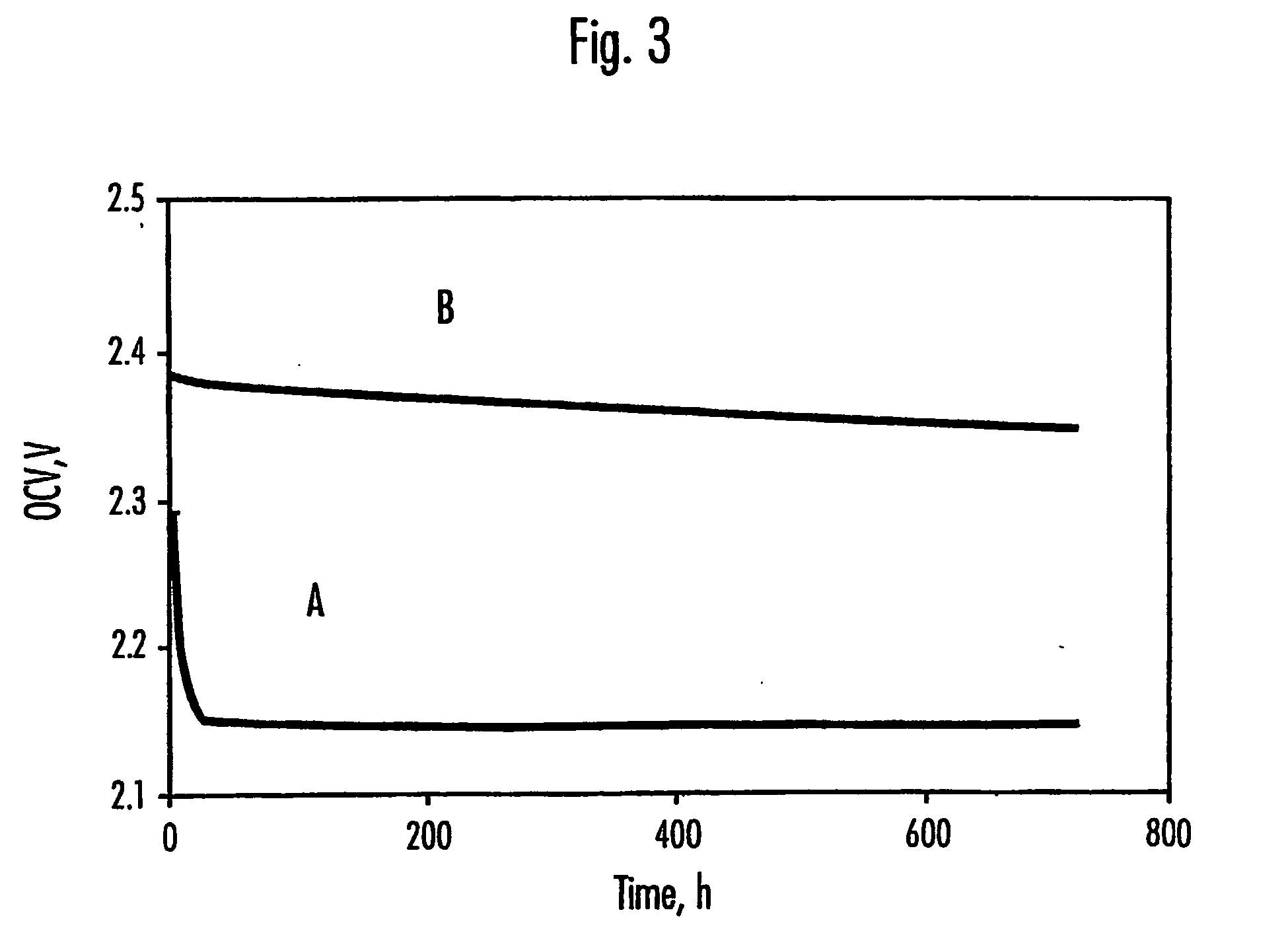
Sulfur utilization efficiency in lithium-sulfur full cells
OCT 14, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium-Sulfur Battery Technology Background and Objectives
Lithium-sulfur (Li-S) batteries have emerged as a promising next-generation energy storage technology due to their theoretical energy density of 2600 Wh/kg, which significantly surpasses that of conventional lithium-ion batteries (typically 100-265 Wh/kg). This remarkable potential stems from sulfur's high theoretical capacity of 1675 mAh/g and its natural abundance, making it both technically attractive and economically viable as a cathode material.
The development of Li-S battery technology can be traced back to the 1960s when the first conceptual designs were proposed. However, significant research momentum only began building in the early 2000s as limitations of traditional lithium-ion chemistries became apparent for high-energy applications. The past decade has witnessed exponential growth in Li-S research publications and patents, reflecting the global recognition of its potential to revolutionize energy storage.
Despite the promising theoretical performance, practical Li-S batteries face several critical challenges that have hindered their commercialization. Chief among these is the low sulfur utilization efficiency in full cells, which drastically reduces the practical energy density to far below theoretical values. This efficiency bottleneck stems from multiple interconnected issues including the insulating nature of sulfur, the formation of soluble lithium polysulfides leading to the "shuttle effect," and significant volume changes during cycling.
Current technological trends focus on addressing these challenges through innovative materials engineering and cell design. Researchers are exploring nanostructured carbon hosts, functional separators, electrolyte additives, and novel cell architectures to enhance sulfur utilization and mitigate capacity fading mechanisms. The evolution of these approaches demonstrates a shift from single-component optimization to integrated system design thinking.
The primary technical objectives in this field include achieving sulfur utilization efficiency exceeding 80% in practical full cells, extending cycle life beyond 1000 cycles with minimal capacity degradation, and developing scalable manufacturing processes compatible with existing battery production infrastructure. Additionally, there is growing emphasis on ensuring the safety and environmental sustainability of Li-S technology throughout its lifecycle.
Looking forward, the technology roadmap for Li-S batteries envisions gradual implementation in niche applications such as aerospace and specialized portable electronics by 2025, followed by broader adoption in electric vehicles and grid storage by 2030-2035. This progression depends critically on overcoming the sulfur utilization efficiency challenge, which represents both the greatest obstacle and opportunity in realizing the full potential of Li-S battery technology.
