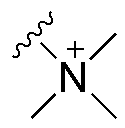Anion exchange membranes for alkaline fuel cells
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Anion Exchange Membrane Technology Background and Objectives
Anion exchange membranes (AEMs) have emerged as a critical component in the development of alkaline fuel cells, representing a significant advancement in clean energy technology. The evolution of AEM technology can be traced back to the mid-20th century, with substantial progress occurring in the past two decades due to increasing global emphasis on sustainable energy solutions. This technology addresses fundamental limitations of traditional proton exchange membrane fuel cells by enabling the use of non-precious metal catalysts and offering improved electrochemical kinetics in alkaline environments.
The trajectory of AEM development has been characterized by progressive improvements in ionic conductivity, chemical stability, and mechanical durability. Early iterations suffered from rapid degradation in alkaline conditions, limiting practical applications. Recent advancements have focused on novel polymer architectures and functional group designs that enhance hydroxide ion transport while maintaining structural integrity under operating conditions.
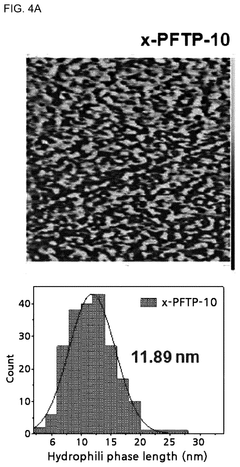
Current technological trends indicate a shift toward multifunctional membrane designs that simultaneously address multiple performance parameters. These include the development of block copolymers with segregated hydrophilic and hydrophobic domains, crosslinked networks for enhanced dimensional stability, and composite materials incorporating inorganic components for improved conductivity and durability.
The primary technical objective in AEM research is to achieve hydroxide conductivity comparable to proton conductivity in acidic systems (>100 mS/cm) while maintaining chemical stability in high pH environments for extended periods (>5000 hours). Secondary objectives include reducing membrane swelling, enhancing mechanical properties under varying hydration conditions, and developing cost-effective manufacturing processes suitable for commercial scale production.
Beyond performance metrics, researchers aim to establish standardized testing protocols for meaningful comparison across different membrane systems. This standardization is crucial for accelerating technology development and facilitating industry adoption. Additionally, there is growing interest in understanding degradation mechanisms at the molecular level to inform rational design strategies for next-generation materials.
The broader goal of AEM technology development extends beyond material properties to enabling complete alkaline membrane electrode assembly architectures that can operate efficiently at lower temperatures (<80°C) and with reduced catalyst loading. This holistic approach recognizes that membrane performance cannot be optimized in isolation but must be considered within the context of the entire fuel cell system.
The trajectory of AEM development has been characterized by progressive improvements in ionic conductivity, chemical stability, and mechanical durability. Early iterations suffered from rapid degradation in alkaline conditions, limiting practical applications. Recent advancements have focused on novel polymer architectures and functional group designs that enhance hydroxide ion transport while maintaining structural integrity under operating conditions.
Current technological trends indicate a shift toward multifunctional membrane designs that simultaneously address multiple performance parameters. These include the development of block copolymers with segregated hydrophilic and hydrophobic domains, crosslinked networks for enhanced dimensional stability, and composite materials incorporating inorganic components for improved conductivity and durability.
The primary technical objective in AEM research is to achieve hydroxide conductivity comparable to proton conductivity in acidic systems (>100 mS/cm) while maintaining chemical stability in high pH environments for extended periods (>5000 hours). Secondary objectives include reducing membrane swelling, enhancing mechanical properties under varying hydration conditions, and developing cost-effective manufacturing processes suitable for commercial scale production.
Beyond performance metrics, researchers aim to establish standardized testing protocols for meaningful comparison across different membrane systems. This standardization is crucial for accelerating technology development and facilitating industry adoption. Additionally, there is growing interest in understanding degradation mechanisms at the molecular level to inform rational design strategies for next-generation materials.
The broader goal of AEM technology development extends beyond material properties to enabling complete alkaline membrane electrode assembly architectures that can operate efficiently at lower temperatures (<80°C) and with reduced catalyst loading. This holistic approach recognizes that membrane performance cannot be optimized in isolation but must be considered within the context of the entire fuel cell system.
