How to Optimize GC-MS Sensitivity for Trace Detection
SEP 22, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
GC-MS Trace Detection Background and Objectives
Gas Chromatography-Mass Spectrometry (GC-MS) has evolved significantly since its inception in the 1950s, becoming an indispensable analytical technique for trace detection across multiple industries. The technology combines the separation capabilities of gas chromatography with the identification power of mass spectrometry, enabling the detection of compounds at extremely low concentrations, often in the parts-per-billion (ppb) or parts-per-trillion (ppt) range.
The evolution of GC-MS technology has been marked by continuous improvements in sensitivity, resolution, and reliability. Early systems were limited by rudimentary ionization techniques and low-resolution mass analyzers. Modern systems incorporate advanced features such as chemical ionization, tandem mass spectrometry, and high-resolution time-of-flight analyzers, dramatically enhancing detection capabilities.
Current technological trends in GC-MS focus on miniaturization, automation, and integration with artificial intelligence for data analysis. These developments aim to address the growing demand for portable, field-deployable systems that maintain laboratory-grade sensitivity while offering real-time analysis capabilities.
The primary objective of optimizing GC-MS sensitivity for trace detection is to achieve reliable identification and quantification of target analytes at increasingly lower concentrations. This is particularly crucial in applications such as environmental monitoring, food safety testing, forensic analysis, and pharmaceutical quality control, where detecting contaminants at trace levels can have significant health, safety, and regulatory implications.
Secondary objectives include improving signal-to-noise ratios, enhancing chromatographic resolution, reducing matrix interference effects, and developing more efficient sample preparation techniques. These factors collectively contribute to the overall sensitivity and reliability of GC-MS analysis for trace detection.
The technical challenges in this field are multifaceted, involving hardware limitations, sample preparation complexities, and data processing hurdles. Overcoming these challenges requires a multidisciplinary approach that integrates advances in materials science, electronics, computer algorithms, and analytical chemistry.
Recent breakthroughs in column technology, detector design, and ionization methods have pushed detection limits to unprecedented levels. However, the theoretical limits of detection have not yet been reached, suggesting significant potential for further optimization through innovative approaches and novel technological combinations.
This technical research aims to comprehensively explore current optimization strategies for GC-MS sensitivity in trace detection applications, identify key technological bottlenecks, and propose innovative solutions that could drive the next generation of ultra-sensitive analytical capabilities.
The evolution of GC-MS technology has been marked by continuous improvements in sensitivity, resolution, and reliability. Early systems were limited by rudimentary ionization techniques and low-resolution mass analyzers. Modern systems incorporate advanced features such as chemical ionization, tandem mass spectrometry, and high-resolution time-of-flight analyzers, dramatically enhancing detection capabilities.
Current technological trends in GC-MS focus on miniaturization, automation, and integration with artificial intelligence for data analysis. These developments aim to address the growing demand for portable, field-deployable systems that maintain laboratory-grade sensitivity while offering real-time analysis capabilities.
The primary objective of optimizing GC-MS sensitivity for trace detection is to achieve reliable identification and quantification of target analytes at increasingly lower concentrations. This is particularly crucial in applications such as environmental monitoring, food safety testing, forensic analysis, and pharmaceutical quality control, where detecting contaminants at trace levels can have significant health, safety, and regulatory implications.
Secondary objectives include improving signal-to-noise ratios, enhancing chromatographic resolution, reducing matrix interference effects, and developing more efficient sample preparation techniques. These factors collectively contribute to the overall sensitivity and reliability of GC-MS analysis for trace detection.
The technical challenges in this field are multifaceted, involving hardware limitations, sample preparation complexities, and data processing hurdles. Overcoming these challenges requires a multidisciplinary approach that integrates advances in materials science, electronics, computer algorithms, and analytical chemistry.
Recent breakthroughs in column technology, detector design, and ionization methods have pushed detection limits to unprecedented levels. However, the theoretical limits of detection have not yet been reached, suggesting significant potential for further optimization through innovative approaches and novel technological combinations.
