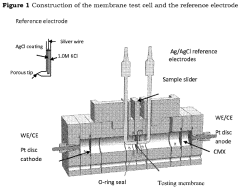
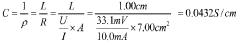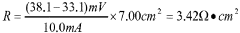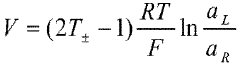Fabrication methods for durable anion exchange membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AEM Fabrication Background and Objectives
Anion exchange membranes (AEMs) have emerged as critical components in various electrochemical technologies, including fuel cells, electrolyzers, and energy storage systems. The development of these membranes dates back to the mid-20th century, but significant advancements have occurred in the past two decades as researchers sought alternatives to proton exchange membrane technologies. The evolution of AEM fabrication methods has been driven by the need to overcome inherent challenges in alkaline environments, particularly chemical and mechanical stability issues.
Traditional fabrication approaches relied heavily on quaternization of polymer backbones with limited consideration for long-term durability. Early AEMs suffered from rapid degradation in alkaline conditions, severely limiting their practical applications. The technological trajectory has since shifted toward developing multi-functional materials that can withstand harsh operating conditions while maintaining high ionic conductivity and mechanical integrity.
Recent advances in polymer chemistry and materials science have enabled more sophisticated fabrication strategies, including cross-linking techniques, block copolymer synthesis, and composite membrane development. These approaches aim to address the fundamental trade-off between ion conductivity and mechanical/chemical stability that has historically plagued AEM development.
The primary objective of current AEM fabrication research is to develop membranes with hydroxide conductivity exceeding 100 mS/cm while maintaining stability for over 5,000 hours under operating conditions. This represents a significant challenge as hydroxide ions are inherently more reactive than protons, leading to accelerated degradation of both the polymer backbone and the functional groups responsible for ion transport.
Emerging trends in AEM fabrication include the incorporation of nanomaterials to enhance mechanical properties, the development of self-healing membranes that can recover from degradation, and the exploration of bio-inspired materials that mimic natural ion transport mechanisms. Additionally, there is growing interest in scalable manufacturing processes that can transition laboratory successes to commercial production.
The global push toward renewable energy and carbon-neutral technologies has intensified research efforts in this field, with particular emphasis on developing fabrication methods that utilize sustainable materials and processes. This aligns with broader environmental goals while potentially reducing production costs, which remains a significant barrier to widespread adoption.
As the field progresses, interdisciplinary approaches combining expertise from polymer chemistry, electrochemistry, and materials engineering are becoming increasingly important. The ultimate goal is to establish standardized fabrication protocols that can reliably produce AEMs with predictable performance characteristics and exceptional durability across various applications.
Traditional fabrication approaches relied heavily on quaternization of polymer backbones with limited consideration for long-term durability. Early AEMs suffered from rapid degradation in alkaline conditions, severely limiting their practical applications. The technological trajectory has since shifted toward developing multi-functional materials that can withstand harsh operating conditions while maintaining high ionic conductivity and mechanical integrity.
Recent advances in polymer chemistry and materials science have enabled more sophisticated fabrication strategies, including cross-linking techniques, block copolymer synthesis, and composite membrane development. These approaches aim to address the fundamental trade-off between ion conductivity and mechanical/chemical stability that has historically plagued AEM development.
The primary objective of current AEM fabrication research is to develop membranes with hydroxide conductivity exceeding 100 mS/cm while maintaining stability for over 5,000 hours under operating conditions. This represents a significant challenge as hydroxide ions are inherently more reactive than protons, leading to accelerated degradation of both the polymer backbone and the functional groups responsible for ion transport.
Emerging trends in AEM fabrication include the incorporation of nanomaterials to enhance mechanical properties, the development of self-healing membranes that can recover from degradation, and the exploration of bio-inspired materials that mimic natural ion transport mechanisms. Additionally, there is growing interest in scalable manufacturing processes that can transition laboratory successes to commercial production.
The global push toward renewable energy and carbon-neutral technologies has intensified research efforts in this field, with particular emphasis on developing fabrication methods that utilize sustainable materials and processes. This aligns with broader environmental goals while potentially reducing production costs, which remains a significant barrier to widespread adoption.
