Exploring Injectable Hydrogel Polyelectrolyte Complexes in Drug Carriers
OCT 15, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrogel Polyelectrolyte Complexes Background and Objectives
Hydrogel polyelectrolyte complexes (HPCs) represent a significant advancement in biomaterial science, emerging from the convergence of polymer chemistry and pharmaceutical technology. These complexes form when oppositely charged polyelectrolytes interact through electrostatic forces, creating three-dimensional networks capable of absorbing substantial amounts of water while maintaining structural integrity. The evolution of HPCs began in the 1960s with rudimentary hydrogel formulations, progressing through significant milestones including the development of stimuli-responsive systems in the 1990s and the integration of biological functionalities in the early 2000s.
The current technological trajectory of HPCs is moving toward increasingly sophisticated, multifunctional systems with enhanced biocompatibility and controlled degradation profiles. Recent innovations have focused on improving mechanical properties, incorporating nanomaterials, and developing "smart" responsive behaviors that react to specific physiological conditions. These advancements have positioned HPCs as promising candidates for next-generation drug delivery platforms.
Injectable HPCs represent a particularly valuable subset of these materials, offering minimally invasive administration while providing sustained drug release capabilities. The in situ gelation mechanism of these systems allows them to transition from a flowable solution to a stable gel upon injection, creating a depot for controlled drug release directly at the target site. This approach addresses several limitations of conventional drug delivery methods, including poor bioavailability, systemic toxicity, and patient compliance issues.
The primary technical objectives for injectable HPC development include: optimizing gelation kinetics to ensure appropriate transition times for clinical applications; enhancing mechanical stability to maintain structural integrity under physiological conditions; improving biodegradability profiles to match therapeutic timelines; and developing predictable drug release kinetics for precise dosing regimens. Additionally, there is significant focus on ensuring batch-to-batch consistency and scalable manufacturing processes to facilitate clinical translation.
From a biological perspective, key objectives include minimizing immunogenicity, enhancing tissue integration, and ensuring complete clearance of degradation products. The biocompatibility of these systems remains paramount, particularly for applications requiring extended contact with tissues or potential for repeated administrations.
Looking forward, the field aims to develop injectable HPCs capable of responding to multiple biological stimuli simultaneously, incorporating theranostic capabilities, and providing personalized release profiles based on individual patient needs. The ultimate goal is to create "intelligent" drug delivery systems that can autonomously adjust their behavior in response to changing physiological conditions, thereby optimizing therapeutic outcomes while minimizing adverse effects.
The current technological trajectory of HPCs is moving toward increasingly sophisticated, multifunctional systems with enhanced biocompatibility and controlled degradation profiles. Recent innovations have focused on improving mechanical properties, incorporating nanomaterials, and developing "smart" responsive behaviors that react to specific physiological conditions. These advancements have positioned HPCs as promising candidates for next-generation drug delivery platforms.
Injectable HPCs represent a particularly valuable subset of these materials, offering minimally invasive administration while providing sustained drug release capabilities. The in situ gelation mechanism of these systems allows them to transition from a flowable solution to a stable gel upon injection, creating a depot for controlled drug release directly at the target site. This approach addresses several limitations of conventional drug delivery methods, including poor bioavailability, systemic toxicity, and patient compliance issues.
The primary technical objectives for injectable HPC development include: optimizing gelation kinetics to ensure appropriate transition times for clinical applications; enhancing mechanical stability to maintain structural integrity under physiological conditions; improving biodegradability profiles to match therapeutic timelines; and developing predictable drug release kinetics for precise dosing regimens. Additionally, there is significant focus on ensuring batch-to-batch consistency and scalable manufacturing processes to facilitate clinical translation.
From a biological perspective, key objectives include minimizing immunogenicity, enhancing tissue integration, and ensuring complete clearance of degradation products. The biocompatibility of these systems remains paramount, particularly for applications requiring extended contact with tissues or potential for repeated administrations.
Looking forward, the field aims to develop injectable HPCs capable of responding to multiple biological stimuli simultaneously, incorporating theranostic capabilities, and providing personalized release profiles based on individual patient needs. The ultimate goal is to create "intelligent" drug delivery systems that can autonomously adjust their behavior in response to changing physiological conditions, thereby optimizing therapeutic outcomes while minimizing adverse effects.
