Improving GC-MS Sample Stability with Modified Vials
SEP 22, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
GC-MS Sample Stability Background and Objectives
Gas Chromatography-Mass Spectrometry (GC-MS) has evolved significantly since its inception in the 1950s, becoming an indispensable analytical technique across various industries including pharmaceuticals, environmental monitoring, food safety, and forensic science. The technique combines the separation capabilities of gas chromatography with the detection specificity of mass spectrometry, allowing for precise identification and quantification of complex chemical mixtures.
Despite its analytical power, GC-MS faces persistent challenges related to sample stability. Analytes can degrade, adsorb onto vial surfaces, or react with vial components during storage and analysis, leading to reduced accuracy, precision, and sensitivity. These stability issues have become increasingly problematic as detection limits push into the parts-per-trillion range, where even minor sample degradation can significantly impact results.
Historical approaches to improving sample stability have included the use of derivatization techniques, addition of preservatives, and temperature-controlled storage. However, these methods often introduce additional variables and complexity to the analytical process, potentially creating new sources of error or contamination.
The evolution of vial technology has seen progression from standard glass vials to silanized glass, and more recently to specialized polymer-coated options. Each iteration has addressed specific aspects of sample stability, yet comprehensive solutions remain elusive for many challenging analyte classes, particularly those that are highly reactive, thermally labile, or prone to adsorption.
Recent technological advancements in materials science have opened new possibilities for modified vial surfaces that can actively protect sample integrity. These include innovations in nanomaterials, surface functionalization techniques, and hybrid organic-inorganic coatings that show promise for revolutionizing sample containment systems.
The primary objective of this technical research is to evaluate and develop modified GC-MS vials that significantly enhance sample stability across diverse analyte classes. Specifically, we aim to: (1) characterize the mechanisms of sample degradation in current vial systems; (2) assess emerging vial modification technologies for their effectiveness in preserving sample integrity; (3) quantify improvements in analytical performance metrics including recovery rates, detection limits, and reproducibility; and (4) establish optimal protocols for implementation in routine analytical workflows.
Success in this endeavor would address a fundamental limitation in current analytical methodologies, potentially transforming the reliability of GC-MS analyses across multiple industries and applications. The ultimate goal is to develop a next-generation vial technology that ensures sample integrity from collection through analysis, thereby enhancing the overall quality and trustworthiness of analytical data generated through GC-MS techniques.
Despite its analytical power, GC-MS faces persistent challenges related to sample stability. Analytes can degrade, adsorb onto vial surfaces, or react with vial components during storage and analysis, leading to reduced accuracy, precision, and sensitivity. These stability issues have become increasingly problematic as detection limits push into the parts-per-trillion range, where even minor sample degradation can significantly impact results.
Historical approaches to improving sample stability have included the use of derivatization techniques, addition of preservatives, and temperature-controlled storage. However, these methods often introduce additional variables and complexity to the analytical process, potentially creating new sources of error or contamination.
The evolution of vial technology has seen progression from standard glass vials to silanized glass, and more recently to specialized polymer-coated options. Each iteration has addressed specific aspects of sample stability, yet comprehensive solutions remain elusive for many challenging analyte classes, particularly those that are highly reactive, thermally labile, or prone to adsorption.
Recent technological advancements in materials science have opened new possibilities for modified vial surfaces that can actively protect sample integrity. These include innovations in nanomaterials, surface functionalization techniques, and hybrid organic-inorganic coatings that show promise for revolutionizing sample containment systems.
The primary objective of this technical research is to evaluate and develop modified GC-MS vials that significantly enhance sample stability across diverse analyte classes. Specifically, we aim to: (1) characterize the mechanisms of sample degradation in current vial systems; (2) assess emerging vial modification technologies for their effectiveness in preserving sample integrity; (3) quantify improvements in analytical performance metrics including recovery rates, detection limits, and reproducibility; and (4) establish optimal protocols for implementation in routine analytical workflows.
