How to Engineer Optimized Processes for Ethyl Acetate Production?
Ethyl Acetate Synthesis Background and Objectives
Ethyl acetate, a versatile organic compound, has been a cornerstone in various industries for decades. Its synthesis and production processes have evolved significantly since its initial discovery in the 19th century. The journey of ethyl acetate production reflects the broader trends in chemical engineering and process optimization, showcasing the industry's continuous pursuit of efficiency and sustainability.
The primary objective in engineering optimized processes for ethyl acetate production is to achieve higher yields, reduce energy consumption, and minimize environmental impact. This aligns with the global shift towards green chemistry and sustainable manufacturing practices. The evolution of ethyl acetate synthesis methods has been driven by these goals, moving from traditional batch processes to more advanced continuous flow systems.
Historically, ethyl acetate was primarily produced through the esterification of ethanol and acetic acid, catalyzed by sulfuric acid. This method, while effective, posed challenges in terms of product purity and process efficiency. As the demand for ethyl acetate grew across industries such as coatings, pharmaceuticals, and food processing, the need for more sophisticated production techniques became apparent.
Recent technological advancements have led to the development of novel catalysts and reactor designs, significantly improving the efficiency of ethyl acetate synthesis. These innovations aim to overcome traditional limitations such as equilibrium constraints and slow reaction rates. The integration of reactive distillation techniques, for instance, has allowed for simultaneous reaction and separation, thereby enhancing overall process efficiency.
The current landscape of ethyl acetate production is characterized by a focus on process intensification and the exploration of alternative feedstocks. Researchers and industry professionals are investigating the use of biomass-derived raw materials as a sustainable alternative to petrochemical-based feedstocks. This shift not only addresses environmental concerns but also aligns with the circular economy principles gaining traction in the chemical industry.
As we look towards the future of ethyl acetate synthesis, the objectives are clear: to develop processes that are not only economically viable but also environmentally benign. This involves exploring novel reaction pathways, designing more efficient catalysts, and leveraging advanced process control strategies. The ultimate goal is to create a production process that maximizes yield and selectivity while minimizing waste and energy consumption.
Market Analysis for Ethyl Acetate Demand
The global market for ethyl acetate has been experiencing steady growth, driven by its versatile applications across various industries. As a key solvent and intermediate in the production of paints, coatings, adhesives, and pharmaceuticals, ethyl acetate's demand is closely tied to the performance of these end-use sectors.
In recent years, the construction and automotive industries have been significant contributors to the increasing demand for ethyl acetate. The growing construction activities, particularly in emerging economies, have fueled the need for paints and coatings, subsequently boosting ethyl acetate consumption. Similarly, the automotive sector's recovery and the trend towards eco-friendly coatings have further propelled market growth.
The packaging industry has also emerged as a major consumer of ethyl acetate, especially in flexible packaging applications. The shift towards sustainable and bio-based packaging solutions has created new opportunities for ethyl acetate derived from renewable sources, aligning with the global push for environmentally friendly products.
Geographically, Asia-Pacific remains the largest market for ethyl acetate, with China and India leading in both production and consumption. The region's rapid industrialization, expanding middle class, and increasing disposable incomes have been key factors driving demand. North America and Europe follow, with mature markets focusing on high-quality, specialized applications in pharmaceuticals and electronics.
Market analysts project the global ethyl acetate market to continue its growth trajectory, with estimates suggesting a compound annual growth rate (CAGR) between 4% to 6% over the next five years. This growth is expected to be fueled by innovations in bio-based production methods, which could potentially reduce costs and improve sustainability profiles.
However, the market faces challenges, including volatility in raw material prices, particularly ethanol and acetic acid. Environmental regulations regarding volatile organic compound (VOC) emissions also pose potential constraints on market expansion in certain regions. Manufacturers are responding by investing in research and development to improve production efficiency and explore alternative feedstocks.
The competitive landscape of the ethyl acetate market is characterized by a mix of large multinational chemical companies and regional players. Key strategies employed by market leaders include capacity expansions, mergers and acquisitions, and partnerships to strengthen their market positions and meet the growing demand.