The development of Li-S battery technology can be traced back to the 1960s when the first conceptual designs were proposed. However, significant research momentum only began building in the early 2000s as limitations of traditional lithium-ion chemistries became apparent for high-energy applications. The past decade has witnessed exponential growth in Li-S research publications and patents, reflecting the global recognition of its potential to revolutionize energy storage.
Despite the promising theoretical performance, practical Li-S batteries face several critical challenges that have hindered their commercialization. Chief among these is the low sulfur utilization efficiency in full cells, which drastically reduces the practical energy density to far below theoretical values. This efficiency bottleneck stems from multiple interconnected issues including the insulating nature of sulfur, the formation of soluble lithium polysulfides leading to the "shuttle effect," and significant volume changes during cycling.
Current technological trends focus on addressing these challenges through innovative materials engineering and cell design. Researchers are exploring nanostructured carbon hosts, functional separators, electrolyte additives, and novel cell architectures to enhance sulfur utilization and mitigate capacity fading mechanisms. The evolution of these approaches demonstrates a shift from single-component optimization to integrated system design thinking.
The primary technical objectives in this field include achieving sulfur utilization efficiency exceeding 80% in practical full cells, extending cycle life beyond 1000 cycles with minimal capacity degradation, and developing scalable manufacturing processes compatible with existing battery production infrastructure. Additionally, there is growing emphasis on ensuring the safety and environmental sustainability of Li-S technology throughout its lifecycle.
Looking forward, the technology roadmap for Li-S batteries envisions gradual implementation in niche applications such as aerospace and specialized portable electronics by 2025, followed by broader adoption in electric vehicles and grid storage by 2030-2035. This progression depends critically on overcoming the sulfur utilization efficiency challenge, which represents both the greatest obstacle and opportunity in realizing the full potential of Li-S battery technology.
Market Analysis for High Energy Density Battery Systems
The high energy density battery market is experiencing unprecedented growth, driven by the expanding electric vehicle (EV) sector, portable electronics, and renewable energy storage systems. Current projections indicate the global high energy density battery market will reach approximately $140 billion by 2030, with a compound annual growth rate exceeding 18% from 2023 to 2030. This remarkable growth trajectory is primarily fueled by stringent environmental regulations, government incentives for clean energy adoption, and increasing consumer demand for longer-lasting portable power solutions.
Lithium-sulfur (Li-S) batteries represent a particularly promising segment within this market due to their theoretical energy density of 2600 Wh/kg, significantly surpassing the 250-300 Wh/kg limit of conventional lithium-ion batteries. This substantial performance advantage positions Li-S technology as a potential game-changer for applications requiring exceptional energy density, including aerospace, defense, and next-generation EVs with extended range capabilities.
Market analysis reveals that the current commercial battery landscape remains dominated by lithium-ion technologies, which command approximately 85% market share. However, industry forecasts suggest Li-S batteries could capture 5-7% of the high-performance battery market by 2028, with accelerated adoption contingent upon overcoming key technical challenges—particularly sulfur utilization efficiency in full cells.
Consumer demand patterns indicate growing preference for batteries offering longer operational times between charges, smaller form factors, and reduced weight. These market requirements align perfectly with the theoretical advantages of Li-S systems, creating a significant commercial opportunity for manufacturers who can successfully address the sulfur utilization efficiency challenge.
Regional market assessment shows Asia-Pacific leading battery manufacturing and innovation, with China, South Korea, and Japan collectively accounting for over 65% of global high-energy battery production. However, substantial R&D investments in North America and Europe are rapidly closing this gap, with particular focus on next-generation technologies like Li-S systems.
Industry stakeholders have identified several key market barriers to Li-S commercialization, including production scalability, cycle life limitations, and cost competitiveness against established technologies. Market research indicates that achieving sulfur utilization efficiency above 70% in commercial-scale Li-S full cells would represent a critical threshold for market viability, potentially unlocking applications worth $25 billion annually by 2032.
Consumer willingness-to-pay analysis demonstrates that premium segments in automotive, aerospace, and specialty electronics markets would readily adopt Li-S technology at a 15-20% price premium over conventional solutions if the promised performance advantages can be reliably delivered in commercial products.
Lithium-sulfur (Li-S) batteries represent a particularly promising segment within this market due to their theoretical energy density of 2600 Wh/kg, significantly surpassing the 250-300 Wh/kg limit of conventional lithium-ion batteries. This substantial performance advantage positions Li-S technology as a potential game-changer for applications requiring exceptional energy density, including aerospace, defense, and next-generation EVs with extended range capabilities.
Market analysis reveals that the current commercial battery landscape remains dominated by lithium-ion technologies, which command approximately 85% market share. However, industry forecasts suggest Li-S batteries could capture 5-7% of the high-performance battery market by 2028, with accelerated adoption contingent upon overcoming key technical challenges—particularly sulfur utilization efficiency in full cells.
Consumer demand patterns indicate growing preference for batteries offering longer operational times between charges, smaller form factors, and reduced weight. These market requirements align perfectly with the theoretical advantages of Li-S systems, creating a significant commercial opportunity for manufacturers who can successfully address the sulfur utilization efficiency challenge.
Regional market assessment shows Asia-Pacific leading battery manufacturing and innovation, with China, South Korea, and Japan collectively accounting for over 65% of global high-energy battery production. However, substantial R&D investments in North America and Europe are rapidly closing this gap, with particular focus on next-generation technologies like Li-S systems.
Industry stakeholders have identified several key market barriers to Li-S commercialization, including production scalability, cycle life limitations, and cost competitiveness against established technologies. Market research indicates that achieving sulfur utilization efficiency above 70% in commercial-scale Li-S full cells would represent a critical threshold for market viability, potentially unlocking applications worth $25 billion annually by 2032.
Consumer willingness-to-pay analysis demonstrates that premium segments in automotive, aerospace, and specialty electronics markets would readily adopt Li-S technology at a 15-20% price premium over conventional solutions if the promised performance advantages can be reliably delivered in commercial products.
