High conductivity polymer backbones for AEM development
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
AEM Polymer Backbone Evolution and Research Objectives
Anion Exchange Membrane (AEM) technology has evolved significantly over the past decades, transitioning from rudimentary polymer structures to sophisticated engineered backbones designed specifically for enhanced ionic conductivity. The historical development of AEM polymer backbones can be traced back to the 1960s with the introduction of basic quaternary ammonium functionalized polystyrene. These early materials suffered from poor chemical stability and limited conductivity, restricting their practical applications in energy conversion devices.
The 1990s marked a pivotal shift with the emergence of poly(arylene ether)s and poly(phenylene oxide) derivatives as potential AEM backbones. These materials offered improved mechanical properties and chemical resistance compared to their predecessors. However, the hydroxide conductivity remained insufficient for commercial viability in fuel cell applications, typically below 10 mS/cm under ambient conditions.
A significant breakthrough occurred in the early 2000s with the development of poly(arylene ether sulfone) backbones, which demonstrated enhanced alkaline stability and mechanical durability. This advancement was followed by the introduction of polyphenylene-based systems in the 2010s, which further improved the balance between mechanical strength and ion transport capabilities.
Current research objectives in AEM polymer backbone development focus on several critical aspects. Primarily, achieving hydroxide conductivity exceeding 100 mS/cm under operating conditions while maintaining mechanical integrity remains the foremost challenge. This target represents approximately a tenfold improvement over many contemporary systems and would position AEM technology as a competitive alternative to proton exchange membrane (PEM) systems.
Another key objective involves enhancing alkaline stability, particularly at elevated temperatures (80-120°C) and high pH environments. Current generation AEM backbones typically degrade through various mechanisms including Hofmann elimination and nucleophilic substitution, significantly limiting operational lifetimes to hundreds of hours rather than the thousands required for commercial viability.
The development of scalable and cost-effective synthesis routes for high-performance polymer backbones constitutes another critical research goal. Many promising laboratory-scale polymers utilize complex multi-step syntheses with expensive reagents, creating barriers to industrial adoption and commercialization.
Additionally, researchers aim to establish clear structure-property relationships that correlate polymer backbone architecture with ion transport mechanisms. Understanding these fundamental relationships would enable rational design approaches rather than empirical optimization, potentially accelerating the discovery of next-generation materials with unprecedented performance characteristics.
The 1990s marked a pivotal shift with the emergence of poly(arylene ether)s and poly(phenylene oxide) derivatives as potential AEM backbones. These materials offered improved mechanical properties and chemical resistance compared to their predecessors. However, the hydroxide conductivity remained insufficient for commercial viability in fuel cell applications, typically below 10 mS/cm under ambient conditions.
A significant breakthrough occurred in the early 2000s with the development of poly(arylene ether sulfone) backbones, which demonstrated enhanced alkaline stability and mechanical durability. This advancement was followed by the introduction of polyphenylene-based systems in the 2010s, which further improved the balance between mechanical strength and ion transport capabilities.
Current research objectives in AEM polymer backbone development focus on several critical aspects. Primarily, achieving hydroxide conductivity exceeding 100 mS/cm under operating conditions while maintaining mechanical integrity remains the foremost challenge. This target represents approximately a tenfold improvement over many contemporary systems and would position AEM technology as a competitive alternative to proton exchange membrane (PEM) systems.
Another key objective involves enhancing alkaline stability, particularly at elevated temperatures (80-120°C) and high pH environments. Current generation AEM backbones typically degrade through various mechanisms including Hofmann elimination and nucleophilic substitution, significantly limiting operational lifetimes to hundreds of hours rather than the thousands required for commercial viability.
The development of scalable and cost-effective synthesis routes for high-performance polymer backbones constitutes another critical research goal. Many promising laboratory-scale polymers utilize complex multi-step syntheses with expensive reagents, creating barriers to industrial adoption and commercialization.
Additionally, researchers aim to establish clear structure-property relationships that correlate polymer backbone architecture with ion transport mechanisms. Understanding these fundamental relationships would enable rational design approaches rather than empirical optimization, potentially accelerating the discovery of next-generation materials with unprecedented performance characteristics.
Market Analysis for High Conductivity AEM Applications
The global market for Anion Exchange Membranes (AEMs) with high conductivity polymer backbones is experiencing robust growth, driven primarily by increasing demand for clean energy technologies. The AEM market is projected to reach $1.2 billion by 2027, growing at a CAGR of 6.8% from 2022 to 2027, with high conductivity variants representing the fastest-growing segment.