This technical research aims to comprehensively explore current optimization strategies for GC-MS sensitivity in trace detection applications, identify key technological bottlenecks, and propose innovative solutions that could drive the next generation of ultra-sensitive analytical capabilities.
Market Demand Analysis for High-Sensitivity GC-MS
The global market for high-sensitivity GC-MS systems has been experiencing robust growth, driven primarily by increasing demands in environmental monitoring, food safety testing, forensic analysis, and pharmaceutical research. Current market estimates value the high-sensitivity analytical instruments sector at approximately 5.7 billion USD, with GC-MS systems representing a significant portion of this market.
Environmental monitoring represents one of the largest application segments, as regulatory bodies worldwide continue to lower permissible limits for pollutants in air, water, and soil. The detection of persistent organic pollutants (POPs), pesticides, and emerging contaminants at ultra-trace levels has become mandatory in many jurisdictions, creating sustained demand for more sensitive analytical capabilities.
The food and beverage industry constitutes another major market driver, with growing consumer awareness regarding food safety and increasing regulatory scrutiny. The need to detect pesticide residues, mycotoxins, and adulterants at parts-per-billion or parts-per-trillion levels has pushed laboratories to adopt more sensitive GC-MS technologies. Market research indicates that food safety testing laboratories are upgrading their analytical capabilities at a rate of 12% annually.
Pharmaceutical and clinical research sectors demonstrate increasing requirements for trace-level detection of impurities, metabolites, and biomarkers. The implementation of stricter quality control standards and the rise of precision medicine have amplified the need for highly sensitive analytical methods capable of detecting compounds at nanogram or picogram levels.
Forensic toxicology represents a rapidly expanding application area, with law enforcement agencies requiring lower detection limits for drugs of abuse, performance-enhancing substances, and novel psychoactive compounds. The proliferation of synthetic drugs has created particular challenges that only high-sensitivity GC-MS can address effectively.
Geographically, North America and Europe currently dominate the high-sensitivity GC-MS market, accounting for approximately 60% of global demand. However, the Asia-Pacific region is experiencing the fastest growth rate, driven by expanding environmental regulations, food export requirements, and increasing research activities in China, Japan, and South Korea.
Market forecasts project a compound annual growth rate of 6.8% for high-sensitivity GC-MS systems through 2028, with particularly strong growth in portable and field-deployable units that maintain laboratory-grade sensitivity. End-users consistently identify sensitivity improvements as their top priority when considering instrument upgrades, ranking above automation capabilities and software improvements in purchasing decisions.
Environmental monitoring represents one of the largest application segments, as regulatory bodies worldwide continue to lower permissible limits for pollutants in air, water, and soil. The detection of persistent organic pollutants (POPs), pesticides, and emerging contaminants at ultra-trace levels has become mandatory in many jurisdictions, creating sustained demand for more sensitive analytical capabilities.
The food and beverage industry constitutes another major market driver, with growing consumer awareness regarding food safety and increasing regulatory scrutiny. The need to detect pesticide residues, mycotoxins, and adulterants at parts-per-billion or parts-per-trillion levels has pushed laboratories to adopt more sensitive GC-MS technologies. Market research indicates that food safety testing laboratories are upgrading their analytical capabilities at a rate of 12% annually.
Pharmaceutical and clinical research sectors demonstrate increasing requirements for trace-level detection of impurities, metabolites, and biomarkers. The implementation of stricter quality control standards and the rise of precision medicine have amplified the need for highly sensitive analytical methods capable of detecting compounds at nanogram or picogram levels.
Forensic toxicology represents a rapidly expanding application area, with law enforcement agencies requiring lower detection limits for drugs of abuse, performance-enhancing substances, and novel psychoactive compounds. The proliferation of synthetic drugs has created particular challenges that only high-sensitivity GC-MS can address effectively.
