Using Barium Hydroxide for Acid Deposition Mitigation Strategies
AUG 1, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Acid Deposition Mitigation Background and Objectives
Acid deposition, commonly known as acid rain, has been a significant environmental concern since the industrial revolution. This phenomenon occurs when sulfur dioxide and nitrogen oxides, primarily emitted from industrial processes and fossil fuel combustion, react with water, oxygen, and other chemicals in the atmosphere. The resulting acidic compounds fall to Earth in various forms, including rain, snow, and fog, causing widespread damage to ecosystems, infrastructure, and human health.
The history of acid deposition research dates back to the 1850s when Robert Angus Smith first observed the relationship between air pollution and acid rain in Manchester, England. However, it wasn't until the 1960s and 1970s that the issue gained widespread attention, particularly in North America and Europe, as scientists began to document the extensive environmental impacts of acid deposition on forests, lakes, and soil.
Over the past few decades, significant efforts have been made to mitigate acid deposition through various strategies, including emissions reduction, use of scrubbers in industrial chimneys, and implementation of stricter environmental regulations. Despite these efforts, acid deposition remains a persistent problem in many parts of the world, necessitating continued research and innovative solutions.
The use of barium hydroxide for acid deposition mitigation represents a novel approach in addressing this long-standing environmental challenge. Barium hydroxide, a strong alkaline compound, has the potential to neutralize acidic substances effectively. Its application in acid deposition mitigation strategies aims to counteract the harmful effects of acidic compounds in the environment, particularly in sensitive ecosystems and areas with high acid deposition rates.
The primary objectives of exploring barium hydroxide as a mitigation strategy are multifaceted. Firstly, researchers aim to assess its efficacy in neutralizing acidic compounds in various environmental matrices, including soil, water bodies, and atmospheric deposition. Secondly, there is a need to evaluate the long-term environmental impacts and sustainability of using barium hydroxide, considering factors such as ecological effects, potential side reactions, and economic viability.
Furthermore, the research seeks to optimize application methods and dosages for maximum effectiveness while minimizing any potential negative impacts. This includes investigating different forms of barium hydroxide application, such as direct soil treatment, water body addition, or atmospheric dispersion techniques. Additionally, the study aims to compare the effectiveness of barium hydroxide with other established acid deposition mitigation methods, providing a comprehensive analysis of its potential as a viable solution in the ongoing battle against acid rain and its detrimental effects on the environment.
The history of acid deposition research dates back to the 1850s when Robert Angus Smith first observed the relationship between air pollution and acid rain in Manchester, England. However, it wasn't until the 1960s and 1970s that the issue gained widespread attention, particularly in North America and Europe, as scientists began to document the extensive environmental impacts of acid deposition on forests, lakes, and soil.
Over the past few decades, significant efforts have been made to mitigate acid deposition through various strategies, including emissions reduction, use of scrubbers in industrial chimneys, and implementation of stricter environmental regulations. Despite these efforts, acid deposition remains a persistent problem in many parts of the world, necessitating continued research and innovative solutions.
The use of barium hydroxide for acid deposition mitigation represents a novel approach in addressing this long-standing environmental challenge. Barium hydroxide, a strong alkaline compound, has the potential to neutralize acidic substances effectively. Its application in acid deposition mitigation strategies aims to counteract the harmful effects of acidic compounds in the environment, particularly in sensitive ecosystems and areas with high acid deposition rates.
The primary objectives of exploring barium hydroxide as a mitigation strategy are multifaceted. Firstly, researchers aim to assess its efficacy in neutralizing acidic compounds in various environmental matrices, including soil, water bodies, and atmospheric deposition. Secondly, there is a need to evaluate the long-term environmental impacts and sustainability of using barium hydroxide, considering factors such as ecological effects, potential side reactions, and economic viability.
