Surface Chemistry Control In Spatial Atomic Layer Deposition
AUG 28, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
SALD Technology Background and Objectives
Spatial Atomic Layer Deposition (SALD) represents a significant evolution in thin film deposition technology, emerging from conventional Atomic Layer Deposition (ALD) techniques that have been fundamental to semiconductor manufacturing since the 1970s. While traditional ALD offers exceptional film quality and conformality, its inherently slow deposition rates have limited broader industrial adoption. SALD addresses this critical limitation by spatially separating the precursor gases rather than temporally separating them, enabling continuous deposition processes with substantially higher throughput.
The development of SALD technology began in the early 2000s, with pioneering work by researchers seeking to overcome ALD's speed constraints while maintaining its precision advantages. The technology has since evolved through several generations, each improving gas delivery systems, substrate handling mechanisms, and precursor chemistry control. This evolution has transformed SALD from a laboratory concept to an industrially viable process for high-volume manufacturing applications.
Surface chemistry control represents the cornerstone of SALD technology advancement. Unlike conventional ALD where surface reactions occur sequentially across the entire substrate, SALD requires precise management of reaction zones with minimal cross-contamination between adjacent precursor regions. This demands sophisticated understanding of gas flow dynamics, surface adsorption kinetics, and reaction mechanisms at the molecular level.
The primary technical objective in SALD development is achieving optimal balance between deposition rate and film quality through enhanced surface chemistry control. This involves developing precursor chemistries with rapid saturation characteristics, designing gas distribution systems that maintain sharp boundaries between reaction zones, and implementing real-time monitoring systems to ensure reaction completeness within the brief exposure times inherent to SALD processes.
Current research aims to expand SALD capabilities beyond traditional metal oxides to include metals, nitrides, sulfides, and complex multi-component materials. This expansion requires fundamental understanding of surface reaction mechanisms for diverse precursor combinations and substrate materials. Additionally, researchers are exploring atmospheric pressure SALD processes to further simplify equipment requirements and reduce manufacturing costs.
The long-term technological trajectory for SALD focuses on enabling roll-to-roll processing for flexible electronics, integrating with additive manufacturing techniques for 3D-printed electronics, and developing selective area deposition capabilities for direct patterning without lithography steps. These advancements would position SALD as a transformative technology for next-generation electronics manufacturing, energy storage devices, and advanced optical coatings.
Achieving these ambitious objectives requires interdisciplinary collaboration between surface chemists, fluid dynamics engineers, materials scientists, and process control specialists to overcome the complex challenges inherent in controlling surface chemistry at the molecular level while maintaining high-throughput continuous processing.
The development of SALD technology began in the early 2000s, with pioneering work by researchers seeking to overcome ALD's speed constraints while maintaining its precision advantages. The technology has since evolved through several generations, each improving gas delivery systems, substrate handling mechanisms, and precursor chemistry control. This evolution has transformed SALD from a laboratory concept to an industrially viable process for high-volume manufacturing applications.
Surface chemistry control represents the cornerstone of SALD technology advancement. Unlike conventional ALD where surface reactions occur sequentially across the entire substrate, SALD requires precise management of reaction zones with minimal cross-contamination between adjacent precursor regions. This demands sophisticated understanding of gas flow dynamics, surface adsorption kinetics, and reaction mechanisms at the molecular level.
The primary technical objective in SALD development is achieving optimal balance between deposition rate and film quality through enhanced surface chemistry control. This involves developing precursor chemistries with rapid saturation characteristics, designing gas distribution systems that maintain sharp boundaries between reaction zones, and implementing real-time monitoring systems to ensure reaction completeness within the brief exposure times inherent to SALD processes.
Current research aims to expand SALD capabilities beyond traditional metal oxides to include metals, nitrides, sulfides, and complex multi-component materials. This expansion requires fundamental understanding of surface reaction mechanisms for diverse precursor combinations and substrate materials. Additionally, researchers are exploring atmospheric pressure SALD processes to further simplify equipment requirements and reduce manufacturing costs.
The long-term technological trajectory for SALD focuses on enabling roll-to-roll processing for flexible electronics, integrating with additive manufacturing techniques for 3D-printed electronics, and developing selective area deposition capabilities for direct patterning without lithography steps. These advancements would position SALD as a transformative technology for next-generation electronics manufacturing, energy storage devices, and advanced optical coatings.
