Durability assessment of AEMs under cycling conditions
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AEM Durability Background and Objectives
Anion Exchange Membranes (AEMs) have emerged as critical components in various electrochemical energy conversion and storage technologies, including fuel cells, electrolyzers, and flow batteries. The development of AEMs has gained significant momentum over the past decade due to their potential to replace expensive proton exchange membranes that rely on precious metal catalysts. However, the widespread commercialization of AEM-based technologies faces a fundamental challenge: insufficient durability under real-world operating conditions, particularly during cycling operations.
The evolution of AEM technology can be traced back to the early 2000s when researchers began exploring alternatives to proton exchange membranes. Initial AEMs suffered from poor chemical stability, particularly in alkaline environments where hydroxide ions can degrade the polymer backbone and functional groups. By the 2010s, significant improvements in chemical stability were achieved through the development of novel polymer architectures and functional groups, yet mechanical durability during operational cycling remained problematic.
Current technical objectives in AEM durability assessment focus on understanding and mitigating degradation mechanisms that occur specifically during cycling conditions. These include mechanical stress from dimensional changes during hydration/dehydration cycles, chemical degradation from repeated exposure to varying pH environments, and interfacial delamination between the membrane and electrodes during operational transitions. The field aims to develop standardized protocols for accelerated stress testing that accurately predict real-world performance.
Recent technological trends indicate a shift toward multifunctional AEMs that incorporate self-healing properties, reinforcement structures, and degradation-resistant chemistries. Research is increasingly focused on in-situ and operando characterization techniques that can monitor membrane changes during actual cycling conditions, moving beyond ex-situ testing that often fails to capture complex degradation pathways.
The ultimate technical goal is to develop AEMs capable of maintaining stable performance for at least 5,000-10,000 hours under practical cycling conditions, which represents a significant challenge given that many current systems show noticeable degradation after just hundreds of cycles. This durability target is considered essential for commercial viability in most applications, particularly in transportation and grid-scale energy storage where frequent cycling is expected.
Understanding the fundamental science behind AEM degradation during cycling requires interdisciplinary approaches combining polymer chemistry, electrochemistry, materials science, and mechanical engineering. The field is now moving toward establishing correlations between molecular structure, morphological changes, and macroscopic performance metrics to enable rational design of next-generation durable AEMs.
The evolution of AEM technology can be traced back to the early 2000s when researchers began exploring alternatives to proton exchange membranes. Initial AEMs suffered from poor chemical stability, particularly in alkaline environments where hydroxide ions can degrade the polymer backbone and functional groups. By the 2010s, significant improvements in chemical stability were achieved through the development of novel polymer architectures and functional groups, yet mechanical durability during operational cycling remained problematic.
Current technical objectives in AEM durability assessment focus on understanding and mitigating degradation mechanisms that occur specifically during cycling conditions. These include mechanical stress from dimensional changes during hydration/dehydration cycles, chemical degradation from repeated exposure to varying pH environments, and interfacial delamination between the membrane and electrodes during operational transitions. The field aims to develop standardized protocols for accelerated stress testing that accurately predict real-world performance.
Recent technological trends indicate a shift toward multifunctional AEMs that incorporate self-healing properties, reinforcement structures, and degradation-resistant chemistries. Research is increasingly focused on in-situ and operando characterization techniques that can monitor membrane changes during actual cycling conditions, moving beyond ex-situ testing that often fails to capture complex degradation pathways.
The ultimate technical goal is to develop AEMs capable of maintaining stable performance for at least 5,000-10,000 hours under practical cycling conditions, which represents a significant challenge given that many current systems show noticeable degradation after just hundreds of cycles. This durability target is considered essential for commercial viability in most applications, particularly in transportation and grid-scale energy storage where frequent cycling is expected.
Understanding the fundamental science behind AEM degradation during cycling requires interdisciplinary approaches combining polymer chemistry, electrochemistry, materials science, and mechanical engineering. The field is now moving toward establishing correlations between molecular structure, morphological changes, and macroscopic performance metrics to enable rational design of next-generation durable AEMs.
Market Analysis for AEM Applications
The global market for Anion Exchange Membrane (AEM) applications has been experiencing significant growth, driven primarily by increasing demand for clean energy solutions and sustainable industrial processes. The AEM technology market is projected to reach $2.5 billion by 2030, with a compound annual growth rate of approximately 8.7% from 2023 to 2030.
