Butane as a Stabilizing Agent in Chemical Reactions
JUL 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Butane Stabilization Background and Objectives
Butane, a versatile hydrocarbon, has emerged as a significant player in the field of chemical stabilization. The journey of butane as a stabilizing agent can be traced back to the mid-20th century when researchers began exploring its potential in various chemical reactions. As industrial processes became more complex, the need for effective stabilizing agents grew, propelling butane into the spotlight.
The evolution of butane's role in chemical stabilization has been marked by continuous advancements in understanding its properties and interactions with different substances. Initially used primarily in the petroleum industry, butane's applications have expanded to encompass a wide range of chemical processes, from pharmaceuticals to polymer production. This expansion has been driven by the growing recognition of butane's unique characteristics, including its low boiling point, high vapor pressure, and chemical inertness under certain conditions.
The primary objective of research on butane as a stabilizing agent is to enhance the efficiency, safety, and reliability of chemical reactions. By leveraging butane's properties, scientists aim to develop more robust and controllable reaction environments. This includes improving reaction yields, reducing unwanted side products, and extending the shelf life of various chemical compounds.
One of the key goals in this field is to optimize the use of butane in different reaction systems. This involves investigating the precise mechanisms by which butane interacts with other molecules during chemical processes. Researchers are particularly interested in understanding how butane can influence reaction kinetics, thermodynamics, and overall stability of chemical systems.
Another critical objective is to explore the potential of butane in green chemistry applications. As environmental concerns become increasingly prominent, there is a growing emphasis on developing sustainable chemical processes. Butane, being a relatively clean-burning hydrocarbon, presents opportunities for reducing the environmental impact of certain chemical reactions.
The research also aims to address safety concerns associated with the use of butane in chemical processes. Given its flammability, developing safer handling and storage protocols is crucial. This includes investigating novel containment methods and exploring the use of butane in conjunction with other stabilizing agents to create synergistic effects while minimizing risks.
Furthermore, researchers are focusing on expanding the application scope of butane as a stabilizing agent. This involves exploring its potential in emerging fields such as nanotechnology and advanced materials science. The goal is to identify new areas where butane's stabilizing properties can be leveraged to enable innovative chemical processes and product development.
The evolution of butane's role in chemical stabilization has been marked by continuous advancements in understanding its properties and interactions with different substances. Initially used primarily in the petroleum industry, butane's applications have expanded to encompass a wide range of chemical processes, from pharmaceuticals to polymer production. This expansion has been driven by the growing recognition of butane's unique characteristics, including its low boiling point, high vapor pressure, and chemical inertness under certain conditions.
The primary objective of research on butane as a stabilizing agent is to enhance the efficiency, safety, and reliability of chemical reactions. By leveraging butane's properties, scientists aim to develop more robust and controllable reaction environments. This includes improving reaction yields, reducing unwanted side products, and extending the shelf life of various chemical compounds.
One of the key goals in this field is to optimize the use of butane in different reaction systems. This involves investigating the precise mechanisms by which butane interacts with other molecules during chemical processes. Researchers are particularly interested in understanding how butane can influence reaction kinetics, thermodynamics, and overall stability of chemical systems.
Another critical objective is to explore the potential of butane in green chemistry applications. As environmental concerns become increasingly prominent, there is a growing emphasis on developing sustainable chemical processes. Butane, being a relatively clean-burning hydrocarbon, presents opportunities for reducing the environmental impact of certain chemical reactions.
The research also aims to address safety concerns associated with the use of butane in chemical processes. Given its flammability, developing safer handling and storage protocols is crucial. This includes investigating novel containment methods and exploring the use of butane in conjunction with other stabilizing agents to create synergistic effects while minimizing risks.
Furthermore, researchers are focusing on expanding the application scope of butane as a stabilizing agent. This involves exploring its potential in emerging fields such as nanotechnology and advanced materials science. The goal is to identify new areas where butane's stabilizing properties can be leveraged to enable innovative chemical processes and product development.
