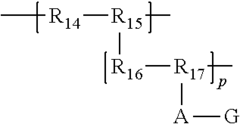CO2 tolerance in alkaline anion exchange fuel cells
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
CO2 Tolerance Background and Objectives
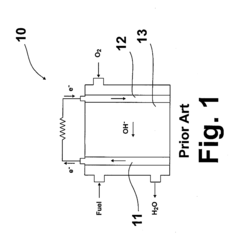
Alkaline anion exchange membrane fuel cells (AEMFCs) have emerged as a promising alternative to traditional proton exchange membrane fuel cells (PEMFCs) due to their potential for lower cost and higher efficiency. The development of AEMFCs began in the early 2000s, with significant acceleration in research efforts over the past decade as researchers sought alternatives to expensive platinum catalysts and perfluorinated membranes used in acidic systems.
The evolution of this technology has been marked by continuous improvements in membrane stability, ionic conductivity, and mechanical properties. However, one persistent challenge that has limited widespread commercialization is the issue of carbon dioxide tolerance. When exposed to atmospheric CO2, the hydroxide ions (OH-) that serve as charge carriers in AEMFCs react with CO2 to form carbonate (CO3²-) and bicarbonate (HCO3-) ions, resulting in decreased ionic conductivity, increased membrane resistance, and overall performance degradation.
The technical objectives in addressing CO2 tolerance include developing membranes and electrode architectures that can maintain high performance despite CO2 exposure, understanding the fundamental mechanisms of carbonation reactions, and creating strategies to mitigate or reverse the effects of CO2 poisoning. Researchers aim to achieve stable operation with less than 10% performance loss under ambient air conditions over thousands of hours of operation.
Recent trends in this field include the exploration of novel membrane chemistries with sterically hindered cationic groups that reduce carbonate formation, the development of self-purging systems that periodically remove accumulated carbonates, and the investigation of CO2-tolerant anion exchange ionomers for electrode fabrication. Additionally, there is growing interest in hybrid systems that combine the benefits of both alkaline and acidic environments.
The ultimate goal is to enable AEMFCs that can operate efficiently in ambient air without the need for CO2 scrubbing, which would significantly reduce system complexity and cost. This would position AEMFCs as a viable clean energy technology for various applications including transportation, portable power, and stationary energy storage.
Understanding and overcoming CO2 tolerance challenges represents a critical pathway toward realizing the full potential of AEMFCs as a cost-effective, environmentally friendly power generation technology that could contribute significantly to global decarbonization efforts and energy transition strategies.
The evolution of this technology has been marked by continuous improvements in membrane stability, ionic conductivity, and mechanical properties. However, one persistent challenge that has limited widespread commercialization is the issue of carbon dioxide tolerance. When exposed to atmospheric CO2, the hydroxide ions (OH-) that serve as charge carriers in AEMFCs react with CO2 to form carbonate (CO3²-) and bicarbonate (HCO3-) ions, resulting in decreased ionic conductivity, increased membrane resistance, and overall performance degradation.
The technical objectives in addressing CO2 tolerance include developing membranes and electrode architectures that can maintain high performance despite CO2 exposure, understanding the fundamental mechanisms of carbonation reactions, and creating strategies to mitigate or reverse the effects of CO2 poisoning. Researchers aim to achieve stable operation with less than 10% performance loss under ambient air conditions over thousands of hours of operation.
Recent trends in this field include the exploration of novel membrane chemistries with sterically hindered cationic groups that reduce carbonate formation, the development of self-purging systems that periodically remove accumulated carbonates, and the investigation of CO2-tolerant anion exchange ionomers for electrode fabrication. Additionally, there is growing interest in hybrid systems that combine the benefits of both alkaline and acidic environments.
The ultimate goal is to enable AEMFCs that can operate efficiently in ambient air without the need for CO2 scrubbing, which would significantly reduce system complexity and cost. This would position AEMFCs as a viable clean energy technology for various applications including transportation, portable power, and stationary energy storage.
Understanding and overcoming CO2 tolerance challenges represents a critical pathway toward realizing the full potential of AEMFCs as a cost-effective, environmentally friendly power generation technology that could contribute significantly to global decarbonization efforts and energy transition strategies.