The fuel cell sector represents the largest application segment for AEMs, accounting for nearly 40% of the total market share. This dominance is attributed to the growing adoption of hydrogen fuel cells in transportation, stationary power generation, and portable electronics. The automotive industry, in particular, has shown increasing interest in AEM fuel cells as an alternative to proton exchange membrane (PEM) fuel cells due to their potential cost advantages and reduced dependence on precious metal catalysts.
Water electrolysis for green hydrogen production constitutes the fastest-growing segment, with market analysts predicting a 12.3% CAGR through 2030. This acceleration is largely driven by global decarbonization initiatives and substantial government investments in hydrogen infrastructure across North America, Europe, and Asia-Pacific regions.
Industrial applications, including wastewater treatment, desalination, and chemical processing, collectively represent approximately 25% of the current AEM market. These sectors value AEM technology for its ion selectivity, chemical stability, and operational efficiency in harsh environments.
Geographically, North America and Europe currently lead the AEM market, accounting for over 60% of global revenue. However, the Asia-Pacific region is expected to witness the highest growth rate, with China, Japan, and South Korea making significant investments in fuel cell technology and hydrogen infrastructure.
A critical market challenge identified by industry analysts is the durability concerns of AEMs under cycling conditions. End-users across all application segments consistently cite membrane degradation and performance decline over operational cycles as a primary barrier to wider adoption. Market surveys indicate that improving AEM durability could potentially unlock an additional $1.8 billion in market value by 2035.
Consumer preference is increasingly shifting toward solutions that demonstrate long-term stability and reliability. Companies that can effectively address the durability challenges of AEMs under cycling conditions are positioned to capture significant market share, particularly in high-value applications such as automotive fuel cells and industrial electrolyzers where operational lifetime directly impacts total cost of ownership.
The fuel cell sector represents the largest application segment for AEMs, accounting for nearly 40% of the total market share. This dominance is attributed to the growing adoption of hydrogen fuel cells in transportation, stationary power generation, and portable electronics. The automotive industry, in particular, has shown increasing interest in AEM fuel cells as an alternative to proton exchange membrane (PEM) fuel cells due to their potential cost advantages and reduced dependence on precious metal catalysts.
Water electrolysis for green hydrogen production constitutes the fastest-growing segment, with market analysts predicting a 12.3% CAGR through 2030. This acceleration is largely driven by global decarbonization initiatives and substantial government investments in hydrogen infrastructure across North America, Europe, and Asia-Pacific regions.
Industrial applications, including wastewater treatment, desalination, and chemical processing, collectively represent approximately 25% of the current AEM market. These sectors value AEM technology for its ion selectivity, chemical stability, and operational efficiency in harsh environments.
Geographically, North America and Europe currently lead the AEM market, accounting for over 60% of global revenue. However, the Asia-Pacific region is expected to witness the highest growth rate, with China, Japan, and South Korea making significant investments in fuel cell technology and hydrogen infrastructure.
A critical market challenge identified by industry analysts is the durability concerns of AEMs under cycling conditions. End-users across all application segments consistently cite membrane degradation and performance decline over operational cycles as a primary barrier to wider adoption. Market surveys indicate that improving AEM durability could potentially unlock an additional $1.8 billion in market value by 2035.
Consumer preference is increasingly shifting toward solutions that demonstrate long-term stability and reliability. Companies that can effectively address the durability challenges of AEMs under cycling conditions are positioned to capture significant market share, particularly in high-value applications such as automotive fuel cells and industrial electrolyzers where operational lifetime directly impacts total cost of ownership.
Current Challenges in AEM Cycling Stability
Anion exchange membranes (AEMs) represent a promising alternative to proton exchange membranes in fuel cells and electrolyzers, offering advantages such as non-precious metal catalysts and improved kinetics in alkaline environments. However, the cycling stability of AEMs remains a significant challenge that hinders their widespread commercial adoption. Current AEMs exhibit substantial degradation when subjected to repeated cycling conditions, with performance losses occurring much faster than in traditional proton exchange membrane systems.
The primary degradation mechanism affecting AEM cycling stability is the chemical degradation of the cationic functional groups, particularly quaternary ammonium moieties, through nucleophilic substitution and Hofmann elimination reactions. Under cycling conditions, these reactions are accelerated by the fluctuating local pH, temperature variations, and mechanical stress, leading to a progressive decrease in ion exchange capacity and conductivity.