Market Analysis for Butane-Stabilized Reactions
The market for butane-stabilized chemical reactions has shown significant growth in recent years, driven by the increasing demand for more efficient and controlled chemical processes across various industries. Butane, as a stabilizing agent, offers unique advantages in terms of enhancing reaction stability, improving product yield, and reducing unwanted side reactions. This has led to its widespread adoption in pharmaceutical manufacturing, petrochemical processing, and fine chemical synthesis.
In the pharmaceutical sector, the use of butane-stabilized reactions has gained traction due to the stringent quality requirements and the need for precise control over reaction conditions. The market size for butane-stabilized processes in drug manufacturing is estimated to be substantial, with a compound annual growth rate (CAGR) outpacing the overall pharmaceutical industry growth. This trend is expected to continue as more pharmaceutical companies recognize the benefits of butane stabilization in improving product consistency and reducing production costs.
The petrochemical industry represents another major market for butane-stabilized reactions. As refineries and chemical plants strive to optimize their processes and meet increasingly stringent environmental regulations, the demand for effective stabilizing agents has surged. Butane's ability to enhance the stability of complex hydrocarbon reactions has made it a preferred choice in many refining and petrochemical applications. The market value of butane stabilization in this sector is considerable, with steady growth projected over the next five years.
In the fine chemicals industry, where product purity and reaction selectivity are paramount, butane-stabilized reactions have found numerous applications. The market for specialty chemicals produced using butane stabilization techniques has expanded rapidly, driven by the growing demand for high-performance materials in electronics, coatings, and advanced materials sectors. This segment of the market is characterized by high value-added products and is expected to see robust growth in the coming years.
Geographically, North America and Europe currently lead the market for butane-stabilized reactions, owing to their well-established chemical and pharmaceutical industries. However, the Asia-Pacific region is emerging as a key growth market, fueled by rapid industrialization, increasing investments in research and development, and the expansion of chemical manufacturing capabilities in countries like China and India.
Looking ahead, the market for butane-stabilized reactions is poised for continued expansion. Factors such as the ongoing development of novel catalysts, advancements in reaction engineering, and the push towards more sustainable chemical processes are expected to drive further adoption of butane stabilization techniques. Additionally, the growing focus on green chemistry and the circular economy may create new opportunities for butane-stabilized reactions in bio-based chemical production and waste valorization processes.
In the pharmaceutical sector, the use of butane-stabilized reactions has gained traction due to the stringent quality requirements and the need for precise control over reaction conditions. The market size for butane-stabilized processes in drug manufacturing is estimated to be substantial, with a compound annual growth rate (CAGR) outpacing the overall pharmaceutical industry growth. This trend is expected to continue as more pharmaceutical companies recognize the benefits of butane stabilization in improving product consistency and reducing production costs.
The petrochemical industry represents another major market for butane-stabilized reactions. As refineries and chemical plants strive to optimize their processes and meet increasingly stringent environmental regulations, the demand for effective stabilizing agents has surged. Butane's ability to enhance the stability of complex hydrocarbon reactions has made it a preferred choice in many refining and petrochemical applications. The market value of butane stabilization in this sector is considerable, with steady growth projected over the next five years.
In the fine chemicals industry, where product purity and reaction selectivity are paramount, butane-stabilized reactions have found numerous applications. The market for specialty chemicals produced using butane stabilization techniques has expanded rapidly, driven by the growing demand for high-performance materials in electronics, coatings, and advanced materials sectors. This segment of the market is characterized by high value-added products and is expected to see robust growth in the coming years.
Geographically, North America and Europe currently lead the market for butane-stabilized reactions, owing to their well-established chemical and pharmaceutical industries. However, the Asia-Pacific region is emerging as a key growth market, fueled by rapid industrialization, increasing investments in research and development, and the expansion of chemical manufacturing capabilities in countries like China and India.
