Potassium-Ion Versus Sodium-Ion: Technology And Cost Comparison
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
K-ion vs Na-ion Battery Technology Background & Objectives
The evolution of battery technology has been a cornerstone of modern technological advancement, with lithium-ion batteries dominating the market for decades. However, concerns regarding lithium's limited global reserves, geopolitical supply chain vulnerabilities, and rising costs have accelerated research into alternative battery chemistries. Potassium-ion (K-ion) and sodium-ion (Na-ion) batteries have emerged as promising candidates due to their abundant raw materials and potential cost advantages.
Historically, lithium-ion battery development began in the 1970s, with commercial introduction in the 1990s. Research into sodium-ion batteries actually predates lithium-ion technology but was deprioritized due to lithium's superior energy density. Potassium-ion battery research gained momentum only in the past decade, representing a relatively newer entrant in the alternative battery landscape.
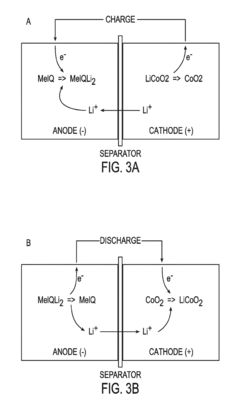
The fundamental principle behind these technologies involves the intercalation of respective ions (K+ or Na+) into electrode materials during charge-discharge cycles. Both potassium and sodium are alkali metals like lithium, sharing similar chemical properties while being significantly more abundant in the Earth's crust. Potassium is approximately 1,000 times more abundant than lithium, while sodium is approximately 1,100 times more abundant.
The primary technical objective of K-ion and Na-ion battery development is to achieve performance metrics comparable to lithium-ion batteries while leveraging cost advantages and supply chain security. Specific goals include improving energy density, cycle life, rate capability, and safety profiles while maintaining the inherent cost benefits of these alternative chemistries.
Current technological trends indicate growing interest in developing specialized electrode materials optimized for the larger ionic radii of potassium (1.38Å) and sodium (1.02Å) compared to lithium (0.76Å). This size difference presents both challenges and opportunities in materials engineering, particularly regarding structural stability during repeated ion insertion and extraction.
The market trajectory for these technologies appears to be initially focused on stationary energy storage applications where energy density is less critical than cost, followed by potential expansion into electric mobility and consumer electronics as the technology matures. Both K-ion and Na-ion technologies are positioned as complementary rather than direct replacements for lithium-ion batteries in many applications.
This comparative analysis aims to evaluate the current state of K-ion versus Na-ion battery technologies, assess their respective advantages and limitations, examine cost structures, and project their potential market penetration timelines across various application segments.
Historically, lithium-ion battery development began in the 1970s, with commercial introduction in the 1990s. Research into sodium-ion batteries actually predates lithium-ion technology but was deprioritized due to lithium's superior energy density. Potassium-ion battery research gained momentum only in the past decade, representing a relatively newer entrant in the alternative battery landscape.
The fundamental principle behind these technologies involves the intercalation of respective ions (K+ or Na+) into electrode materials during charge-discharge cycles. Both potassium and sodium are alkali metals like lithium, sharing similar chemical properties while being significantly more abundant in the Earth's crust. Potassium is approximately 1,000 times more abundant than lithium, while sodium is approximately 1,100 times more abundant.
The primary technical objective of K-ion and Na-ion battery development is to achieve performance metrics comparable to lithium-ion batteries while leveraging cost advantages and supply chain security. Specific goals include improving energy density, cycle life, rate capability, and safety profiles while maintaining the inherent cost benefits of these alternative chemistries.
Current technological trends indicate growing interest in developing specialized electrode materials optimized for the larger ionic radii of potassium (1.38Å) and sodium (1.02Å) compared to lithium (0.76Å). This size difference presents both challenges and opportunities in materials engineering, particularly regarding structural stability during repeated ion insertion and extraction.
The market trajectory for these technologies appears to be initially focused on stationary energy storage applications where energy density is less critical than cost, followed by potential expansion into electric mobility and consumer electronics as the technology matures. Both K-ion and Na-ion technologies are positioned as complementary rather than direct replacements for lithium-ion batteries in many applications.
