Phosphorylation Vs Acetylation: Impact on Protein Function
SEP 23, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PTM Background and Research Objectives
Post-translational modifications (PTMs) represent critical regulatory mechanisms that significantly expand the functional diversity of proteins beyond their primary amino acid sequence. Among these modifications, phosphorylation and acetylation stand as two of the most extensively studied and biologically significant PTMs. The historical trajectory of PTM research began in the mid-20th century with the discovery of protein phosphorylation, which was later recognized as a fundamental regulatory mechanism when Fischer and Krebs elucidated the role of phosphorylase kinase in glycogen metabolism, earning them the Nobel Prize in 1992.
The field has since witnessed exponential growth, with technological advancements in mass spectrometry and proteomics enabling the identification of thousands of modification sites across the proteome. Recent developments in high-throughput techniques have facilitated comprehensive mapping of the "PTM code," revealing intricate crosstalk between different modifications and their collective impact on protein function.
Current trends in PTM research emphasize the integration of multiple modification types within regulatory networks and their roles in cellular signaling cascades. The emergence of systems biology approaches has shifted focus from studying individual modifications to understanding the complex interplay between various PTMs and their cumulative effects on protein behavior, subcellular localization, and interaction networks.
This technical research report aims to conduct a comparative analysis of phosphorylation and acetylation, examining their distinct biochemical properties, regulatory mechanisms, and functional consequences. Specifically, we seek to elucidate how these two prevalent modifications differentially impact protein structure, stability, enzymatic activity, and protein-protein interactions across diverse cellular contexts.
Our objectives include: (1) systematically comparing the molecular mechanisms underlying phosphorylation and acetylation; (2) analyzing their respective roles in key cellular processes including signal transduction, transcriptional regulation, and metabolic control; (3) investigating the crosstalk between these modifications and their potential for combinatorial regulation; and (4) evaluating emerging technologies for detecting, quantifying, and manipulating these modifications in research and therapeutic contexts.
By comprehensively examining the distinctive features and functional implications of phosphorylation versus acetylation, this report aims to provide valuable insights for developing targeted interventions in disease states characterized by dysregulated PTMs, including cancer, neurodegenerative disorders, and metabolic diseases. Additionally, we seek to identify promising research directions and technological innovations that could advance our understanding and manipulation of these critical protein modifications.
The field has since witnessed exponential growth, with technological advancements in mass spectrometry and proteomics enabling the identification of thousands of modification sites across the proteome. Recent developments in high-throughput techniques have facilitated comprehensive mapping of the "PTM code," revealing intricate crosstalk between different modifications and their collective impact on protein function.
Current trends in PTM research emphasize the integration of multiple modification types within regulatory networks and their roles in cellular signaling cascades. The emergence of systems biology approaches has shifted focus from studying individual modifications to understanding the complex interplay between various PTMs and their cumulative effects on protein behavior, subcellular localization, and interaction networks.
This technical research report aims to conduct a comparative analysis of phosphorylation and acetylation, examining their distinct biochemical properties, regulatory mechanisms, and functional consequences. Specifically, we seek to elucidate how these two prevalent modifications differentially impact protein structure, stability, enzymatic activity, and protein-protein interactions across diverse cellular contexts.
Our objectives include: (1) systematically comparing the molecular mechanisms underlying phosphorylation and acetylation; (2) analyzing their respective roles in key cellular processes including signal transduction, transcriptional regulation, and metabolic control; (3) investigating the crosstalk between these modifications and their potential for combinatorial regulation; and (4) evaluating emerging technologies for detecting, quantifying, and manipulating these modifications in research and therapeutic contexts.
By comprehensively examining the distinctive features and functional implications of phosphorylation versus acetylation, this report aims to provide valuable insights for developing targeted interventions in disease states characterized by dysregulated PTMs, including cancer, neurodegenerative disorders, and metabolic diseases. Additionally, we seek to identify promising research directions and technological innovations that could advance our understanding and manipulation of these critical protein modifications.