Current Challenges in Sulfur Utilization Efficiency
Despite significant advancements in lithium-sulfur (Li-S) battery technology, sulfur utilization efficiency remains one of the most critical challenges hindering commercial viability. Current Li-S full cells typically achieve only 30-50% sulfur utilization in practical applications, far below the theoretical capacity of 1675 mAh/g. This efficiency gap represents a substantial barrier to realizing the full potential of this promising energy storage technology.
The polysulfide shuttle effect continues to be a primary obstacle to efficient sulfur utilization. During discharge, long-chain polysulfides (Li2Sx, 4≤x≤8) dissolve into the electrolyte and migrate to the anode, where they are reduced and then diffuse back to the cathode. This cyclical process consumes active material, reduces coulombic efficiency, and causes rapid capacity fading, ultimately limiting sulfur utilization.
High internal resistance within sulfur cathodes presents another significant challenge. The insulating nature of sulfur (5×10^-30 S/cm) and its discharge products (Li2S) creates substantial electronic transport barriers. This resistance issue is particularly problematic at high sulfur loadings (>5 mg/cm²), which are necessary for practical energy density but often result in decreased sulfur utilization due to limited electron access to active material in thicker electrodes.
Volume expansion during cycling (up to 80%) causes mechanical stress that disrupts the cathode structure. This expansion leads to loss of electrical contact between sulfur particles and conductive additives, creating isolated sulfur domains that become electrochemically inaccessible and reducing overall utilization efficiency.
Electrolyte limitations further constrain sulfur utilization. The conventional electrolyte-to-sulfur (E/S) ratio in laboratory cells (>10 μL/mg) is impractically high for commercial applications, which require ratios below 3 μL/mg. At these lower ratios, insufficient electrolyte penetration into the cathode structure significantly reduces sulfur accessibility and utilization.
The formation of insulating Li2S films during discharge creates passivation layers that impede further electrochemical reactions. This kinetic barrier becomes particularly problematic at high discharge rates and high sulfur loadings, resulting in premature discharge termination before complete sulfur utilization is achieved.
Anode-side reactions also impact sulfur utilization efficiency. Lithium metal anodes suffer from dendrite formation and continuous SEI layer growth due to reactions with polysulfides. These processes consume active lithium and electrolyte, creating an imbalance between cathode and anode capacities that ultimately limits the achievable sulfur utilization in full cells.
The polysulfide shuttle effect continues to be a primary obstacle to efficient sulfur utilization. During discharge, long-chain polysulfides (Li2Sx, 4≤x≤8) dissolve into the electrolyte and migrate to the anode, where they are reduced and then diffuse back to the cathode. This cyclical process consumes active material, reduces coulombic efficiency, and causes rapid capacity fading, ultimately limiting sulfur utilization.
High internal resistance within sulfur cathodes presents another significant challenge. The insulating nature of sulfur (5×10^-30 S/cm) and its discharge products (Li2S) creates substantial electronic transport barriers. This resistance issue is particularly problematic at high sulfur loadings (>5 mg/cm²), which are necessary for practical energy density but often result in decreased sulfur utilization due to limited electron access to active material in thicker electrodes.
Volume expansion during cycling (up to 80%) causes mechanical stress that disrupts the cathode structure. This expansion leads to loss of electrical contact between sulfur particles and conductive additives, creating isolated sulfur domains that become electrochemically inaccessible and reducing overall utilization efficiency.
Electrolyte limitations further constrain sulfur utilization. The conventional electrolyte-to-sulfur (E/S) ratio in laboratory cells (>10 μL/mg) is impractically high for commercial applications, which require ratios below 3 μL/mg. At these lower ratios, insufficient electrolyte penetration into the cathode structure significantly reduces sulfur accessibility and utilization.
The formation of insulating Li2S films during discharge creates passivation layers that impede further electrochemical reactions. This kinetic barrier becomes particularly problematic at high discharge rates and high sulfur loadings, resulting in premature discharge termination before complete sulfur utilization is achieved.
Anode-side reactions also impact sulfur utilization efficiency. Lithium metal anodes suffer from dendrite formation and continuous SEI layer growth due to reactions with polysulfides. These processes consume active lithium and electrolyte, creating an imbalance between cathode and anode capacities that ultimately limits the achievable sulfur utilization in full cells.
State-of-the-Art Approaches for Improving Sulfur Utilization
01 Cathode structure optimization for improved sulfur utilization
Optimizing the cathode structure in lithium-sulfur batteries can significantly enhance sulfur utilization efficiency. This includes developing porous carbon structures that can effectively contain sulfur and prevent polysulfide dissolution. Advanced cathode designs with controlled pore size distribution and surface chemistry can trap sulfur species during cycling, leading to higher active material utilization and improved electrochemical performance.- Cathode structure optimization for improved sulfur utilization: Optimizing the cathode structure in lithium-sulfur batteries can significantly enhance sulfur utilization efficiency. This includes designing porous carbon structures that can effectively contain sulfur and prevent polysulfide dissolution. Advanced cathode architectures with controlled pore size distribution and surface functionality can improve the electrochemical accessibility of sulfur, leading to higher utilization rates during cycling. These structural modifications help address the volume expansion issues and maintain electrical contact throughout the charge-discharge process.
- Electrolyte modifications for polysulfide shuttling prevention: Modifying the electrolyte composition can effectively reduce polysulfide shuttling, which is a major cause of low sulfur utilization in lithium-sulfur batteries. Additives that form protective layers on the lithium anode or create chemical bonds with polysulfides can prevent their migration. Functional electrolytes with optimized salt concentrations and solvent ratios can enhance the ionic conductivity while suppressing polysulfide dissolution. These electrolyte engineering approaches lead to improved coulombic efficiency and better sulfur utilization over extended cycling.
- Conductive polymer binders and coatings: Incorporating conductive polymer binders and coatings in lithium-sulfur battery cathodes can enhance sulfur utilization efficiency. These polymers provide multiple benefits: they improve electrical conductivity throughout the cathode, physically trap polysulfides through chemical interactions, and accommodate volume changes during cycling. Polymers with functional groups that can form chemical bonds with sulfur species are particularly effective at maintaining cathode integrity and preventing capacity loss, resulting in higher active material utilization rates.