This comparative analysis aims to evaluate the current state of K-ion versus Na-ion battery technologies, assess their respective advantages and limitations, examine cost structures, and project their potential market penetration timelines across various application segments.
Market Demand Analysis for Post-Lithium Battery Solutions
The global energy storage market is experiencing a paradigm shift as concerns about lithium resource scarcity and geopolitical supply chain vulnerabilities intensify. Post-lithium battery solutions, particularly potassium-ion (K-ion) and sodium-ion (Na-ion) technologies, are emerging as promising alternatives with substantial market potential. Current projections indicate the energy storage market will exceed $300 billion by 2030, with post-lithium technologies potentially capturing 15-20% of this expanding sector.
Industrial applications represent the most immediate market opportunity for Na-ion and K-ion batteries. These sectors prioritize cost-effectiveness and safety over energy density, making them ideal early adopters. The stationary energy storage segment, valued at approximately $27 billion in 2022, is projected to grow at a CAGR of 30% through 2030, creating significant space for post-lithium solutions.
Electric mobility presents another substantial market, particularly in regions with developing economies where affordability outweighs performance requirements. The low-speed electric vehicle market in Asia and budget electric vehicles globally could benefit from Na-ion and K-ion batteries' cost advantages, despite their lower energy density compared to lithium-ion counterparts.
Consumer electronics manufacturers are increasingly exploring diversification of battery technologies to mitigate supply chain risks. This sector values the enhanced safety profiles of Na-ion and K-ion batteries, particularly for applications where thermal runaway risks must be minimized.
Regional market analysis reveals Asia-Pacific as the dominant manufacturing hub and potential largest market for post-lithium batteries, driven by China's aggressive development of sodium-ion technology. Europe follows with strong interest stemming from sustainability initiatives and raw material sovereignty concerns, while North America shows growing investment in research but lags in commercialization efforts.
Market adoption will be significantly influenced by regulatory frameworks, with the EU Battery Directive and similar regulations worldwide increasingly emphasizing sustainability metrics that favor Na-ion and K-ion technologies due to their reduced environmental footprint and reliance on more abundant resources.
Cost sensitivity analysis indicates that markets with high price elasticity will likely be early adopters of these technologies. The projected 20-30% cost advantage of Na-ion batteries over lithium-ion counterparts (when accounting for total cost of ownership) represents a compelling value proposition for price-sensitive segments, potentially accelerating market penetration beyond current forecasts.
Industrial applications represent the most immediate market opportunity for Na-ion and K-ion batteries. These sectors prioritize cost-effectiveness and safety over energy density, making them ideal early adopters. The stationary energy storage segment, valued at approximately $27 billion in 2022, is projected to grow at a CAGR of 30% through 2030, creating significant space for post-lithium solutions.
Electric mobility presents another substantial market, particularly in regions with developing economies where affordability outweighs performance requirements. The low-speed electric vehicle market in Asia and budget electric vehicles globally could benefit from Na-ion and K-ion batteries' cost advantages, despite their lower energy density compared to lithium-ion counterparts.
Consumer electronics manufacturers are increasingly exploring diversification of battery technologies to mitigate supply chain risks. This sector values the enhanced safety profiles of Na-ion and K-ion batteries, particularly for applications where thermal runaway risks must be minimized.
Regional market analysis reveals Asia-Pacific as the dominant manufacturing hub and potential largest market for post-lithium batteries, driven by China's aggressive development of sodium-ion technology. Europe follows with strong interest stemming from sustainability initiatives and raw material sovereignty concerns, while North America shows growing investment in research but lags in commercialization efforts.
Market adoption will be significantly influenced by regulatory frameworks, with the EU Battery Directive and similar regulations worldwide increasingly emphasizing sustainability metrics that favor Na-ion and K-ion technologies due to their reduced environmental footprint and reliance on more abundant resources.
Cost sensitivity analysis indicates that markets with high price elasticity will likely be early adopters of these technologies. The projected 20-30% cost advantage of Na-ion batteries over lithium-ion counterparts (when accounting for total cost of ownership) represents a compelling value proposition for price-sensitive segments, potentially accelerating market penetration beyond current forecasts.
