Sample Tracking And Chain Of Custody In DNA Data Storage
AUG 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
DNA Data Storage Background and Objectives
DNA data storage represents a revolutionary approach to digital information preservation, emerging from the convergence of molecular biology and computer science. This technology leverages DNA's remarkable properties as a storage medium, including its density (capable of storing 455 exabytes per gram), longevity (potentially thousands of years under proper conditions), and energy efficiency. Since the initial demonstration by Church et al. in 2012, who encoded a 659-kilobyte book in DNA, the field has progressed significantly through contributions from academic institutions and technology companies.
The evolution of DNA data storage technology has followed a trajectory of increasing sophistication. Early approaches focused on basic encoding schemes, while recent advancements have addressed error correction, random access capabilities, and scalability challenges. The technology has transitioned from proof-of-concept demonstrations to increasingly practical implementations, with storage capacities growing from kilobytes to megabytes and potentially beyond.
Current technical objectives in the field center on developing robust sample tracking and chain of custody protocols. These systems are essential for ensuring data integrity, security, and accessibility throughout the DNA storage lifecycle. Without reliable tracking mechanisms, the potential of DNA storage cannot be fully realized in commercial or sensitive applications where data provenance is critical.
The specific goals for sample tracking in DNA data storage include establishing tamper-evident custody trails, implementing molecular-level authentication methods, and creating standardized protocols for sample handling. These objectives align with broader aims to make DNA storage commercially viable by addressing concerns related to data security, regulatory compliance, and integration with existing digital infrastructure.
Research efforts are increasingly focused on developing end-to-end solutions that maintain unbroken chains of custody from data encoding through storage and retrieval. This includes exploring blockchain integration for immutable record-keeping, developing DNA barcoding systems for sample identification, and creating physical-digital hybrid security measures to prevent unauthorized access or manipulation.
The technological trajectory suggests that DNA data storage is approaching an inflection point where laboratory demonstrations must evolve into industrial implementations. Sample tracking and chain of custody protocols represent a critical bridge in this transition, enabling the technology to meet the stringent requirements of enterprise data centers, national archives, and other high-value storage applications where data provenance cannot be compromised.
The evolution of DNA data storage technology has followed a trajectory of increasing sophistication. Early approaches focused on basic encoding schemes, while recent advancements have addressed error correction, random access capabilities, and scalability challenges. The technology has transitioned from proof-of-concept demonstrations to increasingly practical implementations, with storage capacities growing from kilobytes to megabytes and potentially beyond.
Current technical objectives in the field center on developing robust sample tracking and chain of custody protocols. These systems are essential for ensuring data integrity, security, and accessibility throughout the DNA storage lifecycle. Without reliable tracking mechanisms, the potential of DNA storage cannot be fully realized in commercial or sensitive applications where data provenance is critical.
The specific goals for sample tracking in DNA data storage include establishing tamper-evident custody trails, implementing molecular-level authentication methods, and creating standardized protocols for sample handling. These objectives align with broader aims to make DNA storage commercially viable by addressing concerns related to data security, regulatory compliance, and integration with existing digital infrastructure.
Research efforts are increasingly focused on developing end-to-end solutions that maintain unbroken chains of custody from data encoding through storage and retrieval. This includes exploring blockchain integration for immutable record-keeping, developing DNA barcoding systems for sample identification, and creating physical-digital hybrid security measures to prevent unauthorized access or manipulation.
The technological trajectory suggests that DNA data storage is approaching an inflection point where laboratory demonstrations must evolve into industrial implementations. Sample tracking and chain of custody protocols represent a critical bridge in this transition, enabling the technology to meet the stringent requirements of enterprise data centers, national archives, and other high-value storage applications where data provenance cannot be compromised.
Market Analysis for DNA Storage Solutions
The DNA data storage market is experiencing significant growth, driven by the exponential increase in global data production and the limitations of conventional storage technologies. Current projections indicate the global DNA data storage market could reach $3.5 billion by 2030, with a compound annual growth rate exceeding 55% between 2023 and 2030. This remarkable growth trajectory is fueled by the inherent advantages of DNA storage, including its unprecedented data density, longevity, and energy efficiency compared to traditional storage media.