- Nanostructured sulfur composites: Developing nanostructured sulfur composites is an effective approach to increase sulfur utilization efficiency in lithium-sulfur batteries. These composites typically involve encapsulating sulfur within nanomaterials such as carbon nanotubes, graphene, or metal oxides. The nanostructuring provides shorter diffusion paths for lithium ions, better electrical contact with sulfur particles, and physical confinement of polysulfides. This results in improved electrochemical accessibility of sulfur, enhanced reaction kinetics, and ultimately higher utilization of the active material during battery operation.
- Interlayers and functional separators: Implementing interlayers and functional separators between the cathode and anode can significantly improve sulfur utilization efficiency. These components act as physical barriers that block polysulfide migration while allowing lithium ion transport. Advanced separator designs incorporate materials with high adsorption capacity for polysulfides, such as carbon materials with tailored porosity or metal compounds with chemical affinity for sulfur species. By effectively containing sulfur species within the cathode region, these interlayers minimize active material loss and maintain higher sulfur utilization throughout battery cycling.
02 Electrolyte modifications for enhanced sulfur utilization
Modifying the electrolyte composition can significantly improve sulfur utilization efficiency in lithium-sulfur batteries. Specialized electrolyte additives can suppress the shuttle effect of polysulfides, while optimized electrolyte formulations can enhance the solubility and conversion of sulfur species. These modifications help maintain active sulfur material within the cathode region, resulting in higher capacity retention and improved cycling stability.Expand Specific Solutions03 Conductive polymer binders and coatings
Incorporating conductive polymer binders and coatings in lithium-sulfur battery cathodes can enhance electrical connectivity between sulfur particles and the conductive network. These materials can effectively trap polysulfides through chemical interactions while maintaining good ionic and electronic conductivity. The improved electrical contact and reduced polysulfide dissolution lead to higher sulfur utilization efficiency and extended cycle life.Expand Specific Solutions04 Nanostructured sulfur composites
Developing nanostructured sulfur composites can dramatically improve sulfur utilization efficiency. These composites typically involve sulfur impregnated into nanoporous materials or chemically bonded to nanostructured hosts. The nanoscale confinement of sulfur shortens lithium ion diffusion paths and provides better electrical contact, while also physically restricting polysulfide dissolution. This approach enables higher active material utilization and improved rate capability.Expand Specific Solutions05 Interlayers and functional separators
Implementing interlayers and functional separators between the cathode and anode can effectively block polysulfide migration while allowing lithium ion transport. These components act as physical and chemical barriers that trap dissolved polysulfides and prevent them from reaching the anode. By keeping sulfur species within the cathode region, these designs significantly improve sulfur utilization efficiency and battery cycle life.Expand Specific Solutions
Leading Companies and Research Institutions in Li-S Battery Field
The lithium-sulfur battery market is currently in an early growth phase, characterized by significant R&D investment but limited commercial deployment. With a projected market size of $2.5-3 billion by 2030, this technology promises 2-5 times higher energy density than conventional lithium-ion batteries. Key players advancing sulfur utilization efficiency include Sion Power with their Licerion® technology, PolyPlus Battery with protected lithium electrode technology, and LG Energy Solution developing commercial applications. Academic institutions like Central South University and the University of California are making significant contributions to fundamental research. The technology remains at TRL 5-6, with challenges in cycle life and sulfur utilization efficiency still requiring breakthrough innovations before widespread commercialization can occur.
Sion Power Corp.
Technical Solution: Sion Power has developed a proprietary lithium-sulfur (Li-S) technology called "Licerion" that significantly improves sulfur utilization efficiency. Their approach incorporates a protected lithium anode technology combined with high-loading sulfur cathodes. The company employs advanced carbon-sulfur composite structures with tailored pore architectures that effectively trap polysulfide intermediates during cycling. Their cathode design includes conductive carbon frameworks with mesoporous structures that facilitate both electron transport and ion diffusion while providing physical confinement of sulfur species. Sion has also implemented specialized electrolyte formulations containing lithium nitrate and other additives that form a stable solid electrolyte interphase (SEI) on the lithium anode, reducing shuttle effects and improving coulombic efficiency to over 99%. Their full cells have demonstrated practical energy densities exceeding 400 Wh/kg with sulfur utilization rates approaching 70% of theoretical capacity.
Strengths: Industry-leading energy density (400+ Wh/kg) with relatively high sulfur utilization. Proprietary protected lithium anode technology effectively mitigates polysulfide shuttle effect. Weaknesses: Higher manufacturing costs compared to conventional lithium-ion batteries. Cycle life still limited to hundreds of cycles rather than thousands needed for certain applications.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed an advanced lithium-sulfur battery system focusing on maximizing sulfur utilization efficiency in full cells. Their approach incorporates a multi-layered cathode architecture with gradient porosity that optimizes sulfur loading while managing volume expansion. The company utilizes a proprietary carbon-sulfur composite with nitrogen and oxygen functional groups that chemically bind polysulfides, significantly reducing the shuttle effect. Their technology employs a dual-functional interlayer between cathode and separator that acts as both a physical barrier and chemical trap for migrating polysulfides. LG has also formulated specialized electrolytes with lithium salt concentrations optimized for sulfur electrochemistry, incorporating additives that stabilize the solid electrolyte interphase on both electrodes. Their full cells demonstrate sulfur utilization efficiency of approximately 65% of theoretical capacity while maintaining stability over extended cycling, achieving energy densities of 350-400 Wh/kg at the cell level.
Strengths: Established large-scale manufacturing capabilities that could enable commercialization at scale. Comprehensive battery management systems that optimize performance and safety. Weaknesses: Still facing challenges with cycle life limitations compared to their commercial lithium-ion offerings. Higher initial self-discharge rates than conventional lithium-ion technology.