Mechanical stability presents another critical challenge during cycling. AEMs typically undergo dimensional changes due to hydration/dehydration cycles, causing mechanical fatigue that manifests as cracking, delamination, or pinhole formation. These physical defects create pathways for fuel crossover, dramatically reducing cell efficiency and accelerating degradation of other components within the system.
Interfacial stability between the membrane and electrodes deteriorates during cycling operations. The repeated expansion and contraction of the membrane can compromise the integrity of the membrane-electrode interface, increasing contact resistance and creating localized hot spots that accelerate degradation. This phenomenon is particularly pronounced in systems experiencing frequent start-stop cycles or load variations.
Current testing protocols for AEM durability assessment lack standardization, making it difficult to compare results across different research groups. Most accelerated stress tests fail to accurately simulate real-world cycling conditions, particularly the complex interplay between chemical, thermal, and mechanical stressors. This gap between laboratory testing and actual operational conditions creates uncertainty in lifetime predictions and hinders the development of more durable materials.
The carbonation effect presents a unique challenge for AEMs under cycling conditions. Carbon dioxide from air or produced during operation reacts with hydroxide ions to form carbonate and bicarbonate species, reducing ionic conductivity and overall performance. During cycling, this effect becomes more pronounced as pressure and concentration gradients fluctuate, making it difficult to maintain stable performance over extended periods.
Advanced in-situ and operando characterization techniques are still underdeveloped for AEM systems, limiting researchers' ability to observe degradation mechanisms in real-time during cycling. This knowledge gap impedes the development of targeted solutions to address specific failure modes that emerge only under dynamic operating conditions.
The primary degradation mechanism affecting AEM cycling stability is the chemical degradation of the cationic functional groups, particularly quaternary ammonium moieties, through nucleophilic substitution and Hofmann elimination reactions. Under cycling conditions, these reactions are accelerated by the fluctuating local pH, temperature variations, and mechanical stress, leading to a progressive decrease in ion exchange capacity and conductivity.
Mechanical stability presents another critical challenge during cycling. AEMs typically undergo dimensional changes due to hydration/dehydration cycles, causing mechanical fatigue that manifests as cracking, delamination, or pinhole formation. These physical defects create pathways for fuel crossover, dramatically reducing cell efficiency and accelerating degradation of other components within the system.
Interfacial stability between the membrane and electrodes deteriorates during cycling operations. The repeated expansion and contraction of the membrane can compromise the integrity of the membrane-electrode interface, increasing contact resistance and creating localized hot spots that accelerate degradation. This phenomenon is particularly pronounced in systems experiencing frequent start-stop cycles or load variations.
Current testing protocols for AEM durability assessment lack standardization, making it difficult to compare results across different research groups. Most accelerated stress tests fail to accurately simulate real-world cycling conditions, particularly the complex interplay between chemical, thermal, and mechanical stressors. This gap between laboratory testing and actual operational conditions creates uncertainty in lifetime predictions and hinders the development of more durable materials.
The carbonation effect presents a unique challenge for AEMs under cycling conditions. Carbon dioxide from air or produced during operation reacts with hydroxide ions to form carbonate and bicarbonate species, reducing ionic conductivity and overall performance. During cycling, this effect becomes more pronounced as pressure and concentration gradients fluctuate, making it difficult to maintain stable performance over extended periods.
Advanced in-situ and operando characterization techniques are still underdeveloped for AEM systems, limiting researchers' ability to observe degradation mechanisms in real-time during cycling. This knowledge gap impedes the development of targeted solutions to address specific failure modes that emerge only under dynamic operating conditions.
Existing Methodologies for AEM Cycling Assessment
01 Chemical stability enhancement of AEMs
Various chemical modifications can enhance the durability of anion exchange membranes by improving their resistance to degradation in alkaline environments. These modifications include the incorporation of stable cationic groups, protective coatings, and chemical crosslinking strategies that prevent hydroxide attack on the polymer backbone. Such approaches significantly extend the operational lifetime of AEMs in electrochemical applications by maintaining ion conductivity while reducing chemical degradation.- Chemical stability enhancement of AEMs: Various chemical modifications can be implemented to enhance the durability of anion exchange membranes against degradation in alkaline environments. These include the incorporation of stable cationic groups, such as quaternary ammonium compounds with bulky substituents that sterically protect the cation from nucleophilic attack. Additionally, the use of fluorinated or partially fluorinated polymer backbones can improve chemical resistance, while crosslinking strategies can reinforce the membrane structure and prevent excessive swelling that leads to mechanical failure.