Current Technical Challenges in K-ion and Na-ion Batteries
Both potassium-ion (K-ion) and sodium-ion (Na-ion) batteries face significant technical challenges that currently limit their widespread commercial adoption despite their promising potential as alternatives to lithium-ion batteries. One of the primary challenges for K-ion batteries relates to electrode materials. The larger ionic radius of potassium (1.38 Å) compared to lithium (0.76 Å) causes substantial volume changes during charge-discharge cycles, leading to structural instability and rapid capacity fading. This volume expansion can reach up to 400% in some anode materials, causing mechanical stress that degrades battery performance over time.
For Na-ion batteries, similar challenges exist with the sodium ion's larger radius (1.02 Å), though to a lesser extent than potassium. Both technologies struggle with finding suitable anode materials that can accommodate these larger ions while maintaining structural integrity. Graphite, the standard anode material for lithium-ion batteries, performs poorly with sodium ions due to insufficient interlayer spacing, while hard carbons show promise but suffer from low initial Coulombic efficiency.
Electrolyte stability presents another major challenge for both battery types. The highly reactive nature of potassium metal creates safety concerns and accelerates electrolyte decomposition. For Na-ion batteries, the solid electrolyte interphase (SEI) formation is less stable than in lithium-ion systems, leading to continuous electrolyte consumption and capacity loss. Researchers are exploring various electrolyte additives and formulations to enhance stability, but optimal solutions remain elusive.
Cathode materials for both technologies face challenges in achieving high energy density. Current K-ion cathodes typically deliver specific capacities below 140 mAh/g, significantly lower than commercial lithium-ion cathodes. Similarly, Na-ion cathodes struggle with lower operating voltages, reducing overall energy density. Prussian blue analogs show promise for both systems but suffer from moisture sensitivity and manufacturing challenges.
Manufacturing scalability represents a significant hurdle, particularly for K-ion batteries. The high reactivity of potassium requires stringent moisture-free production environments, increasing manufacturing complexity and cost. While Na-ion battery production can leverage existing lithium-ion manufacturing infrastructure, specialized equipment modifications are still necessary to handle the different chemical properties of sodium-based materials.
Cycle life limitations affect both technologies, with current prototypes typically achieving 500-1000 cycles before significant capacity degradation occurs—well below the 2000+ cycles of advanced lithium-ion batteries. This performance gap must be addressed before either technology can compete effectively in applications requiring long service life, such as grid storage or electric vehicles.
For Na-ion batteries, similar challenges exist with the sodium ion's larger radius (1.02 Å), though to a lesser extent than potassium. Both technologies struggle with finding suitable anode materials that can accommodate these larger ions while maintaining structural integrity. Graphite, the standard anode material for lithium-ion batteries, performs poorly with sodium ions due to insufficient interlayer spacing, while hard carbons show promise but suffer from low initial Coulombic efficiency.
Electrolyte stability presents another major challenge for both battery types. The highly reactive nature of potassium metal creates safety concerns and accelerates electrolyte decomposition. For Na-ion batteries, the solid electrolyte interphase (SEI) formation is less stable than in lithium-ion systems, leading to continuous electrolyte consumption and capacity loss. Researchers are exploring various electrolyte additives and formulations to enhance stability, but optimal solutions remain elusive.
Cathode materials for both technologies face challenges in achieving high energy density. Current K-ion cathodes typically deliver specific capacities below 140 mAh/g, significantly lower than commercial lithium-ion cathodes. Similarly, Na-ion cathodes struggle with lower operating voltages, reducing overall energy density. Prussian blue analogs show promise for both systems but suffer from moisture sensitivity and manufacturing challenges.
Manufacturing scalability represents a significant hurdle, particularly for K-ion batteries. The high reactivity of potassium requires stringent moisture-free production environments, increasing manufacturing complexity and cost. While Na-ion battery production can leverage existing lithium-ion manufacturing infrastructure, specialized equipment modifications are still necessary to handle the different chemical properties of sodium-based materials.