Geographically, North America and Europe currently dominate the high-sensitivity GC-MS market, accounting for approximately 60% of global demand. However, the Asia-Pacific region is experiencing the fastest growth rate, driven by expanding environmental regulations, food export requirements, and increasing research activities in China, Japan, and South Korea.
Market forecasts project a compound annual growth rate of 6.8% for high-sensitivity GC-MS systems through 2028, with particularly strong growth in portable and field-deployable units that maintain laboratory-grade sensitivity. End-users consistently identify sensitivity improvements as their top priority when considering instrument upgrades, ranking above automation capabilities and software improvements in purchasing decisions.
Current Limitations and Technical Challenges in GC-MS Sensitivity
Despite significant advancements in Gas Chromatography-Mass Spectrometry (GC-MS) technology, several critical limitations continue to challenge the achievement of optimal sensitivity for trace detection applications. The fundamental sensitivity barrier stems from the inherent signal-to-noise ratio constraints in current detector designs, particularly when dealing with analyte concentrations in the sub-ppb (parts per billion) range. Modern GC-MS systems typically achieve detection limits around 1-10 pg, but many environmental, forensic, and clinical applications require detection at femtogram levels.
Sample preparation represents another significant challenge, with analyte loss during extraction, cleanup, and transfer processes often reaching 30-50%. This issue becomes particularly problematic when working with complex matrices such as biological fluids, environmental samples, or food products, where matrix effects can suppress ionization efficiency by up to 80% for certain compounds. The variability in recovery rates introduces substantial uncertainty in quantitative analyses at trace levels.
Ionization efficiency remains a persistent bottleneck, with conventional electron ionization (EI) sources typically converting only 0.01-0.1% of analyte molecules into detectable ions. While chemical ionization (CI) can improve this efficiency for certain compound classes, it introduces selectivity biases and often sacrifices structural information critical for compound identification. The trade-off between sensitivity and specificity continues to challenge method development for trace analysis.
Instrumental drift and calibration stability present additional complications, with sensitivity fluctuations of 5-15% commonly observed during extended analytical sequences. This variability necessitates frequent recalibration and internal standardization, increasing analysis complexity and reducing sample throughput. Temperature-dependent response factors further complicate quantitation, particularly for thermally labile compounds that may degrade during analysis.
Chromatographic resolution limitations also impact sensitivity, as co-eluting compounds can cause ion suppression or enhancement effects. Current column technologies struggle to separate structurally similar isomers or compounds with similar physicochemical properties, leading to potential misidentification or quantification errors at trace levels. Ultra-high resolution columns offer improved separation but often at the cost of increased analysis time and reduced sample capacity.
Data processing algorithms represent an emerging challenge, as current peak detection and deconvolution software struggles with distinguishing true signals from noise at the detection limit threshold. False positive rates can exceed 20% when working near instrumental detection limits, while false negatives may occur when true peaks are algorithmically filtered as noise. Advanced machine learning approaches show promise but require extensive validation across diverse sample types.
Sample preparation represents another significant challenge, with analyte loss during extraction, cleanup, and transfer processes often reaching 30-50%. This issue becomes particularly problematic when working with complex matrices such as biological fluids, environmental samples, or food products, where matrix effects can suppress ionization efficiency by up to 80% for certain compounds. The variability in recovery rates introduces substantial uncertainty in quantitative analyses at trace levels.
Ionization efficiency remains a persistent bottleneck, with conventional electron ionization (EI) sources typically converting only 0.01-0.1% of analyte molecules into detectable ions. While chemical ionization (CI) can improve this efficiency for certain compound classes, it introduces selectivity biases and often sacrifices structural information critical for compound identification. The trade-off between sensitivity and specificity continues to challenge method development for trace analysis.
Instrumental drift and calibration stability present additional complications, with sensitivity fluctuations of 5-15% commonly observed during extended analytical sequences. This variability necessitates frequent recalibration and internal standardization, increasing analysis complexity and reducing sample throughput. Temperature-dependent response factors further complicate quantitation, particularly for thermally labile compounds that may degrade during analysis.