Key Patents and Breakthroughs in Sulfur Cathode Design
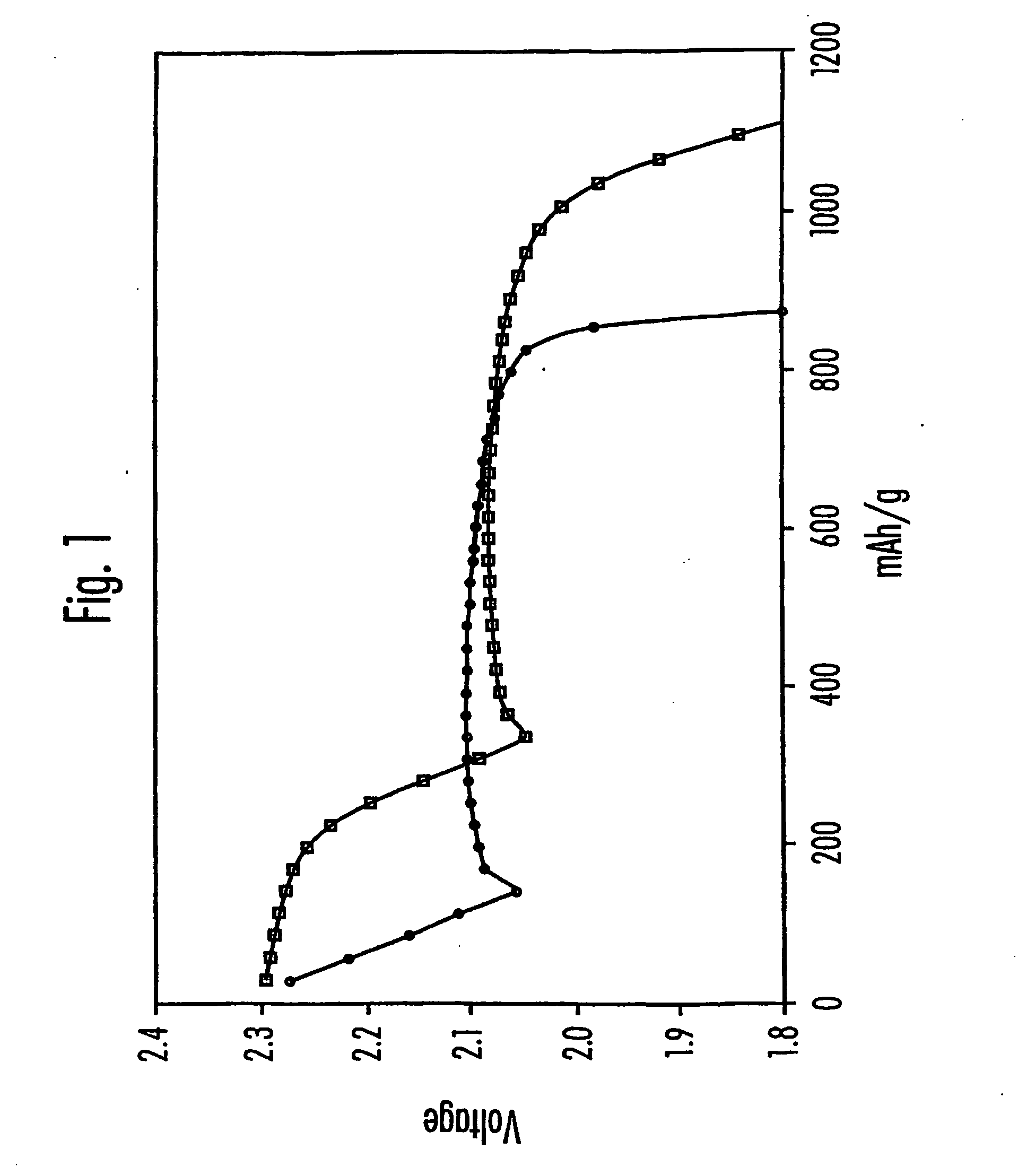
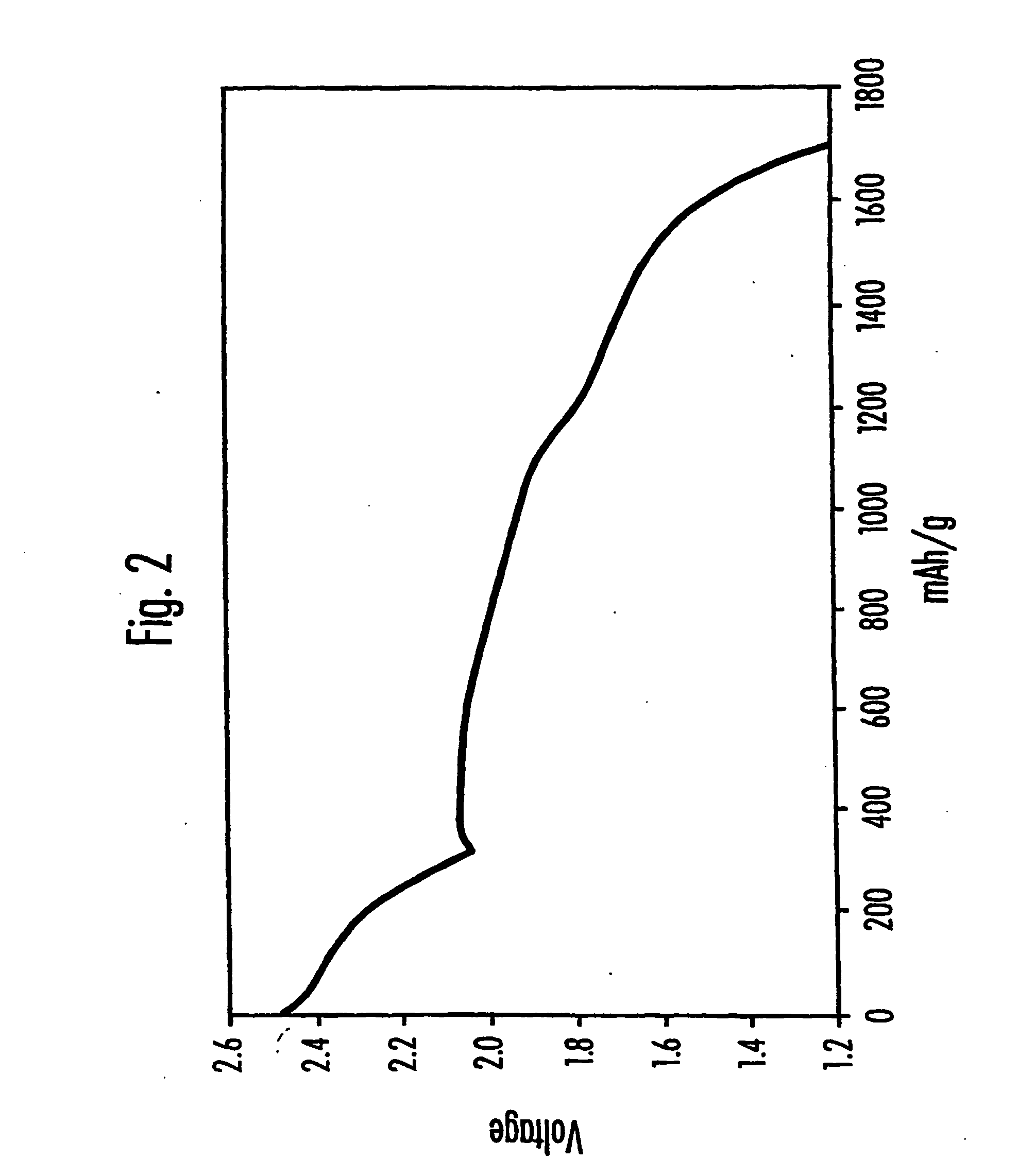
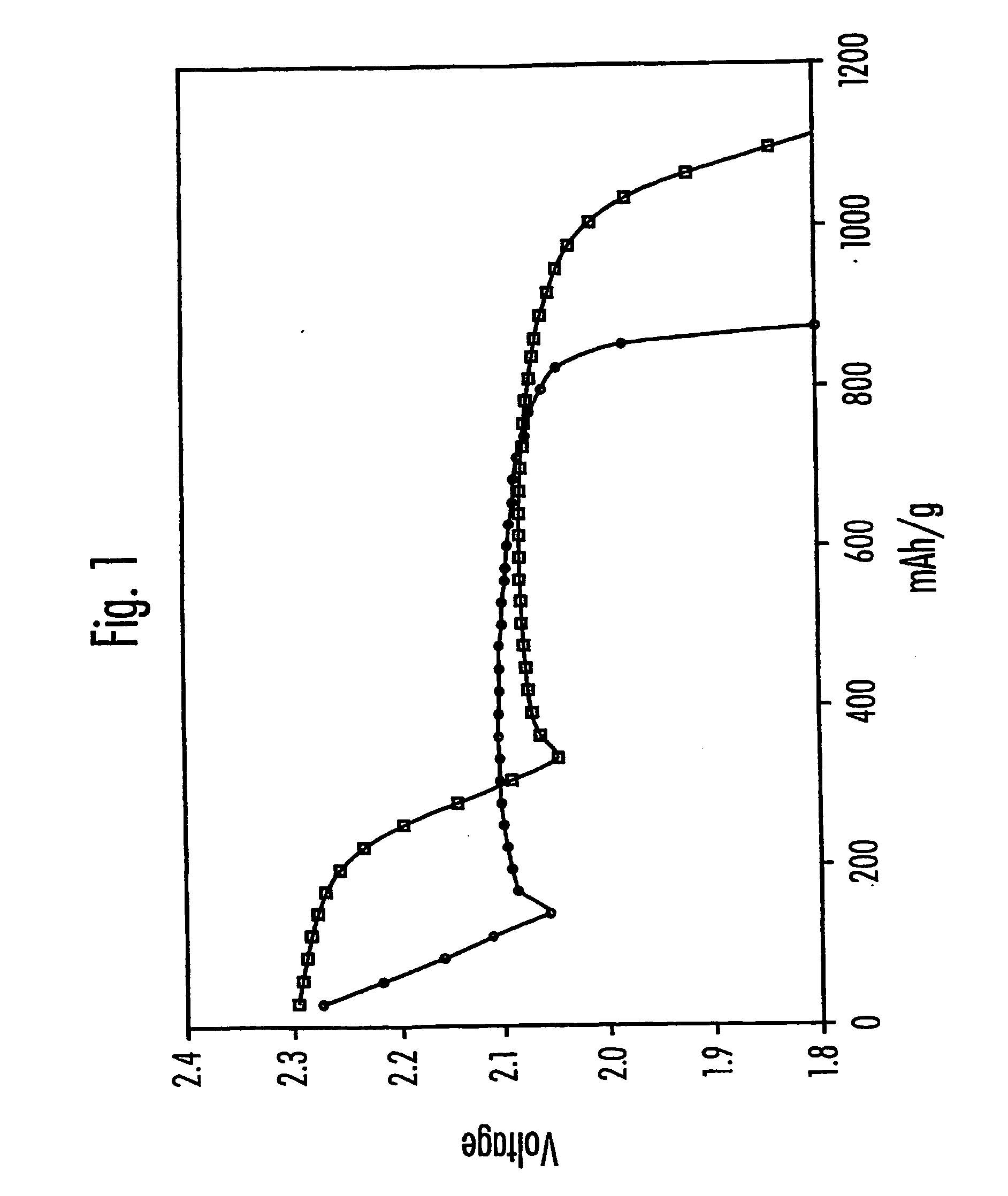
Electrolytes for lithium sulfur cells
PatentInactiveUS20070082270A1
Innovation
- Incorporating N—O compounds as additives in the nonaqueous electrolyte of Li/S cells, which reduce the reactivity between lithium anodes and sulfur-containing cathode reduction products, enhancing sulfur utilization, charge-discharge efficiency, and minimizing self-discharge by inhibiting the redox shuttle mechanism.
