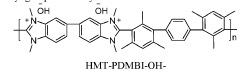
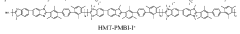
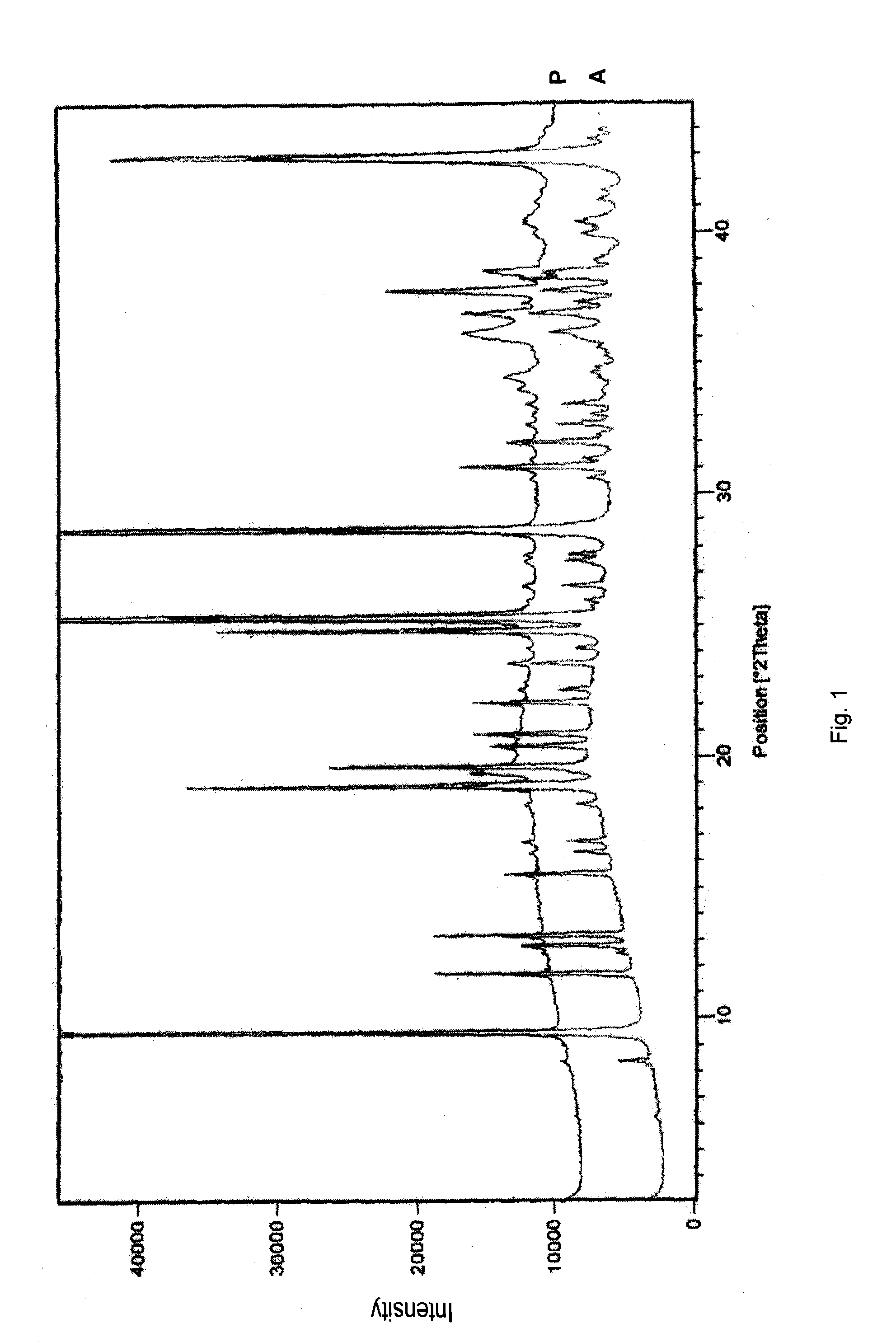
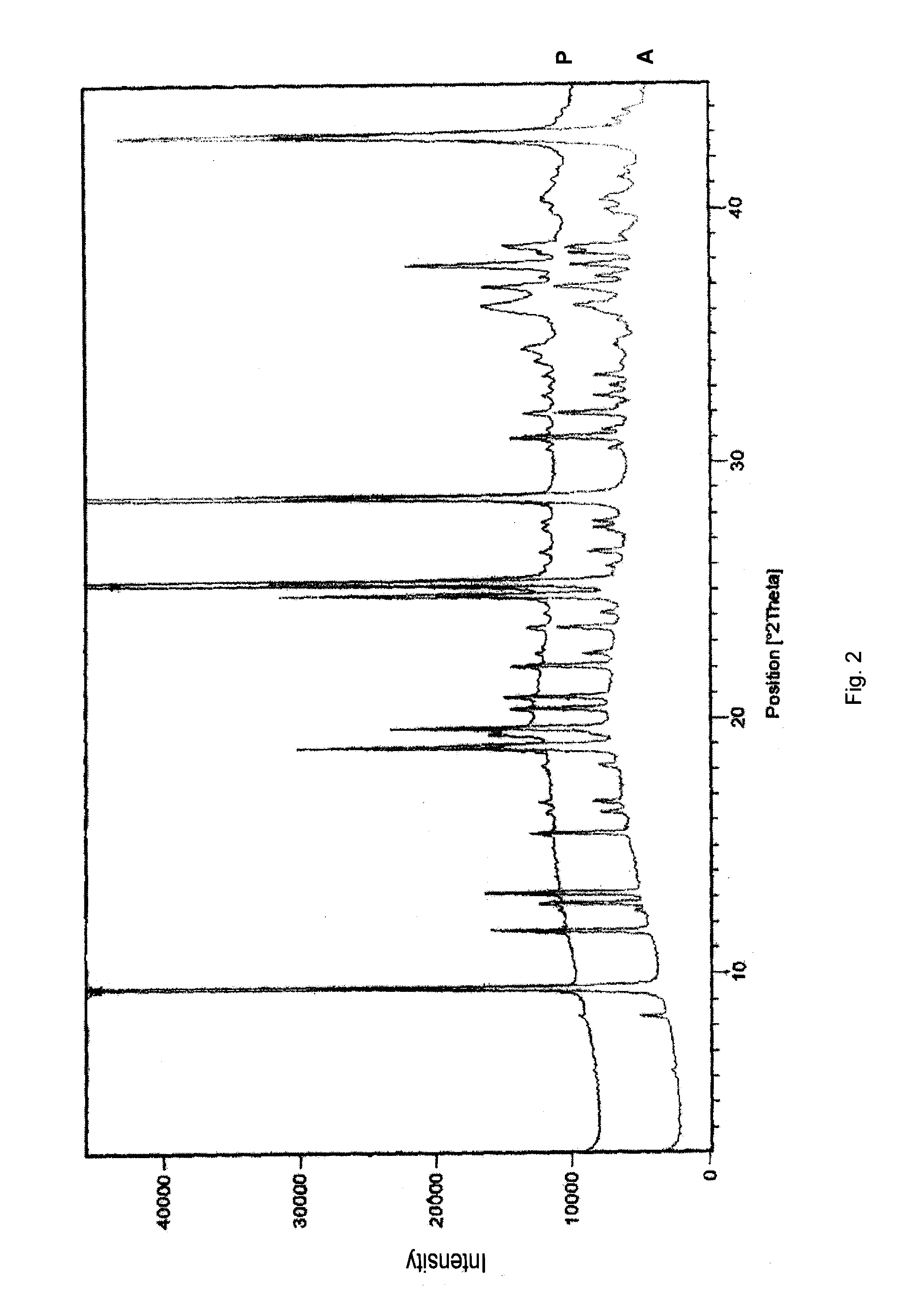
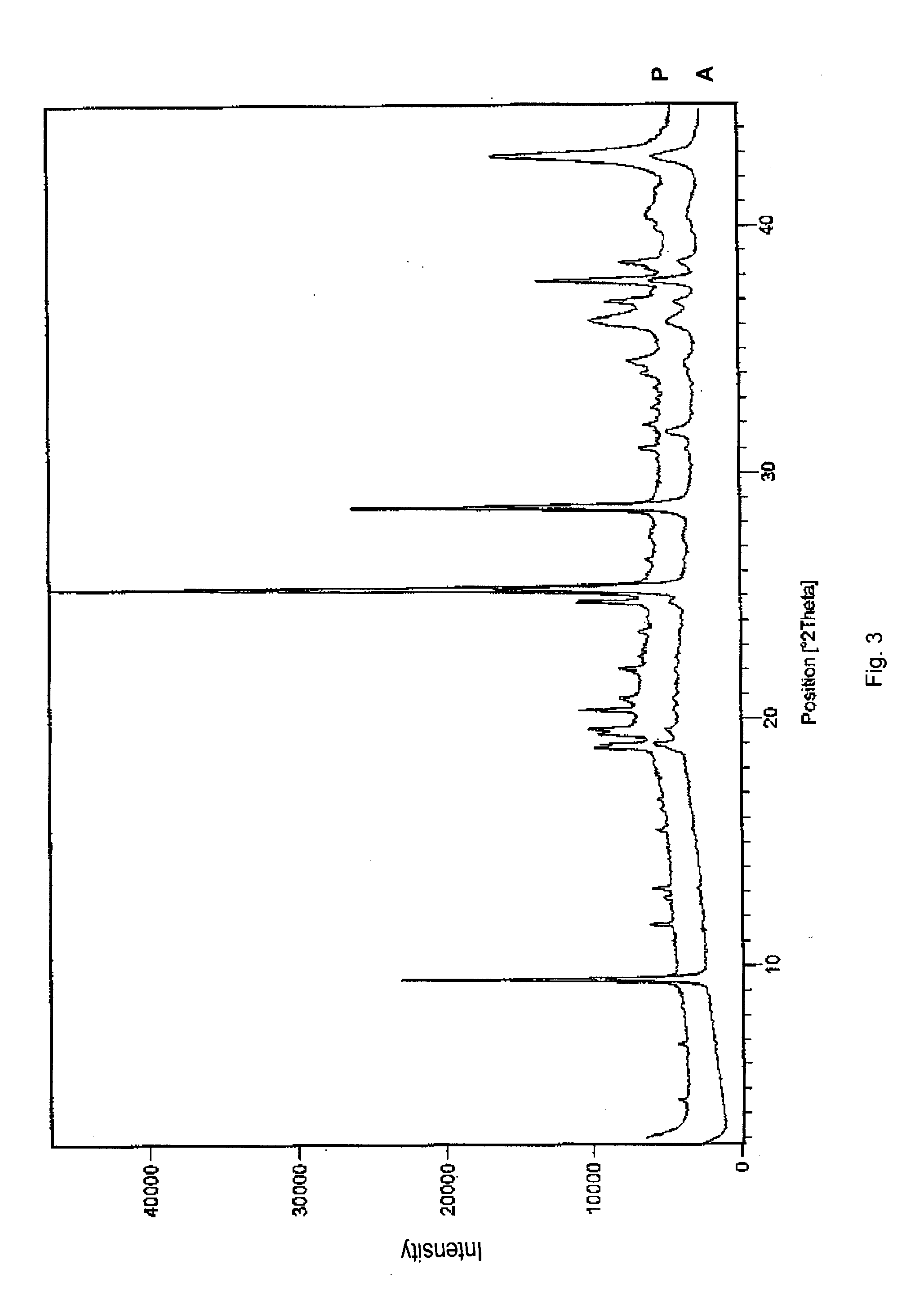
Stability of benzimidazolium cations in harsh alkaline media
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Benzimidazolium Stability Background and Research Objectives
Benzimidazolium compounds have emerged as significant structures in various chemical applications over the past several decades. These heterocyclic organic compounds, characterized by a fused benzene and imidazole ring system, have demonstrated remarkable versatility across multiple industries including pharmaceuticals, materials science, and catalysis. The historical development of benzimidazolium chemistry can be traced back to the mid-20th century, with significant advancements occurring in the 1990s when their potential as ionic liquid precursors and N-heterocyclic carbene ligands was recognized.
The stability of benzimidazolium cations in harsh alkaline environments represents a critical technological challenge that has gained increasing attention in recent years. This interest is primarily driven by their potential applications in anion exchange membranes for fuel cells, electrolyzers, and other electrochemical devices operating under high pH conditions. The alkaline stability of these cations directly impacts the longevity and performance of such devices, making it a key determinant in their commercial viability.