Market Analysis for Injectable Drug Delivery Systems
The injectable drug delivery systems market has witnessed substantial growth in recent years, driven by increasing prevalence of chronic diseases and growing demand for minimally invasive treatment options. Currently valued at approximately 40 billion USD globally, this market is projected to grow at a compound annual growth rate (CAGR) of 11.5% over the next five years, potentially reaching 69 billion USD by 2028.
Injectable hydrogel polyelectrolyte complexes represent a particularly promising segment within this broader market. These advanced drug carriers address several limitations of conventional drug delivery systems, including poor bioavailability, rapid clearance, and inadequate targeting. The demand for such sophisticated delivery systems is especially strong in oncology, diabetes management, and autoimmune disease treatment, where controlled release profiles can significantly improve therapeutic outcomes.
North America currently dominates the injectable drug delivery market with approximately 45% market share, followed by Europe (30%) and Asia-Pacific (20%). However, the Asia-Pacific region is expected to exhibit the highest growth rate, driven by improving healthcare infrastructure, increasing healthcare expenditure, and growing awareness about advanced treatment options in countries like China, India, and Japan.
From an end-user perspective, hospitals remain the largest consumer segment, accounting for 55% of the market. However, home care settings are emerging as the fastest-growing segment due to the increasing shift toward self-administration of injectable medications, particularly for chronic disease management.
Key market drivers include the rising geriatric population, increasing incidence of chronic diseases, technological advancements in drug delivery systems, and growing preference for self-administration devices. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of efficient drug delivery systems and expanding telehealth services.
Regulatory factors significantly influence market dynamics. Stringent approval processes by regulatory bodies like the FDA and EMA ensure product safety but can extend time-to-market. Recent regulatory changes favoring innovative drug delivery systems, including the FDA's fast-track designation for certain novel delivery technologies, are expected to positively impact market growth.
Reimbursement policies also play a crucial role in market adoption. While injectable hydrogel systems often command premium pricing, favorable reimbursement scenarios in developed markets are facilitating uptake. However, cost concerns remain a significant barrier in emerging economies, necessitating strategic pricing approaches for global market penetration.
Injectable hydrogel polyelectrolyte complexes represent a particularly promising segment within this broader market. These advanced drug carriers address several limitations of conventional drug delivery systems, including poor bioavailability, rapid clearance, and inadequate targeting. The demand for such sophisticated delivery systems is especially strong in oncology, diabetes management, and autoimmune disease treatment, where controlled release profiles can significantly improve therapeutic outcomes.
North America currently dominates the injectable drug delivery market with approximately 45% market share, followed by Europe (30%) and Asia-Pacific (20%). However, the Asia-Pacific region is expected to exhibit the highest growth rate, driven by improving healthcare infrastructure, increasing healthcare expenditure, and growing awareness about advanced treatment options in countries like China, India, and Japan.
From an end-user perspective, hospitals remain the largest consumer segment, accounting for 55% of the market. However, home care settings are emerging as the fastest-growing segment due to the increasing shift toward self-administration of injectable medications, particularly for chronic disease management.
Key market drivers include the rising geriatric population, increasing incidence of chronic diseases, technological advancements in drug delivery systems, and growing preference for self-administration devices. The COVID-19 pandemic has further accelerated market growth by highlighting the importance of efficient drug delivery systems and expanding telehealth services.
Regulatory factors significantly influence market dynamics. Stringent approval processes by regulatory bodies like the FDA and EMA ensure product safety but can extend time-to-market. Recent regulatory changes favoring innovative drug delivery systems, including the FDA's fast-track designation for certain novel delivery technologies, are expected to positively impact market growth.
Reimbursement policies also play a crucial role in market adoption. While injectable hydrogel systems often command premium pricing, favorable reimbursement scenarios in developed markets are facilitating uptake. However, cost concerns remain a significant barrier in emerging economies, necessitating strategic pricing approaches for global market penetration.
Current Challenges in Hydrogel-Based Drug Carriers
Despite significant advancements in hydrogel-based drug delivery systems, several critical challenges continue to impede their widespread clinical application. The complexity of injectable hydrogel polyelectrolyte complexes presents multifaceted obstacles that researchers must overcome to realize their full potential as drug carriers.
Mechanical property optimization remains a fundamental challenge, as these systems must maintain structural integrity while allowing controlled degradation. Many current hydrogel formulations exhibit either excessive rigidity, limiting injectability, or insufficient mechanical strength, resulting in premature degradation after administration. This delicate balance between injectability and post-injection stability has proven difficult to achieve consistently across different drug types and therapeutic applications.