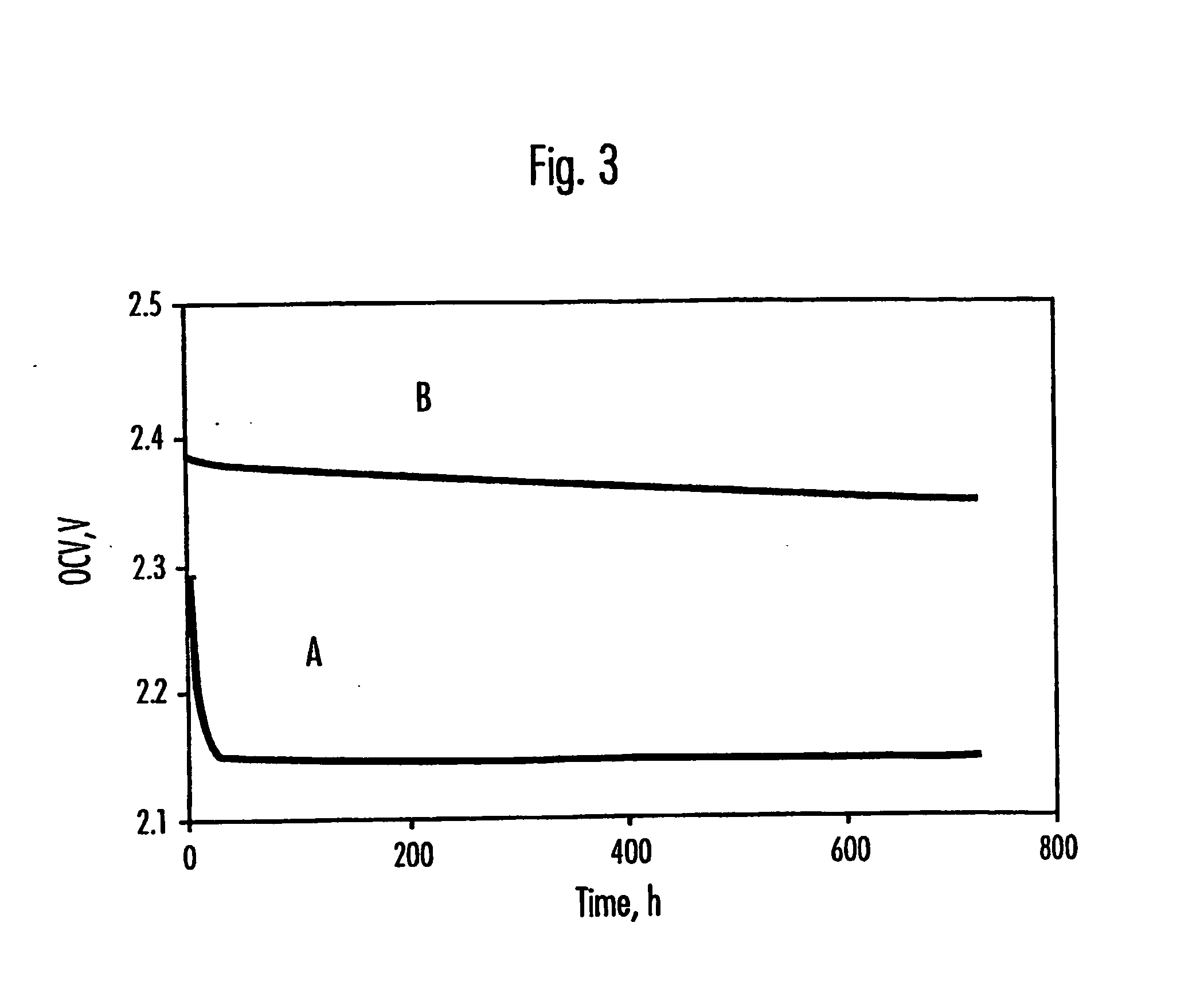
Electrolytes for lithium sulfur cells
PatentInactiveUS20070082264A1
Innovation
- Incorporating N—O compounds as additives in the nonaqueous electrolyte of Li/S cells, which reduce the reactivity between lithium anodes and sulfur-containing cathode reduction products, enhancing sulfur utilization, charge-discharge efficiency, and minimizing self-discharge by inhibiting the redox shuttle mechanism.
Environmental Impact and Sustainability Assessment
The environmental impact of lithium-sulfur (Li-S) batteries represents a critical dimension in evaluating their viability as next-generation energy storage solutions. Unlike conventional lithium-ion batteries that rely on cobalt and nickel, Li-S cells utilize sulfur—an abundant, low-cost byproduct of petroleum refining—potentially reducing the environmental footprint associated with raw material extraction. However, the environmental benefits are directly proportional to sulfur utilization efficiency, as higher efficiency translates to fewer resources consumed per unit of energy stored.
Life cycle assessments indicate that Li-S batteries with high sulfur utilization efficiency (>70%) can achieve up to 30% lower carbon footprint compared to conventional lithium-ion technologies. This reduction stems primarily from decreased energy-intensive mining operations and simplified manufacturing processes. Conversely, low sulfur utilization (<50%) diminishes these advantages, as more cells are required to deliver equivalent energy storage capacity, offsetting the environmental benefits.
Water consumption represents another significant environmental consideration. The production of high-purity lithium metal for anodes remains water-intensive, but improved sulfur utilization reduces the total number of cells needed, thereby decreasing overall water requirements. Research indicates that achieving 80% sulfur utilization could reduce water consumption by approximately 25% compared to current commercial battery technologies.
End-of-life management presents both challenges and opportunities. Sulfur's non-toxic nature simplifies recycling processes compared to conventional batteries containing heavy metals. Advanced recycling technologies specifically designed for Li-S cells have demonstrated recovery rates exceeding 90% for lithium and 95% for sulfur when operating with high-efficiency cells. These recovery rates significantly enhance the sustainability profile of Li-S technology.
The sustainability advantages of Li-S batteries extend beyond environmental metrics to include social dimensions. Reduced dependence on geopolitically sensitive materials like cobalt mitigates supply chain risks and potential human rights concerns associated with mining operations. Furthermore, sulfur's global availability promotes more geographically distributed manufacturing capabilities, potentially democratizing energy storage technology production.