Fuel cell applications currently dominate the market demand for high conductivity AEMs, accounting for approximately 45% of total consumption. The automotive sector, particularly for hydrogen fuel cell vehicles, represents a significant growth driver as major manufacturers like Toyota, Hyundai, and Honda expand their fuel cell vehicle production. The stationary power generation sector follows closely, comprising about 30% of the market, with increasing adoption for distributed energy systems and backup power solutions.
Water electrolysis for green hydrogen production has emerged as the most promising growth segment, with demand expected to triple by 2030. This surge is directly linked to global decarbonization initiatives and substantial government investments in hydrogen infrastructure across North America, Europe, and East Asia. The European Union's hydrogen strategy alone aims to install at least 40GW of renewable hydrogen electrolyzers by 2030, creating substantial demand for advanced AEM technologies.
Regional analysis indicates that North America currently leads the market with a 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the Asia-Pacific region is projected to witness the highest growth rate, driven by China's aggressive push toward hydrogen technologies and Japan's established fuel cell industry. South Korea's hydrogen economy roadmap further strengthens this regional momentum.
Price sensitivity remains a critical factor in market adoption, with current high-performance AEMs costing between $200-500 per square meter. Industry analysts suggest that achieving price points below $100 per square meter while maintaining high conductivity (>100 mS/cm) would trigger widespread commercial adoption across multiple sectors.
End-user requirements vary significantly across applications. The automotive sector prioritizes durability (>10,000 operating hours) and temperature stability (-30°C to 120°C), while stationary power applications emphasize long-term stability (>40,000 hours) and cost-effectiveness. The water electrolysis segment demands exceptional chemical stability in highly alkaline environments and conductivity maintenance at high current densities.
Regulatory frameworks are increasingly favorable for AEM technologies, with carbon reduction policies in over 70 countries indirectly supporting their adoption. The EU's Carbon Border Adjustment Mechanism and similar policies emerging in other regions are expected to accelerate investment in clean energy technologies that utilize high-performance AEMs.
Fuel cell applications currently dominate the market demand for high conductivity AEMs, accounting for approximately 45% of total consumption. The automotive sector, particularly for hydrogen fuel cell vehicles, represents a significant growth driver as major manufacturers like Toyota, Hyundai, and Honda expand their fuel cell vehicle production. The stationary power generation sector follows closely, comprising about 30% of the market, with increasing adoption for distributed energy systems and backup power solutions.
Water electrolysis for green hydrogen production has emerged as the most promising growth segment, with demand expected to triple by 2030. This surge is directly linked to global decarbonization initiatives and substantial government investments in hydrogen infrastructure across North America, Europe, and East Asia. The European Union's hydrogen strategy alone aims to install at least 40GW of renewable hydrogen electrolyzers by 2030, creating substantial demand for advanced AEM technologies.
Regional analysis indicates that North America currently leads the market with a 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the Asia-Pacific region is projected to witness the highest growth rate, driven by China's aggressive push toward hydrogen technologies and Japan's established fuel cell industry. South Korea's hydrogen economy roadmap further strengthens this regional momentum.
Price sensitivity remains a critical factor in market adoption, with current high-performance AEMs costing between $200-500 per square meter. Industry analysts suggest that achieving price points below $100 per square meter while maintaining high conductivity (>100 mS/cm) would trigger widespread commercial adoption across multiple sectors.
End-user requirements vary significantly across applications. The automotive sector prioritizes durability (>10,000 operating hours) and temperature stability (-30°C to 120°C), while stationary power applications emphasize long-term stability (>40,000 hours) and cost-effectiveness. The water electrolysis segment demands exceptional chemical stability in highly alkaline environments and conductivity maintenance at high current densities.
Regulatory frameworks are increasingly favorable for AEM technologies, with carbon reduction policies in over 70 countries indirectly supporting their adoption. The EU's Carbon Border Adjustment Mechanism and similar policies emerging in other regions are expected to accelerate investment in clean energy technologies that utilize high-performance AEMs.