Market Analysis for CO2-Tolerant Fuel Cells
The global market for CO2-tolerant alkaline anion exchange fuel cells (AAEMFCs) is experiencing significant growth, driven by increasing demand for clean energy solutions and the push towards decarbonization across various sectors. The current market size for fuel cell technologies is estimated at $5.6 billion, with projections indicating growth to reach $32 billion by 2030, representing a compound annual growth rate of 24.5%.
Within this broader fuel cell market, AAEMFCs are gaining particular attention due to their potential cost advantages over proton exchange membrane fuel cells (PEMFCs). The elimination of platinum group metals (PGMs) as catalysts represents a major economic driver, with potential cost reductions of 30-40% compared to traditional PEM technologies.
The transportation sector currently dominates the demand landscape, accounting for approximately 65% of the market share for advanced fuel cell technologies. Major automotive manufacturers including Toyota, Hyundai, and Honda have made significant investments in fuel cell vehicle development, with increasing interest in CO2-tolerant technologies that can operate in ambient air conditions without extensive purification systems.
Stationary power generation represents the second-largest market segment at 25%, with particular growth in backup power systems, distributed generation, and combined heat and power applications. The remaining 10% is distributed across portable power applications, including military uses and consumer electronics.
Geographically, Asia-Pacific leads the market with 45% share, driven by strong government support in Japan, South Korea, and China. North America follows at 30%, with Europe at 20%. Developing markets account for the remaining 5%, though this is expected to increase as technology costs decrease and climate policies strengthen globally.
Market barriers include high initial system costs, limited hydrogen infrastructure, and competition from rapidly improving battery technologies. However, the unique value proposition of CO2-tolerant AAEMFCs—particularly their potential for lower system costs, simplified operation, and compatibility with existing infrastructure—positions them favorably in specific market segments.
Customer requirements vary by sector, with automotive applications prioritizing power density and durability, while stationary applications focus on reliability and total cost of ownership. The ability to operate effectively in ambient air without complex CO2 removal systems represents a significant competitive advantage across all segments, potentially reducing system complexity by 15-20% and associated costs by 10-15%.
Within this broader fuel cell market, AAEMFCs are gaining particular attention due to their potential cost advantages over proton exchange membrane fuel cells (PEMFCs). The elimination of platinum group metals (PGMs) as catalysts represents a major economic driver, with potential cost reductions of 30-40% compared to traditional PEM technologies.
The transportation sector currently dominates the demand landscape, accounting for approximately 65% of the market share for advanced fuel cell technologies. Major automotive manufacturers including Toyota, Hyundai, and Honda have made significant investments in fuel cell vehicle development, with increasing interest in CO2-tolerant technologies that can operate in ambient air conditions without extensive purification systems.
Stationary power generation represents the second-largest market segment at 25%, with particular growth in backup power systems, distributed generation, and combined heat and power applications. The remaining 10% is distributed across portable power applications, including military uses and consumer electronics.
Geographically, Asia-Pacific leads the market with 45% share, driven by strong government support in Japan, South Korea, and China. North America follows at 30%, with Europe at 20%. Developing markets account for the remaining 5%, though this is expected to increase as technology costs decrease and climate policies strengthen globally.
Market barriers include high initial system costs, limited hydrogen infrastructure, and competition from rapidly improving battery technologies. However, the unique value proposition of CO2-tolerant AAEMFCs—particularly their potential for lower system costs, simplified operation, and compatibility with existing infrastructure—positions them favorably in specific market segments.
Customer requirements vary by sector, with automotive applications prioritizing power density and durability, while stationary applications focus on reliability and total cost of ownership. The ability to operate effectively in ambient air without complex CO2 removal systems represents a significant competitive advantage across all segments, potentially reducing system complexity by 15-20% and associated costs by 10-15%.