Current Production Challenges and Limitations
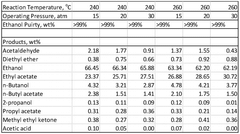
The production of ethyl acetate faces several significant challenges and limitations in current industrial processes. One of the primary issues is the equilibrium limitation inherent in the esterification reaction between ethanol and acetic acid. This equilibrium constraint results in incomplete conversion, typically around 65-70%, necessitating additional separation and recycling steps, which increase production costs and energy consumption.
Another major challenge is the corrosive nature of the reactants and catalysts used in the process. Acetic acid, in particular, can cause severe corrosion to equipment, leading to increased maintenance costs and potential safety hazards. This corrosion issue also limits the choice of materials for reactor construction and piping, often requiring expensive corrosion-resistant alloys or specialized coatings.
The presence of water as a byproduct in the esterification reaction poses additional complications. Water not only shifts the equilibrium unfavorably but also forms azeotropes with ethanol and ethyl acetate, making separation and purification more difficult and energy-intensive. Conventional distillation techniques often struggle to achieve high-purity ethyl acetate due to these azeotropic mixtures.
Furthermore, the catalysts used in traditional processes, such as sulfuric acid, present environmental and safety concerns. These homogeneous catalysts are difficult to separate from the product stream, leading to potential contamination and the need for neutralization steps. This not only adds to the production costs but also generates waste that requires proper disposal.
Energy efficiency is another significant limitation in current ethyl acetate production processes. The multiple distillation and separation steps required to purify the product and recycle unreacted materials consume substantial amounts of energy. This high energy demand not only increases production costs but also contributes to the overall carbon footprint of the process.
Lastly, the optimization of reaction conditions presents a complex challenge. Balancing factors such as temperature, pressure, reactant ratios, and residence time to maximize yield and selectivity while minimizing side reactions and energy consumption requires sophisticated control systems and process modeling. The interdependence of these variables makes it difficult to achieve optimal performance across all aspects of the production process simultaneously.
Addressing these challenges and limitations is crucial for developing more efficient, cost-effective, and environmentally friendly processes for ethyl acetate production. Innovations in catalyst design, reactor technology, separation techniques, and process integration are needed to overcome these obstacles and improve the overall sustainability of ethyl acetate manufacturing.
Existing Optimization Techniques for Ethyl Acetate Production
01 Reaction conditions optimization
Optimizing reaction conditions such as temperature, pressure, and catalyst concentration is crucial for improving ethyl acetate production efficiency. This involves fine-tuning parameters to achieve higher yields, better selectivity, and reduced energy consumption.- Reaction conditions optimization: Optimizing reaction conditions such as temperature, pressure, and catalyst concentration is crucial for improving ethyl acetate production efficiency. This involves careful control of parameters to maximize yield and product quality while minimizing side reactions and energy consumption.
- Catalyst development and selection: Developing and selecting appropriate catalysts can significantly enhance the ethyl acetate production process. This includes exploring novel catalytic materials, improving catalyst stability, and optimizing catalyst regeneration methods to increase overall process efficiency and reduce costs.
- Continuous flow processes: Implementing continuous flow processes for ethyl acetate production can improve efficiency and product quality. This approach allows for better control of reaction parameters, reduced equipment size, and increased throughput compared to batch processes.
- Purification and separation techniques: Enhancing purification and separation techniques is essential for obtaining high-quality ethyl acetate. This includes developing advanced distillation methods, membrane separation technologies, and other innovative purification processes to improve product purity and reduce energy consumption.
- Process monitoring and control systems: Implementing advanced process monitoring and control systems can optimize ethyl acetate production. This involves using real-time data analysis, predictive modeling, and automated control systems to maintain optimal operating conditions and quickly respond to process variations.