Cycle life limitations affect both technologies, with current prototypes typically achieving 500-1000 cycles before significant capacity degradation occurs—well below the 2000+ cycles of advanced lithium-ion batteries. This performance gap must be addressed before either technology can compete effectively in applications requiring long service life, such as grid storage or electric vehicles.
Comparative Analysis of Current K-ion and Na-ion Solutions
01 Electrode materials for sodium-ion batteries
Various materials are being developed for sodium-ion battery electrodes to improve performance and reduce costs. These include novel cathode materials, anode compositions, and electrode structures that enhance sodium ion storage capacity and cycling stability. Research focuses on materials that can effectively intercalate sodium ions while maintaining structural integrity during charge-discharge cycles, addressing the larger ionic radius of sodium compared to lithium.- Electrode materials for sodium-ion batteries: Various materials are being developed for sodium-ion battery electrodes to improve performance and reduce costs. These include carbon-based materials, transition metal oxides, and phosphates that can effectively store and release sodium ions. These materials offer advantages such as abundant raw material supply, good cycling stability, and high energy density, making sodium-ion batteries a cost-effective alternative to lithium-ion batteries for certain applications.
- Potassium-ion battery technology advancements: Potassium-ion batteries represent an emerging technology with potential cost advantages due to the abundance of potassium resources. Recent advancements focus on developing suitable electrode materials that can accommodate the larger potassium ions, improving electrolyte formulations to enhance ionic conductivity, and designing battery structures that maintain stability during cycling. These developments aim to position potassium-ion batteries as viable alternatives to lithium-ion batteries.
- Cost reduction strategies for alkali metal-ion batteries: Several approaches are being explored to reduce the cost of potassium-ion and sodium-ion batteries. These include using earth-abundant materials instead of rare metals, simplifying manufacturing processes, improving battery cycle life to reduce lifetime costs, and developing scalable production methods. The cost advantage of these batteries stems primarily from the lower raw material costs compared to lithium-ion batteries, making them potentially suitable for large-scale energy storage applications.
- Electrolyte innovations for alkali metal-ion batteries: Electrolyte formulations play a crucial role in the performance and safety of potassium-ion and sodium-ion batteries. Recent innovations include developing non-flammable electrolytes, optimizing salt concentrations to improve ionic conductivity, and creating electrolyte additives that form stable solid-electrolyte interfaces. These advancements help address challenges related to dendrite formation, side reactions, and overall battery stability, contributing to longer cycle life and improved safety profiles.
- Comparative performance and applications of alternative battery technologies: Research comparing potassium-ion and sodium-ion batteries with conventional lithium-ion batteries reveals distinct advantages and limitations for different applications. While lithium-ion batteries generally offer higher energy density, sodium and potassium-based alternatives can provide cost benefits for stationary energy storage, grid applications, and other uses where energy density is less critical than cost. The selection between these technologies depends on specific application requirements, including cycling needs, temperature conditions, and economic constraints.