Chromatographic resolution limitations also impact sensitivity, as co-eluting compounds can cause ion suppression or enhancement effects. Current column technologies struggle to separate structurally similar isomers or compounds with similar physicochemical properties, leading to potential misidentification or quantification errors at trace levels. Ultra-high resolution columns offer improved separation but often at the cost of increased analysis time and reduced sample capacity.
Data processing algorithms represent an emerging challenge, as current peak detection and deconvolution software struggles with distinguishing true signals from noise at the detection limit threshold. False positive rates can exceed 20% when working near instrumental detection limits, while false negatives may occur when true peaks are algorithmically filtered as noise. Advanced machine learning approaches show promise but require extensive validation across diverse sample types.
Current Methodologies for GC-MS Sensitivity Optimization
01 Enhancing GC-MS sensitivity through sample preparation techniques
Various sample preparation techniques can significantly enhance the sensitivity of GC-MS analysis. These include extraction methods, concentration procedures, and derivatization approaches that transform analytes into more volatile and thermally stable compounds. Proper sample preparation can reduce matrix interference, increase signal-to-noise ratio, and lower detection limits, thereby improving overall sensitivity of the GC-MS system.- Enhancing GC-MS sensitivity through sample preparation techniques: Various sample preparation techniques can significantly enhance the sensitivity of GC-MS analysis. These include extraction methods, concentration procedures, and derivatization techniques that transform analytes into more volatile compounds suitable for gas chromatography. Proper sample preparation can reduce matrix interference, increase signal-to-noise ratio, and lower detection limits, thereby improving overall sensitivity of the analytical method.
- Instrumental modifications for improved GC-MS sensitivity: Modifications to GC-MS instrumentation can enhance sensitivity through improvements in ionization efficiency, ion transmission, and detector response. These modifications include specialized ion sources, enhanced vacuum systems, optimized ion optics, and advanced detector technologies. Such instrumental enhancements allow for detection of lower analyte concentrations and improve the overall performance of GC-MS systems for trace analysis applications.
- Software and data processing approaches for sensitivity enhancement: Advanced software algorithms and data processing techniques can significantly improve the effective sensitivity of GC-MS analysis. These include noise reduction algorithms, deconvolution methods for overlapping peaks, selective ion monitoring, and machine learning approaches for pattern recognition. By extracting meaningful signals from background noise, these computational methods can lower detection limits and enhance the ability to identify trace compounds in complex matrices.
- Novel column technologies for enhanced separation and sensitivity: Specialized chromatographic columns with advanced stationary phases can improve GC-MS sensitivity by providing better separation of analytes, reducing peak broadening, and minimizing background interference. These column technologies include capillary columns with reduced internal diameter, novel polymer-based stationary phases, and columns with optimized film thickness. Better chromatographic separation leads to improved mass spectral quality and enhanced detection capabilities.
- Tandem and hybrid MS techniques for improved sensitivity: Tandem mass spectrometry (MS/MS) and hybrid mass analyzer configurations can dramatically improve the sensitivity and selectivity of GC-MS analysis. These approaches include triple quadrupole systems, ion trap-time of flight combinations, and high-resolution mass analyzers. By providing additional dimensions of separation and more specific detection of target analytes, these advanced MS techniques can achieve lower detection limits and better discrimination against matrix interference in complex samples.