Current technological trends indicate a shift toward developing more robust benzimidazolium derivatives that can withstand prolonged exposure to strongly basic conditions. Research has evolved from simple structural modifications to sophisticated molecular engineering approaches that consider electronic effects, steric hindrance, and supramolecular interactions to enhance stability. This evolution reflects the growing understanding of degradation mechanisms and structure-stability relationships within this class of compounds.
The primary objective of this technical research is to comprehensively evaluate the current state of benzimidazolium cation stability in alkaline media and identify promising pathways for improvement. Specifically, we aim to: (1) elucidate the fundamental degradation mechanisms affecting benzimidazolium cations under alkaline conditions; (2) assess the effectiveness of various structural modifications in enhancing stability; (3) establish quantitative structure-stability relationships to guide future molecular design; and (4) identify benchmark compounds that demonstrate superior alkaline resistance.
Additionally, this research seeks to explore the correlation between molecular structure and alkaline stability across different benzimidazolium derivatives, with particular emphasis on the impact of substituent patterns, electronic properties, and conformational factors. By establishing these correlations, we anticipate developing predictive models that can accelerate the discovery of highly stable benzimidazolium compounds suitable for next-generation alkaline electrochemical technologies.
The findings from this investigation are expected to contribute significantly to the advancement of anion exchange membrane technology and potentially expand the application scope of benzimidazolium-based materials in other alkaline-operating systems, thereby addressing critical challenges in renewable energy conversion and storage technologies.
The stability of benzimidazolium cations in harsh alkaline environments represents a critical technological challenge that has gained increasing attention in recent years. This interest is primarily driven by their potential applications in anion exchange membranes for fuel cells, electrolyzers, and other electrochemical devices operating under high pH conditions. The alkaline stability of these cations directly impacts the longevity and performance of such devices, making it a key determinant in their commercial viability.
Current technological trends indicate a shift toward developing more robust benzimidazolium derivatives that can withstand prolonged exposure to strongly basic conditions. Research has evolved from simple structural modifications to sophisticated molecular engineering approaches that consider electronic effects, steric hindrance, and supramolecular interactions to enhance stability. This evolution reflects the growing understanding of degradation mechanisms and structure-stability relationships within this class of compounds.
The primary objective of this technical research is to comprehensively evaluate the current state of benzimidazolium cation stability in alkaline media and identify promising pathways for improvement. Specifically, we aim to: (1) elucidate the fundamental degradation mechanisms affecting benzimidazolium cations under alkaline conditions; (2) assess the effectiveness of various structural modifications in enhancing stability; (3) establish quantitative structure-stability relationships to guide future molecular design; and (4) identify benchmark compounds that demonstrate superior alkaline resistance.