Achieving these ambitious objectives requires interdisciplinary collaboration between surface chemists, fluid dynamics engineers, materials scientists, and process control specialists to overcome the complex challenges inherent in controlling surface chemistry at the molecular level while maintaining high-throughput continuous processing.
Market Applications and Demand Analysis
Spatial Atomic Layer Deposition (SALD) technology has witnessed significant market growth due to its ability to enable high-throughput, atmospheric pressure deposition processes. The global market for ALD equipment, including spatial ALD variants, is projected to reach $3.7 billion by 2026, growing at a CAGR of 18.5% from 2021. Surface chemistry control in SALD represents a critical aspect driving this market expansion, as it directly impacts product quality, manufacturing efficiency, and application versatility.
The semiconductor industry remains the primary demand driver, accounting for approximately 65% of the total SALD market. With the continuous miniaturization of electronic components and the transition to 3D architectures, precise surface chemistry control has become essential for manufacturing high-performance integrated circuits. Major semiconductor manufacturers are increasingly adopting SALD for high-k dielectrics, barrier layers, and gate oxides, where atomic-level precision is paramount.
Renewable energy applications represent the fastest-growing segment for SALD technology, with an estimated annual growth rate of 25%. Solar cell manufacturers are particularly interested in surface chemistry-controlled SALD for depositing anti-reflection coatings, passivation layers, and transparent conductive oxides. The ability to process large-area substrates at atmospheric pressure makes SALD especially attractive for photovoltaic production, potentially reducing manufacturing costs by up to 40% compared to conventional vacuum-based ALD.
The display industry has emerged as another significant market for SALD, particularly for manufacturing OLED and flexible displays. The demand for moisture barriers with extremely low water vapor transmission rates has driven adoption of SALD processes with enhanced surface chemistry control. Market analysis indicates that approximately 30% of new display manufacturing lines are incorporating SALD technology for critical barrier layers.
Medical and biomedical applications represent an emerging market segment, with growing interest in SALD for coating implantable devices, biosensors, and drug delivery systems. The biocompatibility of surfaces produced through controlled SALD chemistry has opened new opportunities in this sector, with an estimated market value of $450 million by 2025.
Geographically, Asia-Pacific dominates the SALD market with approximately 55% share, followed by North America (25%) and Europe (15%). This distribution closely follows semiconductor and electronics manufacturing hubs. The demand for advanced surface chemistry control in SALD is particularly strong in regions with established high-tech manufacturing ecosystems, where the technology's benefits in terms of throughput, quality, and cost reduction are most valued.
The semiconductor industry remains the primary demand driver, accounting for approximately 65% of the total SALD market. With the continuous miniaturization of electronic components and the transition to 3D architectures, precise surface chemistry control has become essential for manufacturing high-performance integrated circuits. Major semiconductor manufacturers are increasingly adopting SALD for high-k dielectrics, barrier layers, and gate oxides, where atomic-level precision is paramount.
Renewable energy applications represent the fastest-growing segment for SALD technology, with an estimated annual growth rate of 25%. Solar cell manufacturers are particularly interested in surface chemistry-controlled SALD for depositing anti-reflection coatings, passivation layers, and transparent conductive oxides. The ability to process large-area substrates at atmospheric pressure makes SALD especially attractive for photovoltaic production, potentially reducing manufacturing costs by up to 40% compared to conventional vacuum-based ALD.
The display industry has emerged as another significant market for SALD, particularly for manufacturing OLED and flexible displays. The demand for moisture barriers with extremely low water vapor transmission rates has driven adoption of SALD processes with enhanced surface chemistry control. Market analysis indicates that approximately 30% of new display manufacturing lines are incorporating SALD technology for critical barrier layers.
Medical and biomedical applications represent an emerging market segment, with growing interest in SALD for coating implantable devices, biosensors, and drug delivery systems. The biocompatibility of surfaces produced through controlled SALD chemistry has opened new opportunities in this sector, with an estimated market value of $450 million by 2025.
Geographically, Asia-Pacific dominates the SALD market with approximately 55% share, followed by North America (25%) and Europe (15%). This distribution closely follows semiconductor and electronics manufacturing hubs. The demand for advanced surface chemistry control in SALD is particularly strong in regions with established high-tech manufacturing ecosystems, where the technology's benefits in terms of throughput, quality, and cost reduction are most valued.