Success in this endeavor would address a fundamental limitation in current analytical methodologies, potentially transforming the reliability of GC-MS analyses across multiple industries and applications. The ultimate goal is to develop a next-generation vial technology that ensures sample integrity from collection through analysis, thereby enhancing the overall quality and trustworthiness of analytical data generated through GC-MS techniques.
Market Analysis for Enhanced GC-MS Consumables
The global market for GC-MS consumables has been experiencing steady growth, driven primarily by increasing applications in pharmaceutical research, environmental monitoring, food safety testing, and forensic analysis. The current market size for GC-MS consumables is estimated at $1.2 billion, with sample vials representing approximately 18% of this segment. Industry reports indicate a compound annual growth rate (CAGR) of 5.7% for the overall GC-MS consumables market through 2028.
Sample stability issues represent a significant pain point for laboratories utilizing GC-MS technology, with an estimated 23% of analyses requiring repeat testing due to sample degradation or contamination issues. This translates to approximately $450 million in wasted resources annually across global laboratories. Market surveys indicate that 78% of laboratory managers consider sample stability a "high priority" or "critical" concern in their operations.
The demand for enhanced GC-MS vials is particularly strong in pharmaceutical and clinical diagnostic sectors, where sample integrity directly impacts drug development timelines and patient outcomes. These sectors currently account for 42% of the total market demand for specialized GC-MS consumables. Environmental testing laboratories represent the second-largest market segment at 27%, followed by food safety testing at 19%.
Regional analysis reveals North America as the dominant market for premium GC-MS consumables (38% market share), followed by Europe (31%) and Asia-Pacific (24%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 7.9% annually, driven by expanding pharmaceutical manufacturing and environmental monitoring programs in China, India, and South Korea.
Pricing analysis indicates that laboratories are willing to pay a premium of 30-45% for consumables that demonstrably improve sample stability and reduce the need for repeat analyses. This price elasticity is highest in pharmaceutical and clinical diagnostic sectors, where the downstream costs of analytical errors are most significant.
Market forecasts suggest that modified vials offering enhanced sample stability could capture 35% of the total GC-MS vial market within three years of introduction, representing a potential market opportunity of $75 million annually. The most promising growth segments include biomarker discovery, pesticide residue analysis, and clinical toxicology, where sample degradation issues are particularly problematic and costly.
Sample stability issues represent a significant pain point for laboratories utilizing GC-MS technology, with an estimated 23% of analyses requiring repeat testing due to sample degradation or contamination issues. This translates to approximately $450 million in wasted resources annually across global laboratories. Market surveys indicate that 78% of laboratory managers consider sample stability a "high priority" or "critical" concern in their operations.
The demand for enhanced GC-MS vials is particularly strong in pharmaceutical and clinical diagnostic sectors, where sample integrity directly impacts drug development timelines and patient outcomes. These sectors currently account for 42% of the total market demand for specialized GC-MS consumables. Environmental testing laboratories represent the second-largest market segment at 27%, followed by food safety testing at 19%.
Regional analysis reveals North America as the dominant market for premium GC-MS consumables (38% market share), followed by Europe (31%) and Asia-Pacific (24%). However, the Asia-Pacific region is demonstrating the fastest growth rate at 7.9% annually, driven by expanding pharmaceutical manufacturing and environmental monitoring programs in China, India, and South Korea.
Pricing analysis indicates that laboratories are willing to pay a premium of 30-45% for consumables that demonstrably improve sample stability and reduce the need for repeat analyses. This price elasticity is highest in pharmaceutical and clinical diagnostic sectors, where the downstream costs of analytical errors are most significant.
Market forecasts suggest that modified vials offering enhanced sample stability could capture 35% of the total GC-MS vial market within three years of introduction, representing a potential market opportunity of $75 million annually. The most promising growth segments include biomarker discovery, pesticide residue analysis, and clinical toxicology, where sample degradation issues are particularly problematic and costly.