Furthermore, the research seeks to optimize application methods and dosages for maximum effectiveness while minimizing any potential negative impacts. This includes investigating different forms of barium hydroxide application, such as direct soil treatment, water body addition, or atmospheric dispersion techniques. Additionally, the study aims to compare the effectiveness of barium hydroxide with other established acid deposition mitigation methods, providing a comprehensive analysis of its potential as a viable solution in the ongoing battle against acid rain and its detrimental effects on the environment.
Market Analysis for Acid Rain Neutralization Solutions
The market for acid rain neutralization solutions has been experiencing steady growth due to increasing environmental concerns and stringent regulations aimed at reducing the impact of acid deposition. Barium hydroxide, as a potential solution for acid rain mitigation, is positioned within a broader market of environmental remediation technologies.
The global market for acid rain neutralization solutions is primarily driven by the power generation, manufacturing, and transportation sectors, which are major contributors to sulfur dioxide and nitrogen oxide emissions. These industries are under pressure to adopt cleaner technologies and mitigation strategies, creating a significant demand for effective neutralization solutions.
In recent years, there has been a shift towards more sustainable and cost-effective acid rain mitigation strategies. This trend has opened up opportunities for innovative solutions like barium hydroxide, which offers unique advantages in terms of neutralization capacity and potential for byproduct recovery.
The market for acid rain neutralization solutions is geographically diverse, with North America, Europe, and Asia-Pacific regions being the key markets. Developing economies in Asia, particularly China and India, are expected to witness substantial growth in demand due to rapid industrialization and increasing environmental awareness.
Barium hydroxide, as a specific solution for acid deposition mitigation, competes with other neutralizing agents such as limestone, lime, and sodium bicarbonate. Its market potential is influenced by factors such as effectiveness, cost, availability, and ease of application. While barium hydroxide offers high neutralization capacity, its market adoption may be affected by concerns related to barium toxicity and the need for careful handling.
The regulatory landscape plays a crucial role in shaping the market for acid rain neutralization solutions. Stringent environmental regulations in developed countries and emerging policies in developing nations are expected to drive the adoption of effective mitigation strategies, potentially benefiting solutions like barium hydroxide.
Market growth is also influenced by technological advancements in application methods and monitoring systems. Innovations in delivery systems and real-time pH monitoring can enhance the efficiency and precision of acid rain neutralization processes, potentially increasing the attractiveness of barium hydroxide as a solution.
As sustainability becomes a key focus for industries worldwide, there is growing interest in solutions that not only neutralize acid rain but also offer additional environmental benefits. Barium hydroxide's potential for byproduct recovery and its ability to sequester carbon dioxide could position it favorably in this evolving market landscape.
The global market for acid rain neutralization solutions is primarily driven by the power generation, manufacturing, and transportation sectors, which are major contributors to sulfur dioxide and nitrogen oxide emissions. These industries are under pressure to adopt cleaner technologies and mitigation strategies, creating a significant demand for effective neutralization solutions.
In recent years, there has been a shift towards more sustainable and cost-effective acid rain mitigation strategies. This trend has opened up opportunities for innovative solutions like barium hydroxide, which offers unique advantages in terms of neutralization capacity and potential for byproduct recovery.
The market for acid rain neutralization solutions is geographically diverse, with North America, Europe, and Asia-Pacific regions being the key markets. Developing economies in Asia, particularly China and India, are expected to witness substantial growth in demand due to rapid industrialization and increasing environmental awareness.
Barium hydroxide, as a specific solution for acid deposition mitigation, competes with other neutralizing agents such as limestone, lime, and sodium bicarbonate. Its market potential is influenced by factors such as effectiveness, cost, availability, and ease of application. While barium hydroxide offers high neutralization capacity, its market adoption may be affected by concerns related to barium toxicity and the need for careful handling.
The regulatory landscape plays a crucial role in shaping the market for acid rain neutralization solutions. Stringent environmental regulations in developed countries and emerging policies in developing nations are expected to drive the adoption of effective mitigation strategies, potentially benefiting solutions like barium hydroxide.
Market growth is also influenced by technological advancements in application methods and monitoring systems. Innovations in delivery systems and real-time pH monitoring can enhance the efficiency and precision of acid rain neutralization processes, potentially increasing the attractiveness of barium hydroxide as a solution.