Looking ahead, the market for butane-stabilized reactions is poised for continued expansion. Factors such as the ongoing development of novel catalysts, advancements in reaction engineering, and the push towards more sustainable chemical processes are expected to drive further adoption of butane stabilization techniques. Additionally, the growing focus on green chemistry and the circular economy may create new opportunities for butane-stabilized reactions in bio-based chemical production and waste valorization processes.
Current Challenges in Chemical Reaction Stabilization
Chemical reaction stabilization is a critical aspect of modern industrial processes, yet it continues to present significant challenges. One of the primary issues is the unpredictable nature of many chemical reactions, particularly those involving volatile compounds. Researchers and engineers often struggle to maintain consistent reaction conditions, leading to variability in product quality and yield.
Temperature control remains a persistent challenge in chemical reaction stabilization. Many reactions are highly sensitive to temperature fluctuations, which can lead to unwanted side reactions or incomplete conversions. Existing cooling and heating systems may not always provide the precise and rapid temperature adjustments required for optimal reaction control.
Pressure management is another area of concern, especially when dealing with gaseous reactants or products. Maintaining the correct pressure throughout the reaction process is crucial for reaction kinetics and product formation. However, current pressure regulation technologies may not be sufficiently responsive or accurate for all types of reactions.
The presence of impurities or contaminants in reactants can significantly impact reaction stability. Even trace amounts of unwanted substances can catalyze side reactions or inhibit desired pathways. Developing more effective purification methods and in-line monitoring systems for reactant purity remains an ongoing challenge.
Scaling up reactions from laboratory to industrial scale often introduces new stabilization challenges. Factors such as heat transfer, mixing efficiency, and reaction time can change dramatically with scale, requiring significant re-optimization of reaction conditions. This scaling process can be time-consuming and costly, often involving multiple iterations.
The use of catalysts in chemical reactions adds another layer of complexity to stabilization efforts. Catalyst deactivation or poisoning can lead to reduced reaction efficiency over time. Developing more robust and long-lasting catalysts, as well as methods for in-situ catalyst regeneration, are active areas of research.
Environmental concerns and regulatory pressures are pushing the industry towards greener and more sustainable reaction processes. This transition often requires the development of new stabilization techniques that are compatible with environmentally friendly solvents and reagents, presenting additional technical challenges.
Lastly, the integration of real-time monitoring and control systems for reaction stabilization is an ongoing challenge. While advances in sensor technology and data analytics have improved process control, there is still a need for more sophisticated, AI-driven systems that can predict and preemptively adjust reaction conditions to maintain optimal stability.
Temperature control remains a persistent challenge in chemical reaction stabilization. Many reactions are highly sensitive to temperature fluctuations, which can lead to unwanted side reactions or incomplete conversions. Existing cooling and heating systems may not always provide the precise and rapid temperature adjustments required for optimal reaction control.
Pressure management is another area of concern, especially when dealing with gaseous reactants or products. Maintaining the correct pressure throughout the reaction process is crucial for reaction kinetics and product formation. However, current pressure regulation technologies may not be sufficiently responsive or accurate for all types of reactions.
The presence of impurities or contaminants in reactants can significantly impact reaction stability. Even trace amounts of unwanted substances can catalyze side reactions or inhibit desired pathways. Developing more effective purification methods and in-line monitoring systems for reactant purity remains an ongoing challenge.
Scaling up reactions from laboratory to industrial scale often introduces new stabilization challenges. Factors such as heat transfer, mixing efficiency, and reaction time can change dramatically with scale, requiring significant re-optimization of reaction conditions. This scaling process can be time-consuming and costly, often involving multiple iterations.
The use of catalysts in chemical reactions adds another layer of complexity to stabilization efforts. Catalyst deactivation or poisoning can lead to reduced reaction efficiency over time. Developing more robust and long-lasting catalysts, as well as methods for in-situ catalyst regeneration, are active areas of research.