02 Potassium-ion battery technology advancements
Potassium-ion batteries represent an emerging alternative to lithium-ion batteries, with research focusing on developing suitable electrode materials and electrolytes. These batteries leverage the abundance and lower cost of potassium resources while addressing challenges related to the large ionic radius of potassium. Recent advancements include novel frameworks for potassium ion storage, improved cycling performance, and enhanced energy density through material innovations.Expand Specific Solutions03 Cost reduction strategies for alkali metal-ion batteries
Various approaches are being developed to reduce the manufacturing and material costs of sodium and potassium-ion batteries. These include utilizing earth-abundant materials, simplifying production processes, recycling battery components, and developing scalable manufacturing techniques. Cost advantages over lithium-ion batteries are achieved through the use of more widely available resources and potentially simpler cell designs that don't require expensive materials like cobalt.Expand Specific Solutions04 Electrolyte formulations for enhanced performance
Advanced electrolyte formulations are critical for improving the performance of sodium and potassium-ion batteries. Research focuses on developing electrolytes that provide high ionic conductivity, wide electrochemical stability windows, and compatibility with electrode materials. Novel electrolyte compositions include optimized salt concentrations, solvent mixtures, and additives that form stable solid-electrolyte interphases, reducing unwanted side reactions and improving battery cycle life and safety.Expand Specific Solutions05 Battery system integration and comparative performance
Integration of sodium and potassium-ion batteries into practical energy storage systems requires addressing challenges related to cell design, thermal management, and battery management systems. Research compares the performance metrics of these emerging battery technologies with conventional lithium-ion batteries, including energy density, power capability, cycle life, and temperature performance. Studies also evaluate the economic and environmental impacts of deploying these alternative battery technologies at scale.Expand Specific Solutions
Key Industry Players in K-ion and Na-ion Battery Development
The potassium-ion versus sodium-ion battery technology landscape is currently in an early growth phase, with a market size expanding but still modest compared to lithium-ion technologies. Technologically, sodium-ion batteries are showing greater commercial maturity, with companies like Faradion Ltd. and Shenzhen Zhenhua New Materials achieving early market penetration. Research institutions including Ningbo University, Tongji University, and Centre National de la Recherche Scientifique are advancing fundamental science in both technologies. Major industrial players such as POSCO Holdings, Toyota, and GEM Co. are strategically positioning in this space, particularly focusing on sodium-ion as a near-term alternative. Cost advantages over lithium-ion technologies represent the primary competitive driver, with sodium-ion currently holding an edge in commercialization timeline while potassium-ion potentially offers superior long-term performance characteristics.
Faradion Ltd.
Technical Solution: Faradion has pioneered sodium-ion battery technology as a cost-effective alternative to lithium-ion batteries. Their proprietary Na-ion technology utilizes abundant, low-cost materials including sodium salts and hard carbon anodes derived from sustainable sources. The company has developed a layered oxide cathode chemistry (Na-Ni-Mn-Mg-Ti-O) that delivers energy densities approaching 160 Wh/kg at the cell level. Faradion's cells operate in the same voltage window as Li-ion (3-4.2V) allowing compatibility with existing manufacturing infrastructure. Their technology eliminates the need for copper current collectors, using aluminum instead for both electrodes, significantly reducing material costs. Faradion has demonstrated cycle life exceeding 1,000 cycles with 80% capacity retention and has successfully scaled production to multi-Ah pouch cells.
Strengths: Lower raw material costs (30-40% less than Li-ion), copper-free design, compatibility with existing manufacturing equipment, and use of abundant materials. Weaknesses: Lower energy density compared to advanced Li-ion cells, less mature supply chain, and still developing fast-charging capabilities.
Centre National de la Recherche Scientifique
Technical Solution: CNRS has developed advanced research on both potassium-ion and sodium-ion battery technologies through comprehensive comparative studies. Their potassium-ion battery research focuses on Prussian Blue Analogs (PBAs) as cathode materials, demonstrating capacities of 140 mAh/g with good cycling stability. For sodium-ion, CNRS has pioneered Na3V2(PO4)2F3 cathode materials achieving energy densities of approximately 140 Wh/kg. Their comparative analysis shows that while K-ion batteries benefit from higher operating voltages due to the lower standard potential of K+/K compared to Na+/Na, sodium-ion systems offer better cost economics with only 6-8% lower energy density. CNRS has also developed hard carbon anodes from biomass precursors for both systems, achieving reversible capacities of 300 mAh/g for Na-ion and 270 mAh/g for K-ion batteries. Their economic analysis indicates sodium-ion technology offers a 20-30% cost advantage over lithium-ion, while potassium-ion presents a 10-15% advantage.
Strengths: Comprehensive scientific understanding of both technologies, development of sustainable electrode materials, and detailed cost-performance analysis. Weaknesses: Research primarily at laboratory scale, limited industrial partnerships for commercialization, and challenges in translating academic research to manufacturing processes.
Critical Patents and Research Breakthroughs in Alkali Metal Batteries
Method for producing lithium-ion, sodium-ion and potassium-ion batteries with increased safety
PatentInactiveUS20190058193A1
Innovation
- The production of lithiated pyomelanin (LPM), sodiated pyomelanin (SPM), and potassiated pyomelanin (PPM) through dissolving melanin in alkaline solutions, reducing, titrating, dialyzing, and lyophilizing to create stable, non-aqueous battery components that control lithium ion release and reduce the risk of metallic lithium formation, thereby enhancing safety.