02 Ionization and detection optimization for improved sensitivity
Optimizing ionization parameters and detection systems can substantially improve GC-MS sensitivity. This includes adjustments to electron energy, ion source temperature, and detector voltage. Advanced detection technologies such as electron multipliers, time-of-flight analyzers, and triple quadrupole systems can provide enhanced sensitivity for trace analysis. Fine-tuning these parameters based on target analytes can significantly lower detection limits.Expand Specific Solutions03 Column technology and chromatographic separation improvements
Advancements in column technology play a crucial role in enhancing GC-MS sensitivity. This includes the development of high-efficiency capillary columns with optimized stationary phases, reduced internal diameters, and improved thermal stability. Better chromatographic separation reduces co-elution issues and minimizes ion suppression effects, leading to improved detection of trace compounds and enhanced overall system sensitivity.Expand Specific Solutions04 Data processing and analytical software enhancements
Sophisticated data processing algorithms and analytical software can significantly improve the effective sensitivity of GC-MS systems. These include advanced peak deconvolution techniques, noise reduction algorithms, and automated signal enhancement tools. Machine learning approaches for spectral analysis can help identify compounds at lower concentrations and improve the signal-to-noise ratio without hardware modifications.Expand Specific Solutions05 Specialized GC-MS configurations for ultra-trace analysis
Custom GC-MS system configurations designed specifically for ultra-trace analysis can achieve exceptional sensitivity levels. These include tandem mass spectrometry (GC-MS/MS) setups, cold injection systems, and specialized interfaces. Cryogenic focusing techniques, splitless injection methods, and large volume injection approaches can also significantly enhance the detection capabilities for compounds present at extremely low concentrations.Expand Specific Solutions
Leading Manufacturers and Research Institutions in GC-MS Technology
The GC-MS trace detection market is in a mature growth phase, characterized by established technologies and steady innovation. The global market size for analytical instruments, including GC-MS systems, exceeds $5 billion annually with consistent growth rates of 5-7%. Leading players like Shimadzu, JEOL, and LECO have achieved high technical maturity in sensitivity optimization through innovations in ion source design, detector technology, and sample preparation techniques. Mid-tier competitors such as Micromass UK (Waters subsidiary) and DH Technologies are focusing on specialized applications and software integration. Academic institutions including Beijing University of Technology and Tokyo University of Science are contributing significant research in novel ionization methods and detector improvements, creating a competitive ecosystem balancing commercial innovation with fundamental research advances.
Shimadzu Corp.
Technical Solution: Shimadzu has developed advanced GC-MS sensitivity optimization technologies including their proprietary Advanced Flow Technology (AFT) and high-efficiency ion source designs. Their GCMS-TQ8050 NX triple quadrupole system achieves femtogram-level sensitivity through optimization of the ion optics system and heated multipole interface. The company's Active Time Management system minimizes downtime between analyses, while their Smart SIM technology automatically optimizes dwell times for Selected Ion Monitoring to maximize sensitivity. Shimadzu has also implemented advanced noise reduction algorithms and improved detector technologies that enhance signal-to-noise ratios by up to 30% compared to previous generations. Their dual-stage ion sampling interface effectively reduces chemical noise while maintaining high ion transmission efficiency.
Strengths: Industry-leading sensitivity levels reaching femtogram detection limits; comprehensive software tools for method optimization; robust hardware design ensuring consistent performance. Weaknesses: Higher initial investment cost compared to some competitors; complex systems may require specialized training for optimal operation; proprietary consumables can increase operational costs.
Commissariat à l´énergie atomique et aux énergies Alternatives
Technical Solution: The French Alternative Energies and Atomic Energy Commission (CEA) has developed specialized GC-MS sensitivity enhancement techniques focused on ultra-trace detection for nuclear safety and environmental monitoring applications. Their approach includes cryogenic trapping systems that pre-concentrate volatile analytes before introduction to the GC-MS, achieving enrichment factors of 100-1000×. CEA has pioneered specialized derivatization protocols optimized for specific trace contaminants, enhancing detector response by improving compound volatility and thermal stability. Their research has led to advanced multi-dimensional GC techniques (GC×GC-MS) that separate complex matrices more effectively, reducing chemical noise and improving detection limits. CEA has also developed custom ion source modifications that optimize ionization efficiency for specific compound classes, particularly for halogenated compounds and volatile organics at sub-ppt levels. Their work includes specialized software algorithms for signal processing that can extract meaningful data from near-noise level signals.