As sustainability becomes a key focus for industries worldwide, there is growing interest in solutions that not only neutralize acid rain but also offer additional environmental benefits. Barium hydroxide's potential for byproduct recovery and its ability to sequester carbon dioxide could position it favorably in this evolving market landscape.
Current Status and Challenges in Acid Deposition Control
Acid deposition control has made significant progress over the past few decades, yet it remains a persistent environmental challenge globally. Current mitigation strategies primarily focus on reducing emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx) from industrial and transportation sources. Many countries have implemented stringent regulations and adopted cleaner technologies, resulting in substantial reductions in acid rain precursors.
In developed nations, the implementation of flue gas desulfurization systems in power plants and the widespread use of catalytic converters in vehicles have led to notable improvements in air quality and reduced acid deposition. However, developing countries continue to struggle with balancing economic growth and environmental protection, often resulting in higher emission levels and more severe acid deposition problems.
The use of alternative energy sources, such as renewable energy and nuclear power, has also contributed to the reduction of acid-forming emissions. Additionally, the development of more efficient combustion technologies and the shift towards cleaner fuels have played crucial roles in mitigating acid deposition.
Despite these advancements, several challenges persist in acid deposition control. One major issue is the long-range transport of pollutants, which can cause acid deposition in regions far from the emission sources. This transboundary nature of acid deposition necessitates international cooperation and coordinated efforts to address the problem effectively.
Another significant challenge is the increasing contribution of ammonia emissions to acid deposition. While sulfur and nitrogen emissions have decreased in many regions, ammonia emissions, primarily from agricultural sources, have not seen similar reductions. This shift in the composition of acid deposition requires new strategies and technologies for mitigation.
The slow recovery of ecosystems affected by long-term acid deposition poses another challenge. Even with reduced emissions, many water bodies and soils continue to suffer from the legacy effects of acidification, requiring additional measures such as liming to restore ecological balance.
In the context of using barium hydroxide for acid deposition mitigation, current research is exploring its potential as a neutralizing agent. While promising in laboratory settings, the practical application of barium hydroxide on a large scale faces several hurdles, including cost-effectiveness, potential environmental impacts, and logistical challenges in distribution and application.
Moving forward, addressing these challenges will require a multifaceted approach, combining continued emission reductions, ecosystem restoration efforts, and innovative technologies. The exploration of novel mitigation strategies, such as the use of barium hydroxide, represents an important avenue for future research and development in the field of acid deposition control.
In developed nations, the implementation of flue gas desulfurization systems in power plants and the widespread use of catalytic converters in vehicles have led to notable improvements in air quality and reduced acid deposition. However, developing countries continue to struggle with balancing economic growth and environmental protection, often resulting in higher emission levels and more severe acid deposition problems.
The use of alternative energy sources, such as renewable energy and nuclear power, has also contributed to the reduction of acid-forming emissions. Additionally, the development of more efficient combustion technologies and the shift towards cleaner fuels have played crucial roles in mitigating acid deposition.
Despite these advancements, several challenges persist in acid deposition control. One major issue is the long-range transport of pollutants, which can cause acid deposition in regions far from the emission sources. This transboundary nature of acid deposition necessitates international cooperation and coordinated efforts to address the problem effectively.
Another significant challenge is the increasing contribution of ammonia emissions to acid deposition. While sulfur and nitrogen emissions have decreased in many regions, ammonia emissions, primarily from agricultural sources, have not seen similar reductions. This shift in the composition of acid deposition requires new strategies and technologies for mitigation.
The slow recovery of ecosystems affected by long-term acid deposition poses another challenge. Even with reduced emissions, many water bodies and soils continue to suffer from the legacy effects of acidification, requiring additional measures such as liming to restore ecological balance.
In the context of using barium hydroxide for acid deposition mitigation, current research is exploring its potential as a neutralizing agent. While promising in laboratory settings, the practical application of barium hydroxide on a large scale faces several hurdles, including cost-effectiveness, potential environmental impacts, and logistical challenges in distribution and application.