02 Catalyst development and selection
Developing and selecting appropriate catalysts can significantly enhance the ethyl acetate production process. This includes exploring novel catalytic materials, improving catalyst stability, and optimizing catalyst regeneration methods to increase overall process efficiency.Expand Specific Solutions03 Continuous flow processes
Implementing continuous flow processes for ethyl acetate production can lead to improved productivity and reduced operational costs. This approach allows for better control of reaction parameters, easier scale-up, and enhanced product quality compared to batch processes.Expand Specific Solutions04 Purification and separation techniques
Optimizing purification and separation techniques is essential for obtaining high-quality ethyl acetate. This includes developing efficient distillation methods, implementing membrane separation technologies, and exploring novel purification approaches to reduce energy consumption and increase product purity.Expand Specific Solutions05 Process monitoring and control systems
Implementing advanced process monitoring and control systems can significantly improve ethyl acetate production efficiency. This involves using real-time data analysis, predictive modeling, and automated control systems to optimize process parameters and ensure consistent product quality.Expand Specific Solutions
Key Industry Players and Competitive Landscape
The ethyl acetate production optimization landscape is characterized by a mature market with established players and emerging innovators. The industry is in a growth phase, driven by increasing demand across various sectors such as coatings, pharmaceuticals, and food packaging. The global market size for ethyl acetate is substantial, with projections indicating continued expansion. Technologically, the field is evolving, with companies like Celanese International Corp. and Resonac Corp. leading in traditional production methods. Simultaneously, innovative approaches are being explored by firms such as Viridis Chemical LLC and Greenyug LLC, focusing on renewable and sustainable production processes. Academic institutions like the University of Campinas and Nanjing Tech University are contributing to research and development, potentially influencing future technological advancements in the industry.
Celanese International Corp.
Resonac Corp.
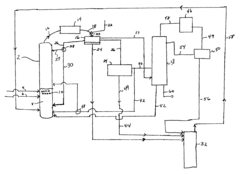
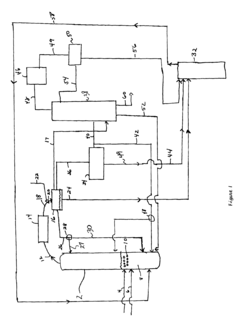
Innovative Catalysts and Reaction Mechanisms
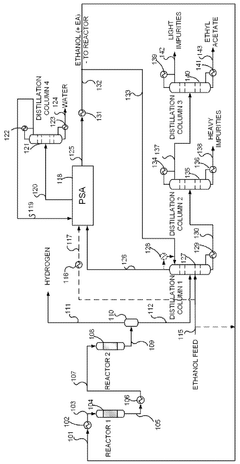
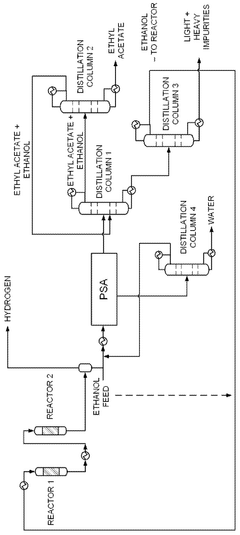
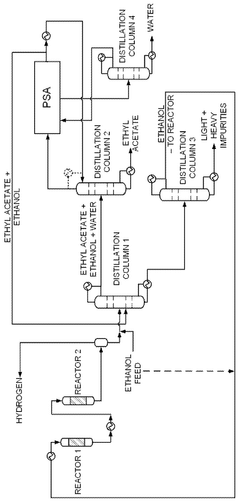
- A novel process that converts ethanol to ethyl acetate through dehydrogenation, followed by selective hydrogenation of byproducts to facilitate easier separation, and utilizes pressure swing adsorption to minimize water content and protect the catalyst.
- The process involves using a membrane separation unit to remove water from the condensed reaction stream, recycling the dried stream back into the production process, and employing an additional distillation zone to produce purified ethyl acetate with minimal acid content, thereby optimizing water management and increasing process capacity.
Environmental Impact and Sustainability Considerations
The production of ethyl acetate, while an essential process in various industries, carries significant environmental implications that demand careful consideration. The primary environmental concerns associated with ethyl acetate production include air pollution, water contamination, and energy consumption. Volatile organic compound (VOC) emissions, particularly during the esterification and purification stages, contribute to air quality degradation and potential health hazards. Implementing advanced emission control technologies, such as regenerative thermal oxidizers or carbon adsorption systems, can substantially reduce these emissions.