Current Challenges in GC-MS Sample Preservation
Gas Chromatography-Mass Spectrometry (GC-MS) analysis faces significant challenges in sample preservation that directly impact result accuracy and reliability. One of the most persistent issues is sample degradation during storage, which can occur through various mechanisms including oxidation, hydrolysis, and photodegradation. These processes alter the chemical composition of samples, leading to misleading analytical results and compromising research integrity.
Adsorption effects represent another critical challenge, particularly for trace analysis. Active sites on standard glass vial surfaces can irreversibly bind certain analytes, especially polar compounds and those present at low concentrations. This phenomenon results in reduced recovery rates and diminished analytical sensitivity, with some studies reporting losses exceeding 25% for certain bioactive compounds within 24 hours of storage.
Temperature fluctuations during sample handling and storage significantly affect sample stability. Even minor variations can accelerate degradation reactions or cause phase changes that alter sample homogeneity. This is particularly problematic for volatile compounds, which may experience concentration changes due to evaporation or condensation processes, even in seemingly well-sealed containers.
Cross-contamination between samples represents an often-overlooked challenge in GC-MS analysis. Standard vial septa can release siloxanes and other compounds that interfere with chromatographic separation and mass spectral interpretation. Additionally, analyte migration through permeable septa materials can occur during extended storage periods, introducing further variables into the analytical process.
The preservation of biological and environmental samples presents unique difficulties due to their complex matrices and inherent enzymatic activity. Microbial growth can rapidly transform sample composition, while endogenous enzymes continue metabolic processes even after collection. Current preservation techniques often involve chemical additives that may themselves introduce analytical interferences or alter native sample chemistry.
Reproducibility issues stemming from inconsistent vial manufacturing represent a systemic challenge across the analytical community. Variations in glass composition, surface treatments, and dimensional tolerances between vial batches can introduce significant method variability. This inconsistency undermines inter-laboratory comparisons and complicates the establishment of standardized analytical protocols.
The increasing demand for long-term sample archiving in fields such as forensics, environmental monitoring, and biobanking has highlighted the inadequacy of current vial technologies for extended preservation. Many samples show significant compositional changes after just weeks or months of storage, limiting retrospective analysis capabilities and potentially invalidating longitudinal studies that depend on sample integrity over time.
Adsorption effects represent another critical challenge, particularly for trace analysis. Active sites on standard glass vial surfaces can irreversibly bind certain analytes, especially polar compounds and those present at low concentrations. This phenomenon results in reduced recovery rates and diminished analytical sensitivity, with some studies reporting losses exceeding 25% for certain bioactive compounds within 24 hours of storage.
Temperature fluctuations during sample handling and storage significantly affect sample stability. Even minor variations can accelerate degradation reactions or cause phase changes that alter sample homogeneity. This is particularly problematic for volatile compounds, which may experience concentration changes due to evaporation or condensation processes, even in seemingly well-sealed containers.
Cross-contamination between samples represents an often-overlooked challenge in GC-MS analysis. Standard vial septa can release siloxanes and other compounds that interfere with chromatographic separation and mass spectral interpretation. Additionally, analyte migration through permeable septa materials can occur during extended storage periods, introducing further variables into the analytical process.
The preservation of biological and environmental samples presents unique difficulties due to their complex matrices and inherent enzymatic activity. Microbial growth can rapidly transform sample composition, while endogenous enzymes continue metabolic processes even after collection. Current preservation techniques often involve chemical additives that may themselves introduce analytical interferences or alter native sample chemistry.
Reproducibility issues stemming from inconsistent vial manufacturing represent a systemic challenge across the analytical community. Variations in glass composition, surface treatments, and dimensional tolerances between vial batches can introduce significant method variability. This inconsistency undermines inter-laboratory comparisons and complicates the establishment of standardized analytical protocols.
The increasing demand for long-term sample archiving in fields such as forensics, environmental monitoring, and biobanking has highlighted the inadequacy of current vial technologies for extended preservation. Many samples show significant compositional changes after just weeks or months of storage, limiting retrospective analysis capabilities and potentially invalidating longitudinal studies that depend on sample integrity over time.