Additionally, this research seeks to explore the correlation between molecular structure and alkaline stability across different benzimidazolium derivatives, with particular emphasis on the impact of substituent patterns, electronic properties, and conformational factors. By establishing these correlations, we anticipate developing predictive models that can accelerate the discovery of highly stable benzimidazolium compounds suitable for next-generation alkaline electrochemical technologies.
The findings from this investigation are expected to contribute significantly to the advancement of anion exchange membrane technology and potentially expand the application scope of benzimidazolium-based materials in other alkaline-operating systems, thereby addressing critical challenges in renewable energy conversion and storage technologies.
Market Applications and Demand Analysis for Alkaline-Stable Cations
The market for alkaline-stable cations, particularly benzimidazolium-based structures, has witnessed significant growth driven by their applications in energy conversion and storage technologies. The global anion exchange membrane (AEM) market, where these cations play a crucial role, was valued at approximately $570 million in 2022 and is projected to grow at a compound annual growth rate of 6.8% through 2030.
The primary demand for alkaline-stable cations comes from the fuel cell industry, specifically anion exchange membrane fuel cells (AEMFCs). This sector has experienced robust growth due to increasing focus on clean energy solutions and hydrogen economy initiatives. Major automotive manufacturers and energy companies have invested substantially in AEMFC technology, recognizing its potential advantages over proton exchange membrane fuel cells (PEMFCs) in terms of cost and catalyst requirements.
Water electrolysis represents another significant market application, with alkaline water electrolyzers requiring stable cationic groups to maintain performance over extended operational periods. The green hydrogen production market, valued at $3.2 billion in 2021, is expected to reach $9.8 billion by 2028, creating substantial demand for advanced alkaline-stable materials.
The pharmaceutical and fine chemical industries also contribute to market demand, utilizing benzimidazolium-based catalysts for various organic transformations under alkaline conditions. These applications require cations that maintain structural integrity in high pH environments to ensure consistent catalytic performance and product quality.
Geographically, North America and Europe lead in research and development of alkaline-stable cations, while Asia-Pacific represents the fastest-growing market due to increasing investments in renewable energy infrastructure and manufacturing capabilities. Japan, South Korea, and China have established national hydrogen strategies that specifically mention the importance of alkaline membrane technologies.
Market analysis indicates that end-users are willing to pay premium prices for cations demonstrating superior alkaline stability, with performance under harsh conditions (pH > 13, temperatures > 80°C) being the primary value driver. Industry surveys show that a 20% improvement in alkaline stability can command up to 40% price premium in specialized applications.
The market faces challenges from competing technologies, particularly traditional proton exchange membranes and solid oxide systems. However, the potential cost advantages and operational benefits of alkaline systems continue to drive research investment and market growth for stable benzimidazolium cations and related structures.
The primary demand for alkaline-stable cations comes from the fuel cell industry, specifically anion exchange membrane fuel cells (AEMFCs). This sector has experienced robust growth due to increasing focus on clean energy solutions and hydrogen economy initiatives. Major automotive manufacturers and energy companies have invested substantially in AEMFC technology, recognizing its potential advantages over proton exchange membrane fuel cells (PEMFCs) in terms of cost and catalyst requirements.
Water electrolysis represents another significant market application, with alkaline water electrolyzers requiring stable cationic groups to maintain performance over extended operational periods. The green hydrogen production market, valued at $3.2 billion in 2021, is expected to reach $9.8 billion by 2028, creating substantial demand for advanced alkaline-stable materials.
The pharmaceutical and fine chemical industries also contribute to market demand, utilizing benzimidazolium-based catalysts for various organic transformations under alkaline conditions. These applications require cations that maintain structural integrity in high pH environments to ensure consistent catalytic performance and product quality.
Geographically, North America and Europe lead in research and development of alkaline-stable cations, while Asia-Pacific represents the fastest-growing market due to increasing investments in renewable energy infrastructure and manufacturing capabilities. Japan, South Korea, and China have established national hydrogen strategies that specifically mention the importance of alkaline membrane technologies.
Market analysis indicates that end-users are willing to pay premium prices for cations demonstrating superior alkaline stability, with performance under harsh conditions (pH > 13, temperatures > 80°C) being the primary value driver. Industry surveys show that a 20% improvement in alkaline stability can command up to 40% price premium in specialized applications.
The market faces challenges from competing technologies, particularly traditional proton exchange membranes and solid oxide systems. However, the potential cost advantages and operational benefits of alkaline systems continue to drive research investment and market growth for stable benzimidazolium cations and related structures.
Current Challenges in Benzimidazolium Stability under Alkaline Conditions
The stability of benzimidazolium cations in alkaline environments presents significant challenges for their application in various technological fields. Despite their promising properties as functional materials, these compounds undergo degradation through multiple pathways when exposed to high pH conditions. The primary degradation mechanism involves nucleophilic attack by hydroxide ions at the C2 position of the imidazolium ring, leading to ring-opening reactions and subsequent decomposition.