As the field progresses, interdisciplinary approaches combining expertise from polymer chemistry, electrochemistry, and materials engineering are becoming increasingly important. The ultimate goal is to establish standardized fabrication protocols that can reliably produce AEMs with predictable performance characteristics and exceptional durability across various applications.
Market Analysis for Durable AEM Applications
The global market for anion exchange membranes (AEMs) is experiencing significant growth, driven primarily by increasing demand for clean energy technologies. The AEM market was valued at approximately $570 million in 2022 and is projected to reach $1.2 billion by 2030, representing a compound annual growth rate (CAGR) of 9.8%. This growth trajectory is largely attributed to the expanding applications of AEMs in fuel cells, electrolyzers, and energy storage systems.
The fuel cell sector represents the largest application segment for durable AEMs, accounting for nearly 45% of the total market share. Within this segment, alkaline fuel cells (AFCs) and anion exchange membrane fuel cells (AEMFCs) are gaining traction due to their potential cost advantages over proton exchange membrane fuel cells (PEMFCs), particularly in transportation and stationary power applications.
Water electrolysis for hydrogen production constitutes the fastest-growing application segment, with a projected CAGR of 12.3% through 2030. This acceleration is driven by global hydrogen strategies and substantial investments in green hydrogen infrastructure across Europe, North America, and parts of Asia. The European Clean Hydrogen Alliance alone has announced over €45 billion in planned investments, creating significant opportunities for advanced AEM technologies.
Geographically, North America currently leads the market with approximately 35% share, followed closely by Europe at 32% and Asia-Pacific at 28%. However, the Asia-Pacific region is expected to witness the highest growth rate, particularly in China, Japan, and South Korea, where government initiatives supporting hydrogen economies are creating favorable market conditions.
End-user industries demonstrate varying adoption rates for durable AEMs. The automotive sector represents a significant growth opportunity, with major manufacturers increasingly incorporating fuel cell technology into their vehicle development roadmaps. The stationary power generation sector follows closely, particularly for backup power systems and distributed energy applications in remote locations.
Market challenges include price sensitivity, with current high-performance durable AEMs costing between $400-800 per square meter, significantly higher than traditional membranes. This price premium necessitates clear demonstration of performance advantages and longevity benefits to justify adoption. Additionally, competition from established proton exchange membrane technologies creates market entry barriers for new AEM products.
Customer requirements are evolving toward membranes with longer operational lifetimes (>20,000 hours), improved alkaline stability, higher hydroxide conductivity (>100 mS/cm), and reduced cost. These market demands are directly influencing research priorities in fabrication methods for more durable and cost-effective AEMs.
The fuel cell sector represents the largest application segment for durable AEMs, accounting for nearly 45% of the total market share. Within this segment, alkaline fuel cells (AFCs) and anion exchange membrane fuel cells (AEMFCs) are gaining traction due to their potential cost advantages over proton exchange membrane fuel cells (PEMFCs), particularly in transportation and stationary power applications.
Water electrolysis for hydrogen production constitutes the fastest-growing application segment, with a projected CAGR of 12.3% through 2030. This acceleration is driven by global hydrogen strategies and substantial investments in green hydrogen infrastructure across Europe, North America, and parts of Asia. The European Clean Hydrogen Alliance alone has announced over €45 billion in planned investments, creating significant opportunities for advanced AEM technologies.
Geographically, North America currently leads the market with approximately 35% share, followed closely by Europe at 32% and Asia-Pacific at 28%. However, the Asia-Pacific region is expected to witness the highest growth rate, particularly in China, Japan, and South Korea, where government initiatives supporting hydrogen economies are creating favorable market conditions.
End-user industries demonstrate varying adoption rates for durable AEMs. The automotive sector represents a significant growth opportunity, with major manufacturers increasingly incorporating fuel cell technology into their vehicle development roadmaps. The stationary power generation sector follows closely, particularly for backup power systems and distributed energy applications in remote locations.
Market challenges include price sensitivity, with current high-performance durable AEMs costing between $400-800 per square meter, significantly higher than traditional membranes. This price premium necessitates clear demonstration of performance advantages and longevity benefits to justify adoption. Additionally, competition from established proton exchange membrane technologies creates market entry barriers for new AEM products.