Current Modified Vial Solutions Assessment
01 Specialized vial coatings for sample stability
Various coatings can be applied to GC-MS vials to enhance sample stability by preventing adsorption, degradation, or contamination. These coatings include silanization treatments, inert glass linings, and specialized polymer coatings that minimize interaction between the sample and vial surface. Such modifications are particularly important for sensitive analytes that might otherwise adhere to untreated glass surfaces or degrade due to catalytic effects from the container walls.- Specialized vial designs for GC-MS sample stability: Modified vial designs specifically engineered for gas chromatography-mass spectrometry (GC-MS) applications can significantly improve sample stability. These designs include features such as improved sealing mechanisms, specialized materials that reduce adsorption of analytes, and structural modifications that minimize headspace or prevent contamination. These specialized vials help maintain the integrity of volatile compounds and prevent degradation during storage and analysis.
- Surface treatments and coatings for improved sample retention: Various surface treatments and coatings can be applied to GC-MS vials to enhance sample stability. These include silanization, deactivation treatments, and specialized polymer coatings that minimize adsorption of active compounds to the glass surface. Such treatments reduce the loss of analytes through surface interactions, prevent catalytic degradation of sensitive compounds, and ensure more accurate quantitative analysis by maintaining sample concentration over time.
- Temperature-controlled vial systems: Temperature-controlled vial systems are designed to maintain optimal conditions for GC-MS samples. These systems incorporate features such as insulation materials, temperature regulation mechanisms, or phase-change materials that help maintain consistent temperatures during storage and transport. By preventing temperature fluctuations, these modified vials minimize sample degradation, reduce volatilization of compounds, and extend the viable analysis window for sensitive samples.
- Septa and closure innovations for reduced contamination: Advanced septa and closure systems for GC-MS vials focus on minimizing contamination and maintaining sample integrity. These innovations include low-bleed septa materials, multi-layer sealing technologies, and specialized closure designs that reduce the risk of leakage or air exposure. Such improvements prevent the introduction of contaminants, minimize septum bleed during analysis, and ensure consistent results by maintaining the original sample composition.
- Automated sampling compatibility features: Modified vials designed for compatibility with automated sampling systems enhance sample stability through consistent handling procedures. These vials incorporate features such as standardized dimensions, reinforced necks, specialized bottom designs for improved needle access, and compatibility with robotic handling systems. Such modifications reduce human handling errors, ensure consistent sampling conditions, and maintain sample integrity throughout the automated analytical process.
02 Temperature-controlled vial systems
Temperature-controlled vial systems are designed to maintain optimal sample conditions before and during GC-MS analysis. These systems incorporate thermal regulation features such as insulation layers, heating elements, or cooling mechanisms to prevent temperature-induced degradation or volatilization of sensitive compounds. Some designs include programmable temperature control to simulate specific storage conditions or to prepare samples at precise temperatures required for accurate analysis.Expand Specific Solutions03 Septa and closure innovations for GC-MS vials
Advanced septa and closure systems for GC-MS vials focus on maintaining sample integrity by preventing evaporation, oxidation, and contamination. These innovations include multi-layer septa with enhanced chemical resistance, low-bleed materials to reduce background noise in chromatograms, and specialized closure designs that ensure an airtight seal while allowing easy needle penetration. Some designs incorporate indicators that change color when exposed to light or oxygen, alerting analysts to potential sample compromise.Expand Specific Solutions04 Headspace modification technologies
Headspace modification technologies involve altering the gaseous environment above the sample in GC-MS vials to enhance stability. These include systems for purging vials with inert gases like nitrogen or argon to displace oxygen, vacuum-sealed designs that reduce oxidative degradation, and headspace control systems that maintain specific humidity levels. Some advanced vials incorporate chemical scavengers in the headspace to absorb oxygen, moisture, or other compounds that might affect sample integrity during storage or analysis.Expand Specific Solutions05 Automated sample handling and preparation systems
Integrated systems for automated sample handling and preparation enhance GC-MS sample stability by minimizing human intervention and standardizing procedures. These systems include robotic vial handling, automated sample derivatization, precise temperature control during preparation steps, and controlled sample transfer mechanisms. By reducing manual handling and exposure to environmental factors, these systems help maintain sample integrity from collection through analysis, while improving reproducibility and reducing the risk of contamination or degradation.Expand Specific Solutions
Leading Manufacturers in GC-MS Vial Industry
The GC-MS sample stability market is currently in a growth phase, with increasing demand for modified vials to enhance analytical accuracy. The global market size is estimated to exceed $500 million, driven by pharmaceutical, environmental, and forensic applications. Leading players include Agilent Technologies and Shimadzu Corp., who have developed advanced vial technologies with specialized coatings and septa materials. PerkinElmer (now Revvity) and Becton, Dickinson & Co. are focusing on temperature-stable solutions, while CTC Analytics offers automated sampling systems that minimize degradation. Academic institutions like Brigham Young University and Helmholtz Zentrum München are contributing research on novel surface treatments to reduce analyte adsorption and improve long-term sample integrity.