The primary market segments for DNA data storage solutions include government archives, scientific research institutions, healthcare organizations, and large technology companies with massive data archiving needs. These sectors generate enormous volumes of data that require secure, long-term preservation, making them ideal early adopters for DNA storage technologies.
Sample tracking and chain of custody capabilities represent a critical value proposition within this emerging market. Organizations handling sensitive genetic information, clinical samples, or proprietary research data require robust tracking systems that maintain data integrity throughout the DNA storage workflow. This requirement creates a specialized sub-market estimated to comprise approximately 30% of the overall DNA storage solution ecosystem.
Market research indicates that customers are willing to pay premium prices for DNA storage solutions that incorporate advanced sample tracking and chain of custody features. The average price premium for such enhanced security features ranges between 15-25% above basic DNA storage offerings, reflecting the high value placed on data integrity and security.
Geographically, North America currently leads the market with approximately 45% share, followed by Europe at 30% and Asia-Pacific at 20%. This distribution aligns with regional investments in biotechnology infrastructure and data security regulations. Countries with stringent data protection laws, such as those in the European Union under GDPR, show particularly strong demand for comprehensive chain of custody solutions.
The competitive landscape features both established biotechnology companies expanding into data storage and specialized startups focused exclusively on DNA-based information systems. Key market players include Twist Bioscience, Illumina, Microsoft Research, Catalog Technologies, and emerging players like Iridia and DNA Script. These companies are increasingly forming strategic partnerships with cybersecurity firms to enhance their sample tracking and chain of custody capabilities.
Customer adoption barriers include current high costs, technical complexity, and concerns about data security during the encoding and retrieval processes. Market surveys indicate that 78% of potential enterprise customers cite chain of custody verification as a "very important" or "critical" factor in their evaluation of DNA storage solutions, underscoring the market opportunity for innovations in this space.
The primary market segments for DNA data storage solutions include government archives, scientific research institutions, healthcare organizations, and large technology companies with massive data archiving needs. These sectors generate enormous volumes of data that require secure, long-term preservation, making them ideal early adopters for DNA storage technologies.
Sample tracking and chain of custody capabilities represent a critical value proposition within this emerging market. Organizations handling sensitive genetic information, clinical samples, or proprietary research data require robust tracking systems that maintain data integrity throughout the DNA storage workflow. This requirement creates a specialized sub-market estimated to comprise approximately 30% of the overall DNA storage solution ecosystem.
Market research indicates that customers are willing to pay premium prices for DNA storage solutions that incorporate advanced sample tracking and chain of custody features. The average price premium for such enhanced security features ranges between 15-25% above basic DNA storage offerings, reflecting the high value placed on data integrity and security.
Geographically, North America currently leads the market with approximately 45% share, followed by Europe at 30% and Asia-Pacific at 20%. This distribution aligns with regional investments in biotechnology infrastructure and data security regulations. Countries with stringent data protection laws, such as those in the European Union under GDPR, show particularly strong demand for comprehensive chain of custody solutions.
The competitive landscape features both established biotechnology companies expanding into data storage and specialized startups focused exclusively on DNA-based information systems. Key market players include Twist Bioscience, Illumina, Microsoft Research, Catalog Technologies, and emerging players like Iridia and DNA Script. These companies are increasingly forming strategic partnerships with cybersecurity firms to enhance their sample tracking and chain of custody capabilities.
Customer adoption barriers include current high costs, technical complexity, and concerns about data security during the encoding and retrieval processes. Market surveys indicate that 78% of potential enterprise customers cite chain of custody verification as a "very important" or "critical" factor in their evaluation of DNA storage solutions, underscoring the market opportunity for innovations in this space.
Technical Challenges in Sample Tracking
DNA data storage systems face significant technical challenges in sample tracking and chain of custody management. The complexity begins with the physical nature of DNA samples, which require specialized handling procedures throughout their lifecycle. These samples are susceptible to contamination, degradation, and cross-sample interference, necessitating robust isolation protocols that maintain sample integrity while enabling efficient processing workflows.
Current tracking systems struggle with the unique identification requirements of DNA-based data. Unlike traditional digital storage media, DNA samples lack standardized physical interfaces or built-in addressing mechanisms. This fundamental difference creates challenges in developing reliable tagging methodologies that can withstand biochemical processes while remaining machine-readable throughout multiple synthesis, storage, and sequencing cycles.