Moving forward, addressing these challenges will require a multifaceted approach, combining continued emission reductions, ecosystem restoration efforts, and innovative technologies. The exploration of novel mitigation strategies, such as the use of barium hydroxide, represents an important avenue for future research and development in the field of acid deposition control.
Existing Barium Hydroxide Application Methods
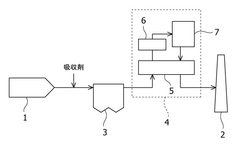
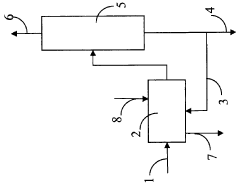
01 Acid gas removal using barium hydroxide
Barium hydroxide is used as an effective agent for removing acid gases from industrial processes. This method involves the reaction of barium hydroxide with acidic components, resulting in the formation of insoluble barium salts. The process can be applied in various industrial settings to reduce harmful emissions and improve air quality.- Acid gas removal using barium hydroxide: Barium hydroxide can be used to remove acid gases from industrial processes. The alkaline nature of barium hydroxide allows it to neutralize acidic components in gas streams, forming barium salts as byproducts. This method is particularly effective for treating flue gases and other emissions containing sulfur dioxide or carbon dioxide.
- Barium hydroxide in soil treatment: Barium hydroxide can be applied to acidic soils to neutralize excess acidity and improve soil quality. This process, known as liming, helps to raise the pH of the soil, making it more suitable for plant growth. The treatment can also enhance nutrient availability and microbial activity in the soil.
- Barium hydroxide in wastewater treatment: Barium hydroxide can be used in wastewater treatment processes to remove various contaminants. It is particularly effective in precipitating sulfates and phosphates from industrial effluents. The high alkalinity of barium hydroxide also aids in pH adjustment and heavy metal removal from wastewater streams.
- Barium hydroxide in air pollution control: Barium hydroxide can be utilized in air pollution control systems to capture and neutralize acidic pollutants. It can be used in scrubbers or as a dry sorbent injection material to remove sulfur dioxide, hydrogen chloride, and other acid gases from industrial emissions, helping to reduce atmospheric acid deposition.
- Barium hydroxide in chemical manufacturing: Barium hydroxide serves as a versatile reagent in various chemical manufacturing processes. It can be used in the production of other barium compounds, as a catalyst in organic synthesis reactions, and as a pH regulator in industrial processes. Its alkaline properties make it useful in neutralizing acidic byproducts in chemical reactions.
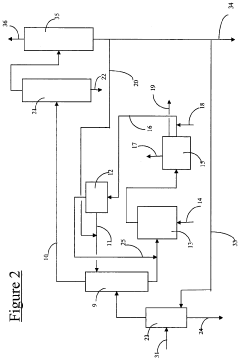
02 Barium hydroxide in flue gas desulfurization
Barium hydroxide is utilized in flue gas desulfurization processes to remove sulfur dioxide from exhaust gases. The barium hydroxide reacts with sulfur dioxide to form barium sulfite or sulfate, which can be easily separated from the gas stream. This method is particularly effective in reducing sulfur emissions from power plants and other industrial facilities.Expand Specific Solutions03 Barium hydroxide in wastewater treatment
Barium hydroxide is employed in wastewater treatment processes to remove various contaminants, including heavy metals and sulfates. The high pH of barium hydroxide solutions facilitates the precipitation of metal hydroxides and the formation of insoluble barium sulfate. This application helps in improving water quality and meeting environmental regulations.Expand Specific Solutions04 Barium hydroxide in soil remediation
Barium hydroxide is used in soil remediation techniques to neutralize acidic soils and immobilize certain contaminants. The alkaline nature of barium hydroxide helps in adjusting soil pH and promoting the precipitation of harmful substances. This application is particularly useful in the restoration of contaminated industrial sites and mining areas.Expand Specific Solutions05 Barium hydroxide in carbon dioxide capture
Barium hydroxide is investigated as a potential agent for carbon dioxide capture and sequestration. The reaction between barium hydroxide and carbon dioxide results in the formation of barium carbonate, which can be easily separated and stored. This method is being explored as a means to reduce greenhouse gas emissions and mitigate climate change.Expand Specific Solutions
Key Players in Environmental Remediation Industry