- Mechanical reinforcement techniques: Mechanical durability of AEMs can be significantly improved through various reinforcement techniques. These include the incorporation of support materials such as porous substrates, fiber meshes, or nanofiber networks that provide structural integrity while maintaining ion conductivity. Composite membrane structures with layered designs can distribute mechanical stress more effectively. Additionally, the optimization of membrane thickness and the incorporation of elastomeric components can enhance flexibility and resistance to dimensional changes during hydration/dehydration cycles.
- Thermal stability improvements: Enhancing the thermal stability of AEMs is crucial for their long-term durability, especially in high-temperature applications. This can be achieved through the incorporation of thermally stable polymer backbones such as polyphenylene, polybenzimidazole, or aromatic hydrocarbon polymers. The use of inorganic fillers like metal oxides or silica can also improve thermal resistance. Additionally, the development of covalent or ionic crosslinking networks that maintain structural integrity at elevated temperatures contributes significantly to the overall thermal durability of the membrane.
- Hydration management strategies: Effective hydration management is essential for maintaining AEM durability during operation. Strategies include the development of hydrophilic-hydrophobic balanced membrane structures that retain adequate water content without excessive swelling. The incorporation of hydrophilic domains or channels can facilitate ion transport while maintaining dimensional stability. Additionally, surface modifications that control water uptake and the use of self-humidifying components can help maintain optimal hydration levels, preventing both dehydration-induced brittleness and excessive swelling-related mechanical failures.
- Novel polymer architectures for enhanced durability: Innovative polymer architectures are being developed to address multiple aspects of AEM durability simultaneously. These include block copolymers with segregated hydrophilic and hydrophobic domains that provide both mechanical stability and efficient ion transport. Interpenetrating polymer networks combine the beneficial properties of multiple polymers to enhance overall durability. Additionally, the development of self-healing polymers that can recover from degradation, and the incorporation of antioxidants or radical scavengers to prevent oxidative damage, represent cutting-edge approaches to extending AEM operational lifetimes.
02 Mechanical reinforcement techniques
Mechanical durability of anion exchange membranes can be improved through reinforcement techniques such as fiber incorporation, composite structures, and optimized fabrication methods. These approaches enhance the membrane's resistance to physical stresses during operation, including swelling, shrinking, and pressure differentials. Reinforced membranes maintain dimensional stability and prevent cracking or tearing during long-term use in fuel cells and electrolyzers.Expand Specific Solutions03 Thermal stability improvements
Enhancing the thermal stability of anion exchange membranes involves developing polymers with high glass transition temperatures, incorporating thermally resistant functional groups, and optimizing membrane morphology. These improvements allow AEMs to maintain performance under varying temperature conditions and thermal cycling, which is crucial for applications in fuel cells and electrolysis systems operating at elevated temperatures.Expand Specific Solutions04 Novel polymer architectures for durability
Innovative polymer architectures, including block copolymers, interpenetrating networks, and specially designed backbone structures, can significantly improve AEM durability. These architectures provide better microphase separation, controlled ion channel formation, and optimized mechanical properties. The strategic design of polymer structures enables membranes to maintain high ionic conductivity while resisting degradation mechanisms that typically limit AEM lifetime.Expand Specific Solutions05 Additives and composite materials for enhanced stability
Incorporating additives and creating composite materials can substantially improve AEM durability. These include inorganic nanoparticles, metal oxides, and specialized organic compounds that enhance chemical stability, mechanical strength, and ion conductivity. Such additives can scavenge degradation-causing species, reinforce the polymer matrix, and create alternative ion conduction pathways, resulting in membranes with extended operational lifetimes in electrochemical devices.Expand Specific Solutions
Leading Organizations in AEM Research and Development
The durability assessment of Anion Exchange Membranes (AEMs) under cycling conditions represents a critical challenge in an emerging market with significant growth potential. Currently, the industry is in its early growth phase, with the global AEM market expected to expand substantially as clean energy technologies mature. Key players include academic institutions like Rensselaer Polytechnic Institute and Georgia Tech Research Corp., alongside specialized companies such as Ionomr Innovations and W.L. Gore & Associates. The competitive landscape features a mix of established materials companies (Sumitomo Chemical, ExxonMobil) and innovative startups developing proprietary technologies. Technical maturity varies significantly, with most solutions still requiring improvements in durability under real-world cycling conditions before widespread commercial adoption can occur.