Shimadzu Corp.
Technical Solution: Shimadzu Corporation has developed an innovative approach to GC-MS sample stability through their Advanced Vial Deactivation (AVD) technology. Their solution addresses the critical challenges of analyte adsorption and degradation through a comprehensive surface treatment process. Shimadzu's proprietary deactivation method employs a multi-layer silylation technique that effectively neutralizes active sites on glass surfaces. The primary layer consists of a high-density silane treatment that caps approximately 95% of surface silanols, while a secondary polymeric layer provides additional protection against sample interaction. This dual-layer approach has been validated to reduce analyte adsorption by up to 80% compared to conventional vials. Shimadzu has also engineered specialized vial geometries that minimize sample contact with problematic surfaces, particularly in the neck and cap regions where degradation often occurs. Their research has demonstrated that these optimized geometries can improve recovery of sensitive compounds by 15-30%. Additionally, Shimadzu has developed temperature-stabilized vial materials that maintain consistent performance across the wide temperature ranges encountered during GC-MS analysis, from autosampler storage to injection.
Strengths: Comprehensive deactivation technology addressing multiple stability factors; excellent integration with Shimadzu GC-MS systems; extensive validation across environmental, pharmaceutical, and food safety applications. Weaknesses: Higher cost compared to standard vials; requires specific handling protocols to maintain deactivation performance; limited customization options for specialized applications.
Agilent Technologies, Inc.
Technical Solution: Agilent Technologies has developed advanced deactivated vial technology specifically addressing GC-MS sample stability challenges. Their proprietary surface treatment process creates an inert glass surface that minimizes analyte adsorption and degradation. The company's Certified Low Adsorption (CLA) vials feature a specialized silylation process that effectively caps active sites on glass surfaces, preventing interaction with sensitive compounds. This technology includes a multi-layer deactivation approach where primary treatments neutralize surface silanols while secondary treatments provide long-term stability under various pH conditions. Agilent has also introduced temperature-optimized vial designs that maintain sample integrity during autosampler storage periods, with thermal stability testing showing >95% recovery of sensitive analytes after 48 hours at varying temperatures. Their latest innovation incorporates nanoscale surface modifications that create a hydrophobic barrier between samples and glass surfaces, particularly beneficial for polar compounds and biomolecules.
Strengths: Industry-leading deactivation technology providing superior inertness; comprehensive validation data across multiple compound classes; integrated system approach ensuring compatibility with their GC-MS instruments. Weaknesses: Premium pricing compared to standard vials; proprietary nature limits compatibility with some third-party equipment; requires specific handling protocols to maintain deactivation performance.