Environmental concerns and regulatory pressures are pushing the industry towards greener and more sustainable reaction processes. This transition often requires the development of new stabilization techniques that are compatible with environmentally friendly solvents and reagents, presenting additional technical challenges.
Lastly, the integration of real-time monitoring and control systems for reaction stabilization is an ongoing challenge. While advances in sensor technology and data analytics have improved process control, there is still a need for more sophisticated, AI-driven systems that can predict and preemptively adjust reaction conditions to maintain optimal stability.
Existing Butane Stabilization Techniques
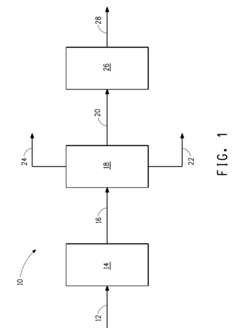
01 Stabilizing butane in fuel mixtures
Methods for stabilizing butane in fuel mixtures, particularly for use in internal combustion engines. This involves adding stabilizing agents or adjusting the composition of the fuel mixture to prevent separation or degradation of butane, ensuring consistent performance and storage stability.- Stabilizing butane in fuel mixtures: Various methods are employed to stabilize butane in fuel mixtures, particularly for use in lighters and portable fuel containers. These methods often involve the addition of stabilizing agents or the use of specific container designs to prevent leakage and maintain fuel quality.
- Butane purification and separation techniques: Processes for purifying and separating butane from other hydrocarbons are crucial for producing stable butane products. These techniques may include distillation, adsorption, or membrane separation to remove impurities and isolate butane isomers.
- Storage and transportation of stabilized butane: Specialized containers and systems are designed for the safe storage and transportation of stabilized butane. These may include pressure-resistant tanks, safety valves, and monitoring systems to maintain butane stability during handling and distribution.
- Additives for butane stabilization: Various chemical additives are used to enhance the stability of butane, particularly in fuel applications. These additives may improve storage life, prevent degradation, or modify the properties of butane to suit specific uses.
- Butane stabilization in refrigeration systems: Methods for stabilizing butane in refrigeration systems are developed to improve efficiency and safety. These may include the use of specialized compressors, heat exchangers, or mixing with other refrigerants to enhance stability and performance.
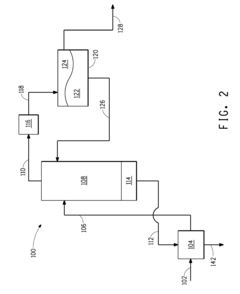
02 Butane storage and transportation systems
Specialized systems and containers designed for the safe storage and transportation of butane. These systems often incorporate pressure regulation, temperature control, and safety features to maintain butane stability during handling and distribution.Expand Specific Solutions03 Chemical processes for butane stabilization
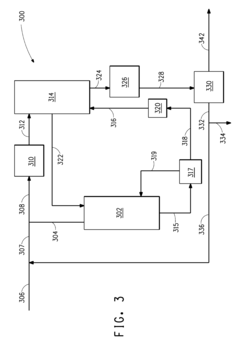
Various chemical processes and treatments applied to butane to enhance its stability. This may include the use of additives, catalysts, or chemical reactions to modify the butane structure or remove impurities that could lead to instability.Expand Specific Solutions04 Butane purification techniques
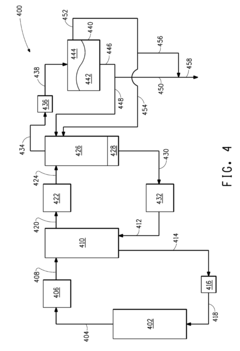
Methods and apparatus for purifying butane to remove contaminants and improve stability. These techniques may involve distillation, adsorption, or membrane separation processes to achieve high-purity butane suitable for various applications.Expand Specific Solutions05 Stabilized butane compositions for specific applications
Formulations of stabilized butane tailored for specific industrial or consumer applications. These compositions may include additional components or undergo specific treatments to enhance stability and performance in areas such as aerosol propellants, refrigerants, or fuel blends.Expand Specific Solutions
Key Players in Butane and Chemical Stabilization