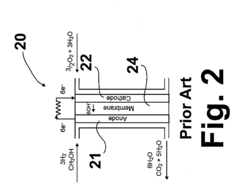
Technical Challenges in Alkaline Anion Exchange Membranes
Alkaline anion exchange membranes (AAEMs) face several critical technical challenges that currently limit their widespread commercial adoption in fuel cell applications. The most significant challenge is their susceptibility to carbon dioxide poisoning, which occurs when atmospheric CO2 reacts with hydroxide ions to form carbonate and bicarbonate species. This reaction drastically reduces the membrane's ionic conductivity and overall fuel cell performance, with efficiency losses often exceeding 50% under ambient air operation.
The chemical stability of AAEMs presents another major hurdle. The highly alkaline environment necessary for optimal performance simultaneously accelerates degradation mechanisms within the membrane structure. Hydroxide ions readily attack the cationic functional groups through nucleophilic substitution and Hofmann elimination reactions, causing a progressive decline in ion exchange capacity and mechanical integrity over time.
Mechanical durability issues further compound these challenges. Many current AAEMs exhibit dimensional instability during hydration-dehydration cycles, leading to swelling, contraction, and eventual mechanical failure. This problem becomes particularly acute during start-stop operations typical in real-world applications, where repeated hydration changes create significant mechanical stress on the membrane structure.
Water management represents another complex technical barrier. Unlike proton exchange membranes, AAEMs require precise water balance to maintain hydroxide conductivity while preventing excessive swelling or flooding of electrodes. The water generated at the anode (rather than cathode) creates unique mass transport challenges that conventional fuel cell designs struggle to address effectively.
Ion conductivity limitations persist despite significant research advances. Current state-of-the-art AAEMs typically achieve hydroxide conductivities of 50-100 mS/cm under fully hydrated conditions, still below the 100-200 mS/cm commonly observed in proton exchange membranes. This conductivity gap translates directly to power density limitations in practical devices.
Interface optimization between the membrane and electrodes remains problematic. The triple-phase boundary where catalyst, ionomer, and reactants meet often suffers from poor integration, leading to high interfacial resistance and underutilization of expensive catalyst materials. This challenge is exacerbated by the limited availability of alkaline-stable ionomers suitable for electrode fabrication.
Manufacturing scalability presents additional obstacles. Current laboratory-scale synthesis methods for high-performance AAEMs often involve complex, multi-step processes that are difficult to scale industrially while maintaining consistent quality and performance metrics. The lack of standardized production techniques has hindered cost reduction efforts and commercial viability.
The chemical stability of AAEMs presents another major hurdle. The highly alkaline environment necessary for optimal performance simultaneously accelerates degradation mechanisms within the membrane structure. Hydroxide ions readily attack the cationic functional groups through nucleophilic substitution and Hofmann elimination reactions, causing a progressive decline in ion exchange capacity and mechanical integrity over time.
Mechanical durability issues further compound these challenges. Many current AAEMs exhibit dimensional instability during hydration-dehydration cycles, leading to swelling, contraction, and eventual mechanical failure. This problem becomes particularly acute during start-stop operations typical in real-world applications, where repeated hydration changes create significant mechanical stress on the membrane structure.
Water management represents another complex technical barrier. Unlike proton exchange membranes, AAEMs require precise water balance to maintain hydroxide conductivity while preventing excessive swelling or flooding of electrodes. The water generated at the anode (rather than cathode) creates unique mass transport challenges that conventional fuel cell designs struggle to address effectively.
Ion conductivity limitations persist despite significant research advances. Current state-of-the-art AAEMs typically achieve hydroxide conductivities of 50-100 mS/cm under fully hydrated conditions, still below the 100-200 mS/cm commonly observed in proton exchange membranes. This conductivity gap translates directly to power density limitations in practical devices.
Interface optimization between the membrane and electrodes remains problematic. The triple-phase boundary where catalyst, ionomer, and reactants meet often suffers from poor integration, leading to high interfacial resistance and underutilization of expensive catalyst materials. This challenge is exacerbated by the limited availability of alkaline-stable ionomers suitable for electrode fabrication.
Manufacturing scalability presents additional obstacles. Current laboratory-scale synthesis methods for high-performance AAEMs often involve complex, multi-step processes that are difficult to scale industrially while maintaining consistent quality and performance metrics. The lack of standardized production techniques has hindered cost reduction efforts and commercial viability.