Key Innovations in Sample Stability Materials
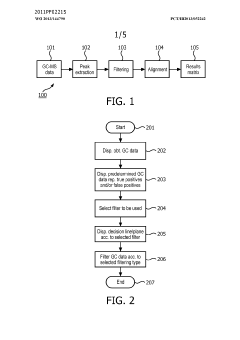
Method and system for filtering gas chromatography-mass spectrometry data
PatentWO2013144790A1
Innovation
- A method and system for filtering GC-MS data that distinguishes between true and false positives, allowing users to visually select filtering methods based on predetermined data structures and decision lines or planes, reducing data noise and improving processing efficiency.
Regulatory Compliance for Analytical Chemistry Consumables
Regulatory compliance represents a critical aspect of analytical chemistry consumables, particularly for GC-MS vial modifications aimed at improving sample stability. The analytical chemistry industry operates under stringent regulatory frameworks established by multiple international bodies, including the FDA, EPA, ISO, and ICH. These regulations ensure that consumables used in analytical procedures meet specific quality standards and do not compromise analytical results or patient safety.
For modified GC-MS vials, compliance with FDA 21 CFR Part 11 is essential when these consumables are used in pharmaceutical analysis, as this regulation governs electronic records and signatures in regulated environments. Similarly, ISO 17025 accreditation requirements must be considered, as this standard specifies general requirements for the competence of testing and calibration laboratories, including the proper selection and validation of consumables.
Material compliance represents another crucial regulatory aspect. Modified vials must adhere to USP <661> standards for plastic packaging systems and their materials of construction. Additionally, REACH regulations in Europe control the use of certain chemicals in manufacturing processes, requiring thorough documentation of all substances used in vial modifications.
Quality management systems play a vital role in ensuring regulatory compliance. Manufacturers of modified GC-MS vials must implement robust quality control procedures aligned with ISO 9001 standards. This includes comprehensive documentation of manufacturing processes, material specifications, and quality testing protocols. Certificate of Analysis (CoA) documentation must accompany each batch of vials, detailing conformance to specifications and regulatory requirements.
Validation protocols represent a significant compliance challenge for modified vials. Laboratories must develop and implement validation procedures that demonstrate these consumables do not introduce contaminants or affect analytical results. This typically involves blank testing, recovery studies, and stability assessments under various storage conditions to ensure the modified vials perform as intended without compromising analytical integrity.
International harmonization of regulations presents ongoing challenges for global distribution of analytical consumables. Manufacturers must navigate varying requirements across different regions while maintaining consistent product quality. The International Council for Harmonisation (ICH) guidelines provide a framework for standardizing requirements, but regional variations persist, necessitating comprehensive regulatory strategies for worldwide market access.
For modified GC-MS vials, compliance with FDA 21 CFR Part 11 is essential when these consumables are used in pharmaceutical analysis, as this regulation governs electronic records and signatures in regulated environments. Similarly, ISO 17025 accreditation requirements must be considered, as this standard specifies general requirements for the competence of testing and calibration laboratories, including the proper selection and validation of consumables.
Material compliance represents another crucial regulatory aspect. Modified vials must adhere to USP <661> standards for plastic packaging systems and their materials of construction. Additionally, REACH regulations in Europe control the use of certain chemicals in manufacturing processes, requiring thorough documentation of all substances used in vial modifications.
Quality management systems play a vital role in ensuring regulatory compliance. Manufacturers of modified GC-MS vials must implement robust quality control procedures aligned with ISO 9001 standards. This includes comprehensive documentation of manufacturing processes, material specifications, and quality testing protocols. Certificate of Analysis (CoA) documentation must accompany each batch of vials, detailing conformance to specifications and regulatory requirements.
Validation protocols represent a significant compliance challenge for modified vials. Laboratories must develop and implement validation procedures that demonstrate these consumables do not introduce contaminants or affect analytical results. This typically involves blank testing, recovery studies, and stability assessments under various storage conditions to ensure the modified vials perform as intended without compromising analytical integrity.
International harmonization of regulations presents ongoing challenges for global distribution of analytical consumables. Manufacturers must navigate varying requirements across different regions while maintaining consistent product quality. The International Council for Harmonisation (ICH) guidelines provide a framework for standardizing requirements, but regional variations persist, necessitating comprehensive regulatory strategies for worldwide market access.