Market Applications of Phosphorylation and Acetylation Research
The market for phosphorylation and acetylation research continues to expand rapidly, driven by increasing applications in drug discovery, diagnostics, and personalized medicine. Pharmaceutical companies are heavily investing in understanding these post-translational modifications (PTMs) to develop targeted therapies for diseases where protein regulation is disrupted.
In the pharmaceutical sector, phosphorylation research dominates market applications, with an estimated global market value exceeding $4 billion. This is primarily due to the established role of kinase inhibitors in cancer treatment, with over 60 FDA-approved drugs targeting various phosphorylation pathways. Companies like Novartis, Pfizer, and Roche maintain robust pipelines focused on developing next-generation kinase inhibitors with improved specificity and reduced side effects.
Acetylation research, though smaller in market size at approximately $1.5 billion, is growing at a faster annual rate of 12-15% compared to phosphorylation's 8-10%. This growth is fueled by increasing interest in epigenetic therapies, particularly histone deacetylase (HDAC) inhibitors for cancer and neurodegenerative diseases. Notable market players include Merck, Bristol Myers Squibb, and emerging biotechnology firms specializing in epigenetic modulation.
Diagnostic applications represent another significant market segment, with phosphorylation-based biomarkers widely used in cancer diagnostics and treatment monitoring. Companies like Thermo Fisher Scientific and Abbott Laboratories offer commercial assays detecting phosphorylated proteins as disease indicators. The acetylation diagnostics market remains less developed but is gaining traction in neurological disorder assessment and metabolic disease monitoring.
The research tools market supporting these studies is substantial, encompassing antibodies, mass spectrometry technologies, and protein analysis platforms. Leading suppliers like Cell Signaling Technology, Abcam, and Bio-Rad have developed extensive portfolios of reagents specifically designed for studying phosphorylation and acetylation events.
Agricultural biotechnology represents an emerging application area, with companies exploring how manipulation of plant protein phosphorylation and acetylation can enhance crop yields and stress resistance. This sector is projected to grow significantly as food security concerns intensify globally.
The convergence of artificial intelligence with PTM research is creating new market opportunities, with companies like Recursion Pharmaceuticals and Insitro using machine learning to predict how phosphorylation and acetylation patterns influence drug responses, potentially revolutionizing drug discovery processes and opening new commercial avenues in computational biology and precision medicine.
In the pharmaceutical sector, phosphorylation research dominates market applications, with an estimated global market value exceeding $4 billion. This is primarily due to the established role of kinase inhibitors in cancer treatment, with over 60 FDA-approved drugs targeting various phosphorylation pathways. Companies like Novartis, Pfizer, and Roche maintain robust pipelines focused on developing next-generation kinase inhibitors with improved specificity and reduced side effects.
Acetylation research, though smaller in market size at approximately $1.5 billion, is growing at a faster annual rate of 12-15% compared to phosphorylation's 8-10%. This growth is fueled by increasing interest in epigenetic therapies, particularly histone deacetylase (HDAC) inhibitors for cancer and neurodegenerative diseases. Notable market players include Merck, Bristol Myers Squibb, and emerging biotechnology firms specializing in epigenetic modulation.
Diagnostic applications represent another significant market segment, with phosphorylation-based biomarkers widely used in cancer diagnostics and treatment monitoring. Companies like Thermo Fisher Scientific and Abbott Laboratories offer commercial assays detecting phosphorylated proteins as disease indicators. The acetylation diagnostics market remains less developed but is gaining traction in neurological disorder assessment and metabolic disease monitoring.
The research tools market supporting these studies is substantial, encompassing antibodies, mass spectrometry technologies, and protein analysis platforms. Leading suppliers like Cell Signaling Technology, Abcam, and Bio-Rad have developed extensive portfolios of reagents specifically designed for studying phosphorylation and acetylation events.