Strengths: Cutting-edge research capabilities; specialized expertise in ultra-trace detection; advanced multi-dimensional separation techniques. Weaknesses: Technologies often require significant customization for commercial applications; systems typically require highly trained operators; solutions may be less standardized than commercial offerings.
Key Innovations in Sample Preparation and Ionization Technologies
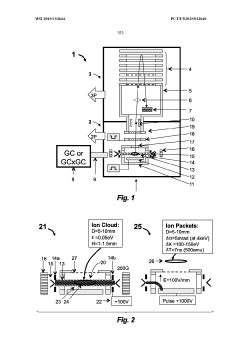
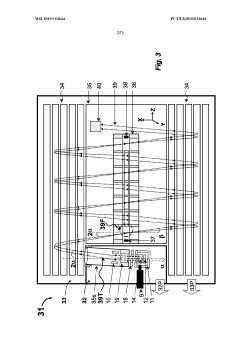
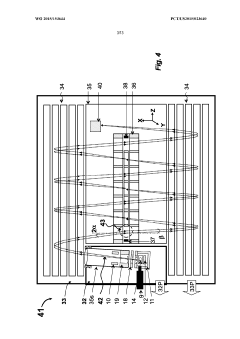
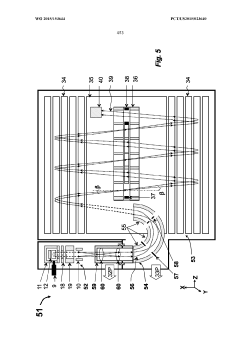
GC-TOF ms with improved detection limit
PatentWO2015153644A1
Innovation
- The implementation of a semi-open electron impact ion source coupled with a high-resolution multi-reflecting time-of-flight analyzer, along with specific ion-optical elements and pulsing techniques, enhances ion transmission and reduces time-of-flight aberrations, allowing for improved differentiation between sample and chemical background.
Gas chromatograph mass spectrometer
PatentWO2017154062A1
Innovation
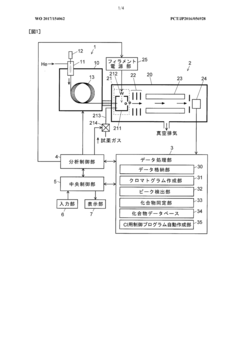
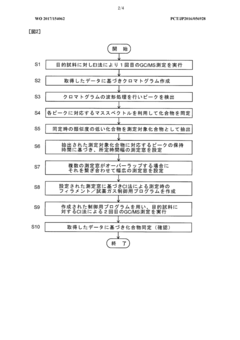
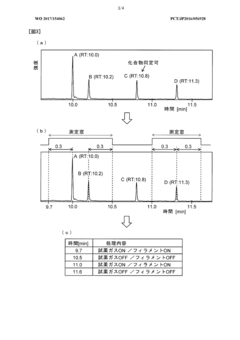
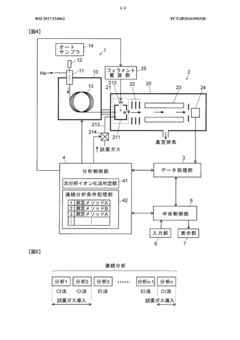
- A GC-MS system with an ion source capable of switching between electron ionization (EI) and chemical ionization (CI) methods, where the reagent gas supply and thermionic electron generation are controlled to minimize reagent gas ion adhesion by only generating ions during specific time ranges corresponding to detected compounds, thereby maintaining high sensitivity and accuracy.
Regulatory Standards for Trace Analysis Applications
Regulatory standards for trace analysis using GC-MS are increasingly stringent across various sectors, reflecting the growing importance of accurate detection at ultra-low concentrations. The Environmental Protection Agency (EPA) has established Method 8270 for semi-volatile organic compounds, requiring detection limits in the low parts-per-billion range, with specific quality control parameters including minimum signal-to-noise ratios of 3:1 for detection and 10:1 for quantification. These standards necessitate optimized GC-MS systems capable of consistently achieving these sensitivity thresholds.