Surface Chemistry Control Challenges
Surface chemistry control represents one of the most critical challenges in Spatial Atomic Layer Deposition (SALD) technology. Unlike conventional ALD which relies on temporal separation of precursors, SALD employs spatial separation, creating unique surface reaction dynamics that demand precise chemical management. The fundamental challenge lies in maintaining self-limiting reactions while operating at atmospheric pressure and higher temperatures, conditions that significantly alter reaction kinetics compared to vacuum-based ALD.
Precursor cross-talk presents a persistent obstacle in SALD systems. Despite physical separation mechanisms such as inert gas barriers, precursor molecules can diffuse across designated zones, leading to CVD-like growth rather than the desired self-limiting ALD reactions. This challenge intensifies with increasing deposition temperatures and when using highly reactive or volatile precursors, requiring sophisticated gas flow engineering solutions.
Surface saturation dynamics differ substantially in SALD compared to conventional ALD. The limited residence time of substrates in each precursor zone necessitates rapid and complete surface reactions. Achieving full surface coverage becomes particularly challenging when working with complex precursors or when targeting high growth rates. Researchers must carefully balance precursor concentration, substrate translation speed, and zone dimensions to ensure complete surface reactions.
Byproduct management presents another significant hurdle. In conventional ALD, purge steps effectively remove reaction byproducts, but SALD's continuous process offers limited time for byproduct evacuation. These residual byproducts can interfere with subsequent surface reactions, potentially causing film contamination, non-uniformity, or defect formation. This challenge becomes more pronounced when depositing materials that generate complex or sticky byproducts.
Temperature gradients across the deposition zones further complicate surface chemistry control. These gradients can cause inconsistent reaction rates and incomplete surface coverage, particularly at zone boundaries. The thermal management challenge is exacerbated when working with temperature-sensitive substrates or when attempting to integrate SALD into roll-to-roll manufacturing processes.
Substrate surface properties introduce additional variables affecting reaction efficiency. Surface hydroxyl density, roughness, and chemical functionality can vary significantly across different substrate materials, requiring tailored precursor chemistries and process parameters. This becomes particularly challenging when developing universal SALD processes intended to work across multiple substrate types.
Recent research has focused on developing specialized precursors optimized for SALD conditions, employing computational chemistry to predict reaction pathways, and implementing real-time monitoring systems to provide feedback on surface reactions. Despite these advances, surface chemistry control remains a fundamental challenge that requires interdisciplinary approaches combining chemical engineering, fluid dynamics, and materials science to fully address.
Precursor cross-talk presents a persistent obstacle in SALD systems. Despite physical separation mechanisms such as inert gas barriers, precursor molecules can diffuse across designated zones, leading to CVD-like growth rather than the desired self-limiting ALD reactions. This challenge intensifies with increasing deposition temperatures and when using highly reactive or volatile precursors, requiring sophisticated gas flow engineering solutions.
Surface saturation dynamics differ substantially in SALD compared to conventional ALD. The limited residence time of substrates in each precursor zone necessitates rapid and complete surface reactions. Achieving full surface coverage becomes particularly challenging when working with complex precursors or when targeting high growth rates. Researchers must carefully balance precursor concentration, substrate translation speed, and zone dimensions to ensure complete surface reactions.
Byproduct management presents another significant hurdle. In conventional ALD, purge steps effectively remove reaction byproducts, but SALD's continuous process offers limited time for byproduct evacuation. These residual byproducts can interfere with subsequent surface reactions, potentially causing film contamination, non-uniformity, or defect formation. This challenge becomes more pronounced when depositing materials that generate complex or sticky byproducts.
Temperature gradients across the deposition zones further complicate surface chemistry control. These gradients can cause inconsistent reaction rates and incomplete surface coverage, particularly at zone boundaries. The thermal management challenge is exacerbated when working with temperature-sensitive substrates or when attempting to integrate SALD into roll-to-roll manufacturing processes.
Substrate surface properties introduce additional variables affecting reaction efficiency. Surface hydroxyl density, roughness, and chemical functionality can vary significantly across different substrate materials, requiring tailored precursor chemistries and process parameters. This becomes particularly challenging when developing universal SALD processes intended to work across multiple substrate types.
Recent research has focused on developing specialized precursors optimized for SALD conditions, employing computational chemistry to predict reaction pathways, and implementing real-time monitoring systems to provide feedback on surface reactions. Despite these advances, surface chemistry control remains a fundamental challenge that requires interdisciplinary approaches combining chemical engineering, fluid dynamics, and materials science to fully address.