Looking forward, improving sulfur utilization efficiency represents not merely a technical challenge but a critical sustainability imperative. Each percentage point improvement in utilization efficiency cascades through the entire life cycle, amplifying environmental benefits. Regulatory frameworks increasingly recognize this relationship, with several jurisdictions developing battery passport systems that will require disclosure of resource utilization efficiency metrics, including sulfur utilization in relevant technologies.
Life cycle assessments indicate that Li-S batteries with high sulfur utilization efficiency (>70%) can achieve up to 30% lower carbon footprint compared to conventional lithium-ion technologies. This reduction stems primarily from decreased energy-intensive mining operations and simplified manufacturing processes. Conversely, low sulfur utilization (<50%) diminishes these advantages, as more cells are required to deliver equivalent energy storage capacity, offsetting the environmental benefits.
Water consumption represents another significant environmental consideration. The production of high-purity lithium metal for anodes remains water-intensive, but improved sulfur utilization reduces the total number of cells needed, thereby decreasing overall water requirements. Research indicates that achieving 80% sulfur utilization could reduce water consumption by approximately 25% compared to current commercial battery technologies.
End-of-life management presents both challenges and opportunities. Sulfur's non-toxic nature simplifies recycling processes compared to conventional batteries containing heavy metals. Advanced recycling technologies specifically designed for Li-S cells have demonstrated recovery rates exceeding 90% for lithium and 95% for sulfur when operating with high-efficiency cells. These recovery rates significantly enhance the sustainability profile of Li-S technology.
The sustainability advantages of Li-S batteries extend beyond environmental metrics to include social dimensions. Reduced dependence on geopolitically sensitive materials like cobalt mitigates supply chain risks and potential human rights concerns associated with mining operations. Furthermore, sulfur's global availability promotes more geographically distributed manufacturing capabilities, potentially democratizing energy storage technology production.
Looking forward, improving sulfur utilization efficiency represents not merely a technical challenge but a critical sustainability imperative. Each percentage point improvement in utilization efficiency cascades through the entire life cycle, amplifying environmental benefits. Regulatory frameworks increasingly recognize this relationship, with several jurisdictions developing battery passport systems that will require disclosure of resource utilization efficiency metrics, including sulfur utilization in relevant technologies.
Scale-up and Manufacturing Considerations
The scale-up of lithium-sulfur (Li-S) battery technology from laboratory prototypes to commercial production presents significant manufacturing challenges that directly impact sulfur utilization efficiency. Current laboratory-scale cells often achieve high sulfur utilization under idealized conditions, but these results rarely translate to large-format cells manufactured at industrial scales.
Material processing represents a critical consideration in scaling Li-S technology. The preparation of sulfur cathodes requires precise control over sulfur distribution within conductive hosts. Industrial-scale mixing and coating processes must be optimized to maintain the intimate contact between sulfur and conductive additives achieved in laboratory settings. Variations in material homogeneity during large-batch processing can create "dead zones" where sulfur becomes electrochemically inaccessible, dramatically reducing overall utilization efficiency.
Electrode fabrication techniques must evolve beyond conventional slurry-based approaches. The high sulfur loading required for commercial viability (>5 mg/cm²) creates thick electrodes that exacerbate mass transport limitations. Advanced manufacturing methods such as 3D printing, electrospinning, or freeze-casting show promise for creating hierarchical electrode architectures that maintain efficient ion transport pathways even at high sulfur loadings.
Electrolyte management presents another manufacturing hurdle. The optimal electrolyte-to-sulfur ratio for maximizing utilization in laboratory cells (often >10 μL/mg) is economically impractical at commercial scales. Manufacturing processes must be developed to achieve adequate sulfur wetting and polysulfide containment with substantially reduced electrolyte volumes (<3 μL/mg), potentially through innovative cell designs or electrolyte infiltration techniques.
Quality control methodologies require significant adaptation for Li-S technology. Traditional battery manufacturing quality metrics may not adequately capture the unique failure modes affecting sulfur utilization. New in-line analytical techniques must be developed to monitor sulfur distribution, electrolyte infiltration, and separator functionality during production.
Cost considerations ultimately dictate manufacturing decisions that impact sulfur utilization. While high-surface-area carbon hosts and specialized separators improve laboratory performance, their cost may be prohibitive at scale. Manufacturing processes must balance performance optimization with economic viability, potentially through the development of lower-cost alternatives or more efficient material utilization strategies.
Material processing represents a critical consideration in scaling Li-S technology. The preparation of sulfur cathodes requires precise control over sulfur distribution within conductive hosts. Industrial-scale mixing and coating processes must be optimized to maintain the intimate contact between sulfur and conductive additives achieved in laboratory settings. Variations in material homogeneity during large-batch processing can create "dead zones" where sulfur becomes electrochemically inaccessible, dramatically reducing overall utilization efficiency.
Electrode fabrication techniques must evolve beyond conventional slurry-based approaches. The high sulfur loading required for commercial viability (>5 mg/cm²) creates thick electrodes that exacerbate mass transport limitations. Advanced manufacturing methods such as 3D printing, electrospinning, or freeze-casting show promise for creating hierarchical electrode architectures that maintain efficient ion transport pathways even at high sulfur loadings.
Electrolyte management presents another manufacturing hurdle. The optimal electrolyte-to-sulfur ratio for maximizing utilization in laboratory cells (often >10 μL/mg) is economically impractical at commercial scales. Manufacturing processes must be developed to achieve adequate sulfur wetting and polysulfide containment with substantially reduced electrolyte volumes (<3 μL/mg), potentially through innovative cell designs or electrolyte infiltration techniques.
Quality control methodologies require significant adaptation for Li-S technology. Traditional battery manufacturing quality metrics may not adequately capture the unique failure modes affecting sulfur utilization. New in-line analytical techniques must be developed to monitor sulfur distribution, electrolyte infiltration, and separator functionality during production.
Cost considerations ultimately dictate manufacturing decisions that impact sulfur utilization. While high-surface-area carbon hosts and specialized separators improve laboratory performance, their cost may be prohibitive at scale. Manufacturing processes must balance performance optimization with economic viability, potentially through the development of lower-cost alternatives or more efficient material utilization strategies.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!