Market Analysis for Alkaline Fuel Cell Applications
The alkaline fuel cell (AFC) market is experiencing significant growth driven by increasing demand for clean energy solutions and the global push towards decarbonization. Current market valuations place the global fuel cell market at approximately 5.8 billion USD in 2023, with projections indicating growth to reach 34.2 billion USD by 2030, representing a compound annual growth rate (CAGR) of 25.4%. Within this broader market, AFCs are gaining traction due to their cost advantages over proton exchange membrane fuel cells (PEMFCs).
The transportation sector represents the largest potential market for alkaline fuel cell applications, particularly in heavy-duty vehicles, buses, and material handling equipment. This segment is expected to grow at a CAGR of 27.8% through 2030, driven by stringent emission regulations and government incentives for zero-emission vehicles. The European Union's commitment to reduce greenhouse gas emissions by 55% by 2030 has created a favorable regulatory environment for AFC adoption in transportation.
Stationary power generation constitutes the second-largest application segment, with particular growth in backup power systems, distributed generation, and off-grid applications. This market is projected to reach 9.7 billion USD by 2030, growing at a CAGR of 23.6%. Regions with unreliable grid infrastructure, particularly in developing economies across Asia-Pacific and Africa, present substantial opportunities for AFC deployment.
Portable applications represent a smaller but rapidly growing segment, with applications in military equipment, remote monitoring systems, and consumer electronics. This segment is expected to grow at a CAGR of 29.2%, reaching 3.8 billion USD by 2030, driven by increasing demand for reliable portable power sources.
Geographically, Asia-Pacific dominates the AFC market, accounting for 42% of global market share, followed by Europe (31%) and North America (21%). China leads the Asia-Pacific region with substantial government support through its hydrogen strategy outlined in the 14th Five-Year Plan. Japan and South Korea have also established ambitious targets for fuel cell deployment, particularly in transportation and residential applications.
Key market drivers include decreasing costs of anion exchange membranes, increasing investment in hydrogen infrastructure, and supportive government policies. However, market challenges persist, including competition from battery electric technologies, hydrogen storage and distribution limitations, and durability concerns with current membrane technologies. The cost of platinum-free catalysts and manufacturing scalability remain critical factors influencing market penetration rates across different application segments.
The transportation sector represents the largest potential market for alkaline fuel cell applications, particularly in heavy-duty vehicles, buses, and material handling equipment. This segment is expected to grow at a CAGR of 27.8% through 2030, driven by stringent emission regulations and government incentives for zero-emission vehicles. The European Union's commitment to reduce greenhouse gas emissions by 55% by 2030 has created a favorable regulatory environment for AFC adoption in transportation.
Stationary power generation constitutes the second-largest application segment, with particular growth in backup power systems, distributed generation, and off-grid applications. This market is projected to reach 9.7 billion USD by 2030, growing at a CAGR of 23.6%. Regions with unreliable grid infrastructure, particularly in developing economies across Asia-Pacific and Africa, present substantial opportunities for AFC deployment.
Portable applications represent a smaller but rapidly growing segment, with applications in military equipment, remote monitoring systems, and consumer electronics. This segment is expected to grow at a CAGR of 29.2%, reaching 3.8 billion USD by 2030, driven by increasing demand for reliable portable power sources.
Geographically, Asia-Pacific dominates the AFC market, accounting for 42% of global market share, followed by Europe (31%) and North America (21%). China leads the Asia-Pacific region with substantial government support through its hydrogen strategy outlined in the 14th Five-Year Plan. Japan and South Korea have also established ambitious targets for fuel cell deployment, particularly in transportation and residential applications.
Key market drivers include decreasing costs of anion exchange membranes, increasing investment in hydrogen infrastructure, and supportive government policies. However, market challenges persist, including competition from battery electric technologies, hydrogen storage and distribution limitations, and durability concerns with current membrane technologies. The cost of platinum-free catalysts and manufacturing scalability remain critical factors influencing market penetration rates across different application segments.
Current Challenges in AEM Development for Alkaline Fuel Cells
Despite significant advancements in anion exchange membrane (AEM) technology for alkaline fuel cells, several critical challenges continue to impede widespread commercialization. The most persistent obstacle remains chemical stability, particularly in highly alkaline environments. Current AEMs suffer from degradation through nucleophilic displacement and Hofmann elimination reactions, which attack the cationic functional groups responsible for anion conductivity. This degradation significantly reduces membrane lifetime under operating conditions, with most contemporary membranes showing performance deterioration after only hundreds of hours rather than the thousands required for commercial viability.