Current research indicates that structural modifications can influence stability, but comprehensive understanding remains limited. Benzimidazolium derivatives with electron-withdrawing substituents show marginally improved resistance to alkaline degradation, yet even these modified structures eventually succumb to decomposition under prolonged exposure to harsh alkaline conditions.
Temperature effects compound these stability issues, with degradation rates increasing exponentially at elevated temperatures. This thermal sensitivity severely restricts applications in high-temperature alkaline environments, such as those found in certain fuel cell systems and industrial catalytic processes. The synergistic effect of temperature and alkalinity creates particularly challenging conditions for maintaining structural integrity.
Concentration effects present another dimension of complexity. Higher hydroxide concentrations predictably accelerate degradation, but the relationship is not strictly linear, suggesting complex degradation kinetics. Studies have shown threshold effects where stability decreases dramatically beyond certain hydroxide concentration levels, indicating potential changes in degradation mechanisms.
The presence of metal ions in alkaline media introduces additional complications. Certain transition metals can coordinate with benzimidazolium structures, either enhancing stability through protective coordination or catalyzing decomposition through facilitated nucleophilic attack. This metal-dependent behavior remains poorly understood and difficult to predict.
Analytical challenges further complicate research efforts. Real-time monitoring of degradation pathways presents technical difficulties, with degradation intermediates often being short-lived and difficult to isolate. Current analytical methods struggle to provide comprehensive mechanistic insights, particularly in complex media relevant to practical applications.
Computational modeling approaches have attempted to predict stability and degradation pathways, but validation against experimental data shows significant discrepancies. The multifaceted nature of the degradation processes, involving solvent effects, ion pairing, and dynamic conformational changes, exceeds the predictive capabilities of current computational methods.
These challenges collectively represent significant barriers to the widespread application of benzimidazolium-based materials in alkaline environments, necessitating innovative approaches to enhance stability while maintaining desired functional properties.
Current research indicates that structural modifications can influence stability, but comprehensive understanding remains limited. Benzimidazolium derivatives with electron-withdrawing substituents show marginally improved resistance to alkaline degradation, yet even these modified structures eventually succumb to decomposition under prolonged exposure to harsh alkaline conditions.
Temperature effects compound these stability issues, with degradation rates increasing exponentially at elevated temperatures. This thermal sensitivity severely restricts applications in high-temperature alkaline environments, such as those found in certain fuel cell systems and industrial catalytic processes. The synergistic effect of temperature and alkalinity creates particularly challenging conditions for maintaining structural integrity.
Concentration effects present another dimension of complexity. Higher hydroxide concentrations predictably accelerate degradation, but the relationship is not strictly linear, suggesting complex degradation kinetics. Studies have shown threshold effects where stability decreases dramatically beyond certain hydroxide concentration levels, indicating potential changes in degradation mechanisms.
The presence of metal ions in alkaline media introduces additional complications. Certain transition metals can coordinate with benzimidazolium structures, either enhancing stability through protective coordination or catalyzing decomposition through facilitated nucleophilic attack. This metal-dependent behavior remains poorly understood and difficult to predict.
Analytical challenges further complicate research efforts. Real-time monitoring of degradation pathways presents technical difficulties, with degradation intermediates often being short-lived and difficult to isolate. Current analytical methods struggle to provide comprehensive mechanistic insights, particularly in complex media relevant to practical applications.
Computational modeling approaches have attempted to predict stability and degradation pathways, but validation against experimental data shows significant discrepancies. The multifaceted nature of the degradation processes, involving solvent effects, ion pairing, and dynamic conformational changes, exceeds the predictive capabilities of current computational methods.
These challenges collectively represent significant barriers to the widespread application of benzimidazolium-based materials in alkaline environments, necessitating innovative approaches to enhance stability while maintaining desired functional properties.
Existing Methodologies for Enhancing Benzimidazolium Alkaline Stability
01 Structural modifications for enhanced stability
Benzimidazolium cations can be stabilized through various structural modifications. These include the introduction of specific substituents at key positions on the benzimidazole ring, incorporation of electron-donating or withdrawing groups, and modification of the N-alkylation pattern. Such structural changes can significantly improve the thermal and chemical stability of benzimidazolium compounds, making them more suitable for various applications including catalysis and electrochemical processes.- Structural modifications for enhanced stability: Various structural modifications can be implemented to enhance the stability of benzimidazolium cations. These include the introduction of specific substituents at key positions of the benzimidazole ring, incorporation of electron-donating or electron-withdrawing groups, and modification of the N-alkylation pattern. These structural changes can significantly improve the thermal, chemical, and photochemical stability of benzimidazolium cations for various applications.
- Stabilization in ionic liquid applications: Benzimidazolium cations are widely used in ionic liquid formulations where their stability is crucial for performance. Specific counter-anions can be paired with benzimidazolium cations to enhance their thermal and electrochemical stability. Additionally, incorporating specific functional groups on the benzimidazolium core can improve stability under various conditions, making these ionic liquids suitable for catalysis, electrochemistry, and separation processes.