Customer requirements are evolving toward membranes with longer operational lifetimes (>20,000 hours), improved alkaline stability, higher hydroxide conductivity (>100 mS/cm), and reduced cost. These market demands are directly influencing research priorities in fabrication methods for more durable and cost-effective AEMs.
Current Challenges in AEM Fabrication Technology
Despite significant advancements in anion exchange membrane (AEM) technology, several critical challenges persist in fabrication methods that limit their widespread commercial adoption. The primary obstacle remains chemical stability, particularly in alkaline environments where hydroxide ions aggressively attack the cationic functional groups, leading to membrane degradation and shortened operational lifespans. This degradation occurs through multiple mechanisms including Hofmann elimination, nucleophilic substitution, and ylide formation, which collectively compromise membrane integrity over time.
Mechanical durability presents another significant challenge, as many AEMs exhibit dimensional instability during hydration-dehydration cycles. This manifests as excessive swelling, which weakens mechanical properties and creates physical stress that can lead to cracking and membrane failure. The trade-off between ion conductivity and mechanical strength remains difficult to optimize, as higher ion exchange capacity typically correlates with greater water uptake and consequent dimensional instability.
Scalable manufacturing represents a substantial hurdle for AEM commercialization. Current laboratory-scale fabrication methods often involve complex multi-step processes that are difficult to translate to industrial production. Techniques such as electrospinning, phase inversion, and radiation grafting face challenges in maintaining consistent quality and performance characteristics when scaled up. Additionally, many fabrication approaches utilize environmentally problematic solvents or require energy-intensive processing steps, raising sustainability concerns.
The integration of functional groups presents technical difficulties in achieving uniform distribution throughout the membrane matrix. Heterogeneous distribution of ionic sites creates localized areas of varying conductivity and mechanical properties, leading to inconsistent performance and potential failure points. Current methods struggle to precisely control the degree of functionalization while maintaining other desirable membrane properties.
Cost-effectiveness remains a significant barrier, with many high-performance AEMs requiring expensive precursors or complex synthesis routes. The multi-step fabrication processes increase production costs substantially compared to proton exchange membranes, limiting economic viability for mass-market applications. Furthermore, quality control and reproducibility issues persist across different fabrication batches, creating challenges for consistent performance in commercial applications.
Interfacial compatibility between the membrane and electrode materials presents additional complications, as poor adhesion can lead to increased interfacial resistance and reduced device efficiency. Current fabrication approaches often fail to adequately address this interface engineering aspect, resulting in suboptimal performance in assembled devices.
Mechanical durability presents another significant challenge, as many AEMs exhibit dimensional instability during hydration-dehydration cycles. This manifests as excessive swelling, which weakens mechanical properties and creates physical stress that can lead to cracking and membrane failure. The trade-off between ion conductivity and mechanical strength remains difficult to optimize, as higher ion exchange capacity typically correlates with greater water uptake and consequent dimensional instability.
Scalable manufacturing represents a substantial hurdle for AEM commercialization. Current laboratory-scale fabrication methods often involve complex multi-step processes that are difficult to translate to industrial production. Techniques such as electrospinning, phase inversion, and radiation grafting face challenges in maintaining consistent quality and performance characteristics when scaled up. Additionally, many fabrication approaches utilize environmentally problematic solvents or require energy-intensive processing steps, raising sustainability concerns.
The integration of functional groups presents technical difficulties in achieving uniform distribution throughout the membrane matrix. Heterogeneous distribution of ionic sites creates localized areas of varying conductivity and mechanical properties, leading to inconsistent performance and potential failure points. Current methods struggle to precisely control the degree of functionalization while maintaining other desirable membrane properties.
Cost-effectiveness remains a significant barrier, with many high-performance AEMs requiring expensive precursors or complex synthesis routes. The multi-step fabrication processes increase production costs substantially compared to proton exchange membranes, limiting economic viability for mass-market applications. Furthermore, quality control and reproducibility issues persist across different fabrication batches, creating challenges for consistent performance in commercial applications.
Interfacial compatibility between the membrane and electrode materials presents additional complications, as poor adhesion can lead to increased interfacial resistance and reduced device efficiency. Current fabrication approaches often fail to adequately address this interface engineering aspect, resulting in suboptimal performance in assembled devices.