Controlled drug release kinetics presents another significant hurdle. The heterogeneous nature of polyelectrolyte complex hydrogels often leads to unpredictable release profiles, with initial burst release followed by suboptimal sustained delivery. This inconsistency compromises therapeutic efficacy and potentially increases side effects, particularly for drugs with narrow therapeutic windows. Engineering precise control over release mechanisms that respond appropriately to physiological conditions remains an ongoing challenge.
Biocompatibility and immunogenicity concerns persist despite advances in biomaterial design. Some polyelectrolyte components trigger inflammatory responses or immune reactions when injected, limiting their clinical utility. Additionally, the degradation products of these complex systems may accumulate in tissues or organs, potentially causing long-term toxicity that is difficult to predict during preclinical testing phases.
Scalable manufacturing represents a substantial industrial challenge. The production of injectable hydrogel polyelectrolyte complexes with consistent quality, sterility, and batch-to-batch reproducibility remains problematic. Current synthesis methods often involve complex procedures that are difficult to scale while maintaining precise control over critical quality attributes such as crosslinking density, pore size distribution, and polyelectrolyte interaction strength.
Regulatory hurdles further complicate clinical translation. The complex, multicomponent nature of these drug delivery systems creates challenges in regulatory approval pathways, as authorities require comprehensive characterization and stability data. The lack of standardized testing protocols specifically designed for injectable hydrogel polyelectrolyte complexes further complicates the regulatory landscape.
Storage stability and shelf-life limitations present practical challenges for commercialization. Many promising formulations exhibit premature gelation, phase separation, or loss of functional properties during storage, necessitating complex preservation methods or reconstitution protocols that complicate clinical use and patient compliance.
Mechanical property optimization remains a fundamental challenge, as these systems must maintain structural integrity while allowing controlled degradation. Many current hydrogel formulations exhibit either excessive rigidity, limiting injectability, or insufficient mechanical strength, resulting in premature degradation after administration. This delicate balance between injectability and post-injection stability has proven difficult to achieve consistently across different drug types and therapeutic applications.
Controlled drug release kinetics presents another significant hurdle. The heterogeneous nature of polyelectrolyte complex hydrogels often leads to unpredictable release profiles, with initial burst release followed by suboptimal sustained delivery. This inconsistency compromises therapeutic efficacy and potentially increases side effects, particularly for drugs with narrow therapeutic windows. Engineering precise control over release mechanisms that respond appropriately to physiological conditions remains an ongoing challenge.
Biocompatibility and immunogenicity concerns persist despite advances in biomaterial design. Some polyelectrolyte components trigger inflammatory responses or immune reactions when injected, limiting their clinical utility. Additionally, the degradation products of these complex systems may accumulate in tissues or organs, potentially causing long-term toxicity that is difficult to predict during preclinical testing phases.
Scalable manufacturing represents a substantial industrial challenge. The production of injectable hydrogel polyelectrolyte complexes with consistent quality, sterility, and batch-to-batch reproducibility remains problematic. Current synthesis methods often involve complex procedures that are difficult to scale while maintaining precise control over critical quality attributes such as crosslinking density, pore size distribution, and polyelectrolyte interaction strength.
Regulatory hurdles further complicate clinical translation. The complex, multicomponent nature of these drug delivery systems creates challenges in regulatory approval pathways, as authorities require comprehensive characterization and stability data. The lack of standardized testing protocols specifically designed for injectable hydrogel polyelectrolyte complexes further complicates the regulatory landscape.
Storage stability and shelf-life limitations present practical challenges for commercialization. Many promising formulations exhibit premature gelation, phase separation, or loss of functional properties during storage, necessitating complex preservation methods or reconstitution protocols that complicate clinical use and patient compliance.