In the pharmaceutical industry, ICH Q3 guidelines govern impurity analysis, mandating identification and quantification of impurities at 0.05% levels relative to the active pharmaceutical ingredient. This requires GC-MS systems to demonstrate exceptional sensitivity and reproducibility, with validation protocols that verify system performance at these trace levels. The FDA further enforces these requirements through cGMP regulations that demand robust analytical methods for product release testing.
Food safety regulations, particularly EU Regulation 396/2005 and the FDA's Food Safety Modernization Act, establish maximum residue limits (MRLs) for pesticides often at sub-ppb levels. These regulations require laboratories to implement highly sensitive GC-MS methods with documented validation procedures demonstrating capability to detect contaminants at regulatory thresholds. Method performance criteria typically include recovery rates of 70-120% and relative standard deviations below 20% at the limit of quantification.
Forensic toxicology applications face unique regulatory challenges, with standards set by organizations such as SWGTOX (Scientific Working Group for Forensic Toxicology) requiring detection capabilities in complex matrices at pg/mL levels. Chain-of-custody documentation and defensibility of analytical results place additional demands on GC-MS sensitivity optimization protocols, requiring comprehensive validation studies that demonstrate method robustness.
Emerging regulations for persistent organic pollutants under the Stockholm Convention have established global monitoring requirements with detection limits in the fg/g range for certain compounds. These ultra-trace requirements push conventional GC-MS technology to its limits, necessitating advanced sample preparation techniques and instrument modifications to achieve compliance. Laboratories must demonstrate proficiency through successful participation in international interlaboratory comparison studies.
Regulatory compliance also extends to laboratory accreditation standards such as ISO/IEC 17025, which requires documented evidence of method validation, uncertainty estimation, and ongoing quality control measures. This includes regular sensitivity verification through analysis of certified reference materials and participation in proficiency testing programs that evaluate a laboratory's capability to detect trace analytes at regulatory thresholds.
In the pharmaceutical industry, ICH Q3 guidelines govern impurity analysis, mandating identification and quantification of impurities at 0.05% levels relative to the active pharmaceutical ingredient. This requires GC-MS systems to demonstrate exceptional sensitivity and reproducibility, with validation protocols that verify system performance at these trace levels. The FDA further enforces these requirements through cGMP regulations that demand robust analytical methods for product release testing.
Food safety regulations, particularly EU Regulation 396/2005 and the FDA's Food Safety Modernization Act, establish maximum residue limits (MRLs) for pesticides often at sub-ppb levels. These regulations require laboratories to implement highly sensitive GC-MS methods with documented validation procedures demonstrating capability to detect contaminants at regulatory thresholds. Method performance criteria typically include recovery rates of 70-120% and relative standard deviations below 20% at the limit of quantification.
Forensic toxicology applications face unique regulatory challenges, with standards set by organizations such as SWGTOX (Scientific Working Group for Forensic Toxicology) requiring detection capabilities in complex matrices at pg/mL levels. Chain-of-custody documentation and defensibility of analytical results place additional demands on GC-MS sensitivity optimization protocols, requiring comprehensive validation studies that demonstrate method robustness.
Emerging regulations for persistent organic pollutants under the Stockholm Convention have established global monitoring requirements with detection limits in the fg/g range for certain compounds. These ultra-trace requirements push conventional GC-MS technology to its limits, necessitating advanced sample preparation techniques and instrument modifications to achieve compliance. Laboratories must demonstrate proficiency through successful participation in international interlaboratory comparison studies.
Regulatory compliance also extends to laboratory accreditation standards such as ISO/IEC 17025, which requires documented evidence of method validation, uncertainty estimation, and ongoing quality control measures. This includes regular sensitivity verification through analysis of certified reference materials and participation in proficiency testing programs that evaluate a laboratory's capability to detect trace analytes at regulatory thresholds.