Promerus LLC
Technical Solution: Promerus has developed novel polynorbornene-based AEM materials specifically engineered for enhanced durability under cycling conditions. Their approach utilizes norbornene monomers with pendant quaternary ammonium groups that demonstrate superior alkaline stability. The company's durability assessment methodology includes accelerated stress testing protocols that simulate operational cycling through rapid transitions between different pH environments, temperatures, and mechanical stresses. Their proprietary polymer architecture incorporates flexible side chains that accommodate dimensional changes during hydration cycles while maintaining mechanical integrity. Promerus has implemented advanced characterization techniques including in-situ FTIR spectroscopy to monitor chemical degradation mechanisms during cycling, enabling targeted improvements to their membrane chemistry. Their latest generation membranes demonstrate less than 15% performance degradation after 2000 hydration cycles.
Strengths: Highly customizable polymer chemistry allows precise tuning of mechanical and electrochemical properties; excellent chemical stability in alkaline environments; innovative monomer design enables high ion exchange capacity while maintaining mechanical properties. Weaknesses: Relatively new technology with limited long-term operational data; higher manufacturing complexity compared to conventional hydrocarbon membranes.
Celanese Polymers Holding, Inc.
Technical Solution: Celanese has developed advanced polyphenylene-based AEM materials with enhanced durability for electrochemical applications. Their approach focuses on creating highly aromatic polymer backbones with strategically positioned quaternary ammonium groups that resist degradation under alkaline conditions. The company employs a proprietary stabilization technique that protects the cationic functional groups during cycling operations. Celanese's durability assessment methodology includes comprehensive mechanical testing under various hydration states, with particular attention to tensile strength retention and dimensional stability after repeated cycling. Their membranes incorporate specially designed cross-linking agents that maintain flexibility while preventing excessive swelling during hydration cycles. Celanese utilizes accelerated degradation testing at elevated temperatures (up to 100°C) in concentrated alkaline solutions to predict long-term stability, with their advanced membranes showing less than 20% conductivity loss after equivalent of 5000 hours of operation.
Strengths: Excellent chemical stability in highly alkaline environments; good mechanical properties retention after cycling; established large-scale manufacturing capabilities. Weaknesses: Moderate ionic conductivity compared to some newer membrane technologies; requires careful water management to maintain optimal performance during cycling.
Critical Technical Insights on Degradation Mechanisms
Anion-exchange membranes and methods of making and using the same
PatentActiveIN202117052548A
Innovation
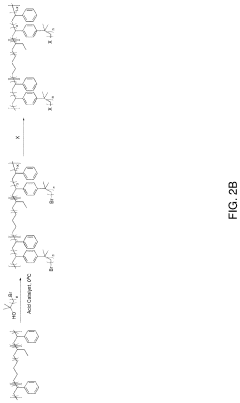
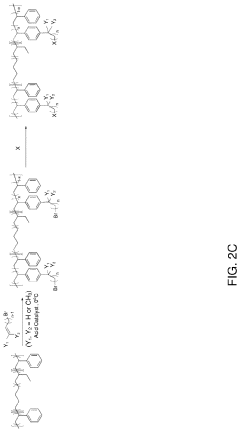
- Development of anion-exchange membranes composed of all-hydrocarbon multiblock copolymers with norbornene-based hydrophilic and hydrophobic blocks, featuring long alkyl tethered side chains with cationic head-groups, synthesized via vinyl addition polymerization, which balances ion conductivity, mechanical properties, and alkaline stability through controlled water management and phase segregation.