Current Injectable Hydrogel Polyelectrolyte Formulations
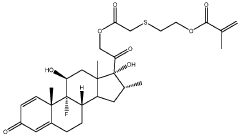
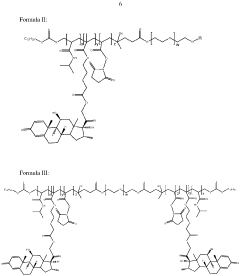
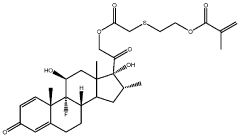
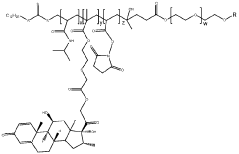
01 Composition and structure of injectable hydrogel polyelectrolyte complexes
Injectable hydrogel polyelectrolyte complexes are formed through interactions between oppositely charged polymers. These complexes can be designed with specific structural properties to control their mechanical strength, stability, and injectability. The composition typically includes cationic and anionic polymers that form a three-dimensional network through electrostatic interactions. The structure can be modified by adjusting the ratio of the polyelectrolytes, the ionic strength of the solution, and the molecular weight of the polymers.- Composition and structure of injectable hydrogel polyelectrolyte complexes: Injectable hydrogel polyelectrolyte complexes are formed through interactions between oppositely charged polymers. These complexes can be designed with specific structural properties to control their mechanical strength, stability, and injectability. The composition typically includes cationic and anionic polymers that form a three-dimensional network through electrostatic interactions. The structure can be modified by adjusting polymer concentration, molecular weight, and charge density to achieve desired properties for various biomedical applications.
- Biomedical applications of injectable hydrogel polyelectrolyte complexes: Injectable hydrogel polyelectrolyte complexes have diverse applications in biomedicine, including drug delivery systems, tissue engineering scaffolds, and wound healing materials. These hydrogels can encapsulate therapeutic agents for controlled release, support cell growth and differentiation, and provide a moist environment for wound healing. Their injectability allows for minimally invasive administration, making them suitable for delivering cells, growth factors, and other bioactive molecules to specific sites in the body.
- Stimuli-responsive properties of hydrogel polyelectrolyte complexes: Hydrogel polyelectrolyte complexes can be engineered to respond to various stimuli such as pH, temperature, ionic strength, and specific biomolecules. These stimuli-responsive properties enable smart functionalities including controlled drug release, self-healing capabilities, and adaptive mechanical properties. By incorporating specific functional groups or responsive polymers, these hydrogels can undergo reversible transitions between different states, allowing for precise control over their behavior in biological environments.
- Fabrication methods for injectable hydrogel polyelectrolyte complexes: Various fabrication techniques are employed to prepare injectable hydrogel polyelectrolyte complexes with controlled properties. These methods include simple mixing of polyelectrolyte solutions, in situ gelation, microfluidic approaches, and electrospinning. The fabrication process can be optimized to control parameters such as gelation time, pore size, and mechanical properties. Advanced manufacturing techniques allow for the creation of complex structures with spatial gradients in composition and properties.
- Modification strategies to enhance performance of polyelectrolyte hydrogels: Various modification strategies are employed to enhance the performance of injectable hydrogel polyelectrolyte complexes. These include chemical crosslinking to improve mechanical stability, incorporation of nanoparticles for enhanced functionality, grafting of bioactive molecules to promote cell adhesion and proliferation, and hybridization with other materials to create composite systems. These modifications can address limitations such as poor mechanical properties, rapid degradation, or insufficient bioactivity, thereby expanding the range of applications for these hydrogels.