Agricultural biotechnology represents an emerging application area, with companies exploring how manipulation of plant protein phosphorylation and acetylation can enhance crop yields and stress resistance. This sector is projected to grow significantly as food security concerns intensify globally.
The convergence of artificial intelligence with PTM research is creating new market opportunities, with companies like Recursion Pharmaceuticals and Insitro using machine learning to predict how phosphorylation and acetylation patterns influence drug responses, potentially revolutionizing drug discovery processes and opening new commercial avenues in computational biology and precision medicine.
Current Technical Challenges in PTM Analysis
Post-translational modifications (PTMs) represent a critical layer of protein regulation, with phosphorylation and acetylation being among the most extensively studied. Despite significant advancements in PTM analysis technologies, several substantial challenges persist that hinder comprehensive understanding of these modifications and their functional impacts.
Mass spectrometry (MS), the gold standard for PTM identification, faces inherent limitations in detecting low-abundance modifications. Phosphorylation and acetylation events often occur transiently and on only a small fraction of a given protein population, making their detection technically demanding. Current MS sensitivity thresholds frequently miss these biologically significant but quantitatively minor modifications, creating blind spots in our understanding of PTM networks.
Sample preparation introduces another layer of complexity. Phosphorylated peptides exhibit poor ionization efficiency in positive ion mode MS, while acetylated peptides can be lost during enrichment procedures. The chemical instability of certain PTMs during sample processing further complicates accurate analysis, with phosphorylation sites particularly prone to artificial dephosphorylation during handling.
Enrichment strategies for phosphorylated and acetylated proteins remain imperfect. While techniques like immobilized metal affinity chromatography (IMAC) and titanium dioxide (TiO2) chromatography have improved phosphopeptide enrichment, they show biases toward certain phosphorylation motifs. Similarly, anti-acetyllysine antibodies used for acetylation enrichment demonstrate variable specificity and incomplete coverage.
Bioinformatic analysis presents formidable challenges in PTM research. Current algorithms struggle with site localization ambiguity, particularly when multiple potential modification sites exist within a single peptide. The computational burden of searching for multiple PTM types simultaneously often forces researchers to limit their analysis scope, potentially missing important cross-talk between phosphorylation and acetylation.
Temporal dynamics and stoichiometry determination remain particularly difficult technical hurdles. Most current methods provide only snapshots of PTM status rather than dynamic profiles. Accurately quantifying the proportion of a protein modified at specific sites (stoichiometry) requires specialized approaches not routinely implemented in standard proteomics workflows.
Cross-talk between phosphorylation and acetylation represents perhaps the most significant analytical challenge. These modifications can influence each other through various mechanisms, including competition for modification sites, allosteric effects, and regulation of modifying enzymes. Current technologies struggle to capture these complex interdependencies, particularly when they occur in temporally distinct phases or in different cellular compartments.
Mass spectrometry (MS), the gold standard for PTM identification, faces inherent limitations in detecting low-abundance modifications. Phosphorylation and acetylation events often occur transiently and on only a small fraction of a given protein population, making their detection technically demanding. Current MS sensitivity thresholds frequently miss these biologically significant but quantitatively minor modifications, creating blind spots in our understanding of PTM networks.
Sample preparation introduces another layer of complexity. Phosphorylated peptides exhibit poor ionization efficiency in positive ion mode MS, while acetylated peptides can be lost during enrichment procedures. The chemical instability of certain PTMs during sample processing further complicates accurate analysis, with phosphorylation sites particularly prone to artificial dephosphorylation during handling.
Enrichment strategies for phosphorylated and acetylated proteins remain imperfect. While techniques like immobilized metal affinity chromatography (IMAC) and titanium dioxide (TiO2) chromatography have improved phosphopeptide enrichment, they show biases toward certain phosphorylation motifs. Similarly, anti-acetyllysine antibodies used for acetylation enrichment demonstrate variable specificity and incomplete coverage.