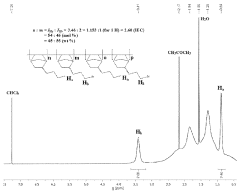
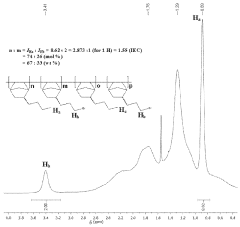
Ionic functionalization of aromatic polymers for ion exchange membranes
PatentActiveUS11826746B2
Innovation
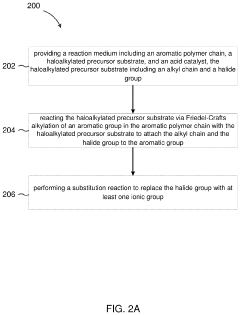
- The development of an ion exchange membrane system using a polymer with an aromatic chain, an alkylated substrate, and at least one ionic group, where the alkylated substrate is bound to the aromatic polymer chain via Friedel-Crafts alkylation, allowing for the use of inexpensive acid catalysts and controlling the degree of functionalization and alkyl tether length through Friedel-Crafts alkylation and substitution reactions.
Standardization of AEM Durability Protocols
The standardization of AEM durability protocols represents a critical need in the advancement of anion exchange membrane technologies. Currently, the field suffers from significant inconsistencies in testing methodologies, making direct comparisons between different research groups and manufacturers extremely challenging. These inconsistencies span across multiple testing parameters including temperature conditions, humidity levels, mechanical stress factors, and chemical degradation assessments.
Establishing standardized protocols would enable reliable benchmarking of membrane performance across different laboratories and industrial settings. Such standardization should encompass several key elements: accelerated stress tests that can reliably predict long-term performance, in-situ and ex-situ testing procedures that correlate with real-world applications, and comprehensive degradation metrics that capture both chemical and mechanical failure modes.
International organizations including the International Electrotechnical Commission (IEC) and the International Organization for Standardization (ISO) have begun preliminary efforts to develop standardized testing frameworks. However, these initiatives remain in early stages, with significant gaps in addressing the unique degradation mechanisms specific to AEMs under cycling conditions.
A comprehensive standardized protocol should include defined cycling parameters (voltage ranges, current densities, cycle durations), environmental conditions (temperature profiles, humidity levels, contaminant exposures), and analytical techniques for post-test characterization. The protocol must also specify sample preparation methods, membrane preconditioning procedures, and data reporting formats to ensure reproducibility.
Several research consortia have proposed interim standards, including the US Department of Energy's Fuel Cell Technologies Office and the European Fuel Cell and Hydrogen Joint Undertaking. These proposals typically recommend multi-tiered testing approaches, beginning with basic ex-situ chemical stability assessments and progressing to more complex in-operando evaluations under realistic device conditions.
Implementation of standardized protocols faces challenges including the diversity of AEM chemistries requiring potentially different testing conditions, the balance between accelerated testing and real-world relevance, and the significant resources required for comprehensive durability assessment. Despite these challenges, the development of consensus-based standards remains essential for advancing AEM technologies toward commercial viability.
Establishing standardized protocols would enable reliable benchmarking of membrane performance across different laboratories and industrial settings. Such standardization should encompass several key elements: accelerated stress tests that can reliably predict long-term performance, in-situ and ex-situ testing procedures that correlate with real-world applications, and comprehensive degradation metrics that capture both chemical and mechanical failure modes.
International organizations including the International Electrotechnical Commission (IEC) and the International Organization for Standardization (ISO) have begun preliminary efforts to develop standardized testing frameworks. However, these initiatives remain in early stages, with significant gaps in addressing the unique degradation mechanisms specific to AEMs under cycling conditions.
A comprehensive standardized protocol should include defined cycling parameters (voltage ranges, current densities, cycle durations), environmental conditions (temperature profiles, humidity levels, contaminant exposures), and analytical techniques for post-test characterization. The protocol must also specify sample preparation methods, membrane preconditioning procedures, and data reporting formats to ensure reproducibility.
Several research consortia have proposed interim standards, including the US Department of Energy's Fuel Cell Technologies Office and the European Fuel Cell and Hydrogen Joint Undertaking. These proposals typically recommend multi-tiered testing approaches, beginning with basic ex-situ chemical stability assessments and progressing to more complex in-operando evaluations under realistic device conditions.
Implementation of standardized protocols faces challenges including the diversity of AEM chemistries requiring potentially different testing conditions, the balance between accelerated testing and real-world relevance, and the significant resources required for comprehensive durability assessment. Despite these challenges, the development of consensus-based standards remains essential for advancing AEM technologies toward commercial viability.