02 Biomedical applications of injectable hydrogel polyelectrolyte complexes
Injectable hydrogel polyelectrolyte complexes have various biomedical applications, including drug delivery systems, tissue engineering scaffolds, and wound healing materials. These complexes can encapsulate therapeutic agents and provide controlled release over time. They can also serve as scaffolds for cell growth and tissue regeneration. The injectability of these hydrogels allows for minimally invasive administration, making them suitable for various clinical applications.Expand Specific Solutions03 Stimuli-responsive injectable hydrogel polyelectrolyte complexes
Stimuli-responsive injectable hydrogel polyelectrolyte complexes can change their properties in response to external stimuli such as pH, temperature, or ionic strength. These smart materials can undergo reversible sol-gel transitions, allowing them to be injected as a solution and form a gel at the target site. The stimuli-responsive behavior can be utilized for controlled drug release, where the release rate can be modulated by applying specific stimuli. This feature enhances the versatility and applicability of these hydrogels in various biomedical settings.Expand Specific Solutions04 Fabrication methods for injectable hydrogel polyelectrolyte complexes
Various fabrication methods can be employed to prepare injectable hydrogel polyelectrolyte complexes. These include simple mixing of oppositely charged polymers, layer-by-layer assembly, and in situ gelation techniques. The fabrication process can be optimized to control the physical properties of the resulting hydrogels, such as porosity, swelling behavior, and degradation rate. Advanced techniques like microfluidics and 3D printing can also be used to create hydrogels with complex architectures and spatial distributions of functional components.Expand Specific Solutions05 Functionalization of injectable hydrogel polyelectrolyte complexes
Injectable hydrogel polyelectrolyte complexes can be functionalized with various bioactive molecules to enhance their performance for specific applications. Functionalization can involve the incorporation of growth factors, cell adhesion peptides, or antimicrobial agents. The polyelectrolyte backbone can also be modified with functional groups to enable crosslinking or to introduce specific biological activities. These modifications can improve the biocompatibility, biodegradability, and therapeutic efficacy of the hydrogels.Expand Specific Solutions
Leading Companies in Hydrogel Polyelectrolyte Research
The injectable hydrogel polyelectrolyte complexes market for drug carriers is currently in an early growth phase, characterized by intensive research and emerging commercial applications. The global market size is estimated to reach approximately $2.5 billion by 2027, driven by increasing demand for targeted drug delivery systems. Technical maturity varies significantly across players, with established pharmaceutical companies like Merck & Co. and Baxter International leading commercial development, while academic institutions such as University of Florida and Rutgers University contribute fundamental research breakthroughs. Specialized firms like Contraline and Surmodics are advancing niche applications, while Asian players including Sunshine Lake Pharma and Shanghai Laiyi Biopharmaceutical are rapidly closing the technology gap through strategic R&D investments in novel hydrogel formulations and manufacturing processes.
Merck & Co., Inc.
Technical Solution: Merck has developed advanced injectable hydrogel polyelectrolyte complexes (PECs) for controlled drug delivery systems. Their technology utilizes oppositely charged polyelectrolytes that form stable complexes through electrostatic interactions, creating a three-dimensional network capable of encapsulating various therapeutic agents. The company's approach incorporates temperature-responsive polymers that remain liquid at room temperature but form solid hydrogels at body temperature, enabling minimally invasive injection. Merck's PEC systems feature tunable degradation rates through incorporation of enzymatically cleavable crosslinks, allowing for customized drug release profiles ranging from days to months. Their formulations include specific combinations of polycationic chitosan derivatives and polyanionic hyaluronic acid that optimize biocompatibility while maintaining structural integrity in physiological environments. Recent innovations include incorporation of stimuli-responsive elements that can trigger drug release in response to specific disease biomarkers or external stimuli.
Strengths: Extensive pharmaceutical development expertise and regulatory experience accelerates clinical translation. Their global manufacturing infrastructure ensures consistent quality and scalability. Weaknesses: Higher production costs compared to conventional delivery systems may limit application in cost-sensitive markets. Their proprietary formulations may require specialized handling and storage conditions.
Baxter International, Inc.
Technical Solution: Baxter International has pioneered injectable hydrogel PEC technology specifically designed for sustained release of biologics and small molecule drugs. Their proprietary platform combines synthetic and natural polyelectrolytes to create hydrogels with precisely controlled mechanical properties and degradation kinetics. The company's approach utilizes a dual-component system where oppositely charged polyelectrolytes are stored separately and form a hydrogel network immediately upon mixing during the injection process. This in-situ gelation mechanism prevents premature degradation of sensitive therapeutic cargo. Baxter's technology incorporates specialized polysaccharide derivatives with enhanced mucoadhesive properties, improving residence time at target tissues. Their formulations demonstrate remarkable shear-thinning behavior, flowing easily during injection but rapidly recovering structural integrity afterward. Recent developments include integration of imaging agents within the hydrogel matrix to enable real-time monitoring of gel degradation and drug release in vivo, providing valuable feedback for clinical applications.
Strengths: Extensive experience in medical device and pharmaceutical development provides strong regulatory pathway expertise. Their established distribution networks facilitate rapid commercialization. Weaknesses: Their systems may have limited application for hydrophobic drugs due to the hydrophilic nature of most polyelectrolyte components. Some formulations may require refrigeration, complicating storage and handling.
Key Patents and Breakthroughs in Polyelectrolyte Complexes
Injectable hydrogel for drug delivery
PatentWO2024061673A1
Innovation
- Development of an injectable hydrogel based on AB and/or ABA block co-polymers crosslinked via disulphide bonds, allowing for biodegradable and minimally invasive delivery of dexamethasone through a 30G needle, with self-healing properties and controlled release over several months, facilitated by redox-sensitive degradation in the vitreous environment.