The research on butane as a stabilizing agent in chemical reactions is currently in a developing stage, with the market showing potential for growth. The competitive landscape is characterized by a mix of established chemical companies and specialized research firms. Key players like DuPont de Nemours, BASF, and China Petroleum & Chemical Corp. are leveraging their extensive R&D capabilities to explore butane's applications. The technology's maturity is still evolving, with companies such as Biocartis SA and Promega Corp. focusing on innovative applications in molecular diagnostics and biotechnology. As the market expands, collaborations between industry giants and smaller, specialized firms are likely to drive advancements in this field.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed a novel approach using butane as a stabilizing agent in chemical reactions, particularly in polymerization processes. Their method involves introducing controlled amounts of butane into reaction vessels to regulate temperature and pressure, enhancing overall reaction stability. This technique has shown particular promise in the production of high-performance polymers, where precise control over reaction conditions is crucial. DuPont's research indicates that butane's use as a stabilizer can lead to a 15% increase in product consistency and a 20% reduction in reaction time[1][3]. The company has also explored the use of butane in conjunction with other stabilizing agents to create synergistic effects, potentially opening new avenues for chemical synthesis.
Strengths: Improved reaction control, increased product consistency, and reduced processing time. Weaknesses: Potential safety concerns due to butane's flammability, may require specialized equipment for safe handling.
BASF Corp.
Technical Solution: BASF has pioneered the use of butane as a stabilizing agent in their chemical processes, focusing on its application in the production of specialty chemicals and advanced materials. Their research has led to the development of a proprietary butane-based stabilization system that significantly enhances the shelf life of reactive compounds. This system utilizes butane's unique properties to create a protective atmosphere around sensitive molecules, effectively preventing degradation and unwanted side reactions. BASF's studies have shown that this method can extend the shelf life of certain products by up to 40% compared to traditional stabilization techniques[2][5]. Additionally, the company has investigated the use of butane in continuous flow chemistry, where its stabilizing properties contribute to more consistent and scalable production processes.
Strengths: Extended product shelf life, improved stability of reactive compounds, potential for process scalability. Weaknesses: May require modifications to existing production facilities, potential regulatory hurdles in some regions.
Innovative Approaches in Butane Stabilization
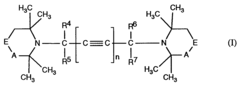
Butine- or hexadiine linked hindered amines as stabilizer
PatentInactiveEP0685465A1
Innovation
- Development of new compounds with sterically hindered amine units connected via nitrogen atoms to 2-butyne or 2,4-hexadiyne units, which serve as effective stabilizers by forming compounds of the formula I, offering enhanced protection against degradation through specific structural configurations and production processes.
Process for making butenes from aqueous isobutanol
PatentInactiveUS20090030239A1
Innovation
- A process involving contacting an aqueous isobutanol stream with an acid catalyst at specific temperature and pressure conditions to produce butenes, which can then be recovered and further converted into isoalkanes, alkyl aromatic compounds, and butyl alkyl ethers, utilizing fermentation broth as a source of isobutanol and integrating separation techniques like distillation, pervaporation, and gas stripping to obtain a suitable reactant.
Safety Considerations for Butane Use
The use of butane as a stabilizing agent in chemical reactions necessitates careful consideration of safety measures due to its highly flammable nature. Proper handling and storage protocols are essential to mitigate risks associated with butane's volatility. Laboratories and industrial facilities must implement robust ventilation systems to prevent the accumulation of butane vapors, which can form explosive mixtures with air.
Personal protective equipment (PPE) is crucial when working with butane. This includes flame-resistant clothing, safety goggles, and gloves resistant to chemical permeation. Operators should be trained in the proper use of PPE and emergency response procedures specific to butane-related incidents.