- Stability in biological and pharmaceutical applications: Benzimidazolium cations can be stabilized for biological and pharmaceutical applications through specific formulation strategies. These include pH control, addition of antioxidants, selection of appropriate counter-ions, and incorporation into delivery systems. The stability of these cations is critical for maintaining their biological activity, including antimicrobial, antifungal, and anticancer properties, as well as ensuring adequate shelf-life of pharmaceutical formulations.
- Environmental and chemical resistance: Benzimidazolium cations can be engineered to resist degradation under harsh environmental and chemical conditions. This includes stability against oxidation, hydrolysis, high temperatures, and exposure to light. Specific structural features, such as bulky substituents and delocalized electronic systems, can protect the cationic center from nucleophilic attack and other degradation pathways, making these compounds suitable for applications in extreme environments.
- Stabilization techniques for industrial applications: Various stabilization techniques can be employed to enhance the durability of benzimidazolium cations in industrial settings. These include encapsulation, complexation with metal ions, incorporation into polymeric matrices, and use of specific additives. These approaches can significantly improve the thermal stability, chemical resistance, and long-term performance of benzimidazolium-based materials in applications such as catalysis, material science, and energy storage systems.
02 Stabilization in ionic liquid applications
Benzimidazolium cations are widely used in ionic liquid formulations where their stability is crucial. Research has focused on enhancing their stability in these applications through counter-ion selection, structural design, and formulation techniques. Stable benzimidazolium-based ionic liquids demonstrate improved performance in electrochemical devices, catalytic processes, and separation technologies, with reduced degradation under operating conditions.Expand Specific Solutions03 Environmental factors affecting stability
The stability of benzimidazolium cations is significantly influenced by environmental factors such as temperature, pH, light exposure, and presence of oxidizing agents. Research has identified optimal conditions for maintaining stability and methods to protect these compounds from degradation. Understanding these environmental influences has led to improved formulations and handling protocols that extend the shelf life and functional performance of benzimidazolium-containing products.Expand Specific Solutions04 Stabilization through complexation and coordination
Benzimidazolium cations can be stabilized through complexation with metal ions or coordination with specific ligands. These interactions can significantly enhance the thermal and chemical stability of the cations by providing additional structural support and electronic stabilization. Metal-benzimidazolium complexes often exhibit improved stability profiles compared to the free cations, making them valuable in catalytic applications, material science, and pharmaceutical development.Expand Specific Solutions05 Analytical methods for stability assessment
Various analytical techniques have been developed to assess and monitor the stability of benzimidazolium cations under different conditions. These include spectroscopic methods (NMR, UV-Vis, IR), chromatographic techniques, thermal analysis, and electrochemical measurements. These analytical approaches enable researchers to quantify degradation rates, identify breakdown products, and optimize formulations for enhanced stability, supporting the development of more robust benzimidazolium-based technologies.Expand Specific Solutions
Leading Research Groups and Industrial Players in Cationic Stability
The stability of benzimidazolium cations in harsh alkaline media represents an emerging technical challenge at the intersection of materials science and industrial chemistry. The market is currently in its growth phase, with increasing applications in pharmaceuticals and specialty chemicals. Key players include academic institutions like Dalian University of Technology and Queen's University Belfast conducting fundamental research, alongside pharmaceutical giants such as Bayer AG, Dr. Reddy's Laboratories, and Eisai Co. who are developing practical applications. The technology shows moderate maturity with established companies like Teva Pharmaceuticals and Boehringer Ingelheim advancing industrial implementations, while research institutions continue exploring novel stabilization methods. This competitive landscape reflects a balanced ecosystem of academic innovation and commercial development driving progress in alkaline-stable benzimidazolium-based materials.
Dalian University of Technology
Technical Solution: Dalian University of Technology has developed innovative approaches to enhance benzimidazolium cation stability in alkaline environments through structural modification strategies. Their research focuses on introducing electron-withdrawing substituents at specific positions of the benzimidazolium ring to reduce electron density at the C2 position, thereby increasing resistance to nucleophilic attack by hydroxide ions. They have successfully synthesized benzimidazolium derivatives with fluorinated alkyl chains and cyano groups that demonstrate significantly improved half-lives in 1M NaOH solutions (>200 hours compared to <10 hours for unmodified variants). Additionally, they've pioneered the development of sterically hindered benzimidazolium frameworks where bulky substituents physically shield the vulnerable C2 position from hydroxide attack. Their computational studies have provided fundamental insights into degradation mechanisms, enabling rational design of more stable derivatives for applications in anion exchange membranes and catalysis under alkaline conditions.
Strengths: Strong fundamental research capabilities combining computational and experimental approaches; innovative structural modification strategies that address the core stability issues. Weaknesses: Limited industrial-scale production capabilities; research primarily remains at laboratory scale without extensive real-world application testing in commercial devices.