State-of-the-Art AEM Fabrication Techniques
01 Chemical modifications to enhance durability
Various chemical modifications can be applied to anion exchange membranes to improve their durability. These include crosslinking techniques, incorporation of stable functional groups, and chemical treatments that enhance resistance to degradation. Such modifications help to maintain membrane integrity and ion conductivity over extended periods of operation, particularly in alkaline environments where traditional membranes tend to degrade.- Chemical modifications for enhanced durability: Various chemical modifications can be applied to anion exchange membranes to improve their durability. These include crosslinking techniques, incorporation of stable functional groups, and chemical treatments that enhance resistance to degradation. Such modifications help to maintain membrane integrity and ion conductivity over extended periods of operation, particularly in alkaline environments where traditional anion exchange membranes tend to degrade.
- Polymer backbone reinforcement strategies: Reinforcing the polymer backbone of anion exchange membranes significantly improves their mechanical strength and chemical stability. This can be achieved through the use of aromatic polymers, fluorinated materials, or composite structures. These reinforced membranes exhibit superior resistance to physical stress and chemical degradation, leading to extended operational lifetimes in various applications including fuel cells and electrolysis systems.
- Composite and hybrid membrane structures: Composite and hybrid membrane structures combine different materials to overcome the limitations of single-component membranes. These may include inorganic-organic hybrids, layered structures, or reinforced membranes with specialized fillers. Such composite designs can simultaneously address multiple durability challenges, including mechanical strength, chemical stability, and resistance to dimensional changes during hydration cycles.
- Temperature and humidity resistance improvements: Enhancing the temperature and humidity resistance of anion exchange membranes is crucial for their durability in varying operational conditions. This can be achieved through the incorporation of thermally stable components, hydrophilic-hydrophobic balanced structures, and specialized additives that maintain membrane performance across wide temperature and humidity ranges. These improvements prevent membrane degradation due to thermal stress and hydration-dehydration cycles.
- Alkaline stability enhancement techniques: Improving the alkaline stability of anion exchange membranes is essential for their durability in high pH environments. This can be achieved through the development of novel cationic groups that resist degradation, protective coatings, and specialized polymer architectures. These techniques minimize the common degradation pathways such as Hofmann elimination and nucleophilic substitution that typically limit membrane lifetime in alkaline conditions.
02 Polymer backbone reinforcement strategies
Reinforcing the polymer backbone of anion exchange membranes is crucial for improving durability. This can be achieved through the use of robust aromatic polymers, fluorinated backbones, or by incorporating rigid structural elements. These reinforcement strategies help to prevent mechanical failure and chemical degradation during operation, resulting in membranes with extended service life and consistent performance.Expand Specific Solutions03 Composite and hybrid membrane structures
Composite and hybrid membrane structures combine different materials to enhance durability. These may include inorganic-organic hybrids, layered structures, or reinforced membranes with supportive matrices. By integrating complementary materials, these membranes achieve improved mechanical strength, chemical stability, and resistance to swelling, addressing multiple durability challenges simultaneously.Expand Specific Solutions04 Alkaline stability enhancement techniques
Specific techniques to enhance alkaline stability are essential for anion exchange membrane durability. These include the development of quaternary ammonium groups with steric protection, incorporation of alkaline-resistant functional groups, and design of membrane architectures that minimize hydroxide attack. These approaches specifically target the common failure mode of degradation in high pH environments.Expand Specific Solutions05 Testing and characterization methods for durability assessment
Advanced testing and characterization methods are crucial for evaluating anion exchange membrane durability. These include accelerated aging tests, in-situ performance monitoring, spectroscopic analysis of degradation mechanisms, and mechanical property evaluations under operating conditions. Such methods provide insights into failure modes and enable the development of more durable membrane materials through systematic improvement.Expand Specific Solutions
Leading Manufacturers and Research Institutions
The fabrication of durable anion exchange membranes (AEMs) is currently in a growth phase, with the global market expanding as these materials become critical for clean energy applications. The market is characterized by a mix of established chemical corporations and emerging research institutions. Companies like Tokuyama Corp., Nitto Denko, and Kuraray lead commercial development with mature manufacturing capabilities, while academic institutions such as Tsinghua University and KAIST drive fundamental innovation. Specialized firms like Dioxide Materials and Enapter are advancing AEM technology specifically for electrolyzer applications. The technology is approaching commercial maturity but still faces challenges in balancing ionic conductivity, chemical stability, and mechanical durability. Recent collaborations between industry leaders and research institutions indicate accelerating development toward cost-effective, high-performance membranes for renewable energy systems.