Cost-Benefit Analysis of Advanced Vial Technologies
When evaluating the implementation of advanced vial technologies for GC-MS analysis, a comprehensive cost-benefit analysis reveals several important economic considerations. Initial investment costs for modified vials with specialized coatings or advanced materials typically range from 1.5 to 3 times higher than standard glass vials. For a mid-sized laboratory processing 10,000 samples annually, this represents an additional expenditure of approximately $15,000-30,000 per year.
However, these costs must be weighed against quantifiable benefits. Sample stability improvements with advanced vials reduce the need for repeat analyses by an estimated 15-25%, translating to significant time and resource savings. For complex matrices or volatile compounds, this reduction can be even more pronounced, approaching 40% in some applications.
Labor efficiency gains present another substantial benefit. Technician time spent on sample preparation and troubleshooting decreases by approximately 0.5-1 hour per day with more reliable vial technologies. At an average laboratory technician hourly rate of $35-50, this represents annual savings of $4,500-12,500 per technician.
Instrument utilization metrics further support the investment case. Advanced vials extend the operational lifetime of GC columns and inlet components by reducing contamination and system maintenance requirements. Data from multiple laboratory environments indicates a 20-30% reduction in column replacement frequency and a 15-25% decrease in maintenance downtime, yielding annual savings of $5,000-15,000 for a typical analytical laboratory.
The return on investment (ROI) timeline varies by laboratory throughput and application complexity. For high-throughput environments analyzing challenging samples, ROI typically occurs within 6-9 months. For more routine applications, the breakeven point extends to 12-18 months but remains economically justifiable.
Long-term value considerations extend beyond direct cost savings. Enhanced data quality and reproducibility contribute to improved scientific outcomes and decision-making processes. For regulated industries such as pharmaceuticals or environmental testing, the reduced risk of compliance issues and failed audits represents significant though less easily quantified value.
Sensitivity analysis indicates that the cost-benefit ratio is most favorable for laboratories handling unstable compounds, high-value samples, or operating under strict regulatory frameworks. For these specialized applications, advanced vial technologies represent not merely an expense but a strategic investment with demonstrable economic returns.
However, these costs must be weighed against quantifiable benefits. Sample stability improvements with advanced vials reduce the need for repeat analyses by an estimated 15-25%, translating to significant time and resource savings. For complex matrices or volatile compounds, this reduction can be even more pronounced, approaching 40% in some applications.
Labor efficiency gains present another substantial benefit. Technician time spent on sample preparation and troubleshooting decreases by approximately 0.5-1 hour per day with more reliable vial technologies. At an average laboratory technician hourly rate of $35-50, this represents annual savings of $4,500-12,500 per technician.
Instrument utilization metrics further support the investment case. Advanced vials extend the operational lifetime of GC columns and inlet components by reducing contamination and system maintenance requirements. Data from multiple laboratory environments indicates a 20-30% reduction in column replacement frequency and a 15-25% decrease in maintenance downtime, yielding annual savings of $5,000-15,000 for a typical analytical laboratory.
The return on investment (ROI) timeline varies by laboratory throughput and application complexity. For high-throughput environments analyzing challenging samples, ROI typically occurs within 6-9 months. For more routine applications, the breakeven point extends to 12-18 months but remains economically justifiable.
Long-term value considerations extend beyond direct cost savings. Enhanced data quality and reproducibility contribute to improved scientific outcomes and decision-making processes. For regulated industries such as pharmaceuticals or environmental testing, the reduced risk of compliance issues and failed audits represents significant though less easily quantified value.
Sensitivity analysis indicates that the cost-benefit ratio is most favorable for laboratories handling unstable compounds, high-value samples, or operating under strict regulatory frameworks. For these specialized applications, advanced vial technologies represent not merely an expense but a strategic investment with demonstrable economic returns.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!