Injectable drug carrier comprising layered double hydroxide
PatentInactiveUS9017719B2
Innovation
- The development of a non-toxic, injectable Layered Double Hydroxide (LDH) drug carrier with controlled particle size and shape, synthesized using divalent and trivalent metal salts and a base solution, allowing for the intercalation of negatively charged drugs, such as methotrexate and vitamins, to enhance drug delivery efficiency and safety.
Biocompatibility and Immunological Considerations
Biocompatibility remains a critical factor in the development of injectable hydrogel polyelectrolyte complexes (PECs) for drug delivery applications. These systems must interact harmoniously with biological tissues without triggering adverse reactions that could compromise therapeutic efficacy or patient safety. Current research indicates that PEC hydrogels generally exhibit favorable biocompatibility profiles due to their hydrophilic nature and structural similarity to natural extracellular matrices.
The immunological response to injectable PEC hydrogels varies significantly based on their composition and surface properties. Natural polymer-based PECs, such as those incorporating chitosan, alginate, or hyaluronic acid, typically demonstrate superior immunocompatibility compared to synthetic alternatives. However, recent advancements in synthetic polymer design have yielded materials with remarkably low immunogenicity through careful molecular engineering and surface modification strategies.
Foreign body responses represent a significant challenge for injectable PEC systems. Upon implantation, these materials may trigger a cascade of inflammatory events, including protein adsorption, leukocyte recruitment, and potential fibrous encapsulation. Research indicates that modulating the charge density, hydrophilicity, and degradation rate of PEC hydrogels can significantly mitigate these responses. Particularly promising are zwitterionic PEC formulations that resist protein adsorption through their balanced charge distribution.
Degradation byproducts from PEC hydrogels warrant careful consideration in biocompatibility assessments. As these materials break down in vivo, they must yield non-toxic metabolites that can be safely processed and eliminated by the body. Studies have demonstrated that PECs composed of naturally derived polymers generally produce biocompatible degradation products, while certain synthetic systems may require additional design considerations to ensure safe clearance pathways.
The integration of immunomodulatory components within PEC hydrogels represents an emerging strategy to enhance biocompatibility. By incorporating anti-inflammatory agents or immunosuppressive factors, researchers have developed "smart" PEC systems capable of actively managing the local immune environment. These advanced formulations not only minimize adverse reactions but can potentially promote healing processes and improve therapeutic outcomes in inflammatory disease contexts.
Standardized evaluation protocols for assessing the biocompatibility of injectable PEC hydrogels remain somewhat inconsistent across the field. While ISO 10993 provides general guidelines for biomedical materials, PEC-specific testing regimens that account for their unique physicochemical properties and biological interactions are still evolving. Comprehensive assessment typically involves in vitro cytotoxicity screening, hemolysis testing, and in vivo implantation studies to evaluate tissue response over clinically relevant timeframes.
The immunological response to injectable PEC hydrogels varies significantly based on their composition and surface properties. Natural polymer-based PECs, such as those incorporating chitosan, alginate, or hyaluronic acid, typically demonstrate superior immunocompatibility compared to synthetic alternatives. However, recent advancements in synthetic polymer design have yielded materials with remarkably low immunogenicity through careful molecular engineering and surface modification strategies.
Foreign body responses represent a significant challenge for injectable PEC systems. Upon implantation, these materials may trigger a cascade of inflammatory events, including protein adsorption, leukocyte recruitment, and potential fibrous encapsulation. Research indicates that modulating the charge density, hydrophilicity, and degradation rate of PEC hydrogels can significantly mitigate these responses. Particularly promising are zwitterionic PEC formulations that resist protein adsorption through their balanced charge distribution.
Degradation byproducts from PEC hydrogels warrant careful consideration in biocompatibility assessments. As these materials break down in vivo, they must yield non-toxic metabolites that can be safely processed and eliminated by the body. Studies have demonstrated that PECs composed of naturally derived polymers generally produce biocompatible degradation products, while certain synthetic systems may require additional design considerations to ensure safe clearance pathways.
The integration of immunomodulatory components within PEC hydrogels represents an emerging strategy to enhance biocompatibility. By incorporating anti-inflammatory agents or immunosuppressive factors, researchers have developed "smart" PEC systems capable of actively managing the local immune environment. These advanced formulations not only minimize adverse reactions but can potentially promote healing processes and improve therapeutic outcomes in inflammatory disease contexts.