Tokuyama Corp.
Technical Solution: Tokuyama Corporation has developed sophisticated fabrication methods for durable anion exchange membranes based on their extensive experience in ion exchange technologies. Their approach utilizes fluorinated and partially fluorinated polymer backbones with strategically attached quaternary ammonium groups to achieve high ionic conductivity while maintaining chemical stability. Tokuyama employs a proprietary reinforcement technique where the membrane is supported by an engineered substrate that enhances mechanical durability without compromising ion transport. Their manufacturing process involves precision extrusion and lamination steps followed by controlled functionalization reactions to achieve uniform distribution of ion exchange sites. The company has pioneered a post-treatment process that significantly reduces membrane swelling while preserving conductivity, addressing one of the key challenges in AEM technology. Tokuyama's membranes demonstrate exceptional dimensional stability with less than 10% dimensional change when transitioning between dry and fully hydrated states, while maintaining hydroxide conductivity above 80 mS/cm at operating temperatures. Their latest generation membranes show remarkable alkaline stability with less than 5% degradation in ion exchange capacity after 2000 hours in 1M KOH at 60°C.
Strengths: Superior dimensional stability during hydration cycles; excellent balance between conductivity and mechanical strength; established mass production capabilities ensuring consistent quality and competitive pricing. Weaknesses: Relatively complex manufacturing process requiring precise control of multiple parameters; potential environmental concerns with fluorinated polymer components; limited flexibility for customization in specialized applications.
Giner, Inc.
Technical Solution: Giner has developed innovative fabrication methods for highly durable anion exchange membranes specifically designed for electrochemical applications. Their approach centers on radiation-grafted membranes where ion-conducting groups are introduced to pre-formed polymer films through controlled radiation exposure followed by grafting reactions. This technique allows precise control over the distribution and density of functional groups throughout the membrane structure. Giner employs a proprietary stabilization process that incorporates sterically hindered quaternary ammonium groups, significantly enhancing alkaline stability compared to conventional AEMs. Their manufacturing process includes a specialized annealing step that optimizes the microphase separation between hydrophilic and hydrophobic domains, creating efficient ion transport channels while maintaining structural integrity. Giner has pioneered composite membrane architectures where different functional layers are combined to create membranes with gradient properties that address specific electrochemical requirements. Their membranes demonstrate hydroxide conductivity exceeding 90 mS/cm at 80°C while maintaining mechanical integrity and dimensional stability. Recent advancements include the incorporation of self-healing components that can mitigate degradation mechanisms, extending membrane lifetime in aggressive alkaline environments by up to 300% compared to conventional AEMs.
Strengths: Exceptional performance in electrochemical applications like fuel cells and electrolyzers; highly customizable properties through radiation grafting parameters; excellent chemical stability in aggressive operating conditions. Weaknesses: Higher manufacturing costs due to specialized radiation processing equipment; potential scalability challenges for high-volume production; limited shelf life for certain membrane formulations before installation.
Key Patents and Scientific Breakthroughs
Anion exchange membranes and process for making
PatentActiveEP3626341A1
Innovation
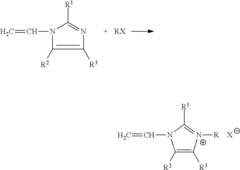
- The development of anion exchange membranes using functional monomers with tertiary amine groups and quaternizing agents, polymerized with crosslinking monomers and initiators within porous substrates, to create cross-linked polymers with low resistance and high permeability, suitable for a broad operational pH range.
High-performance anion exchange membranes and methods of making same
PatentActiveUS20180057370A1
Innovation
- A method for producing homogenous ion exchange membranes involves mixing a tertiary amine monomer with a quaternization agent, followed by a cross-linking agent and solvent to form a monomeric solution, which is then cast onto a polymeric microporous substrate, resulting in membranes with enhanced electrochemical properties like low resistivity and high permselectivity, and allowing for a more efficient and cost-effective production process.