Current Surface Chemistry Control Methods
01 Precursor chemistry in spatial ALD
Spatial atomic layer deposition (ALD) relies on specific precursor chemistries to achieve controlled surface reactions. The selection of precursors significantly impacts film quality, growth rate, and uniformity. Various metal-organic compounds, halides, and other reactive species are used as precursors in spatial ALD processes. The surface chemistry between these precursors and substrates determines the deposition characteristics and final film properties.- Precursor chemistry in spatial ALD: Spatial atomic layer deposition (ALD) relies on specific precursor chemistries to achieve controlled surface reactions. The selection of precursors significantly impacts film quality, growth rate, and uniformity. Various metal-organic compounds, halides, and other reactive species are used as precursors in spatial ALD processes. The surface chemistry between these precursors and substrates determines the atomic-level control of film deposition, with reaction mechanisms involving chemisorption, ligand exchange, and surface saturation.
- Reaction zone separation techniques: Spatial ALD differs from conventional ALD by physically separating reaction zones rather than temporally separating them. This separation is achieved through specialized reactor designs that maintain distinct precursor regions while moving the substrate between them. Various techniques are employed to ensure effective separation, including gas curtains, physical barriers, and precise gas flow management. These separation methods prevent precursor mixing and cross-contamination, which is essential for maintaining self-limiting surface reactions and achieving high-quality thin films.
- Surface functionalization and modification: Surface functionalization plays a crucial role in spatial ALD processes by preparing substrates for optimal precursor reactions. Various pre-treatment methods are used to modify surface properties, including plasma treatments, chemical functionalization, and thermal conditioning. These modifications create specific binding sites for precursors, enhance nucleation, and improve film adhesion. The controlled introduction of functional groups on substrate surfaces enables precise engineering of interfacial chemistry, which is essential for achieving desired film properties and growth characteristics.
- High-throughput deposition processes: Spatial ALD enables high-throughput deposition processes compared to conventional ALD methods. By eliminating the need for time-consuming purge steps between precursor exposures, spatial ALD significantly increases deposition rates while maintaining atomic-level precision. Various approaches to enhance throughput include multi-substrate processing, continuous substrate movement systems, and optimized precursor delivery mechanisms. These high-throughput capabilities make spatial ALD particularly suitable for industrial applications requiring large-area thin film deposition with precise thickness control.
- Novel materials and applications: Spatial ALD surface chemistry enables the deposition of various novel materials for advanced applications. These include high-k dielectrics, barrier layers, transparent conductive oxides, and semiconductor materials. The precise control over surface reactions allows for the creation of complex multilayer structures, doped films, and nanocomposites with tailored properties. Applications span from microelectronics and photovoltaics to flexible electronics, optical coatings, and energy storage devices. The ability to deposit conformal films on complex geometries and temperature-sensitive substrates expands the application range of spatial ALD technology.
02 Reaction mechanisms in spatial ALD surface chemistry
The surface chemistry in spatial ALD involves complex reaction mechanisms including chemisorption, ligand exchange, and surface saturation. These mechanisms control how precursor molecules interact with substrate surfaces and with each other in sequential deposition steps. Understanding these reaction pathways is crucial for optimizing deposition parameters and achieving desired film characteristics. The spatial separation of precursors in this technique allows for unique reaction control compared to conventional ALD.Expand Specific Solutions03 Surface modification techniques for enhanced spatial ALD
Various surface modification techniques are employed to enhance spatial ALD processes. These include plasma treatments, functionalization with specific chemical groups, and surface activation methods. Such modifications can improve precursor adsorption, reaction efficiency, and film nucleation. By controlling the surface chemistry through these modifications, it becomes possible to tailor the growth behavior and properties of deposited films for specific applications.Expand Specific Solutions04 Temperature effects on spatial ALD surface reactions
Temperature plays a critical role in spatial ALD surface chemistry by influencing reaction kinetics, precursor adsorption, and desorption processes. The temperature window for effective spatial ALD must be carefully controlled to ensure complete surface reactions while preventing precursor decomposition or condensation. Different temperature regimes can lead to varying growth modes, film morphologies, and material properties, making temperature management a key parameter in spatial ALD process optimization.Expand Specific Solutions05 Novel materials and applications enabled by spatial ALD surface chemistry
Spatial ALD surface chemistry enables the deposition of novel materials and thin films for advanced applications. These include high-k dielectrics, barrier layers, transparent conductive oxides, and functional coatings. The precise control over surface reactions in spatial ALD allows for the creation of complex multilayer structures, doped materials, and nanocomposites with tailored properties. These capabilities have expanded the application scope of ALD to areas such as flexible electronics, energy storage devices, and optical coatings.Expand Specific Solutions
Leading Companies and Research Institutions
Surface Chemistry Control in Spatial Atomic Layer Deposition (SALD) is currently in a growth phase, with the market expanding due to increasing demand for high-precision thin film deposition in semiconductor and electronics industries. The global SALD technology market is experiencing significant growth, driven by applications in flexible electronics, solar cells, and advanced packaging. Leading companies like Applied Materials, Lam Research, and SPTS Technologies have established strong positions through advanced equipment development, while academic institutions such as Stanford University, California Institute of Technology, and Huazhong University of Science & Technology contribute significant research innovations. The technology is approaching maturity in certain applications but continues to evolve with new materials and processes being developed by companies like NAURA Microelectronics and Piotech, creating a competitive landscape balanced between established equipment manufacturers and emerging specialized solution providers.