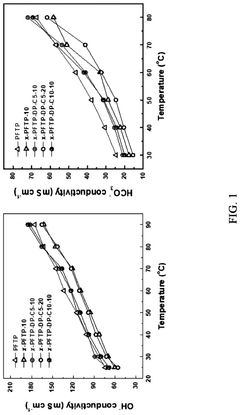
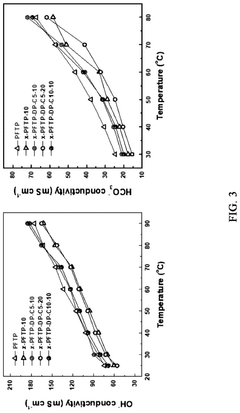
Ion conductivity presents another major challenge, as most AEMs exhibit hydroxide conductivity values between 10-100 mS/cm, substantially lower than the proton conductivity achieved in perfluorosulfonic acid membranes used in acidic fuel cells (approximately 100-200 mS/cm). This conductivity gap necessitates operation at higher temperatures or increased membrane hydration, both introducing additional system complexities.
Water management remains particularly problematic for AEM systems. The membrane must maintain sufficient hydration for ion transport while preventing flooding of electrodes. Unlike proton exchange membranes where water moves with proton transport, in AEMs, water and hydroxide ions move in opposite directions, creating complex hydration gradients that are difficult to manage during operation.
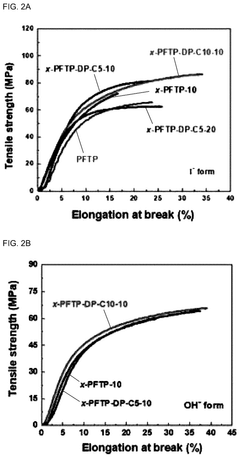
Mechanical stability under repeated hydration-dehydration cycles continues to challenge researchers. Many high-performance AEMs exhibit excessive swelling when hydrated, leading to dimensional instability that compromises fuel cell assembly integrity and increases the likelihood of mechanical failure during operation. Current membranes typically show 20-40% dimensional changes between dry and fully hydrated states, whereas commercial viability requires limiting this to below 10%.
Carbonation effects further complicate AEM operation, as atmospheric CO2 readily reacts with hydroxide ions to form carbonate and bicarbonate species with significantly lower mobility. This phenomenon reduces effective ionic conductivity and overall cell performance, particularly in systems exposed to air. Studies indicate conductivity losses of 30-60% when operating in ambient air versus CO2-free environments.
Manufacturing scalability presents additional barriers, as many laboratory-scale membranes utilize synthesis methods that are difficult to scale industrially. Complex multi-step reactions, expensive reagents, and environmentally problematic solvents limit commercial translation. The development of simplified, environmentally benign synthesis routes compatible with roll-to-roll processing remains an active research priority.
Cost considerations also pose significant challenges, with current high-performance AEMs utilizing expensive fluorinated polymers or complex architectures that increase material costs substantially above commercial targets. Achieving performance parity with less expensive materials represents a critical research direction for market viability.
Ion conductivity presents another major challenge, as most AEMs exhibit hydroxide conductivity values between 10-100 mS/cm, substantially lower than the proton conductivity achieved in perfluorosulfonic acid membranes used in acidic fuel cells (approximately 100-200 mS/cm). This conductivity gap necessitates operation at higher temperatures or increased membrane hydration, both introducing additional system complexities.
Water management remains particularly problematic for AEM systems. The membrane must maintain sufficient hydration for ion transport while preventing flooding of electrodes. Unlike proton exchange membranes where water moves with proton transport, in AEMs, water and hydroxide ions move in opposite directions, creating complex hydration gradients that are difficult to manage during operation.
Mechanical stability under repeated hydration-dehydration cycles continues to challenge researchers. Many high-performance AEMs exhibit excessive swelling when hydrated, leading to dimensional instability that compromises fuel cell assembly integrity and increases the likelihood of mechanical failure during operation. Current membranes typically show 20-40% dimensional changes between dry and fully hydrated states, whereas commercial viability requires limiting this to below 10%.