Current Limitations in Polymer Backbone Conductivity
Despite significant advancements in anion exchange membrane (AEM) technology, polymer backbone conductivity remains a critical bottleneck limiting overall performance. Current polymer backbones used in AEMs typically achieve hydroxide ion conductivities between 10-100 mS/cm under optimal conditions, which falls significantly short of the 200+ mS/cm achieved by proton exchange membranes. This conductivity gap represents one of the most significant barriers to widespread AEM adoption in fuel cells and electrolyzers.
The inherent molecular structure of conventional polymer backbones presents fundamental limitations. Most existing systems rely on quaternary ammonium functional groups attached to hydrocarbon or partially fluorinated backbones, which create suboptimal ion channels. These structures often result in tortuous pathways for ion transport, increasing the effective distance ions must travel and reducing overall conductivity.
Water management presents another significant challenge. Unlike proton transport mechanisms that benefit from the Grotthuss hopping mechanism, hydroxide ion transport requires more substantial water content for effective conduction. Current polymer backbones struggle to maintain adequate hydration levels while preventing excessive swelling that compromises mechanical stability. The hydrophobic-hydrophilic balance remains difficult to optimize with existing materials.
Chemical stability under alkaline conditions continues to plague polymer backbone development. The high pH environment necessary for hydroxide conduction accelerates degradation through various mechanisms including Hofmann elimination, nucleophilic substitution, and oxidative attacks. Most current backbones exhibit significant conductivity loss after just 500-1000 hours of operation, far below the 5000+ hours required for commercial viability.
Temperature sensitivity further limits practical applications. Many promising polymer backbones demonstrate acceptable conductivity only within narrow temperature ranges, typically 60-80°C. Performance drops precipitously at lower temperatures due to reduced ion mobility, while higher temperatures accelerate chemical degradation and cause excessive membrane dehydration.
Manufacturing scalability presents additional challenges. Complex synthesis routes for high-performance polymer backbones often involve multiple reaction steps with expensive catalysts and stringent reaction conditions. Current production methods typically yield small quantities of material with batch-to-batch variability, making commercial-scale production economically unfeasible.
The trade-off between mechanical properties and ion conductivity remains unresolved. Increasing the ionic group concentration improves conductivity but typically weakens mechanical integrity through excessive swelling. Conversely, reinforcing the backbone structure to enhance mechanical stability often creates barriers to ion transport, reducing overall conductivity.
The inherent molecular structure of conventional polymer backbones presents fundamental limitations. Most existing systems rely on quaternary ammonium functional groups attached to hydrocarbon or partially fluorinated backbones, which create suboptimal ion channels. These structures often result in tortuous pathways for ion transport, increasing the effective distance ions must travel and reducing overall conductivity.
Water management presents another significant challenge. Unlike proton transport mechanisms that benefit from the Grotthuss hopping mechanism, hydroxide ion transport requires more substantial water content for effective conduction. Current polymer backbones struggle to maintain adequate hydration levels while preventing excessive swelling that compromises mechanical stability. The hydrophobic-hydrophilic balance remains difficult to optimize with existing materials.
Chemical stability under alkaline conditions continues to plague polymer backbone development. The high pH environment necessary for hydroxide conduction accelerates degradation through various mechanisms including Hofmann elimination, nucleophilic substitution, and oxidative attacks. Most current backbones exhibit significant conductivity loss after just 500-1000 hours of operation, far below the 5000+ hours required for commercial viability.
Temperature sensitivity further limits practical applications. Many promising polymer backbones demonstrate acceptable conductivity only within narrow temperature ranges, typically 60-80°C. Performance drops precipitously at lower temperatures due to reduced ion mobility, while higher temperatures accelerate chemical degradation and cause excessive membrane dehydration.
Manufacturing scalability presents additional challenges. Complex synthesis routes for high-performance polymer backbones often involve multiple reaction steps with expensive catalysts and stringent reaction conditions. Current production methods typically yield small quantities of material with batch-to-batch variability, making commercial-scale production economically unfeasible.
The trade-off between mechanical properties and ion conductivity remains unresolved. Increasing the ionic group concentration improves conductivity but typically weakens mechanical integrity through excessive swelling. Conversely, reinforcing the backbone structure to enhance mechanical stability often creates barriers to ion transport, reducing overall conductivity.