SPTS Technologies Ltd.
Technical Solution: SPTS Technologies has developed a spatial ALD platform called Delta that specifically addresses surface chemistry control challenges. Their approach utilizes a unique oscillating substrate mechanism that moves wafers between separated precursor zones while maintaining precise gas boundaries through carefully engineered gas curtains. The system incorporates multiple temperature control zones to optimize surface reactions for different precursors, allowing tailored activation energies for each deposition step. SPTS has implemented advanced precursor delivery systems with ultra-fast switching valves (response times <50ms) that enable precise timing of surface reactions. Their technology includes specialized surface preparation modules that can modify substrate surface properties through plasma treatments immediately before deposition. The Delta platform achieves deposition rates of approximately 2-10 nm/min while maintaining excellent thickness uniformity (<±1.5%) across 200mm substrates.[4][7]
Strengths: Excellent process control for complex multi-layer structures; highly efficient precursor utilization; flexible platform design that accommodates various substrate sizes. Weaknesses: Somewhat lower throughput compared to some competing spatial ALD technologies; more complex system integration requirements; higher initial process development costs.
Applied Materials, Inc.
Technical Solution: Applied Materials has developed advanced spatial ALD systems that utilize precise gas flow control mechanisms to achieve optimal surface chemistry control. Their technology employs separated reaction zones with carefully designed gas injection and exhaust systems that maintain distinct precursor regions while allowing substrate movement between zones. The company's approach incorporates real-time monitoring systems that analyze surface reactions during deposition, enabling dynamic adjustment of process parameters to maintain optimal growth conditions. Applied Materials has also pioneered multi-element doping techniques within spatial ALD processes, allowing precise control of material composition through carefully timed precursor exposures and surface activation steps. Their systems can achieve deposition rates up to 10 times faster than conventional ALD while maintaining excellent film uniformity (±2%) across 300mm wafers.[1][3]
Strengths: Industry-leading throughput capabilities while maintaining high-quality film properties; sophisticated in-situ monitoring systems for real-time process control; extensive experience in scaling technologies to production environments. Weaknesses: Higher capital equipment costs compared to conventional ALD systems; requires more complex facility infrastructure; process optimization can be challenging for new material systems.
Key Patents and Innovations in SALD
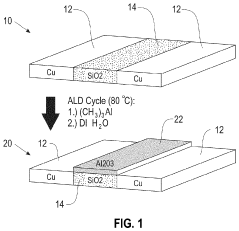
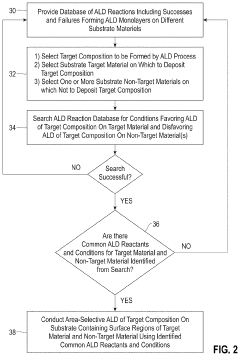
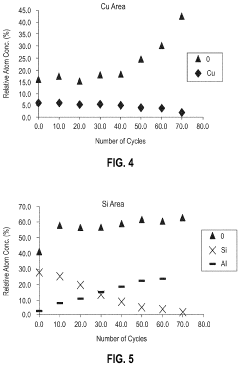
Methods of area-selective atomic layer deposition
PatentInactiveUS20200354834A1
Innovation
- The method involves contacting a substrate with a silicon dioxide surface and a zero-valent metal-containing surface with an organoaluminum compound at low temperatures, followed by exposure to water, to selectively form alumina on silicon dioxide surfaces without depositing on copper surfaces, using a database of ALD reactions to determine optimal conditions for selective deposition.