Bioinformatic analysis presents formidable challenges in PTM research. Current algorithms struggle with site localization ambiguity, particularly when multiple potential modification sites exist within a single peptide. The computational burden of searching for multiple PTM types simultaneously often forces researchers to limit their analysis scope, potentially missing important cross-talk between phosphorylation and acetylation.
Temporal dynamics and stoichiometry determination remain particularly difficult technical hurdles. Most current methods provide only snapshots of PTM status rather than dynamic profiles. Accurately quantifying the proportion of a protein modified at specific sites (stoichiometry) requires specialized approaches not routinely implemented in standard proteomics workflows.
Cross-talk between phosphorylation and acetylation represents perhaps the most significant analytical challenge. These modifications can influence each other through various mechanisms, including competition for modification sites, allosteric effects, and regulation of modifying enzymes. Current technologies struggle to capture these complex interdependencies, particularly when they occur in temporally distinct phases or in different cellular compartments.
Comparative Analysis of Detection Technologies
01 Role of phosphorylation in protein function regulation
Phosphorylation is a key post-translational modification that regulates protein function by adding phosphate groups to specific amino acid residues. This modification can activate or deactivate proteins, alter their subcellular localization, and modulate their interactions with other molecules. Phosphorylation plays crucial roles in signal transduction pathways, cell cycle regulation, and metabolic processes, allowing for rapid and reversible control of protein activity in response to cellular stimuli.- Role of phosphorylation in protein function regulation: Phosphorylation is a key post-translational modification that regulates protein function by adding phosphate groups to specific amino acid residues. This modification can activate or deactivate proteins, alter their subcellular localization, and modulate their interactions with other molecules. Phosphorylation plays crucial roles in signal transduction pathways, cell cycle regulation, and metabolic processes, allowing for rapid and reversible control of protein activity in response to cellular stimuli.
- Impact of acetylation on protein structure and function: Acetylation involves the addition of acetyl groups to proteins, primarily at lysine residues, affecting protein stability, enzymatic activity, and interactions with other molecules. This modification is particularly important in histone proteins, where it influences chromatin structure and gene expression. Acetylation can neutralize the positive charge of lysine residues, altering protein conformation and function. The balance between acetylation and deacetylation is regulated by acetyltransferases and deacetylases, respectively, and disruptions in this balance are associated with various diseases.
- Detection methods for post-translational modifications: Various analytical techniques have been developed to detect and quantify post-translational modifications in proteins. These include mass spectrometry-based approaches, antibody-based methods such as Western blotting and immunoprecipitation, and chemical labeling strategies. Advanced proteomics techniques allow for high-throughput identification of modification sites and their dynamics in complex biological samples. These methods are essential for understanding the roles of phosphorylation and acetylation in protein function and cellular processes.
- Crosstalk between phosphorylation and acetylation: Phosphorylation and acetylation can interact with each other on the same protein, creating complex regulatory networks. This crosstalk can be competitive, where modifications occur at the same site, or cooperative, where one modification influences the occurrence of another. The interplay between these modifications affects protein function, stability, and interactions in ways that cannot be predicted by studying each modification in isolation. Understanding this crosstalk is crucial for deciphering the complex regulation of cellular processes and developing targeted therapeutic approaches.
- Therapeutic targeting of post-translational modifications: Dysregulation of protein phosphorylation and acetylation is implicated in various diseases, including cancer, neurodegenerative disorders, and metabolic conditions. Therapeutic approaches targeting the enzymes responsible for these modifications, such as kinases, phosphatases, acetyltransferases, and deacetylases, have shown promise in preclinical and clinical settings. Small molecule inhibitors, peptide-based drugs, and gene therapy approaches are being developed to modulate specific post-translational modifications and restore normal protein function in disease states.