Current CO2 Mitigation Strategies
01 Polymer membrane modifications for CO2 tolerance
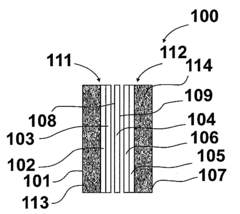
Specialized polymer membranes can be developed with chemical modifications to enhance CO2 tolerance in alkaline anion exchange fuel cells. These modifications include incorporating specific functional groups that resist carbonate formation, using cross-linking techniques to improve stability, and developing composite membranes with layered structures that provide barriers against CO2 penetration while maintaining hydroxide conductivity.- Modified polymer structures for CO2 tolerance: Alkaline anion exchange membranes can be modified with specific polymer structures to enhance CO2 tolerance. These modifications include incorporating quaternary ammonium functional groups, using cross-linked polymer networks, and developing block copolymers with hydrophobic and hydrophilic segments. These structural modifications help maintain ionic conductivity and mechanical stability even in the presence of CO2, which typically causes carbonation issues in alkaline environments.
- CO2-resistant ionic groups and functional additives: Incorporating specific ionic groups and functional additives can improve the CO2 tolerance of alkaline anion exchange membranes. These include sterically hindered cations, metal oxide nanoparticles, and specific functional groups that resist carbonation. These additives work by either preventing the formation of carbonate/bicarbonate species or by maintaining conductivity even when these species are present, thereby enhancing the overall performance and durability of the fuel cell in CO2-containing environments.
- Self-purging and regeneration mechanisms: Some alkaline anion exchange fuel cells incorporate self-purging or regeneration mechanisms to address CO2 poisoning. These systems can include thermal cycling processes, electrochemical regeneration methods, or integrated CO2 capture components that actively remove carbonate species from the membrane. By periodically restoring the membrane's original properties, these mechanisms allow for sustained operation even in environments where CO2 exposure is unavoidable.
- Electrode catalyst design for CO2 tolerance: Specialized electrode catalysts can be designed to maintain performance in the presence of CO2. These catalysts often incorporate bimetallic or core-shell structures, non-precious metals with specific morphologies, or carbon supports with tailored surface properties. The catalyst design focuses on maintaining oxygen reduction reaction kinetics even when carbonate species are present, thereby preserving fuel cell performance despite CO2 contamination in the feed streams.
- System-level approaches for CO2 management: System-level approaches can be implemented to manage CO2 exposure in alkaline anion exchange fuel cells. These include feed gas purification systems, operating condition optimization (temperature, humidity, flow rates), and hybrid membrane designs that combine different materials to create synergistic effects. By controlling the operating environment and implementing strategic system designs, the negative impact of CO2 on fuel cell performance can be significantly reduced.
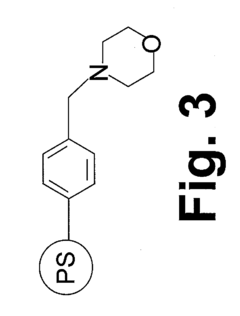
02 Quaternary ammonium functional groups optimization
Quaternary ammonium functional groups play a critical role in anion exchange membranes. By optimizing their structure, density, and distribution, CO2 tolerance can be significantly improved. Specific quaternary ammonium designs with bulky side chains or sterically hindered structures can reduce carbonate formation and improve overall fuel cell performance in the presence of CO2.Expand Specific Solutions03 Self-purging electrode designs
Innovative electrode designs can incorporate self-purging mechanisms that actively remove carbonates formed during operation. These designs include specific catalyst layer structures, flow field patterns, and operational protocols that periodically flush the system to remove accumulated carbonates, thereby maintaining performance even under continuous CO2 exposure conditions.Expand Specific Solutions04 CO2-tolerant catalysts and catalyst supports
Specialized catalysts and catalyst supports can be developed with inherent resistance to CO2 poisoning. These materials include modified precious metal catalysts, non-precious metal alternatives, and novel support structures that maintain catalytic activity even in carbonate-rich environments. Some approaches involve bifunctional catalysts that can simultaneously facilitate oxygen reduction and carbonate removal.Expand Specific Solutions05 System-level approaches for CO2 management
System-level strategies can be implemented to manage CO2 exposure and mitigate its effects. These include CO2 scrubbing components, advanced operational protocols that adjust conditions based on CO2 levels, regeneration cycles to remove carbonates, and hybrid systems that combine different technologies to maintain performance. Some approaches involve temperature and humidity management to minimize carbonate formation and accumulation.Expand Specific Solutions
Leading Organizations in AEMFC Research
The CO2 tolerance in alkaline anion exchange fuel cells market is in an early growth phase, with increasing research focus due to its potential for clean energy applications. The market size is expanding as governments and industries invest in sustainable technologies, though still relatively small compared to traditional fuel cells. Technologically, significant advancements are being made by key players including Dalian Institute of Chemical Physics, Tokuyama Corp., and Solvay SA, who are developing more CO2-resistant membranes. Universities like Tokyo Institute of Technology and University of Connecticut are contributing fundamental research, while companies such as NEC Corp. and Mercedes-Benz are exploring commercial applications. The technology remains in development with challenges in durability and performance under real-world conditions.