The scale of DNA data storage compounds these challenges considerably. As systems move toward practical implementation, they must handle potentially millions of distinct DNA fragments, each representing portions of the stored data. Traditional laboratory sample management approaches become inadequate at this scale, creating bottlenecks in throughput and increasing error probabilities during handling operations.
Error detection and correction mechanisms present another significant hurdle. While computational approaches exist for detecting data corruption, physically tracking the provenance and handling history of samples requires novel solutions that bridge molecular biology and information technology domains. Current systems lack integrated approaches that can validate both data integrity and sample handling compliance simultaneously.
Regulatory frameworks add another layer of complexity to sample tracking solutions. As DNA data storage moves toward commercial applications, systems must comply with diverse regulations governing biological materials, data privacy, and chain of custody documentation. These requirements vary across jurisdictions and application domains, necessitating flexible yet comprehensive tracking architectures.
Interoperability between different laboratory systems represents a persistent challenge. The DNA data storage workflow typically spans multiple specialized equipment platforms from different manufacturers, each with proprietary data formats and control systems. Developing unified tracking protocols that maintain continuity across these heterogeneous environments requires significant standardization efforts that the industry has yet to fully address.
Authentication mechanisms for verifying sample identity and handling authorization remain underdeveloped. Unlike digital systems with established cryptographic solutions, DNA-based authentication must function within the constraints of biochemical processes while providing comparable security guarantees. Current approaches often sacrifice either security rigor or practical usability, creating vulnerabilities in the chain of custody.
Human Engineering
Current tracking systems struggle with the unique identification requirements of DNA-based data. Unlike traditional digital storage media, DNA samples lack standardized physical interfaces or built-in addressing mechanisms. This fundamental difference creates challenges in developing reliable tagging methodologies that can withstand biochemical processes while remaining machine-readable throughout multiple synthesis, storage, and sequencing cycles.
The scale of DNA data storage compounds these challenges considerably. As systems move toward practical implementation, they must handle potentially millions of distinct DNA fragments, each representing portions of the stored data. Traditional laboratory sample management approaches become inadequate at this scale, creating bottlenecks in throughput and increasing error probabilities during handling operations.
Error detection and correction mechanisms present another significant hurdle. While computational approaches exist for detecting data corruption, physically tracking the provenance and handling history of samples requires novel solutions that bridge molecular biology and information technology domains. Current systems lack integrated approaches that can validate both data integrity and sample handling compliance simultaneously.
Regulatory frameworks add another layer of complexity to sample tracking solutions. As DNA data storage moves toward commercial applications, systems must comply with diverse regulations governing biological materials, data privacy, and chain of custody documentation. These requirements vary across jurisdictions and application domains, necessitating flexible yet comprehensive tracking architectures.
Interoperability between different laboratory systems represents a persistent challenge. The DNA data storage workflow typically spans multiple specialized equipment platforms from different manufacturers, each with proprietary data formats and control systems. Developing unified tracking protocols that maintain continuity across these heterogeneous environments requires significant standardization efforts that the industry has yet to fully address.
Authentication mechanisms for verifying sample identity and handling authorization remain underdeveloped. Unlike digital systems with established cryptographic solutions, DNA-based authentication must function within the constraints of biochemical processes while providing comparable security guarantees. Current approaches often sacrifice either security rigor or practical usability, creating vulnerabilities in the chain of custody.
Human Engineering
Current Chain of Custody Methodologies
01 DNA-based tracking and authentication systems
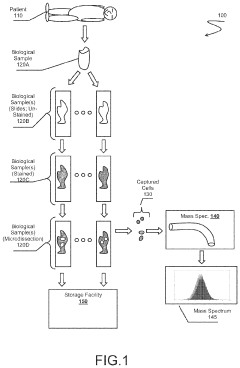
DNA can be used as a unique identifier for tracking and authenticating samples throughout their lifecycle. These systems incorporate DNA markers or signatures that can be embedded into samples, allowing for verification of origin and maintaining chain of custody. The DNA signatures provide tamper-evident security features that are difficult to counterfeit, ensuring sample integrity and authenticity throughout the tracking process.- DNA-based tracking systems for sample management: DNA-based technologies can be implemented for tracking biological samples throughout their lifecycle. These systems encode unique identifiers into DNA molecules that can be attached to or embedded within samples, providing tamper-evident tracking capabilities. The DNA markers serve as molecular barcodes that can be read at any point in the chain of custody, ensuring sample authenticity and preventing unauthorized substitution or tampering. These systems are particularly valuable in forensic applications, clinical trials, and biobanking where sample integrity is critical.