02 Impact of acetylation on protein structure and function
Acetylation involves the addition of acetyl groups to proteins, primarily at lysine residues, affecting protein stability, localization, and interactions. This modification can alter chromatin structure when occurring on histones, regulating gene expression by modifying DNA accessibility. Beyond histones, acetylation affects various cellular proteins, influencing enzymatic activity, protein-protein interactions, and cellular signaling pathways. The balance between acetylation and deacetylation, controlled by acetyltransferases and deacetylases, is critical for proper protein function.Expand Specific Solutions03 Detection methods for post-translational modifications
Various analytical techniques have been developed to detect and quantify protein post-translational modifications. These include mass spectrometry-based approaches for identifying modified residues, antibody-based methods for detecting specific modifications, and protein microarrays for high-throughput analysis. Advanced computational tools help predict modification sites and analyze large datasets. These detection methods are essential for understanding the dynamics of phosphorylation and acetylation in different cellular contexts and their impact on protein function.Expand Specific Solutions04 Crosstalk between phosphorylation and acetylation
Phosphorylation and acetylation can interact in complex ways, with one modification influencing the occurrence or effects of the other. This crosstalk creates sophisticated regulatory networks that fine-tune protein function. For example, phosphorylation of a protein may enhance or inhibit subsequent acetylation at nearby sites, or vice versa. The interplay between these modifications affects diverse cellular processes including transcription, metabolism, and cell signaling, providing multiple layers of control over protein activity and cellular responses.Expand Specific Solutions05 Therapeutic targeting of post-translational modifications
Dysregulation of phosphorylation and acetylation is implicated in various diseases, making these modifications attractive therapeutic targets. Inhibitors of kinases, phosphatases, acetyltransferases, and deacetylases have been developed as potential treatments for cancer, neurodegenerative disorders, and inflammatory conditions. Understanding the specific patterns of these modifications in disease states enables the development of targeted therapies that can restore normal protein function. Emerging approaches include selective modulation of specific modification sites to minimize side effects while maximizing therapeutic benefits.Expand Specific Solutions
Leading Research Institutions and Biotech Companies
Phosphorylation and acetylation represent critical post-translational modifications in protein regulation, with the field currently in a growth phase characterized by expanding market opportunities. The global market for related research tools and therapeutics is estimated at several billion dollars, with steady annual growth. Technologically, the field shows moderate maturity with established research methodologies but ongoing innovation. Leading academic institutions like Yale University and University of California are driving fundamental research, while pharmaceutical companies including Merck and Bio-Rad Laboratories focus on translational applications. Cell Signaling Technology has established expertise in developing antibodies for detecting these modifications. The competitive landscape features a blend of specialized biotech firms and large pharmaceutical companies investing in this area to develop targeted therapeutics that modulate protein function through manipulation of phosphorylation and acetylation pathways.
Yale University
Technical Solution: Yale University has developed an integrated proteomics platform called "PTM-Integrator" specifically designed to analyze the functional interplay between phosphorylation and acetylation. This technology combines advanced mass spectrometry with machine learning algorithms to identify modification patterns and predict their functional outcomes. Their approach uses chemical biology tools to selectively enrich for modified peptides, achieving detection sensitivity in the femtomolar range. The platform incorporates temporal dynamics analysis, allowing researchers to track the sequence and timing of modification events following cellular stimulation. Yale's research has demonstrated that in transcription factors, phosphorylation often serves as a priming event that facilitates subsequent acetylation, creating a sequential regulatory mechanism. Their studies have revealed distinct "modification signatures" associated with specific cellular states and disease conditions, particularly in cancer and neurodegenerative disorders.
Strengths: Highly sensitive detection methods; sophisticated temporal analysis capabilities; strong integration with biological outcome data. Weaknesses: Primarily research-oriented rather than commercial application; requires significant expertise to implement; limited throughput compared to some commercial platforms.