Bayer AG
Technical Solution: Bayer AG has developed proprietary technology for stabilizing benzimidazolium cations in alkaline environments, primarily for applications in agricultural formulations and specialty chemicals. Their approach involves creating hybrid molecular structures where benzimidazolium units are integrated into polymer backbones with protective hydrophobic domains that shield the cationic centers from hydroxide attack. Their patented "AlkaShield" technology incorporates benzimidazolium derivatives with specially designed counter-anions that form tight ion pairs, reducing susceptibility to nucleophilic degradation. Bayer has also pioneered the use of supramolecular protection strategies where the benzimidazolium moieties are encapsulated within cyclodextrin-based host structures, creating a physical barrier against hydroxide ions while maintaining the functional properties of the cation. Their formulations have demonstrated stability at pH values up to 13 for extended periods (>6 months), enabling applications in highly alkaline industrial processes and cleaning formulations where conventional quaternary ammonium compounds rapidly degrade.
Strengths: Extensive industrial-scale manufacturing capabilities; comprehensive intellectual property portfolio; proven track record in commercializing specialty chemical technologies. Weaknesses: Solutions are often application-specific rather than providing fundamental breakthroughs; relatively high production costs compared to conventional alternatives; some approaches require specialized handling procedures.
Key Scientific Breakthroughs in Heterocyclic Cation Stabilization
Crosslinking of hydroxide stable, polybenzimidazoliums and polyimidazoliums membranes and ionomers
PatentWO2018026743A1
Innovation
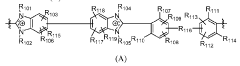
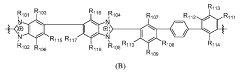
- Development of crosslinked polymers with specific repeating units, such as those described in Formulas A, B, C, and D, which enhance chemical and mechanical stability while maintaining solvent processability by forming crosslinks between benzimidazolium and imidazolium units, thereby stabilizing the membranes in aqueous solutions and improving ionic conductivity.
Pharmaceutical formulations comprising substituted benzimidazole derivatives
PatentInactiveUS20120058194A1
Innovation
- The development of modified release pharmaceutical formulations comprising a core with a substituted benzimidazole derivative or its salt, a stabilizer, and pharmaceutically acceptable excipients, surrounded by pH-dependently soluble release-controlled coating layers that dissolve in specific pH ranges to stabilize the active agent and control drug release.
Environmental Impact and Sustainability Considerations
The environmental implications of benzimidazolium cations in alkaline media extend beyond their chemical stability, encompassing broader ecological and sustainability considerations. These compounds, when deployed in various applications such as anion exchange membranes for fuel cells or electrolyzers, interact with environmental systems throughout their lifecycle. The degradation pathways of benzimidazolium cations under alkaline conditions can lead to the formation of potentially harmful byproducts that may enter aquatic ecosystems, affecting water quality and aquatic life.
Manufacturing processes for benzimidazolium-based materials often involve organic solvents and energy-intensive synthesis routes, contributing to their overall environmental footprint. Life cycle assessments indicate that the production phase accounts for significant carbon emissions and resource consumption, particularly when petroleum-derived precursors are utilized. However, recent advances in green chemistry approaches have demonstrated potential for reducing these impacts through solvent-free synthesis methods and bio-based feedstock alternatives.
The end-of-life management of materials containing benzimidazolium cations presents additional environmental challenges. Their persistence in the environment after disposal may lead to long-term accumulation, particularly concerning for materials with limited biodegradability. Research into recovery and recycling strategies remains underdeveloped, highlighting a critical gap in the circular economy approach for these materials.
From a sustainability perspective, the alkaline stability of benzimidazolium cations directly influences the operational lifespan of devices incorporating these compounds. Enhanced stability translates to longer-lasting materials, reducing replacement frequency and associated resource consumption. This relationship between chemical durability and product longevity represents a key sustainability parameter that must be optimized through molecular design strategies.
Regulatory frameworks increasingly emphasize the importance of comprehensive environmental risk assessments for novel materials. The REACH regulation in Europe and similar initiatives globally now require detailed toxicological and ecotoxicological data for chemical registration. Benzimidazolium-based compounds, particularly those designed for enhanced alkaline stability through structural modifications, must navigate these regulatory landscapes to ensure environmental compliance throughout their commercial lifecycle.
The transition toward more sustainable alternatives has prompted exploration of bio-inspired benzimidazolium derivatives synthesized from renewable resources. These approaches aim to maintain the desired alkaline stability while reducing dependence on fossil-based raw materials. Preliminary studies suggest that certain naturally-derived benzimidazolium structures may offer comparable performance with significantly reduced environmental impact, though scale-up challenges remain substantial.
Manufacturing processes for benzimidazolium-based materials often involve organic solvents and energy-intensive synthesis routes, contributing to their overall environmental footprint. Life cycle assessments indicate that the production phase accounts for significant carbon emissions and resource consumption, particularly when petroleum-derived precursors are utilized. However, recent advances in green chemistry approaches have demonstrated potential for reducing these impacts through solvent-free synthesis methods and bio-based feedstock alternatives.
The end-of-life management of materials containing benzimidazolium cations presents additional environmental challenges. Their persistence in the environment after disposal may lead to long-term accumulation, particularly concerning for materials with limited biodegradability. Research into recovery and recycling strategies remains underdeveloped, highlighting a critical gap in the circular economy approach for these materials.