Perfusion medium
PatentWO2018178069A1
Innovation
- A serum-free perfusion medium with a potassium concentration of 30-250 mM and a sodium to potassium molar ratio of less than 1 is used to inhibit cell proliferation, reducing cell bleed and increasing protein production by maintaining a high viable cell density without affecting viability.
Cost Structure Analysis and Economic Viability Comparison
The cost structure of both potassium-ion batteries (PIBs) and sodium-ion batteries (SIBs) reveals significant differences that impact their economic viability in various applications. Raw material costs represent a fundamental advantage for both technologies compared to lithium-ion batteries (LIBs). Sodium resources are approximately 1,000 times more abundant than lithium, with global reserves widely distributed across multiple regions, reducing geopolitical supply risks. Similarly, potassium is the seventh most abundant element in the Earth's crust, with reserves estimated at 240 billion tons globally.
Manufacturing cost analysis indicates that SIBs can achieve production costs approximately 20-30% lower than conventional LIBs, primarily due to the substitution of copper current collectors with aluminum in both electrodes. PIBs maintain a similar cost advantage, with estimates suggesting 15-25% lower production costs compared to LIBs. However, PIBs currently face higher processing costs due to potassium's higher reactivity with electrolytes, requiring more sophisticated manufacturing environments.
Capital expenditure requirements for establishing production facilities show interesting patterns. Existing LIB manufacturing infrastructure can be modified for both SIB and PIB production with relatively modest investments, estimated at 30-40% of the cost of building new dedicated facilities. This adaptability significantly reduces market entry barriers and accelerates potential commercialization timelines.
Lifecycle economic assessment reveals that SIBs currently demonstrate better overall economic viability with an estimated levelized cost of storage (LCOS) of $0.12-0.18/kWh for grid applications, compared to $0.15-0.22/kWh for PIBs. This difference stems primarily from the more mature state of SIB technology and existing commercial deployment. However, PIBs show promising long-term economic potential due to their higher theoretical energy density capabilities.
Market sensitivity analysis indicates that both technologies' economic competitiveness is highly responsive to scale effects. Production volume increases from 1 GWh to 10 GWh annually could reduce costs by approximately 35-45% for both technologies. Additionally, the economic case for both battery types strengthens considerably in scenarios where lithium prices exceed $25/kg, creating natural market hedging opportunities.
Considering total cost of ownership across a 10-year operational period, SIBs currently offer better returns for stationary storage applications, while PIBs may eventually prove more economical for applications requiring higher energy density, such as certain electric vehicle segments, once technology maturation advances further.
Manufacturing cost analysis indicates that SIBs can achieve production costs approximately 20-30% lower than conventional LIBs, primarily due to the substitution of copper current collectors with aluminum in both electrodes. PIBs maintain a similar cost advantage, with estimates suggesting 15-25% lower production costs compared to LIBs. However, PIBs currently face higher processing costs due to potassium's higher reactivity with electrolytes, requiring more sophisticated manufacturing environments.
Capital expenditure requirements for establishing production facilities show interesting patterns. Existing LIB manufacturing infrastructure can be modified for both SIB and PIB production with relatively modest investments, estimated at 30-40% of the cost of building new dedicated facilities. This adaptability significantly reduces market entry barriers and accelerates potential commercialization timelines.
Lifecycle economic assessment reveals that SIBs currently demonstrate better overall economic viability with an estimated levelized cost of storage (LCOS) of $0.12-0.18/kWh for grid applications, compared to $0.15-0.22/kWh for PIBs. This difference stems primarily from the more mature state of SIB technology and existing commercial deployment. However, PIBs show promising long-term economic potential due to their higher theoretical energy density capabilities.
Market sensitivity analysis indicates that both technologies' economic competitiveness is highly responsive to scale effects. Production volume increases from 1 GWh to 10 GWh annually could reduce costs by approximately 35-45% for both technologies. Additionally, the economic case for both battery types strengthens considerably in scenarios where lithium prices exceed $25/kg, creating natural market hedging opportunities.