- Blockchain integration with DNA data storage for chain of custody: Blockchain technology can be integrated with DNA data storage systems to create immutable records of sample handling and transfers. Each transaction or access event is recorded as a block in the chain, creating a transparent, distributed ledger that cannot be altered retroactively. This approach provides cryptographic verification of each step in the sample's journey, from collection to analysis and storage. The combination of DNA's physical stability with blockchain's digital immutability creates a robust system for maintaining chain of custody in sensitive applications like clinical research, forensics, and pharmaceutical development.
- RFID and barcode systems for DNA sample tracking: Radio-frequency identification (RFID) tags and barcode systems can be implemented for tracking DNA samples and associated data throughout laboratory workflows. These technologies allow for automated scanning and recording of sample movements, reducing human error and creating digital audit trails. Advanced systems may incorporate temperature sensors to monitor storage conditions and ensure sample integrity. When combined with secure database systems, these tracking methods enable real-time location monitoring and comprehensive chain of custody documentation for DNA samples in research, clinical, and forensic settings.
- Secure database systems for DNA sample custody management: Specialized database systems can be implemented to manage the chain of custody for DNA samples and associated data. These systems incorporate access controls, encryption, and comprehensive audit logging to track who accessed samples or data, when, and for what purpose. They may include features for managing consent, documenting sample processing steps, and maintaining regulatory compliance. Some implementations use distributed database architectures to prevent single points of failure or tampering, ensuring that custody records remain intact and verifiable throughout the sample lifecycle.
- Biometric authentication for DNA sample access control: Biometric authentication systems can be integrated into DNA sample tracking workflows to verify the identity of personnel handling sensitive materials. These systems may use fingerprints, facial recognition, iris scans, or other unique biological identifiers to ensure that only authorized individuals can access samples or modify custody records. By creating an additional layer of security beyond traditional passwords or key cards, biometric controls help prevent unauthorized sample access and maintain the integrity of the chain of custody. This approach is particularly valuable in forensic applications where evidence handling must meet strict legal standards.
02 Blockchain technology for secure DNA data storage
Blockchain technology provides a secure, immutable ledger system for tracking DNA samples and maintaining chain of custody records. By recording each transaction or transfer of a DNA sample on a blockchain, organizations can create an unalterable audit trail that verifies sample handling history. This approach enhances security, transparency, and trust in the chain of custody process while preventing unauthorized modifications to sample tracking data.Expand Specific Solutions03 RFID and electronic tracking systems for DNA samples
Radio-frequency identification (RFID) and other electronic tracking technologies can be integrated with DNA sample management systems to automate and secure the chain of custody process. These systems use electronic tags, barcodes, or other identifiers to track sample containers throughout their journey, recording handling events, timestamps, and personnel information. The electronic tracking provides real-time visibility and reduces the risk of human error in the documentation process.Expand Specific Solutions04 Database management systems for DNA sample tracking
Specialized database management systems are designed to store, organize, and manage information related to DNA samples throughout their lifecycle. These systems maintain comprehensive records of sample collection, processing, storage, and transfer events, creating a complete chain of custody documentation. Advanced database architectures incorporate security features, access controls, and audit trails to ensure data integrity and compliance with regulatory requirements.Expand Specific Solutions05 Integrated biometric and DNA sample management
Systems that combine biometric identification technologies with DNA sample tracking provide enhanced security and authentication throughout the chain of custody process. These integrated approaches link the identity of handlers to specific sample management actions through fingerprints, facial recognition, or other biometric markers. By creating this connection between personnel and samples, organizations can verify who accessed samples at each stage, reducing the risk of tampering or unauthorized access.Expand Specific Solutions
Key Industry Players and Competitors
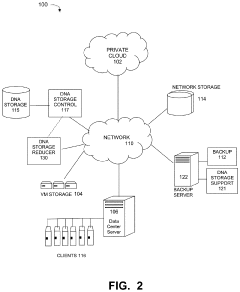
The DNA data storage market is in an early development stage, characterized by significant research activity but limited commercial deployment. The global market size remains relatively small, estimated at under $100 million, but with projected rapid growth as the technology matures. Key players represent diverse sectors: technology giants (Microsoft, Huawei), specialized biotechnology firms (Iridia, Molecular Assemblies), and research institutions (Tsinghua University, BGI Research). The technology maturity varies across the sample tracking and chain of custody aspects, with companies like SampleServe and Genisyss focusing on sample management systems, while Microsoft and Iridia are developing fundamental DNA storage technologies. Western Digital and QIAGEN are leveraging their expertise in data storage and biotechnology respectively to address the emerging challenges in DNA data custody and tracking.