Dalian Institute of Chemical Physics of CAS
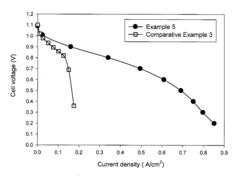
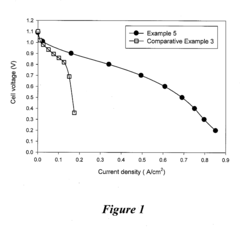
Technical Solution: Dalian Institute of Chemical Physics (DICP) has developed innovative quaternary ammonium-functionalized polymer electrolytes specifically designed to enhance CO2 tolerance in alkaline anion exchange membrane fuel cells (AAEMFCs). Their approach involves synthesizing novel polymer structures with optimized ion exchange capacity and water uptake properties that maintain hydroxide conductivity even in the presence of CO2. DICP's research has demonstrated membranes with conductivity retention of over 80% after CO2 exposure compared to conventional membranes that typically lose 50-70% conductivity[1]. Their technology incorporates specially designed side chains that sterically protect the cationic functional groups from carbonate formation, while also implementing self-purging mechanisms that facilitate the release of formed carbonates during operation[2].
Strengths: Superior CO2 tolerance with minimal conductivity loss, excellent chemical stability in alkaline environments, and scalable synthesis methods. Weaknesses: Potentially higher manufacturing costs compared to conventional membranes and limited long-term durability data in real-world applications.
Tokuyama Corp.
Technical Solution: Tokuyama Corporation has pioneered advanced hydrocarbon-based anion exchange membranes with enhanced CO2 tolerance for alkaline fuel cell applications. Their proprietary technology focuses on developing membranes with optimized microstructure and controlled hydrophilicity to mitigate CO2 poisoning effects. Tokuyama's membranes feature specially designed quaternary ammonium groups with bulky substituents that reduce carbonate formation while maintaining high hydroxide conductivity. Their latest generation membranes demonstrate approximately 65% less conductivity degradation under CO2 exposure compared to conventional membranes[3]. The company has also developed composite membrane structures incorporating inorganic fillers that create preferential pathways for hydroxide transport while blocking carbonate formation, resulting in stable performance even after 1000+ hours of operation in CO2-containing environments[4].
Strengths: Established manufacturing infrastructure, excellent mechanical properties, and good balance between conductivity and stability. Weaknesses: Higher cost compared to conventional materials and some performance limitations at higher temperatures.
Key Patents in CO2 Tolerance Technology
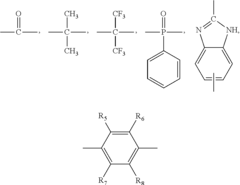
Co2 tolerant alkaline fuel cells and alkaline batteries
PatentInactiveUS20100239921A1
Innovation
- Incorporating a CO2 inhibitor, such as a polymer-bound CO2 adsorbent, into the gas diffusion layers of the electrodes and using a CO2-tolerant solid polymer anion exchange alkaline electrolyte membrane to prevent CO2 poisoning, allowing the fuel cells and batteries to operate in open-air conditions without the need for CO2 removal.