Surface diffusion-induced atomic layer deposition
PatentActiveKR1020120058723A
Innovation
- A surface diffusion-induced atomic layer deposition method that involves supplying a precursor, inducing surface diffusion with an inert gas, purging unadsorbed precursors and reaction by-products, and reacting the adsorbed precursor with a reactive gas to form an atomic layer.
Environmental Impact and Sustainability
Spatial Atomic Layer Deposition (SALD) processes, while offering significant advantages in manufacturing efficiency, present important environmental considerations that must be addressed for sustainable implementation. The environmental footprint of SALD is notably lower than conventional ALD and other deposition techniques due to its atmospheric pressure operation, which eliminates the need for energy-intensive vacuum systems. This reduction in energy consumption directly translates to decreased carbon emissions associated with the manufacturing process.
The precursor utilization efficiency in SALD systems represents a critical environmental advantage. Through optimized surface chemistry control, SALD can achieve precursor utilization rates of up to 90%, significantly higher than conventional ALD methods. This efficiency reduces chemical waste and minimizes the environmental impact of precursor production and disposal, particularly important when considering the toxic and reactive nature of many ALD precursors.
Water consumption presents another sustainability consideration for SALD processes. While traditional semiconductor manufacturing can consume thousands of liters of ultra-pure water, SALD's controlled chemistry approach requires substantially less water for purging and cleaning steps. Recent innovations in closed-loop precursor delivery systems have demonstrated water usage reductions of 40-60% compared to conventional deposition techniques.
The atmospheric operation of SALD also eliminates the need for certain greenhouse gases commonly used in vacuum-based processes. Many traditional semiconductor manufacturing processes utilize perfluorocarbons (PFCs) and sulfur hexafluoride (SF6), which have global warming potentials thousands of times greater than CO2. SALD processes typically avoid these compounds, further reducing their environmental impact.
End-of-life considerations for SALD-manufactured products show promising sustainability metrics. The precise atomic-level control enables the creation of thinner, more efficient functional layers that can reduce material consumption throughout the product lifecycle. Additionally, the uniform, defect-free nature of SALD films can extend product lifespans, reducing waste generation and resource consumption associated with product replacement.
Recent life cycle assessment (LCA) studies comparing SALD to conventional deposition techniques have demonstrated potential environmental impact reductions of 30-45% across categories including global warming potential, acidification, and resource depletion. These environmental advantages position SALD as a key technology for sustainable manufacturing, particularly as industries face increasing regulatory pressure and consumer demand for environmentally responsible production methods.
The precursor utilization efficiency in SALD systems represents a critical environmental advantage. Through optimized surface chemistry control, SALD can achieve precursor utilization rates of up to 90%, significantly higher than conventional ALD methods. This efficiency reduces chemical waste and minimizes the environmental impact of precursor production and disposal, particularly important when considering the toxic and reactive nature of many ALD precursors.
Water consumption presents another sustainability consideration for SALD processes. While traditional semiconductor manufacturing can consume thousands of liters of ultra-pure water, SALD's controlled chemistry approach requires substantially less water for purging and cleaning steps. Recent innovations in closed-loop precursor delivery systems have demonstrated water usage reductions of 40-60% compared to conventional deposition techniques.
The atmospheric operation of SALD also eliminates the need for certain greenhouse gases commonly used in vacuum-based processes. Many traditional semiconductor manufacturing processes utilize perfluorocarbons (PFCs) and sulfur hexafluoride (SF6), which have global warming potentials thousands of times greater than CO2. SALD processes typically avoid these compounds, further reducing their environmental impact.
End-of-life considerations for SALD-manufactured products show promising sustainability metrics. The precise atomic-level control enables the creation of thinner, more efficient functional layers that can reduce material consumption throughout the product lifecycle. Additionally, the uniform, defect-free nature of SALD films can extend product lifespans, reducing waste generation and resource consumption associated with product replacement.
Recent life cycle assessment (LCA) studies comparing SALD to conventional deposition techniques have demonstrated potential environmental impact reductions of 30-45% across categories including global warming potential, acidification, and resource depletion. These environmental advantages position SALD as a key technology for sustainable manufacturing, particularly as industries face increasing regulatory pressure and consumer demand for environmentally responsible production methods.