Microsoft Technology Licensing LLC
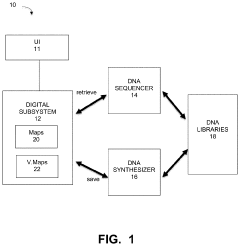
Technical Solution: Microsoft has pioneered significant advancements in DNA data storage with their comprehensive sample tracking and chain of custody system. Their solution integrates blockchain technology to create immutable records of DNA sample handling, ensuring data integrity throughout the storage lifecycle. Microsoft's platform employs a multi-layered authentication system that requires biometric verification at critical handling points, generating cryptographically signed timestamps for each sample transfer. Their proprietary encoding algorithms allow for error detection and correction capabilities specifically designed for the unique challenges of DNA storage media. The system includes automated sample indexing that correlates physical DNA samples with their digital counterparts through QR-coded microfluidic containers, enabling real-time location tracking and handling history for each sample. Microsoft has demonstrated storage densities of 1 exabyte per cubic inch with their DNA storage technology, while maintaining complete chain of custody records.
Strengths: Industry-leading integration with cloud infrastructure provides seamless data management across physical and digital domains. Their established security protocols from enterprise systems have been effectively adapted to biological sample tracking. Weaknesses: The system requires significant computational resources for blockchain verification, potentially limiting deployment in resource-constrained environments. Higher implementation costs compared to traditional sample tracking methods.
Iridia, Inc.
Technical Solution: Iridia has developed a specialized DNA data storage platform with integrated sample tracking and chain of custody features designed specifically for long-term archival applications. Their system utilizes electronic nanopore technology combined with proprietary molecular identifiers to create a seamless tracking solution from DNA synthesis through storage and retrieval. Iridia's platform incorporates a multi-tiered authentication system that combines physical security features with molecular verification methods, ensuring sample integrity throughout the storage lifecycle. Their "DNA Fingerprint" technology embeds cryptographic signatures within non-coding regions of DNA data strands, allowing for sample authentication without compromising stored data. The company has implemented a comprehensive environmental monitoring system that continuously tracks temperature, humidity, and other critical parameters, with automatic alerts for any conditions that might compromise sample integrity. Iridia's chain of custody solution includes detailed documentation of all sample handling events, with secure digital records that meet regulatory requirements for sensitive data storage applications.
Strengths: Purpose-built system specifically designed for DNA data storage applications provides optimized workflows and specialized tracking features. Their electronic nanopore technology enables rapid verification of sample identity without destructive testing. Weaknesses: As a smaller company, they may have limited resources for large-scale deployment and support compared to larger competitors. Their specialized approach may create compatibility challenges with existing laboratory information management systems.
Critical Patents in DNA Sample Tracking
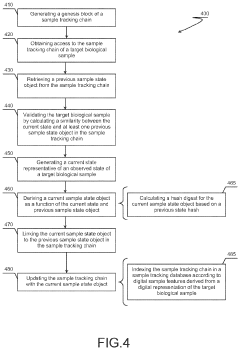
Sample Tracking Via Sample Tracking Chains, Systems And Methods
PatentPendingUS20240242795A1
Innovation
- A biological sample tracking system that utilizes intrinsic properties of the sample to generate a digital chain of states, such as a blockchain, to create a sample-specific audit trail, allowing for accurate and reliable tracking through a workflow without relying solely on extrinsic information.