The use of barium hydroxide for acid deposition mitigation strategies is in a relatively early stage of development, with the market still emerging. The global market for acid rain mitigation technologies is projected to grow significantly in the coming years, driven by increasing environmental concerns and stricter regulations. While the technology shows promise, its maturity level varies among key players. Companies like China Petroleum & Chemical Corp. and SINOPEC Beijing Research Institute of Chemical Industry are at the forefront of research and development in this area, leveraging their expertise in chemical processes. Academic institutions such as Jiangxi University of Science & Technology and Nanchang University are also contributing to advancements in this field, indicating a collaborative approach between industry and academia to address this environmental challenge.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed a comprehensive acid deposition mitigation strategy using barium hydroxide. Their approach involves injecting a barium hydroxide solution into flue gas streams to neutralize sulfur dioxide and other acidic compounds[1]. The process includes a novel scrubber design that enhances contact between the flue gas and the barium hydroxide solution, improving efficiency. Additionally, Sinopec has implemented a closed-loop system for barium recovery and regeneration, reducing waste and operational costs[3]. The company has also integrated this technology with their existing desulfurization units, creating a multi-stage treatment process that achieves over 95% removal of acid-forming compounds[5].
Strengths: High efficiency in acid removal, integrated with existing systems, closed-loop barium recovery. Weaknesses: High initial investment, potential barium contamination risks if not properly managed.
SINOPEC Beijing Research Institute of Chemical Industry
Technical Solution: SINOPEC Beijing Research Institute of Chemical Industry has developed an innovative approach to using barium hydroxide for acid deposition mitigation. Their method involves a two-stage process: first, a fine mist of barium hydroxide solution is sprayed into the flue gas stream in a specially designed reaction chamber[2]. This initial contact neutralizes a significant portion of the acidic compounds. In the second stage, the partially treated gas passes through a packed bed scrubber containing barium hydroxide-impregnated porous media, ensuring thorough neutralization[4]. The institute has also developed a proprietary catalyst that enhances the reaction rate between barium hydroxide and acidic compounds, reducing the overall treatment time and improving efficiency[6].
Strengths: High neutralization efficiency, reduced treatment time, innovative catalyst use. Weaknesses: Complex system design, potential for equipment fouling due to barium sulfate formation.
Core Innovations in Barium-based Neutralization
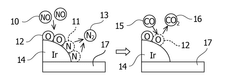
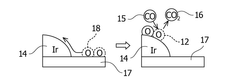
Exhaust gas denitration catalyst, co oxidation catalyst, exhaust gas treatment system, and exhaust gas treatment method
PatentWO2018021511A1
Innovation
- The use of barium sulfate supported with iridium as a catalyst, which maintains denitrification performance across varying temperatures and suppresses CO oxidation, allowing for effective NOx reduction and CO oxidation without promoting CO oxidation, even in high CO concentration exhaust gases.
Process and plant for dehydration by a deliquescent substance
PatentWO2012010754A1
Innovation
- A method and installation utilizing deliquescent dehydrating agents in the form of soda or potash pads for dehydration, where the resulting brine is recycled for neutralization, reducing the need for fresh alkaline solutions and minimizing effluent waste, and allowing for in-process recycling and reuse.
Environmental Impact Assessment of Barium Hydroxide Use
The use of barium hydroxide for acid deposition mitigation strategies necessitates a comprehensive environmental impact assessment. This evaluation aims to identify and analyze the potential effects of barium hydroxide application on various environmental components.
Atmospheric impacts are a primary concern. When barium hydroxide is used to neutralize acidic compounds in the air, it may lead to the formation of barium-containing particulates. These particles could contribute to air pollution and potentially affect air quality in the treated areas. Long-term exposure to elevated levels of barium in the air may pose health risks to both humans and wildlife, necessitating careful monitoring and control measures.