From a sustainability perspective, the alkaline stability of benzimidazolium cations directly influences the operational lifespan of devices incorporating these compounds. Enhanced stability translates to longer-lasting materials, reducing replacement frequency and associated resource consumption. This relationship between chemical durability and product longevity represents a key sustainability parameter that must be optimized through molecular design strategies.
Regulatory frameworks increasingly emphasize the importance of comprehensive environmental risk assessments for novel materials. The REACH regulation in Europe and similar initiatives globally now require detailed toxicological and ecotoxicological data for chemical registration. Benzimidazolium-based compounds, particularly those designed for enhanced alkaline stability through structural modifications, must navigate these regulatory landscapes to ensure environmental compliance throughout their commercial lifecycle.
The transition toward more sustainable alternatives has prompted exploration of bio-inspired benzimidazolium derivatives synthesized from renewable resources. These approaches aim to maintain the desired alkaline stability while reducing dependence on fossil-based raw materials. Preliminary studies suggest that certain naturally-derived benzimidazolium structures may offer comparable performance with significantly reduced environmental impact, though scale-up challenges remain substantial.
Scalability and Industrial Implementation Challenges
The scaling of benzimidazolium-based technologies from laboratory to industrial scale presents significant challenges due to the harsh alkaline environments in which these cations must remain stable. Current industrial implementation faces several critical bottlenecks that require systematic solutions before widespread adoption can occur.
Material production scalability represents a primary concern, as synthesizing benzimidazolium cations with consistent quality at industrial volumes demands precise control over reaction conditions. Temperature, concentration, and pH variations in large-scale reactors can lead to inconsistent stability profiles, affecting the performance of end products. Manufacturing processes must be optimized to maintain uniform cation stability characteristics across production batches.
Equipment compatibility issues further complicate industrial implementation. The corrosive nature of harsh alkaline media necessitates specialized materials for reactors, pipes, and storage vessels. Standard industrial equipment often experiences accelerated degradation when exposed to these conditions, leading to increased maintenance costs and potential safety hazards. The development of cost-effective, corrosion-resistant materials compatible with both benzimidazolium cations and alkaline environments remains an ongoing challenge.
Process integration represents another significant hurdle. Incorporating benzimidazolium-based technologies into existing industrial workflows requires careful consideration of upstream and downstream processes. The stability of these cations can be compromised by contaminants from previous processing steps, while their degradation products may adversely affect subsequent operations. Comprehensive process engineering is needed to ensure seamless integration without compromising stability or efficiency.
Quality control and monitoring systems must be adapted for industrial-scale operations. Current analytical methods for assessing benzimidazolium cation stability often require sophisticated laboratory equipment and are difficult to implement in real-time production environments. The development of robust, inline monitoring techniques capable of providing immediate feedback on cation stability is essential for maintaining product quality during continuous manufacturing processes.
Economic considerations ultimately determine industrial feasibility. The cost of scaling benzimidazolium cation production while maintaining stability in alkaline media must be balanced against performance benefits. Current synthesis routes often involve expensive precursors or complex purification steps that become prohibitively expensive at industrial scales. Process intensification and alternative synthetic pathways must be explored to improve economic viability while preserving the essential stability characteristics in harsh alkaline environments.
Material production scalability represents a primary concern, as synthesizing benzimidazolium cations with consistent quality at industrial volumes demands precise control over reaction conditions. Temperature, concentration, and pH variations in large-scale reactors can lead to inconsistent stability profiles, affecting the performance of end products. Manufacturing processes must be optimized to maintain uniform cation stability characteristics across production batches.
Equipment compatibility issues further complicate industrial implementation. The corrosive nature of harsh alkaline media necessitates specialized materials for reactors, pipes, and storage vessels. Standard industrial equipment often experiences accelerated degradation when exposed to these conditions, leading to increased maintenance costs and potential safety hazards. The development of cost-effective, corrosion-resistant materials compatible with both benzimidazolium cations and alkaline environments remains an ongoing challenge.
Process integration represents another significant hurdle. Incorporating benzimidazolium-based technologies into existing industrial workflows requires careful consideration of upstream and downstream processes. The stability of these cations can be compromised by contaminants from previous processing steps, while their degradation products may adversely affect subsequent operations. Comprehensive process engineering is needed to ensure seamless integration without compromising stability or efficiency.
Quality control and monitoring systems must be adapted for industrial-scale operations. Current analytical methods for assessing benzimidazolium cation stability often require sophisticated laboratory equipment and are difficult to implement in real-time production environments. The development of robust, inline monitoring techniques capable of providing immediate feedback on cation stability is essential for maintaining product quality during continuous manufacturing processes.
Economic considerations ultimately determine industrial feasibility. The cost of scaling benzimidazolium cation production while maintaining stability in alkaline media must be balanced against performance benefits. Current synthesis routes often involve expensive precursors or complex purification steps that become prohibitively expensive at industrial scales. Process intensification and alternative synthetic pathways must be explored to improve economic viability while preserving the essential stability characteristics in harsh alkaline environments.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!