Water usage and wastewater management present another critical environmental challenge. The production process typically requires substantial amounts of water for cooling and separation processes. Implementing closed-loop cooling systems and water recycling technologies can significantly reduce freshwater consumption and minimize wastewater discharge. Additionally, treating wastewater to remove organic contaminants before release is crucial to protect aquatic ecosystems.
Energy efficiency is a key factor in reducing the carbon footprint of ethyl acetate production. Optimizing reaction conditions, improving heat recovery systems, and utilizing energy-efficient equipment can lead to substantial energy savings. The integration of renewable energy sources, such as solar or wind power, into the production facility can further reduce reliance on fossil fuels and decrease greenhouse gas emissions.
Raw material sourcing also plays a vital role in the sustainability of ethyl acetate production. Exploring bio-based feedstocks, such as ethanol derived from renewable sources, can reduce dependence on petrochemical-based materials and contribute to a more circular economy. However, the environmental impact of these alternative feedstocks must be carefully evaluated to ensure they offer genuine sustainability benefits.
Waste reduction and management strategies are essential for minimizing the environmental footprint of ethyl acetate production. Implementing lean manufacturing principles, optimizing production schedules, and improving process efficiency can significantly reduce waste generation. Additionally, exploring opportunities for byproduct valorization and developing closed-loop material cycles can transform waste streams into valuable resources.
Lifecycle assessment (LCA) is a crucial tool for comprehensively evaluating the environmental impact of ethyl acetate production. By analyzing the entire production chain from raw material extraction to end-of-life disposal, manufacturers can identify hotspots for environmental improvement and make informed decisions to enhance overall sustainability. This holistic approach enables the development of more environmentally friendly production processes and products.
Economic Feasibility of Optimized Production Methods
The economic feasibility of optimized production methods for ethyl acetate is a critical consideration for manufacturers seeking to enhance their competitive edge in the market. A comprehensive analysis of the cost-benefit ratio associated with implementing advanced production techniques is essential for informed decision-making.
One of the primary factors influencing the economic viability of optimized processes is the reduction in raw material consumption. By improving reaction efficiency and minimizing waste, manufacturers can significantly lower their input costs. For instance, the implementation of reactive distillation technology has shown potential to reduce raw material usage by up to 20% compared to conventional batch processes.
Energy efficiency is another crucial aspect that impacts the economic feasibility of optimized production methods. Advanced heat integration systems and energy recovery techniques can lead to substantial reductions in utility costs. Studies have demonstrated that optimized processes can achieve energy savings of 30-40% compared to traditional methods, translating to significant operational cost reductions over time.
Capital investment requirements for process optimization must be carefully evaluated against projected long-term benefits. While the initial outlay for advanced equipment and control systems may be substantial, the payback period can be relatively short due to improved productivity and reduced operating costs. For example, the installation of a continuous flow reactor system for ethyl acetate production may require a capital investment of $5-10 million, but can result in annual savings of $2-3 million through increased throughput and reduced energy consumption.
The impact on product quality and consistency is another economic consideration. Optimized processes often lead to higher purity products with fewer impurities, potentially commanding premium prices in the market. Additionally, improved process control can result in more consistent product quality, reducing the costs associated with off-spec batches and rework.
Labor costs can also be significantly affected by process optimization. Automated control systems and continuous production methods typically require fewer operators per unit of output, leading to reduced labor expenses. However, this must be balanced against the need for more skilled personnel to manage and maintain advanced production systems.
Market dynamics play a crucial role in determining the economic feasibility of optimized production methods. The ability to rapidly adjust production volumes in response to market demand can provide a significant competitive advantage. Flexible, modular production systems that can be easily scaled up or down may offer superior economic benefits in volatile market conditions.
In conclusion, the economic feasibility of optimized production methods for ethyl acetate is generally favorable, with potential for substantial cost savings and improved market positioning. However, a thorough analysis of specific production scenarios, market conditions, and investment capabilities is essential for individual manufacturers to determine the most economically viable optimization strategies for their operations.