State-of-the-Art Polymer Backbone Architectures
01 Conjugated polymer backbones for enhanced conductivity
Conjugated polymers with alternating single and double bonds in their backbone structure provide pathways for electron movement, resulting in high electrical conductivity. These polymers, such as polyacetylene, polythiophene, and polyaniline, have delocalized π-electrons that can move along the polymer chain. The conductivity can be further enhanced through doping processes that introduce charge carriers into the polymer system.- Conjugated polymer backbones for enhanced conductivity: Conjugated polymers with extended π-electron systems in their backbones exhibit high electrical conductivity. These polymers feature alternating single and double bonds that allow for electron delocalization throughout the structure. The conjugated backbone provides a pathway for charge carriers, resulting in improved conductivity. Modifications to the conjugated structure, such as incorporating heteroatoms or functional groups, can further enhance the conductivity properties.
- Doping strategies for polymer conductivity enhancement: Doping is a critical process for increasing the conductivity of polymer backbones. By introducing dopants such as iodine, acids, or metal salts into the polymer matrix, the charge carrier concentration can be significantly increased. The dopants create additional charge carriers by oxidizing or reducing the polymer chains, leading to enhanced electrical conductivity. Different doping methods, including chemical, electrochemical, and photochemical approaches, can be employed to achieve optimal conductivity levels.
- Nanocomposite polymer systems for high conductivity: Incorporating nanomaterials such as carbon nanotubes, graphene, or metal nanoparticles into polymer matrices creates nanocomposite systems with enhanced electrical conductivity. These nanomaterials form conductive networks within the polymer backbone, providing additional pathways for charge transport. The synergistic effect between the polymer backbone and the nanomaterials results in conductivity values that exceed those of the individual components. The interface between the nanomaterials and the polymer matrix plays a crucial role in determining the overall conductivity of the composite.
- Self-assembled conductive polymer structures: Self-assembly techniques can be used to create ordered polymer structures with enhanced conductivity. By designing polymer backbones with specific functional groups that promote self-assembly through non-covalent interactions such as hydrogen bonding, π-π stacking, or ionic interactions, highly ordered structures can be formed. These ordered arrangements facilitate charge transport along and between polymer chains, resulting in improved conductivity. Control over the self-assembly process allows for the creation of various morphologies with tailored conductive properties.
- Ionic conductivity in polymer electrolyte systems: Polymer backbones can be designed to facilitate ionic conductivity, which is particularly important for applications in batteries and fuel cells. By incorporating polar functional groups or ionic moieties into the polymer backbone, the movement of ions through the polymer matrix is enhanced. The flexibility of the polymer chains, the presence of amorphous regions, and the interaction between the polymer backbone and the mobile ions all contribute to the overall ionic conductivity. Strategies such as cross-linking, plasticization, and the addition of ionic liquids can further improve the ionic conductivity of these polymer systems.
02 Doping techniques for polymer conductivity enhancement
Various doping methods can significantly increase the conductivity of polymer backbones. Chemical doping involves the addition of electron donors or acceptors that create charge carriers within the polymer structure. Electrochemical doping applies an electrical potential to introduce ions into the polymer matrix. These techniques can transform insulating polymers into semiconducting or even metallic-like conducting materials, with conductivity improvements of several orders of magnitude.Expand Specific Solutions03 Composite polymer systems with conductive fillers
Incorporating conductive fillers such as carbon nanotubes, graphene, or metal nanoparticles into polymer matrices creates composite materials with enhanced electrical conductivity. These fillers form conductive networks within the polymer backbone, providing pathways for electron transport. The conductivity of these composites depends on the filler concentration, dispersion quality, and interfacial interactions between the filler and polymer matrix.Expand Specific Solutions04 Self-assembled conductive polymer structures
Self-assembly techniques can be used to create ordered polymer structures with improved charge transport properties. Block copolymers that self-organize into nanoscale domains, supramolecular assemblies formed through non-covalent interactions, and layer-by-layer deposition methods can all produce polymer systems with enhanced conductivity. These ordered structures minimize charge trapping and provide efficient pathways for electron movement.Expand Specific Solutions05 Ionic conductivity in polymer electrolytes
Polymer electrolytes achieve high ionic conductivity through the incorporation of mobile ions within the polymer backbone or matrix. These systems typically contain polar functional groups that can coordinate with ions and facilitate their movement. The conductivity depends on factors such as the polymer's glass transition temperature, free volume, and the concentration of charge carriers. Applications include batteries, fuel cells, and other electrochemical devices where ion transport is critical.Expand Specific Solutions
Leading Research Groups and Industry Stakeholders
The anion exchange membrane (AEM) development market is currently in a growth phase, with increasing research focus on high conductivity polymer backbones. The global market is expanding rapidly due to rising demand for clean energy technologies, particularly in fuel cells and electrolyzers. Academic institutions dominate the research landscape, with significant contributions from Georgia Tech Research Corp., MIT, Rensselaer Polytechnic Institute, and Cornell University. Industry players like Promerus LLC, 3M Innovative Properties, and Advent Technologies are advancing commercialization efforts. Technical challenges remain in balancing conductivity, stability, and mechanical properties, with research concentrated in North America and Asia. The technology is approaching commercial viability but requires further optimization for widespread adoption in energy applications.