Environmental Impact and Sustainability Considerations
The fabrication of anion exchange membranes (AEMs) presents significant environmental considerations that must be addressed for sustainable implementation. Traditional manufacturing processes often involve toxic solvents such as dimethylformamide (DMF), N-methyl-2-pyrrolidone (NMP), and chloroform, which pose substantial environmental hazards and health risks. Recent advancements have focused on developing greener fabrication routes utilizing less harmful alternatives like ethanol, water-based systems, and supercritical CO2 as processing media, substantially reducing the environmental footprint of AEM production.
Life cycle assessment (LCA) studies indicate that the environmental impact of AEMs extends beyond manufacturing to include raw material extraction, energy consumption during fabrication, and end-of-life disposal. Particularly concerning is the widespread use of fluorinated polymers in high-performance AEMs, which demonstrate exceptional durability but present significant environmental persistence issues. These materials can potentially degrade into perfluoroalkyl substances (PFAS), which are recognized as persistent environmental contaminants with bioaccumulative properties.
Emerging sustainable approaches include bio-based precursors for AEM fabrication, such as chitosan, cellulose derivatives, and lignin-based materials. These renewable resources not only reduce dependence on petroleum-derived polymers but also often exhibit inherent biodegradability. For instance, quaternized chitosan-based membranes have demonstrated promising ionic conductivity while offering improved environmental compatibility compared to conventional synthetic alternatives.
Energy efficiency in fabrication processes represents another critical sustainability factor. Traditional thermal crosslinking methods consume substantial energy, whereas emerging techniques like UV-initiated crosslinking and room-temperature ionic liquid-assisted processing significantly reduce energy requirements. Additionally, additive manufacturing approaches, particularly 3D printing of AEMs, minimize material waste through precise deposition, though these technologies remain in early development stages for ion exchange membrane applications.
Recyclability and circular economy principles are increasingly incorporated into AEM design strategies. Researchers are developing membranes with reversible crosslinking mechanisms that allow for component recovery and reprocessing at end-of-life. Furthermore, modular membrane designs facilitate selective replacement of degraded components rather than entire membrane assemblies, extending functional lifetimes and reducing waste generation.
Water consumption during fabrication presents another environmental challenge, particularly in phase inversion processes. Closed-loop water recycling systems and solvent recovery technologies are being implemented to minimize freshwater requirements and prevent contamination of water bodies with processing chemicals. These approaches align with broader industrial sustainability goals while potentially reducing production costs through resource recovery.
Life cycle assessment (LCA) studies indicate that the environmental impact of AEMs extends beyond manufacturing to include raw material extraction, energy consumption during fabrication, and end-of-life disposal. Particularly concerning is the widespread use of fluorinated polymers in high-performance AEMs, which demonstrate exceptional durability but present significant environmental persistence issues. These materials can potentially degrade into perfluoroalkyl substances (PFAS), which are recognized as persistent environmental contaminants with bioaccumulative properties.
Emerging sustainable approaches include bio-based precursors for AEM fabrication, such as chitosan, cellulose derivatives, and lignin-based materials. These renewable resources not only reduce dependence on petroleum-derived polymers but also often exhibit inherent biodegradability. For instance, quaternized chitosan-based membranes have demonstrated promising ionic conductivity while offering improved environmental compatibility compared to conventional synthetic alternatives.
Energy efficiency in fabrication processes represents another critical sustainability factor. Traditional thermal crosslinking methods consume substantial energy, whereas emerging techniques like UV-initiated crosslinking and room-temperature ionic liquid-assisted processing significantly reduce energy requirements. Additionally, additive manufacturing approaches, particularly 3D printing of AEMs, minimize material waste through precise deposition, though these technologies remain in early development stages for ion exchange membrane applications.
Recyclability and circular economy principles are increasingly incorporated into AEM design strategies. Researchers are developing membranes with reversible crosslinking mechanisms that allow for component recovery and reprocessing at end-of-life. Furthermore, modular membrane designs facilitate selective replacement of degraded components rather than entire membrane assemblies, extending functional lifetimes and reducing waste generation.