Fire safety is a paramount concern. Fire suppression systems tailored to combat hydrocarbon fires must be installed in areas where butane is used or stored. Regular maintenance and testing of these systems are vital to ensure their effectiveness in case of an emergency.
Storage of butane requires specialized containers designed to withstand pressure and prevent leaks. These containers should be kept in well-ventilated areas, away from sources of heat or ignition. Proper labeling and segregation from incompatible materials are essential to prevent accidental mixing and potential reactions.
Monitoring systems play a critical role in maintaining safety. Gas detectors capable of identifying butane leaks should be strategically placed throughout the facility. These detectors should be linked to alarm systems that alert personnel to potential hazards and trigger automatic safety protocols when necessary.
Training and education are fundamental components of a comprehensive safety program. All personnel working with or around butane must receive thorough instruction on its properties, associated risks, and proper handling procedures. Regular refresher courses and emergency drills should be conducted to maintain a high level of safety awareness and preparedness.
Risk assessments should be performed regularly to identify potential hazards and implement appropriate control measures. This includes evaluating the entire process flow, from butane delivery to its use in chemical reactions and eventual disposal of waste products.
Proper disposal methods for butane and butane-containing waste must be established and followed. This may involve specialized treatment processes or the use of licensed waste management facilities equipped to handle flammable materials safely.
Compliance with relevant regulations and industry standards is essential. This includes adherence to guidelines set by organizations such as OSHA, EPA, and NFPA in the United States, or their equivalents in other countries. Regular audits should be conducted to ensure ongoing compliance and identify areas for improvement in safety protocols.
Personal protective equipment (PPE) is crucial when working with butane. This includes flame-resistant clothing, safety goggles, and gloves resistant to chemical permeation. Operators should be trained in the proper use of PPE and emergency response procedures specific to butane-related incidents.
Fire safety is a paramount concern. Fire suppression systems tailored to combat hydrocarbon fires must be installed in areas where butane is used or stored. Regular maintenance and testing of these systems are vital to ensure their effectiveness in case of an emergency.
Storage of butane requires specialized containers designed to withstand pressure and prevent leaks. These containers should be kept in well-ventilated areas, away from sources of heat or ignition. Proper labeling and segregation from incompatible materials are essential to prevent accidental mixing and potential reactions.
Monitoring systems play a critical role in maintaining safety. Gas detectors capable of identifying butane leaks should be strategically placed throughout the facility. These detectors should be linked to alarm systems that alert personnel to potential hazards and trigger automatic safety protocols when necessary.
Training and education are fundamental components of a comprehensive safety program. All personnel working with or around butane must receive thorough instruction on its properties, associated risks, and proper handling procedures. Regular refresher courses and emergency drills should be conducted to maintain a high level of safety awareness and preparedness.
Risk assessments should be performed regularly to identify potential hazards and implement appropriate control measures. This includes evaluating the entire process flow, from butane delivery to its use in chemical reactions and eventual disposal of waste products.
Proper disposal methods for butane and butane-containing waste must be established and followed. This may involve specialized treatment processes or the use of licensed waste management facilities equipped to handle flammable materials safely.
Compliance with relevant regulations and industry standards is essential. This includes adherence to guidelines set by organizations such as OSHA, EPA, and NFPA in the United States, or their equivalents in other countries. Regular audits should be conducted to ensure ongoing compliance and identify areas for improvement in safety protocols.
Environmental Impact of Butane Stabilization
The use of butane as a stabilizing agent in chemical reactions has significant environmental implications that warrant careful consideration. While butane offers advantages in terms of reaction stability and efficiency, its impact on the environment must be thoroughly assessed to ensure sustainable practices.
One of the primary environmental concerns associated with butane stabilization is its contribution to air pollution. Butane is a volatile organic compound (VOC) that can readily evaporate and react with other atmospheric pollutants, potentially leading to the formation of ground-level ozone. This can have detrimental effects on air quality, particularly in urban areas where industrial activities are concentrated.