Soil and water ecosystems are also likely to be affected by barium hydroxide application. As the compound settles or is washed out of the atmosphere, it can alter soil chemistry and pH levels. This may impact soil microbial communities, plant growth, and nutrient cycling processes. In aquatic environments, increased barium concentrations could affect water quality and potentially disrupt aquatic ecosystems, including impacts on fish and other aquatic organisms.
The potential for bioaccumulation of barium in food chains is another critical aspect to consider. Plants may absorb barium from treated soils, potentially leading to increased concentrations in crops and wild vegetation. This could have implications for herbivorous animals and, subsequently, for higher trophic levels in the ecosystem.
Groundwater contamination is a significant concern, particularly in areas with high rainfall or where barium hydroxide is applied extensively. Barium ions can leach into groundwater, potentially affecting drinking water sources and requiring additional water treatment processes to ensure safe consumption.
The production and transportation of barium hydroxide also carry environmental implications. Mining and processing of barium compounds can lead to habitat disruption and generate waste products that require proper management. Additionally, the transportation of large quantities of barium hydroxide to application sites may contribute to increased carbon emissions and other transportation-related environmental impacts.
Long-term ecological effects must be carefully studied. While barium hydroxide may provide short-term benefits in mitigating acid deposition, its prolonged use could lead to unforeseen changes in ecosystem dynamics, biodiversity, and overall environmental health. Comprehensive monitoring programs and long-term studies are essential to fully understand these potential impacts.
In conclusion, while barium hydroxide shows promise as a strategy for acid deposition mitigation, its environmental impacts are complex and multifaceted. A thorough environmental impact assessment is crucial to ensure that the benefits of acid neutralization are not outweighed by unintended negative consequences on the environment and human health.
Atmospheric impacts are a primary concern. When barium hydroxide is used to neutralize acidic compounds in the air, it may lead to the formation of barium-containing particulates. These particles could contribute to air pollution and potentially affect air quality in the treated areas. Long-term exposure to elevated levels of barium in the air may pose health risks to both humans and wildlife, necessitating careful monitoring and control measures.
Soil and water ecosystems are also likely to be affected by barium hydroxide application. As the compound settles or is washed out of the atmosphere, it can alter soil chemistry and pH levels. This may impact soil microbial communities, plant growth, and nutrient cycling processes. In aquatic environments, increased barium concentrations could affect water quality and potentially disrupt aquatic ecosystems, including impacts on fish and other aquatic organisms.
The potential for bioaccumulation of barium in food chains is another critical aspect to consider. Plants may absorb barium from treated soils, potentially leading to increased concentrations in crops and wild vegetation. This could have implications for herbivorous animals and, subsequently, for higher trophic levels in the ecosystem.
Groundwater contamination is a significant concern, particularly in areas with high rainfall or where barium hydroxide is applied extensively. Barium ions can leach into groundwater, potentially affecting drinking water sources and requiring additional water treatment processes to ensure safe consumption.
The production and transportation of barium hydroxide also carry environmental implications. Mining and processing of barium compounds can lead to habitat disruption and generate waste products that require proper management. Additionally, the transportation of large quantities of barium hydroxide to application sites may contribute to increased carbon emissions and other transportation-related environmental impacts.
Long-term ecological effects must be carefully studied. While barium hydroxide may provide short-term benefits in mitigating acid deposition, its prolonged use could lead to unforeseen changes in ecosystem dynamics, biodiversity, and overall environmental health. Comprehensive monitoring programs and long-term studies are essential to fully understand these potential impacts.
In conclusion, while barium hydroxide shows promise as a strategy for acid deposition mitigation, its environmental impacts are complex and multifaceted. A thorough environmental impact assessment is crucial to ensure that the benefits of acid neutralization are not outweighed by unintended negative consequences on the environment and human health.