Considering total cost of ownership across a 10-year operational period, SIBs currently offer better returns for stationary storage applications, while PIBs may eventually prove more economical for applications requiring higher energy density, such as certain electric vehicle segments, once technology maturation advances further.
Raw Material Supply Chain and Sustainability Considerations
The raw material supply chains for potassium-ion batteries (PIBs) and sodium-ion batteries (SIBs) present significant differences that directly impact their economic viability and environmental footprint. Potassium resources are abundantly distributed globally, with approximately 240 billion tons available in seawater and 31 billion tons in the Earth's crust. This widespread availability translates to lower geopolitical risks compared to lithium, which is concentrated in the "Lithium Triangle" of South America and a few other regions.
Sodium resources are even more abundant than potassium, constituting about 2.6% of the Earth's crust and existing in virtually unlimited quantities in seawater. This exceptional abundance gives sodium-ion technology a distinct advantage in terms of raw material security and potentially lower extraction costs. The global distribution of sodium resources also minimizes supply chain vulnerabilities associated with geographically concentrated materials.
From a sustainability perspective, both PIBs and SIBs offer advantages over lithium-ion batteries (LIBs). The extraction of potassium and sodium generally has lower environmental impacts compared to lithium mining, which often involves extensive water usage in arid regions and potential contamination of local ecosystems. The carbon footprint of potassium and sodium extraction is estimated to be 30-40% lower than that of lithium, contributing to better lifecycle assessment outcomes.
Supply chain resilience represents another critical consideration. The established infrastructure for potassium extraction, primarily developed for fertilizer production, provides PIBs with an advantage in terms of immediate scalability. However, sodium's greater abundance may ultimately enable more diversified and robust supply networks as SIB technology matures and demand increases.
Processing requirements also differ between these materials. Potassium compounds typically require more energy-intensive purification processes than sodium compounds, potentially offsetting some of the cost advantages of potassium's established supply chain. Sodium salt purification generally involves simpler processes with lower energy requirements, contributing to potentially lower manufacturing costs for SIBs.
Recycling infrastructure represents a developing aspect of both technologies. Currently, neither PIBs nor SIBs benefit from the established recycling systems that exist for LIBs. However, the simpler chemistry and lower material value of both battery types may actually facilitate the development of more streamlined, cost-effective recycling processes in the future, further enhancing their sustainability credentials as the technologies mature and achieve greater market penetration.
Sodium resources are even more abundant than potassium, constituting about 2.6% of the Earth's crust and existing in virtually unlimited quantities in seawater. This exceptional abundance gives sodium-ion technology a distinct advantage in terms of raw material security and potentially lower extraction costs. The global distribution of sodium resources also minimizes supply chain vulnerabilities associated with geographically concentrated materials.
From a sustainability perspective, both PIBs and SIBs offer advantages over lithium-ion batteries (LIBs). The extraction of potassium and sodium generally has lower environmental impacts compared to lithium mining, which often involves extensive water usage in arid regions and potential contamination of local ecosystems. The carbon footprint of potassium and sodium extraction is estimated to be 30-40% lower than that of lithium, contributing to better lifecycle assessment outcomes.
Supply chain resilience represents another critical consideration. The established infrastructure for potassium extraction, primarily developed for fertilizer production, provides PIBs with an advantage in terms of immediate scalability. However, sodium's greater abundance may ultimately enable more diversified and robust supply networks as SIB technology matures and demand increases.
Processing requirements also differ between these materials. Potassium compounds typically require more energy-intensive purification processes than sodium compounds, potentially offsetting some of the cost advantages of potassium's established supply chain. Sodium salt purification generally involves simpler processes with lower energy requirements, contributing to potentially lower manufacturing costs for SIBs.
Recycling infrastructure represents a developing aspect of both technologies. Currently, neither PIBs nor SIBs benefit from the established recycling systems that exist for LIBs. However, the simpler chemistry and lower material value of both battery types may actually facilitate the development of more streamlined, cost-effective recycling processes in the future, further enhancing their sustainability credentials as the technologies mature and achieve greater market penetration.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!