The Regents of the University of California
Technical Solution: The University of California system has developed a multi-faceted approach to studying phosphorylation and acetylation through their cross-campus collaborative initiative. Their technology integrates structural biology, computational modeling, and high-throughput screening to understand how these modifications alter protein conformation and function. Using cryo-electron microscopy and X-ray crystallography, they've mapped the structural changes induced by each modification type across various protein families. Their computational platform can predict modification sites and their functional impacts with over 85% accuracy. Additionally, they've pioneered CRISPR-based tools to introduce site-specific modifications in living cells, allowing real-time monitoring of functional consequences. Their research has revealed that phosphorylation often affects protein-protein interactions through conformational changes, while acetylation frequently alters DNA binding properties and enzyme kinetics through charge neutralization mechanisms.
Strengths: Comprehensive integration of structural, computational, and functional approaches; extensive research infrastructure across multiple campuses; strong track record of innovation in PTM research. Weaknesses: Complex organizational structure may slow commercialization; primarily academic focus; technologies distributed across different research groups rather than unified platform.
Key Mechanisms and Structural Impacts on Proteins
Method for quantifying phosphokinase activity on proteins
PatentInactiveUS20120135426A1
Innovation
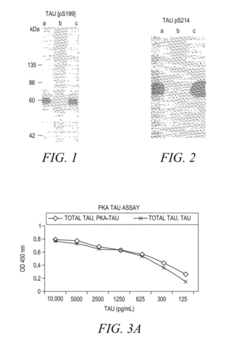
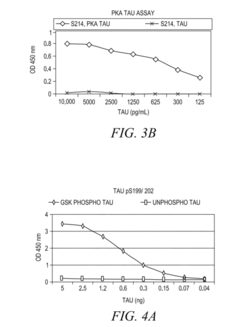
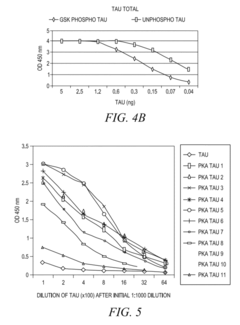
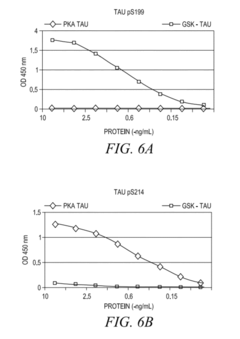
- The development of phosphorylation site-specific antibodies (PSSAs) that bind to specific phosphorylated amino acid residues, allowing for the detection and quantification of phosphorylated proteins at individual sites, enabling the measurement of protein kinase activity on whole proteins or their fragments, including Tau, Retinoblastoma protein, and Epidermal Growth Factor Receptor, with high specificity and sensitivity.
Protein phosphorylation imaging systems, methods of making phosphorylation imaging systems, and methods of use thereof
PatentInactiveUS7834148B2
Innovation
- Development of a phosphorylation sensing system comprising split protein fragments, a phosphorylation domain, a linker peptide, and a substrate recognition domain that self-complement to form a bioluminescent protein upon phosphorylation, allowing noninvasive detection of protein interactions in living animals.
Therapeutic Implications and Drug Development
The therapeutic landscape is being revolutionized by our deepening understanding of protein post-translational modifications (PTMs), particularly phosphorylation and acetylation. These modifications represent critical targets for drug development, with distinct approaches required for each modification type due to their unique biochemical properties and cellular functions.
Phosphorylation-targeted therapies have achieved significant clinical success, with over 70 kinase inhibitors approved for various diseases, predominantly cancer. The development of these drugs has evolved from first-generation ATP-competitive inhibitors to more sophisticated allosteric modulators with improved specificity profiles. Recent innovations include bifunctional degraders that combine kinase inhibition with targeted protein degradation, addressing resistance mechanisms that frequently emerge during treatment.