Cost-Benefit Analysis of Barium Hydroxide Solutions
The cost-benefit analysis of barium hydroxide solutions for acid deposition mitigation strategies reveals a complex interplay of economic, environmental, and technical factors. Initial implementation costs for barium hydroxide-based systems can be substantial, primarily due to the expense of the chemical itself and the necessary infrastructure for its application. However, these upfront investments must be weighed against the long-term benefits of reduced acid deposition and its associated environmental impacts.
One significant advantage of barium hydroxide solutions is their high efficiency in neutralizing acidic compounds. This effectiveness translates to potentially lower overall chemical usage compared to alternative mitigation methods, which can lead to cost savings over time. Additionally, the reduced environmental damage from acid deposition can result in substantial economic benefits, including improved agricultural yields, reduced corrosion of infrastructure, and preservation of ecosystems.
The operational costs of barium hydroxide systems vary depending on factors such as the scale of application, local environmental conditions, and the specific acid deposition challenges being addressed. Ongoing expenses include chemical replenishment, monitoring and maintenance of application systems, and potential waste management considerations. These operational costs must be carefully evaluated against the projected long-term benefits to determine the overall economic viability of the solution.
Environmental benefits of barium hydroxide solutions extend beyond direct acid neutralization. By mitigating acid deposition, these systems can help preserve biodiversity, protect water resources, and maintain soil health. These ecological improvements can have cascading positive effects on various economic sectors, including tourism, fisheries, and forestry. However, it is crucial to consider potential environmental risks associated with barium hydroxide use, such as the impact on local ecosystems if not properly managed.
The cost-benefit analysis must also account for regulatory compliance and potential future policy changes. As environmental regulations become more stringent, the value proposition of barium hydroxide solutions may increase, potentially offsetting initial implementation costs. Furthermore, the adoption of such mitigation strategies could position organizations favorably in terms of corporate social responsibility and environmental stewardship.
Technological advancements in barium hydroxide production and application methods may further improve the cost-benefit ratio over time. Research into more efficient production processes, innovative application techniques, and synergistic combinations with other mitigation strategies could enhance the economic viability of these solutions. As such, the cost-benefit analysis should incorporate a forward-looking perspective that considers potential technological improvements and their impact on long-term economic feasibility.
One significant advantage of barium hydroxide solutions is their high efficiency in neutralizing acidic compounds. This effectiveness translates to potentially lower overall chemical usage compared to alternative mitigation methods, which can lead to cost savings over time. Additionally, the reduced environmental damage from acid deposition can result in substantial economic benefits, including improved agricultural yields, reduced corrosion of infrastructure, and preservation of ecosystems.
The operational costs of barium hydroxide systems vary depending on factors such as the scale of application, local environmental conditions, and the specific acid deposition challenges being addressed. Ongoing expenses include chemical replenishment, monitoring and maintenance of application systems, and potential waste management considerations. These operational costs must be carefully evaluated against the projected long-term benefits to determine the overall economic viability of the solution.
Environmental benefits of barium hydroxide solutions extend beyond direct acid neutralization. By mitigating acid deposition, these systems can help preserve biodiversity, protect water resources, and maintain soil health. These ecological improvements can have cascading positive effects on various economic sectors, including tourism, fisheries, and forestry. However, it is crucial to consider potential environmental risks associated with barium hydroxide use, such as the impact on local ecosystems if not properly managed.
The cost-benefit analysis must also account for regulatory compliance and potential future policy changes. As environmental regulations become more stringent, the value proposition of barium hydroxide solutions may increase, potentially offsetting initial implementation costs. Furthermore, the adoption of such mitigation strategies could position organizations favorably in terms of corporate social responsibility and environmental stewardship.
Technological advancements in barium hydroxide production and application methods may further improve the cost-benefit ratio over time. Research into more efficient production processes, innovative application techniques, and synergistic combinations with other mitigation strategies could enhance the economic viability of these solutions. As such, the cost-benefit analysis should incorporate a forward-looking perspective that considers potential technological improvements and their impact on long-term economic feasibility.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!