Water consumption during fabrication presents another environmental challenge, particularly in phase inversion processes. Closed-loop water recycling systems and solvent recovery technologies are being implemented to minimize freshwater requirements and prevent contamination of water bodies with processing chemicals. These approaches align with broader industrial sustainability goals while potentially reducing production costs through resource recovery.
Scalability and Cost Analysis of Advanced AEM Production
The economic viability of advanced anion exchange membrane (AEM) production remains a critical factor in determining widespread adoption. Current manufacturing processes for durable AEMs face significant challenges when transitioning from laboratory to industrial scale. The cost structure analysis reveals that raw materials constitute approximately 40-60% of total production expenses, with specialized monomers and reinforcement materials being the primary cost drivers.
Production scaling follows distinct pathways depending on membrane architecture. Roll-to-roll processing shows promising economics for thin-film composite AEMs, potentially reducing per-unit costs by 65-70% when scaling from pilot to commercial volumes. Conversely, block copolymer membranes benefit more from batch scaling approaches, with cost reductions plateauing at approximately 45% regardless of further volume increases.
Energy consumption during fabrication represents another significant cost factor, particularly for membranes requiring precise thermal cycling or solvent removal steps. Advanced manufacturing techniques like radiation-induced grafting demonstrate up to 30% lower energy requirements compared to conventional chemical synthesis routes, though equipment capitalization costs remain substantially higher.
Material waste management constitutes a growing proportion of production expenses as environmental regulations tighten. Solvent recovery systems now represent an essential capital investment, adding 15-20% to initial setup costs but reducing operational expenses by 25-30% over a five-year period. Circular economy approaches, including monomer recycling from rejected membranes, show promising economic returns but remain technically challenging.
Labor costs vary significantly by production method, with highly automated processes reducing direct labor inputs by up to 80% compared to manual fabrication techniques. However, these automated systems require specialized maintenance and calibration personnel, partially offsetting direct labor savings with higher-skilled workforce requirements.
Quality control measures represent a non-negotiable cost center, typically accounting for 8-12% of production expenses. Advanced inline characterization techniques offer potential savings by reducing waste and rework, though implementation barriers remain high due to technical complexity and capital requirements.
The economic analysis indicates that achieving price parity with established technologies requires production volumes exceeding 50,000 m² annually. Current market demand supports approximately 15,000-20,000 m² production capacity, creating a challenging economic environment for manufacturers. Strategic partnerships with end-users in emerging applications present the most viable pathway to achieve necessary scale economies in the near term.
Production scaling follows distinct pathways depending on membrane architecture. Roll-to-roll processing shows promising economics for thin-film composite AEMs, potentially reducing per-unit costs by 65-70% when scaling from pilot to commercial volumes. Conversely, block copolymer membranes benefit more from batch scaling approaches, with cost reductions plateauing at approximately 45% regardless of further volume increases.
Energy consumption during fabrication represents another significant cost factor, particularly for membranes requiring precise thermal cycling or solvent removal steps. Advanced manufacturing techniques like radiation-induced grafting demonstrate up to 30% lower energy requirements compared to conventional chemical synthesis routes, though equipment capitalization costs remain substantially higher.
Material waste management constitutes a growing proportion of production expenses as environmental regulations tighten. Solvent recovery systems now represent an essential capital investment, adding 15-20% to initial setup costs but reducing operational expenses by 25-30% over a five-year period. Circular economy approaches, including monomer recycling from rejected membranes, show promising economic returns but remain technically challenging.
Labor costs vary significantly by production method, with highly automated processes reducing direct labor inputs by up to 80% compared to manual fabrication techniques. However, these automated systems require specialized maintenance and calibration personnel, partially offsetting direct labor savings with higher-skilled workforce requirements.
Quality control measures represent a non-negotiable cost center, typically accounting for 8-12% of production expenses. Advanced inline characterization techniques offer potential savings by reducing waste and rework, though implementation barriers remain high due to technical complexity and capital requirements.
The economic analysis indicates that achieving price parity with established technologies requires production volumes exceeding 50,000 m² annually. Current market demand supports approximately 15,000-20,000 m² production capacity, creating a challenging economic environment for manufacturers. Strategic partnerships with end-users in emerging applications present the most viable pathway to achieve necessary scale economies in the near term.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!