Scalability and Industrial Implementation
The scalability of Spatial Atomic Layer Deposition (SALD) represents a critical advantage over conventional ALD techniques, enabling significant throughput improvements while maintaining precise surface chemistry control. Industrial implementation of SALD has progressed substantially in recent years, with production-scale systems now operational across multiple sectors including photovoltaics, display manufacturing, and semiconductor packaging.
Current industrial SALD systems demonstrate deposition rates 10-100 times faster than conventional ALD, with some configurations achieving throughputs exceeding 1200 wafers per hour for specific applications. This dramatic improvement stems from the spatial separation of precursor zones rather than the temporal separation used in traditional ALD, eliminating the need for time-consuming purge cycles.
The implementation architecture varies significantly based on application requirements. Roll-to-roll configurations have gained traction for flexible electronics and barrier films, while rotating drum designs serve high-volume batch processing needs. Linear systems with multiple deposition heads arranged in series have become prevalent in solar cell manufacturing, where uniform zinc oxide and aluminum oxide layers are critical for device performance.
Surface chemistry control at industrial scales presents unique challenges compared to laboratory environments. Gas flow dynamics in large-scale reactors require sophisticated computational fluid dynamics modeling to ensure precursor separation and uniform exposure. Temperature uniformity across large substrates demands advanced heating solutions and thermal management systems to maintain optimal reaction conditions for consistent film growth.
Precursor delivery systems have evolved to accommodate industrial volumes, with liquid delivery systems and heated precursor lines becoming standard for less volatile compounds. Automated process control systems with real-time monitoring capabilities now integrate multiple sensors to track precursor consumption, substrate temperature, and chamber pressure, enabling adaptive adjustments to maintain optimal surface chemistry conditions.
Economic considerations have driven significant innovation in precursor utilization efficiency. Recovery and recycling systems for expensive precursors have been implemented in several commercial SALD platforms, reducing operational costs while minimizing environmental impact. These systems typically capture unreacted precursors through specialized cold traps or absorption media, which are then purified for reuse.
The integration of SALD into existing manufacturing lines presents both opportunities and challenges. While the atmospheric pressure operation simplifies integration compared to vacuum-based techniques, the precise control of environmental conditions around deposition zones requires careful engineering. Leading equipment manufacturers have developed modular SALD systems that can be configured to match specific production requirements and facility constraints.
Current industrial SALD systems demonstrate deposition rates 10-100 times faster than conventional ALD, with some configurations achieving throughputs exceeding 1200 wafers per hour for specific applications. This dramatic improvement stems from the spatial separation of precursor zones rather than the temporal separation used in traditional ALD, eliminating the need for time-consuming purge cycles.
The implementation architecture varies significantly based on application requirements. Roll-to-roll configurations have gained traction for flexible electronics and barrier films, while rotating drum designs serve high-volume batch processing needs. Linear systems with multiple deposition heads arranged in series have become prevalent in solar cell manufacturing, where uniform zinc oxide and aluminum oxide layers are critical for device performance.
Surface chemistry control at industrial scales presents unique challenges compared to laboratory environments. Gas flow dynamics in large-scale reactors require sophisticated computational fluid dynamics modeling to ensure precursor separation and uniform exposure. Temperature uniformity across large substrates demands advanced heating solutions and thermal management systems to maintain optimal reaction conditions for consistent film growth.
Precursor delivery systems have evolved to accommodate industrial volumes, with liquid delivery systems and heated precursor lines becoming standard for less volatile compounds. Automated process control systems with real-time monitoring capabilities now integrate multiple sensors to track precursor consumption, substrate temperature, and chamber pressure, enabling adaptive adjustments to maintain optimal surface chemistry conditions.
Economic considerations have driven significant innovation in precursor utilization efficiency. Recovery and recycling systems for expensive precursors have been implemented in several commercial SALD platforms, reducing operational costs while minimizing environmental impact. These systems typically capture unreacted precursors through specialized cold traps or absorption media, which are then purified for reuse.
The integration of SALD into existing manufacturing lines presents both opportunities and challenges. While the atmospheric pressure operation simplifies integration compared to vacuum-based techniques, the precise control of environmental conditions around deposition zones requires careful engineering. Leading equipment manufacturers have developed modular SALD systems that can be configured to match specific production requirements and facility constraints.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!