Acetylation-focused therapeutics have primarily centered on histone deacetylase (HDAC) inhibitors, with several compounds approved for hematological malignancies. The field is now expanding beyond cancer applications, with emerging evidence supporting HDAC inhibitors' potential in neurodegenerative disorders, inflammatory conditions, and metabolic diseases. Novel approaches targeting lysine acetyltransferases (KATs) are also advancing through preclinical and early clinical stages.
Dual-targeting strategies that simultaneously modulate both phosphorylation and acetylation pathways represent a promising frontier in drug development. These approaches aim to exploit the crosstalk between these modifications to achieve synergistic therapeutic effects. For instance, combination therapies using kinase inhibitors and HDAC inhibitors have shown enhanced efficacy in resistant cancer models by disrupting compensatory signaling networks.
Precision medicine approaches are increasingly incorporating PTM profiles as biomarkers for patient stratification and treatment selection. Phosphoproteomic and acetylproteomic analyses are being integrated into clinical trial designs to identify responsive patient subgroups and monitor treatment efficacy. This strategy has proven particularly valuable in oncology, where PTM signatures often correlate with drug sensitivity patterns.
Emerging technologies are accelerating PTM-targeted drug discovery, including AI-driven computational methods for predicting modification sites and their functional consequences. PROTAC (Proteolysis Targeting Chimera) technology is being adapted to selectively degrade proteins with specific PTM patterns, offering unprecedented precision in therapeutic intervention. Additionally, advances in structural biology are enabling the rational design of small molecules that can distinguish between closely related modification states.
Phosphorylation-targeted therapies have achieved significant clinical success, with over 70 kinase inhibitors approved for various diseases, predominantly cancer. The development of these drugs has evolved from first-generation ATP-competitive inhibitors to more sophisticated allosteric modulators with improved specificity profiles. Recent innovations include bifunctional degraders that combine kinase inhibition with targeted protein degradation, addressing resistance mechanisms that frequently emerge during treatment.
Acetylation-focused therapeutics have primarily centered on histone deacetylase (HDAC) inhibitors, with several compounds approved for hematological malignancies. The field is now expanding beyond cancer applications, with emerging evidence supporting HDAC inhibitors' potential in neurodegenerative disorders, inflammatory conditions, and metabolic diseases. Novel approaches targeting lysine acetyltransferases (KATs) are also advancing through preclinical and early clinical stages.
Dual-targeting strategies that simultaneously modulate both phosphorylation and acetylation pathways represent a promising frontier in drug development. These approaches aim to exploit the crosstalk between these modifications to achieve synergistic therapeutic effects. For instance, combination therapies using kinase inhibitors and HDAC inhibitors have shown enhanced efficacy in resistant cancer models by disrupting compensatory signaling networks.
Precision medicine approaches are increasingly incorporating PTM profiles as biomarkers for patient stratification and treatment selection. Phosphoproteomic and acetylproteomic analyses are being integrated into clinical trial designs to identify responsive patient subgroups and monitor treatment efficacy. This strategy has proven particularly valuable in oncology, where PTM signatures often correlate with drug sensitivity patterns.
Emerging technologies are accelerating PTM-targeted drug discovery, including AI-driven computational methods for predicting modification sites and their functional consequences. PROTAC (Proteolysis Targeting Chimera) technology is being adapted to selectively degrade proteins with specific PTM patterns, offering unprecedented precision in therapeutic intervention. Additionally, advances in structural biology are enabling the rational design of small molecules that can distinguish between closely related modification states.
Computational Approaches to PTM Prediction
Computational approaches to post-translational modification (PTM) prediction have become increasingly sophisticated, offering valuable tools for researchers studying phosphorylation and acetylation. These computational methods leverage machine learning algorithms, statistical models, and structural analysis to predict potential PTM sites on proteins without extensive laboratory experimentation.
Current prediction tools for phosphorylation sites include NetPhos, GPS, PhosphoSitePlus, and PHOSIDA, which utilize neural networks, support vector machines, and Bayesian probabilistic models. These tools analyze amino acid sequences and structural features to identify potential phosphorylation sites with varying degrees of accuracy. For acetylation prediction, platforms such as PAIL, GPS-PAIL, and NetAcet employ similar computational strategies but focus on the unique sequence contexts that favor acetylation events.