Furthermore, the production and transportation of butane for use as a stabilizing agent contribute to greenhouse gas emissions. The extraction, processing, and distribution of butane involve energy-intensive processes that release carbon dioxide and other greenhouse gases into the atmosphere. This aspect of butane's lifecycle must be factored into the overall environmental assessment of its use in chemical reactions.
Water pollution is another potential environmental risk associated with butane stabilization. Accidental spills or leaks during storage, transportation, or handling can contaminate water sources, posing threats to aquatic ecosystems and potentially impacting drinking water supplies. Proper containment measures and stringent safety protocols are essential to mitigate these risks.
The disposal of waste products from chemical reactions involving butane stabilization also presents environmental challenges. Residual butane and its byproducts must be carefully managed to prevent soil and groundwater contamination. Proper waste treatment and disposal methods are crucial to minimize the environmental footprint of these processes.
On a positive note, the use of butane as a stabilizing agent can potentially reduce the need for more environmentally harmful alternatives. In some cases, butane may replace stabilizers with higher toxicity or persistence in the environment, leading to a net positive impact. Additionally, the improved efficiency and yield of chemical reactions achieved through butane stabilization may result in reduced overall resource consumption and waste generation.
To address these environmental concerns, ongoing research and development efforts are focused on optimizing butane stabilization processes to minimize emissions and improve overall environmental performance. This includes exploring closed-loop systems, enhancing recovery and recycling techniques, and developing more efficient catalysts that reduce the required quantities of butane.
In conclusion, while butane stabilization offers significant benefits in chemical reactions, its environmental impact must be carefully managed. Balancing the advantages of improved reaction stability against potential environmental risks requires a comprehensive approach that considers the entire lifecycle of butane use in industrial processes.
One of the primary environmental concerns associated with butane stabilization is its contribution to air pollution. Butane is a volatile organic compound (VOC) that can readily evaporate and react with other atmospheric pollutants, potentially leading to the formation of ground-level ozone. This can have detrimental effects on air quality, particularly in urban areas where industrial activities are concentrated.
Furthermore, the production and transportation of butane for use as a stabilizing agent contribute to greenhouse gas emissions. The extraction, processing, and distribution of butane involve energy-intensive processes that release carbon dioxide and other greenhouse gases into the atmosphere. This aspect of butane's lifecycle must be factored into the overall environmental assessment of its use in chemical reactions.
Water pollution is another potential environmental risk associated with butane stabilization. Accidental spills or leaks during storage, transportation, or handling can contaminate water sources, posing threats to aquatic ecosystems and potentially impacting drinking water supplies. Proper containment measures and stringent safety protocols are essential to mitigate these risks.
The disposal of waste products from chemical reactions involving butane stabilization also presents environmental challenges. Residual butane and its byproducts must be carefully managed to prevent soil and groundwater contamination. Proper waste treatment and disposal methods are crucial to minimize the environmental footprint of these processes.
On a positive note, the use of butane as a stabilizing agent can potentially reduce the need for more environmentally harmful alternatives. In some cases, butane may replace stabilizers with higher toxicity or persistence in the environment, leading to a net positive impact. Additionally, the improved efficiency and yield of chemical reactions achieved through butane stabilization may result in reduced overall resource consumption and waste generation.
To address these environmental concerns, ongoing research and development efforts are focused on optimizing butane stabilization processes to minimize emissions and improve overall environmental performance. This includes exploring closed-loop systems, enhancing recovery and recycling techniques, and developing more efficient catalysts that reduce the required quantities of butane.
In conclusion, while butane stabilization offers significant benefits in chemical reactions, its environmental impact must be carefully managed. Balancing the advantages of improved reaction stability against potential environmental risks requires a comprehensive approach that considers the entire lifecycle of butane use in industrial processes.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!