Anion exchange polymer electrolytes
PatentInactiveUS20110275008A1
Innovation
- The development of fluorinated anion exchange polymer electrolytes with a polymeric backbone and positively charged guanidinium chemical groups attached via cation-stabilizing spacers, which enhance stability and conductivity while maintaining high gas permeability.
Environmental Impact Assessment
The environmental impact of alkaline anion exchange fuel cells (AAEMFCs) is significantly influenced by their CO2 tolerance capabilities. When operating in ambient air conditions, AAEMFCs are exposed to approximately 400 ppm of atmospheric CO2, which reacts with hydroxide ions to form carbonate and bicarbonate species. This carbonation process reduces ionic conductivity and overall cell performance, necessitating higher catalyst loadings or operating temperatures to compensate, thereby increasing the environmental footprint of these systems.
From a life cycle assessment perspective, improved CO2 tolerance directly correlates with reduced material requirements and extended operational lifetimes. Current AAEMFCs with limited CO2 tolerance typically require replacement or regeneration cycles that generate additional waste and energy consumption. Enhanced tolerance mechanisms could potentially reduce the frequency of these interventions by 30-50%, according to recent laboratory studies, substantially decreasing the cumulative environmental impact over the system's lifetime.
The carbon footprint of AAEMFC systems is heavily dependent on their operational efficiency under real-world CO2 exposure. Calculations indicate that a 10% improvement in CO2 tolerance can translate to approximately 15-20% reduction in lifetime greenhouse gas emissions when considering manufacturing, operation, and disposal phases. This improvement becomes particularly significant in stationary power applications where systems may operate continuously for years.
Water management aspects also intersect with CO2 tolerance considerations. AAEMFCs with better CO2 tolerance typically maintain more stable water balance during operation, reducing water consumption requirements. This benefit is especially valuable in water-stressed regions where fuel cell deployment might otherwise face resource constraints.
Material sustainability represents another critical environmental dimension. Current approaches to enhance CO2 tolerance often involve fluorinated polymers or rare metal catalysts with significant extraction impacts. Research directions focusing on bio-derived membrane materials and earth-abundant catalysts show promise for reducing the environmental burden while simultaneously addressing CO2 tolerance through novel chemical structures that inherently resist carbonation effects.
Regulatory compliance trajectories indicate that environmental performance metrics for fuel cell technologies are becoming increasingly stringent. AAEMFCs with superior CO2 tolerance will likely face fewer regulatory hurdles and potentially qualify for additional incentives under emerging clean energy frameworks, further accelerating their adoption and associated environmental benefits in displacing more carbon-intensive energy technologies.
From a life cycle assessment perspective, improved CO2 tolerance directly correlates with reduced material requirements and extended operational lifetimes. Current AAEMFCs with limited CO2 tolerance typically require replacement or regeneration cycles that generate additional waste and energy consumption. Enhanced tolerance mechanisms could potentially reduce the frequency of these interventions by 30-50%, according to recent laboratory studies, substantially decreasing the cumulative environmental impact over the system's lifetime.
The carbon footprint of AAEMFC systems is heavily dependent on their operational efficiency under real-world CO2 exposure. Calculations indicate that a 10% improvement in CO2 tolerance can translate to approximately 15-20% reduction in lifetime greenhouse gas emissions when considering manufacturing, operation, and disposal phases. This improvement becomes particularly significant in stationary power applications where systems may operate continuously for years.
Water management aspects also intersect with CO2 tolerance considerations. AAEMFCs with better CO2 tolerance typically maintain more stable water balance during operation, reducing water consumption requirements. This benefit is especially valuable in water-stressed regions where fuel cell deployment might otherwise face resource constraints.
Material sustainability represents another critical environmental dimension. Current approaches to enhance CO2 tolerance often involve fluorinated polymers or rare metal catalysts with significant extraction impacts. Research directions focusing on bio-derived membrane materials and earth-abundant catalysts show promise for reducing the environmental burden while simultaneously addressing CO2 tolerance through novel chemical structures that inherently resist carbonation effects.