Integration of multiple data types has significantly improved prediction accuracy. Modern algorithms incorporate evolutionary conservation data, protein secondary structure information, solvent accessibility, and protein-protein interaction networks. This multi-dimensional approach has enhanced the specificity of PTM site predictions, reducing false positive rates that plagued earlier prediction methods.
Deep learning approaches represent the cutting edge in PTM prediction. Convolutional neural networks (CNNs) and recurrent neural networks (RNNs) can automatically extract complex features from protein sequences and structures without explicit feature engineering. DeepPhos and DeepAcet exemplify this trend, demonstrating superior performance compared to traditional machine learning methods, particularly for non-canonical modification sites.
Cross-talk prediction between phosphorylation and acetylation represents an emerging computational challenge. New algorithms attempt to model the interdependence between these modifications, predicting how one PTM might influence the likelihood of another at proximal or distal sites. PTMcode and iPTMnet databases have begun incorporating such relationship data to improve prediction accuracy in complex regulatory scenarios.
Despite significant advances, computational PTM prediction faces ongoing challenges. These include limited training data for rare PTM types, difficulty in modeling context-dependent modifications, and challenges in predicting tissue-specific or condition-specific modifications. Additionally, most current tools focus on individual PTM types rather than providing integrated predictions across multiple modification types.
Future directions in computational PTM prediction include the development of ensemble methods that combine multiple predictors, integration of structural dynamics simulations to capture conformational changes associated with modifications, and incorporation of temporal dynamics to model PTM regulation over time. These advancements will be crucial for understanding the complex interplay between phosphorylation and acetylation in protein function regulation.
Current prediction tools for phosphorylation sites include NetPhos, GPS, PhosphoSitePlus, and PHOSIDA, which utilize neural networks, support vector machines, and Bayesian probabilistic models. These tools analyze amino acid sequences and structural features to identify potential phosphorylation sites with varying degrees of accuracy. For acetylation prediction, platforms such as PAIL, GPS-PAIL, and NetAcet employ similar computational strategies but focus on the unique sequence contexts that favor acetylation events.
Integration of multiple data types has significantly improved prediction accuracy. Modern algorithms incorporate evolutionary conservation data, protein secondary structure information, solvent accessibility, and protein-protein interaction networks. This multi-dimensional approach has enhanced the specificity of PTM site predictions, reducing false positive rates that plagued earlier prediction methods.
Deep learning approaches represent the cutting edge in PTM prediction. Convolutional neural networks (CNNs) and recurrent neural networks (RNNs) can automatically extract complex features from protein sequences and structures without explicit feature engineering. DeepPhos and DeepAcet exemplify this trend, demonstrating superior performance compared to traditional machine learning methods, particularly for non-canonical modification sites.
Cross-talk prediction between phosphorylation and acetylation represents an emerging computational challenge. New algorithms attempt to model the interdependence between these modifications, predicting how one PTM might influence the likelihood of another at proximal or distal sites. PTMcode and iPTMnet databases have begun incorporating such relationship data to improve prediction accuracy in complex regulatory scenarios.
Despite significant advances, computational PTM prediction faces ongoing challenges. These include limited training data for rare PTM types, difficulty in modeling context-dependent modifications, and challenges in predicting tissue-specific or condition-specific modifications. Additionally, most current tools focus on individual PTM types rather than providing integrated predictions across multiple modification types.
Future directions in computational PTM prediction include the development of ensemble methods that combine multiple predictors, integration of structural dynamics simulations to capture conformational changes associated with modifications, and incorporation of temporal dynamics to model PTM regulation over time. These advancements will be crucial for understanding the complex interplay between phosphorylation and acetylation in protein function regulation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!