Cost-Benefit Analysis of Advanced GC-MS Sensitivity Solutions
When evaluating advanced sensitivity solutions for GC-MS trace detection, a comprehensive cost-benefit analysis reveals significant financial considerations that organizations must address. Initial capital expenditure for high-sensitivity GC-MS systems typically ranges from $150,000 to $500,000, depending on configuration and sensitivity specifications. This represents a substantial investment that must be justified through operational benefits and analytical capabilities.
Operational costs present another critical dimension, with advanced systems requiring specialized maintenance contracts ranging from $10,000 to $30,000 annually. Additionally, these systems often demand higher-purity carrier gases and more frequent replacement of consumables, increasing routine operational expenses by approximately 30-40% compared to standard systems.
Training requirements constitute a frequently overlooked cost factor. Technical staff require specialized training for optimal operation of high-sensitivity systems, with initial training programs costing $5,000-$15,000 per technician. Organizations must also account for productivity losses during the learning curve period, typically 2-3 months before achieving optimal efficiency.
Against these costs, the benefits of enhanced sensitivity present compelling value propositions. Detection limits improved by 1-3 orders of magnitude enable identification of compounds previously below detection thresholds, creating new analytical capabilities and potential revenue streams. For environmental monitoring applications, this translates to compliance with increasingly stringent regulatory requirements that standard systems cannot meet.
Laboratory throughput improvements represent another significant benefit, with advanced systems reducing sample preparation requirements and enabling direct analysis of lower concentration samples. This can increase laboratory capacity by 20-40% for certain applications, effectively amortizing the higher capital costs across more billable analyses.
Return on investment calculations indicate that organizations performing high-volume trace analysis typically achieve ROI within 2-4 years, while specialized research facilities may realize returns through grant funding opportunities specifically targeting advanced analytical capabilities. The intangible benefits of enhanced scientific reputation and competitive advantage in securing contracts for challenging analytical projects further strengthen the value proposition.
For organizations considering implementation, a phased approach often optimizes the cost-benefit ratio, beginning with targeted upgrades to existing systems before committing to comprehensive platform replacements. This strategy allows for validation of sensitivity improvements in specific applications while distributing capital expenditures over a longer timeframe.
Operational costs present another critical dimension, with advanced systems requiring specialized maintenance contracts ranging from $10,000 to $30,000 annually. Additionally, these systems often demand higher-purity carrier gases and more frequent replacement of consumables, increasing routine operational expenses by approximately 30-40% compared to standard systems.
Training requirements constitute a frequently overlooked cost factor. Technical staff require specialized training for optimal operation of high-sensitivity systems, with initial training programs costing $5,000-$15,000 per technician. Organizations must also account for productivity losses during the learning curve period, typically 2-3 months before achieving optimal efficiency.
Against these costs, the benefits of enhanced sensitivity present compelling value propositions. Detection limits improved by 1-3 orders of magnitude enable identification of compounds previously below detection thresholds, creating new analytical capabilities and potential revenue streams. For environmental monitoring applications, this translates to compliance with increasingly stringent regulatory requirements that standard systems cannot meet.
Laboratory throughput improvements represent another significant benefit, with advanced systems reducing sample preparation requirements and enabling direct analysis of lower concentration samples. This can increase laboratory capacity by 20-40% for certain applications, effectively amortizing the higher capital costs across more billable analyses.
Return on investment calculations indicate that organizations performing high-volume trace analysis typically achieve ROI within 2-4 years, while specialized research facilities may realize returns through grant funding opportunities specifically targeting advanced analytical capabilities. The intangible benefits of enhanced scientific reputation and competitive advantage in securing contracts for challenging analytical projects further strengthen the value proposition.
For organizations considering implementation, a phased approach often optimizes the cost-benefit ratio, beginning with targeted upgrades to existing systems before committing to comprehensive platform replacements. This strategy allows for validation of sensitivity improvements in specific applications while distributing capital expenditures over a longer timeframe.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!