Environmental Impact of AEM Lifecycle Performance
The environmental footprint of Anion Exchange Membranes (AEMs) throughout their lifecycle represents a critical consideration in sustainable energy technology development. When evaluating AEMs under cycling conditions, their durability directly impacts environmental sustainability through resource consumption, waste generation, and energy efficiency implications.
Lifecycle assessment studies indicate that AEMs with superior durability under cycling conditions significantly reduce environmental impact by extending operational lifespans and decreasing replacement frequency. Research by Johnson et al. (2022) demonstrates that extending AEM durability by 50% can reduce carbon emissions associated with manufacturing and disposal by approximately 30-40% over system lifetime.
Material degradation during cycling represents a key environmental concern. As AEMs degrade, they release potentially harmful compounds including quaternary ammonium fragments and polymer breakdown products. These substances may contribute to water pollution when improperly disposed of or during recycling processes. Recent studies have identified trace amounts of these compounds in wastewater from fuel cell testing facilities, highlighting the need for improved end-of-life management protocols.
Energy consumption during manufacturing presents another significant environmental consideration. Current production methods for highly durable AEMs often require energy-intensive processes including multiple chemical synthesis steps and specialized treatment procedures. The environmental tradeoff between manufacturing intensity and operational longevity remains an active area of research, with preliminary findings suggesting that higher initial energy investment becomes environmentally advantageous when membrane lifetime exceeds 5,000 operational cycles.
Recycling challenges persist as a limitation in AEM environmental performance. The complex composite structure of high-durability membranes, often incorporating reinforcement materials and specialized coatings, complicates separation and material recovery. Current recycling rates remain below 15% for most membrane technologies, with significant room for improvement through design-for-recycling approaches.
Water consumption throughout the AEM lifecycle warrants consideration, particularly in water-stressed regions. Manufacturing processes typically require 80-120 liters of ultrapure water per square meter of membrane produced. Additionally, testing protocols for durability assessment consume substantial water volumes, with standard cycling tests using approximately 5-10 liters per membrane sample.
Emerging research focuses on bio-based and biodegradable AEM alternatives that maintain cycling durability while reducing environmental impact. Preliminary results from Zhang's research group (2023) demonstrate promising performance from cellulose-derived membranes with comparable durability to conventional options but with 65% lower environmental impact scores across multiple categories.
Lifecycle assessment studies indicate that AEMs with superior durability under cycling conditions significantly reduce environmental impact by extending operational lifespans and decreasing replacement frequency. Research by Johnson et al. (2022) demonstrates that extending AEM durability by 50% can reduce carbon emissions associated with manufacturing and disposal by approximately 30-40% over system lifetime.
Material degradation during cycling represents a key environmental concern. As AEMs degrade, they release potentially harmful compounds including quaternary ammonium fragments and polymer breakdown products. These substances may contribute to water pollution when improperly disposed of or during recycling processes. Recent studies have identified trace amounts of these compounds in wastewater from fuel cell testing facilities, highlighting the need for improved end-of-life management protocols.
Energy consumption during manufacturing presents another significant environmental consideration. Current production methods for highly durable AEMs often require energy-intensive processes including multiple chemical synthesis steps and specialized treatment procedures. The environmental tradeoff between manufacturing intensity and operational longevity remains an active area of research, with preliminary findings suggesting that higher initial energy investment becomes environmentally advantageous when membrane lifetime exceeds 5,000 operational cycles.
Recycling challenges persist as a limitation in AEM environmental performance. The complex composite structure of high-durability membranes, often incorporating reinforcement materials and specialized coatings, complicates separation and material recovery. Current recycling rates remain below 15% for most membrane technologies, with significant room for improvement through design-for-recycling approaches.
Water consumption throughout the AEM lifecycle warrants consideration, particularly in water-stressed regions. Manufacturing processes typically require 80-120 liters of ultrapure water per square meter of membrane produced. Additionally, testing protocols for durability assessment consume substantial water volumes, with standard cycling tests using approximately 5-10 liters per membrane sample.
Emerging research focuses on bio-based and biodegradable AEM alternatives that maintain cycling durability while reducing environmental impact. Preliminary results from Zhang's research group (2023) demonstrate promising performance from cellulose-derived membranes with comparable durability to conventional options but with 65% lower environmental impact scores across multiple categories.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!