Promerus LLC
Technical Solution: Promerus LLC has developed innovative norbornene-based polymer backbones for AEMs that offer a unique combination of mechanical robustness and chemical stability. Their proprietary polynorbornene platform utilizes ring-opening metathesis polymerization (ROMP) to create polymers with rigid cycloaliphatic structures that resist degradation in alkaline environments[1]. The company has engineered these polymers to incorporate quaternary ammonium groups through side chains that are strategically positioned to minimize nucleophilic attack by hydroxide ions. Their most advanced materials feature a block copolymer architecture where hydrophilic ion-conducting blocks are separated from hydrophobic mechanical support blocks, creating well-defined nanophase-separated morphologies[3]. This approach has yielded membranes with hydroxide conductivities reaching 70-85 mS/cm at 80°C while maintaining dimensional stability. Promerus has also developed specialized crosslinking chemistries that enhance the mechanical properties and durability of their membranes without compromising ion conductivity, addressing a common trade-off in AEM development.
Strengths: Unique norbornene-based chemistry provides excellent alkaline stability; proprietary ROMP technology enables precise control over polymer architecture; effective balance of mechanical properties and ion conductivity. Weaknesses: Relatively complex and potentially costly synthesis procedures; limited long-term performance data in actual fuel cell operating conditions; potential challenges in scaling production.
Massachusetts Institute of Technology
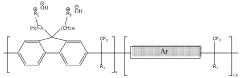
Technical Solution: MIT has developed novel high conductivity polymer backbones for anion exchange membranes (AEMs) based on poly(arylene ether)s containing quaternary ammonium groups. Their approach involves synthesizing polymers with rigid aromatic backbones that provide mechanical stability while incorporating pendant quaternary ammonium groups for ion conductivity. MIT researchers have pioneered the use of block copolymer architectures where hydrophilic blocks containing ion-conducting groups are segregated from hydrophobic blocks that provide mechanical strength[1]. This microphase separation creates well-defined ion-conducting channels, significantly enhancing hydroxide ion conductivity while maintaining dimensional stability. Their recent work has achieved hydroxide conductivities exceeding 100 mS/cm at 80°C while demonstrating improved alkaline stability through strategic placement of the cationic groups away from the polymer backbone[3]. MIT has also developed computational models to predict polymer degradation pathways, enabling rational design of more stable structures.
Strengths: Superior microphase separation leading to well-defined ion-conducting channels; excellent balance between conductivity and mechanical properties; advanced computational capabilities for polymer design. Weaknesses: Complex synthesis procedures may limit large-scale production; some designs still face challenges with long-term alkaline stability at elevated temperatures.
Key Patents and Scientific Breakthroughs in AEM Design
Anion exchange polymers and anion exchange membranes
PatentWO2023049459A1
Innovation
- Development of an anion conducting co-polymer with a poly(phenylene) backbone and aromatic compound linkages, incorporating quaternary ammonium or n-methyl piperidine functional groups, and a porous scaffold support for reinforcement, allowing for thin, mechanically stable, and chemically robust anion exchange membranes.
Anion exchange polymers and membranes for electrolysis
PatentActiveUS11980879B2
Innovation
- Development of novel anion exchange polymers with phenolic hydroxyl groups, featuring stable hydrophobic polymer backbones, hydrophilic quaternary ammonium cationic groups, and hydrophilic phenolic hydroxyl groups on side chains, enhancing OH- conductivity, chemical stability, and mechanical strength, enabling efficient operation in water or CO2 electrolysis and fuel cell applications.