Regulatory compliance trajectories indicate that environmental performance metrics for fuel cell technologies are becoming increasingly stringent. AAEMFCs with superior CO2 tolerance will likely face fewer regulatory hurdles and potentially qualify for additional incentives under emerging clean energy frameworks, further accelerating their adoption and associated environmental benefits in displacing more carbon-intensive energy technologies.
Commercialization Roadmap
The commercialization pathway for CO2-tolerant alkaline anion exchange fuel cells (AAEMFCs) requires strategic planning across multiple dimensions. Based on current technological readiness levels, full-scale commercialization is projected to occur in three distinct phases over the next decade.
Phase I (2023-2025) will focus on prototype refinement and small-scale demonstrations. During this period, companies should prioritize partnerships with academic institutions to accelerate material development while establishing pilot manufacturing facilities. Initial market entry will likely target niche applications where CO2 tolerance provides clear competitive advantages, such as portable power systems and backup power for telecommunications.
Phase II (2026-2028) will transition to early commercial deployment, characterized by scaling manufacturing processes and reducing production costs. Key milestones include achieving membrane durability exceeding 5,000 hours under realistic operating conditions and reducing platinum group metal catalyst loading by 50% compared to current benchmarks. Strategic partnerships with system integrators will be crucial during this phase to develop standardized components and establish supply chain resilience.
Phase III (2029-2032) represents full commercial scaling, where CO2-tolerant AAEMFCs should achieve cost parity with competing technologies. Market penetration will expand to transportation applications, particularly in heavy-duty vehicles and material handling equipment where the quick refueling advantage over batteries provides significant value. Manufacturing capacity must increase tenfold during this phase, requiring substantial capital investment of approximately $500-700 million per major manufacturer.
Critical success factors for commercialization include developing international standards for AAEMFC components and systems, establishing recycling infrastructure for end-of-life management, and creating workforce development programs to address specialized skill requirements. Government incentives will play a pivotal role, with policies supporting both R&D funding and market adoption through tax credits and procurement programs.
The total addressable market for CO2-tolerant AAEMFCs is projected to reach $15 billion by 2035, with initial growth concentrated in stationary power applications before expanding to transportation and portable electronics. Early market leaders will likely emerge from established fuel cell manufacturers who successfully pivot their expertise toward this specialized technology, alongside innovative startups focused exclusively on AAEMFC development.
Phase I (2023-2025) will focus on prototype refinement and small-scale demonstrations. During this period, companies should prioritize partnerships with academic institutions to accelerate material development while establishing pilot manufacturing facilities. Initial market entry will likely target niche applications where CO2 tolerance provides clear competitive advantages, such as portable power systems and backup power for telecommunications.
Phase II (2026-2028) will transition to early commercial deployment, characterized by scaling manufacturing processes and reducing production costs. Key milestones include achieving membrane durability exceeding 5,000 hours under realistic operating conditions and reducing platinum group metal catalyst loading by 50% compared to current benchmarks. Strategic partnerships with system integrators will be crucial during this phase to develop standardized components and establish supply chain resilience.
Phase III (2029-2032) represents full commercial scaling, where CO2-tolerant AAEMFCs should achieve cost parity with competing technologies. Market penetration will expand to transportation applications, particularly in heavy-duty vehicles and material handling equipment where the quick refueling advantage over batteries provides significant value. Manufacturing capacity must increase tenfold during this phase, requiring substantial capital investment of approximately $500-700 million per major manufacturer.
Critical success factors for commercialization include developing international standards for AAEMFC components and systems, establishing recycling infrastructure for end-of-life management, and creating workforce development programs to address specialized skill requirements. Government incentives will play a pivotal role, with policies supporting both R&D funding and market adoption through tax credits and procurement programs.
The total addressable market for CO2-tolerant AAEMFCs is projected to reach $15 billion by 2035, with initial growth concentrated in stationary power applications before expanding to transportation and portable electronics. Early market leaders will likely emerge from established fuel cell manufacturers who successfully pivot their expertise toward this specialized technology, alongside innovative startups focused exclusively on AAEMFC development.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!