Storing digital data in DNA storage using blockchain and destination-side deduplication using smart contracts
PatentPendingUS20220237470A1
Innovation
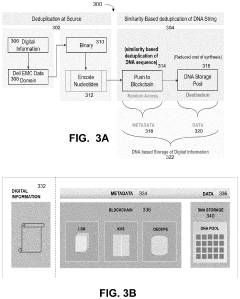
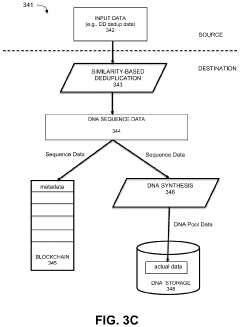
- Implementing a system that uses blockchain for decentralized, immutable storage of metadata and smart contracts for similarity-based deduplication, combined with locality similarity hashing (LSH) for random access and delta encoding, to reduce the number of nucleotides required for data representation and enhance data security.
Regulatory Framework for Biological Data Storage
The regulatory landscape for DNA data storage systems presents a complex intersection of data protection laws, biosafety regulations, and emerging standards specific to biological computing. Current frameworks primarily address either traditional electronic data storage or biological materials separately, creating significant regulatory gaps for this hybrid technology.
In the United States, the FDA has begun developing preliminary guidelines for biological data storage systems, focusing on containment protocols and potential environmental impacts of engineered DNA. These guidelines emphasize the need for robust sample tracking and chain of custody documentation throughout the data lifecycle. The European Union's approach under the GDPR considers DNA-stored data as potentially containing personal information, requiring enhanced security measures and explicit consent mechanisms when human-derived sequences are involved.
International standards bodies, including ISO and IEEE, are developing specialized protocols for DNA data storage, with working groups focused specifically on chain of custody requirements. The proposed ISO/IEC 23092 standard includes provisions for maintaining verifiable records of sample handling, processing, and storage conditions that could significantly impact data integrity.
Biosafety regulations present another critical dimension, with most jurisdictions requiring registration and monitoring of facilities handling synthetic DNA above certain complexity thresholds. The International Gene Synthesis Consortium has established screening protocols that may apply to DNA data storage implementations, particularly when storing potentially sensitive information.
Emerging regulatory considerations include the need for specialized audit trails that document both physical sample handling and digital encoding/decoding processes. Several jurisdictions are exploring blockchain-based verification systems to maintain immutable records of sample custody throughout the DNA data lifecycle.
Industry self-regulation has emerged as an interim solution, with the DNA Data Storage Alliance proposing a comprehensive framework for sample tracking that includes standardized labeling, transfer documentation, and custody verification protocols. These voluntary standards often exceed current regulatory requirements but provide valuable guidance for organizations implementing DNA storage solutions.
The regulatory environment remains highly dynamic, with significant variations between jurisdictions creating compliance challenges for global implementations. Organizations developing DNA data storage systems must establish robust tracking mechanisms that can adapt to evolving requirements while maintaining the chain of custody integrity essential for data validation and regulatory acceptance.
In the United States, the FDA has begun developing preliminary guidelines for biological data storage systems, focusing on containment protocols and potential environmental impacts of engineered DNA. These guidelines emphasize the need for robust sample tracking and chain of custody documentation throughout the data lifecycle. The European Union's approach under the GDPR considers DNA-stored data as potentially containing personal information, requiring enhanced security measures and explicit consent mechanisms when human-derived sequences are involved.
International standards bodies, including ISO and IEEE, are developing specialized protocols for DNA data storage, with working groups focused specifically on chain of custody requirements. The proposed ISO/IEC 23092 standard includes provisions for maintaining verifiable records of sample handling, processing, and storage conditions that could significantly impact data integrity.
Biosafety regulations present another critical dimension, with most jurisdictions requiring registration and monitoring of facilities handling synthetic DNA above certain complexity thresholds. The International Gene Synthesis Consortium has established screening protocols that may apply to DNA data storage implementations, particularly when storing potentially sensitive information.