Sustainability Aspects of Advanced AEM Materials
The development of sustainable Anion Exchange Membrane (AEM) materials represents a critical frontier in advancing clean energy technologies. As environmental concerns intensify globally, the sustainability profile of AEM materials has become increasingly important alongside their performance characteristics. Current high-conductivity polymer backbones for AEMs often rely on petroleum-derived precursors and energy-intensive synthesis processes, creating significant environmental footprints.
Life cycle assessment (LCA) studies reveal that traditional AEM production generates substantial carbon emissions, primarily during monomer synthesis and polymerization stages. Advanced polymer backbones utilizing bio-based precursors, such as those derived from lignin or cellulose, offer promising alternatives with reduced environmental impact. Recent research demonstrates that these bio-derived backbones can achieve conductivity values approaching 100 mS/cm while reducing carbon footprint by up to 40% compared to conventional materials.
Water consumption represents another critical sustainability metric for AEM development. Conventional synthesis methods typically require 80-120 liters of water per square meter of membrane produced. Novel green chemistry approaches incorporating supercritical CO2 processing and ionic liquid-based synthesis have demonstrated potential to reduce water usage by 60-75% while maintaining high ionic conductivity in the resulting polymer backbones.
End-of-life considerations present both challenges and opportunities for sustainable AEM development. Current high-performance fluorinated backbones exhibit exceptional durability but poor biodegradability. Emerging research into degradable linkages within high-conductivity backbones shows promise for creating materials that maintain performance during operational lifetimes but decompose under controlled conditions post-use. Polybenzimidazole-based systems incorporating strategically placed ester bonds demonstrate this balance, achieving 85 mS/cm conductivity while enabling eventual polymer breakdown.
Regulatory frameworks increasingly influence AEM material development trajectories. The European Union's REACH regulations and similar initiatives worldwide are restricting certain chemical components common in high-performance polymer backbones. Forward-looking research focuses on developing compliant alternatives that eliminate substances of very high concern (SVHCs) while maintaining conductivity performance. Quaternary phosphonium-based systems represent one promising direction, offering comparable conductivity to ammonium counterparts with improved regulatory profiles.
Economic sustainability must complement environmental considerations for widespread adoption. Cost modeling indicates that while bio-based and green chemistry approaches currently carry 15-30% price premiums, scaling effects and process optimization could achieve cost parity with conventional materials within 5-7 years, creating economically viable pathways to sustainable high-conductivity AEM materials.
Life cycle assessment (LCA) studies reveal that traditional AEM production generates substantial carbon emissions, primarily during monomer synthesis and polymerization stages. Advanced polymer backbones utilizing bio-based precursors, such as those derived from lignin or cellulose, offer promising alternatives with reduced environmental impact. Recent research demonstrates that these bio-derived backbones can achieve conductivity values approaching 100 mS/cm while reducing carbon footprint by up to 40% compared to conventional materials.
Water consumption represents another critical sustainability metric for AEM development. Conventional synthesis methods typically require 80-120 liters of water per square meter of membrane produced. Novel green chemistry approaches incorporating supercritical CO2 processing and ionic liquid-based synthesis have demonstrated potential to reduce water usage by 60-75% while maintaining high ionic conductivity in the resulting polymer backbones.
End-of-life considerations present both challenges and opportunities for sustainable AEM development. Current high-performance fluorinated backbones exhibit exceptional durability but poor biodegradability. Emerging research into degradable linkages within high-conductivity backbones shows promise for creating materials that maintain performance during operational lifetimes but decompose under controlled conditions post-use. Polybenzimidazole-based systems incorporating strategically placed ester bonds demonstrate this balance, achieving 85 mS/cm conductivity while enabling eventual polymer breakdown.
Regulatory frameworks increasingly influence AEM material development trajectories. The European Union's REACH regulations and similar initiatives worldwide are restricting certain chemical components common in high-performance polymer backbones. Forward-looking research focuses on developing compliant alternatives that eliminate substances of very high concern (SVHCs) while maintaining conductivity performance. Quaternary phosphonium-based systems represent one promising direction, offering comparable conductivity to ammonium counterparts with improved regulatory profiles.
Economic sustainability must complement environmental considerations for widespread adoption. Cost modeling indicates that while bio-based and green chemistry approaches currently carry 15-30% price premiums, scaling effects and process optimization could achieve cost parity with conventional materials within 5-7 years, creating economically viable pathways to sustainable high-conductivity AEM materials.