Standardized evaluation protocols for assessing the biocompatibility of injectable PEC hydrogels remain somewhat inconsistent across the field. While ISO 10993 provides general guidelines for biomedical materials, PEC-specific testing regimens that account for their unique physicochemical properties and biological interactions are still evolving. Comprehensive assessment typically involves in vitro cytotoxicity screening, hemolysis testing, and in vivo implantation studies to evaluate tissue response over clinically relevant timeframes.
Regulatory Approval Pathways for Injectable Biomaterials
The regulatory landscape for injectable hydrogel polyelectrolyte complexes as drug carriers involves multiple pathways across different global jurisdictions. In the United States, the FDA's regulatory framework categorizes these biomaterials primarily under combination products, requiring comprehensive evaluation through either the Center for Drug Evaluation and Research (CDER) or the Center for Biologics Evaluation and Research (CBER), depending on the primary mode of action.
For injectable hydrogels, the premarket approval (PMA) pathway typically applies when the material serves as a significant component of the therapeutic effect. This process demands rigorous clinical trials demonstrating both safety and efficacy, with particular attention to biocompatibility, degradation profiles, and drug release kinetics specific to polyelectrolyte complex formulations.
The 510(k) clearance pathway may be applicable for certain injectable biomaterials when substantial equivalence to a predicate device can be established. However, the novel nature of many polyelectrolyte complex hydrogels often necessitates the more stringent PMA route due to their unique physicochemical properties and drug delivery mechanisms.
In the European Union, injectable biomaterials fall under the Medical Device Regulation (MDR) or the medicinal products directive, depending on their primary intended function. The conformity assessment procedure typically requires clinical evaluation reports and technical documentation demonstrating compliance with General Safety and Performance Requirements, with specific consideration for the polyelectrolyte interactions and their impact on drug delivery profiles.
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has established the Sakigake designation system for innovative medical products, which could accelerate approval for novel injectable hydrogel systems. This pathway offers priority consultation and review for technologies demonstrating significant medical value.
International harmonization efforts through the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) have established guidelines for quality, safety, and efficacy that influence regulatory decisions for injectable biomaterials globally. These standards are particularly relevant for polyelectrolyte complex hydrogels due to their complex physicochemical characteristics.
Post-market surveillance requirements represent a critical component of regulatory compliance for injectable biomaterials, with manufacturers required to monitor long-term safety profiles and report adverse events. For polyelectrolyte complex hydrogels, particular attention must be paid to potential immunogenicity, degradation product toxicity, and unexpected drug release pattern variations in diverse patient populations.
For injectable hydrogels, the premarket approval (PMA) pathway typically applies when the material serves as a significant component of the therapeutic effect. This process demands rigorous clinical trials demonstrating both safety and efficacy, with particular attention to biocompatibility, degradation profiles, and drug release kinetics specific to polyelectrolyte complex formulations.
The 510(k) clearance pathway may be applicable for certain injectable biomaterials when substantial equivalence to a predicate device can be established. However, the novel nature of many polyelectrolyte complex hydrogels often necessitates the more stringent PMA route due to their unique physicochemical properties and drug delivery mechanisms.
In the European Union, injectable biomaterials fall under the Medical Device Regulation (MDR) or the medicinal products directive, depending on their primary intended function. The conformity assessment procedure typically requires clinical evaluation reports and technical documentation demonstrating compliance with General Safety and Performance Requirements, with specific consideration for the polyelectrolyte interactions and their impact on drug delivery profiles.
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has established the Sakigake designation system for innovative medical products, which could accelerate approval for novel injectable hydrogel systems. This pathway offers priority consultation and review for technologies demonstrating significant medical value.
International harmonization efforts through the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) have established guidelines for quality, safety, and efficacy that influence regulatory decisions for injectable biomaterials globally. These standards are particularly relevant for polyelectrolyte complex hydrogels due to their complex physicochemical characteristics.
Post-market surveillance requirements represent a critical component of regulatory compliance for injectable biomaterials, with manufacturers required to monitor long-term safety profiles and report adverse events. For polyelectrolyte complex hydrogels, particular attention must be paid to potential immunogenicity, degradation product toxicity, and unexpected drug release pattern variations in diverse patient populations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!