Emerging regulatory considerations include the need for specialized audit trails that document both physical sample handling and digital encoding/decoding processes. Several jurisdictions are exploring blockchain-based verification systems to maintain immutable records of sample custody throughout the DNA data lifecycle.
Industry self-regulation has emerged as an interim solution, with the DNA Data Storage Alliance proposing a comprehensive framework for sample tracking that includes standardized labeling, transfer documentation, and custody verification protocols. These voluntary standards often exceed current regulatory requirements but provide valuable guidance for organizations implementing DNA storage solutions.
The regulatory environment remains highly dynamic, with significant variations between jurisdictions creating compliance challenges for global implementations. Organizations developing DNA data storage systems must establish robust tracking mechanisms that can adapt to evolving requirements while maintaining the chain of custody integrity essential for data validation and regulatory acceptance.
Security and Privacy Considerations
DNA data storage systems present unique security and privacy challenges that extend beyond traditional digital storage methods. The biological nature of DNA as a storage medium introduces novel vulnerabilities that must be addressed through comprehensive security frameworks. DNA samples containing encoded data require protection against unauthorized access, tampering, and theft throughout their lifecycle - from initial encoding to long-term storage and eventual retrieval.
Encryption methodologies for DNA data storage must be adapted to accommodate the constraints of biological systems while maintaining robust security. Current approaches include molecular-level encryption techniques that integrate cryptographic principles directly into the DNA encoding process. These methods ensure that even if physical access to DNA samples is obtained, the data remains indecipherable without proper decryption keys.
Privacy considerations are particularly critical when DNA storage systems potentially contain sensitive information. The persistence of DNA molecules raises concerns about long-term data exposure risks, as encoded information could theoretically remain intact for thousands of years. This temporal dimension necessitates forward-thinking privacy protocols that anticipate future technological capabilities for DNA sequencing and decryption.
Biohacking represents an emerging threat vector specific to DNA data storage. Malicious actors could potentially introduce synthetic DNA sequences designed to compromise storage systems or extract unauthorized information. Defensive measures must include biological authentication mechanisms and contamination detection protocols to maintain data integrity.
Regulatory frameworks for DNA data storage security remain underdeveloped, creating compliance uncertainties for organizations implementing these technologies. International standards bodies are beginning to address this gap by developing specialized guidelines that account for the unique characteristics of biological storage media.
Access control systems for DNA repositories require multi-layered approaches combining physical security, biometric authentication, and digital safeguards. The chain of custody documentation must be tamper-evident and cryptographically secured to prevent unauthorized manipulation of access records.
Quantum computing poses a significant future threat to current DNA encryption methods. As quantum capabilities advance, organizations must develop quantum-resistant encryption protocols specifically designed for biological storage systems to ensure long-term data protection.
Encryption methodologies for DNA data storage must be adapted to accommodate the constraints of biological systems while maintaining robust security. Current approaches include molecular-level encryption techniques that integrate cryptographic principles directly into the DNA encoding process. These methods ensure that even if physical access to DNA samples is obtained, the data remains indecipherable without proper decryption keys.
Privacy considerations are particularly critical when DNA storage systems potentially contain sensitive information. The persistence of DNA molecules raises concerns about long-term data exposure risks, as encoded information could theoretically remain intact for thousands of years. This temporal dimension necessitates forward-thinking privacy protocols that anticipate future technological capabilities for DNA sequencing and decryption.
Biohacking represents an emerging threat vector specific to DNA data storage. Malicious actors could potentially introduce synthetic DNA sequences designed to compromise storage systems or extract unauthorized information. Defensive measures must include biological authentication mechanisms and contamination detection protocols to maintain data integrity.
Regulatory frameworks for DNA data storage security remain underdeveloped, creating compliance uncertainties for organizations implementing these technologies. International standards bodies are beginning to address this gap by developing specialized guidelines that account for the unique characteristics of biological storage media.
Access control systems for DNA repositories require multi-layered approaches combining physical security, biometric authentication, and digital safeguards. The chain of custody documentation must be tamper-evident and cryptographically secured to prevent unauthorized manipulation of access records.
Quantum computing poses a significant future threat to current DNA encryption methods. As quantum capabilities advance, organizations must develop quantum-resistant encryption protocols specifically designed for biological storage systems to ensure long-term data protection.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!