Performance Benchmarking Methodologies
Establishing standardized performance benchmarking methodologies is crucial for evaluating the effectiveness of high conductivity polymer backbones in anion exchange membrane (AEM) development. These methodologies must encompass multiple parameters to ensure comprehensive assessment and facilitate meaningful comparisons across different polymer systems.
The primary performance metrics for AEM polymer backbones include ionic conductivity, chemical stability, mechanical strength, and water uptake characteristics. Conductivity measurements should be performed using impedance spectroscopy under controlled temperature and humidity conditions, with standardized sample preparation protocols to ensure reproducibility. The four-point probe method has emerged as the gold standard for accurate conductivity assessment, minimizing contact resistance effects that can skew results.
Chemical stability testing requires accelerated aging protocols in alkaline environments, typically using concentrated KOH or NaOH solutions at elevated temperatures (60-80°C). Degradation should be monitored through changes in conductivity, mechanical properties, and chemical structure analysis via FTIR and NMR spectroscopy. Establishing standardized exposure times (100, 500, 1000 hours) enables direct comparison between different polymer backbone chemistries.
Mechanical property evaluation must include tensile testing to determine Young's modulus, elongation at break, and tensile strength. These measurements should be conducted under both dry and hydrated conditions to understand how water uptake affects mechanical integrity. Dimensional stability during hydration/dehydration cycles represents another critical parameter, as excessive swelling can compromise membrane performance in actual devices.
Water uptake and ion exchange capacity (IEC) measurements require standardized protocols involving precise weighing before and after hydration, with controlled humidity exposure. The relationship between IEC, water uptake, and conductivity provides crucial insights into the efficiency of ion transport mechanisms within the polymer backbone.
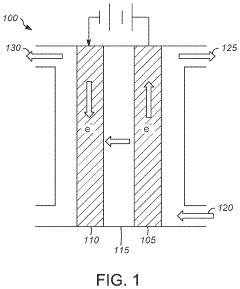
For practical application assessment, membrane electrode assembly (MEA) testing in actual fuel cell or electrolyzer configurations provides the most relevant performance data. Standardized testing conditions including current density, operating temperature, and feed compositions must be established to enable fair comparisons between different polymer systems.
Round-robin testing involving multiple laboratories using identical samples and protocols can significantly enhance data reliability and establish confidence in performance benchmarks. This collaborative approach helps identify procedural variables that may influence results and establishes a more robust foundation for comparing innovative polymer backbone chemistries for next-generation AEM applications.
The primary performance metrics for AEM polymer backbones include ionic conductivity, chemical stability, mechanical strength, and water uptake characteristics. Conductivity measurements should be performed using impedance spectroscopy under controlled temperature and humidity conditions, with standardized sample preparation protocols to ensure reproducibility. The four-point probe method has emerged as the gold standard for accurate conductivity assessment, minimizing contact resistance effects that can skew results.
Chemical stability testing requires accelerated aging protocols in alkaline environments, typically using concentrated KOH or NaOH solutions at elevated temperatures (60-80°C). Degradation should be monitored through changes in conductivity, mechanical properties, and chemical structure analysis via FTIR and NMR spectroscopy. Establishing standardized exposure times (100, 500, 1000 hours) enables direct comparison between different polymer backbone chemistries.
Mechanical property evaluation must include tensile testing to determine Young's modulus, elongation at break, and tensile strength. These measurements should be conducted under both dry and hydrated conditions to understand how water uptake affects mechanical integrity. Dimensional stability during hydration/dehydration cycles represents another critical parameter, as excessive swelling can compromise membrane performance in actual devices.
Water uptake and ion exchange capacity (IEC) measurements require standardized protocols involving precise weighing before and after hydration, with controlled humidity exposure. The relationship between IEC, water uptake, and conductivity provides crucial insights into the efficiency of ion transport mechanisms within the polymer backbone.
For practical application assessment, membrane electrode assembly (MEA) testing in actual fuel cell or electrolyzer configurations provides the most relevant performance data. Standardized testing conditions including current density, operating temperature, and feed compositions must be established to enable fair comparisons between different polymer systems.
Round-robin testing involving multiple laboratories using identical samples and protocols can significantly enhance data reliability and establish confidence in performance benchmarks. This collaborative approach helps identify procedural variables that may influence results and establishes a more robust foundation for comparing innovative polymer backbone chemistries for next-generation AEM applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!