Carbonation effects further complicate AEM operation, as atmospheric CO2 readily reacts with hydroxide ions to form carbonate and bicarbonate species with significantly lower mobility. This phenomenon reduces effective ionic conductivity and overall cell performance, particularly in systems exposed to air. Studies indicate conductivity losses of 30-60% when operating in ambient air versus CO2-free environments.
Manufacturing scalability presents additional barriers, as many laboratory-scale membranes utilize synthesis methods that are difficult to scale industrially. Complex multi-step reactions, expensive reagents, and environmentally problematic solvents limit commercial translation. The development of simplified, environmentally benign synthesis routes compatible with roll-to-roll processing remains an active research priority.
Cost considerations also pose significant challenges, with current high-performance AEMs utilizing expensive fluorinated polymers or complex architectures that increase material costs substantially above commercial targets. Achieving performance parity with less expensive materials represents a critical research direction for market viability.
Current AEM Material Solutions and Fabrication Techniques
01 Polymer-based anion exchange membranes
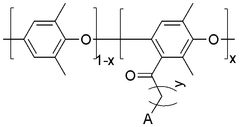
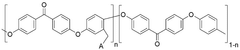
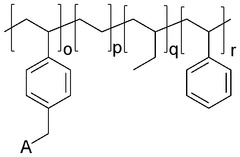
Polymer-based anion exchange membranes are developed using various polymer materials that provide ion conductivity while maintaining mechanical stability. These membranes typically incorporate quaternary ammonium functional groups that facilitate anion transport. The polymer backbone can be modified to enhance properties such as alkaline stability, conductivity, and durability. These membranes find applications in fuel cells, electrolyzers, and other electrochemical devices.- Polymer-based anion exchange membranes: Polymer-based anion exchange membranes are developed using various polymer materials that provide high ionic conductivity and chemical stability. These membranes incorporate functional groups that facilitate anion transport while maintaining mechanical strength. The polymer backbone structure can be modified to enhance durability and performance in different applications such as fuel cells and electrolysis systems.
- Functionalized anion exchange membranes: Functionalization techniques are employed to improve the properties of anion exchange membranes. This involves introducing specific functional groups onto the membrane structure to enhance ion conductivity, selectivity, and stability. Various chemical modifications can be applied to optimize membrane performance for specific applications, resulting in improved ion exchange capacity and reduced membrane swelling.
- Composite anion exchange membranes: Composite anion exchange membranes combine different materials to achieve enhanced properties that cannot be obtained from single-component membranes. These membranes typically consist of a polymer matrix reinforced with inorganic fillers or other supporting materials to improve mechanical strength, thermal stability, and ion conductivity. The composite structure helps to overcome limitations of conventional membranes while maintaining high performance in various electrochemical applications.
- Anion exchange membranes for water treatment: Anion exchange membranes designed specifically for water treatment applications focus on selective removal of anions from water sources. These membranes are engineered to have high selectivity for target contaminants such as nitrates, sulfates, or arsenic compounds. The membrane structure is optimized for durability in water treatment conditions while maintaining high flux rates and resistance to fouling during extended operation periods.
- Alkaline-stable anion exchange membranes: Developing anion exchange membranes with enhanced stability in alkaline environments is crucial for applications such as alkaline fuel cells and electrolyzers. These membranes incorporate specialized chemical structures that resist degradation under high pH conditions while maintaining ion conductivity. Various approaches include the use of sterically hindered cationic groups, reinforced polymer backbones, and protective coatings to extend membrane lifetime in aggressive alkaline environments.
02 Composite anion exchange membranes
Composite anion exchange membranes combine different materials to achieve enhanced performance characteristics. These membranes often incorporate inorganic fillers, such as metal oxides or nanoparticles, within a polymer matrix to improve mechanical strength, thermal stability, and ion conductivity. The composite structure can help reduce membrane swelling while maintaining high ion exchange capacity, making them suitable for applications requiring long-term stability under harsh conditions.Expand Specific Solutions03 Fabrication methods for anion exchange membranes
Various fabrication techniques are employed to produce anion exchange membranes with desired properties. These methods include solution casting, electrospinning, phase inversion, and radiation-induced grafting. The fabrication process significantly influences membrane morphology, which in turn affects properties such as ion conductivity, mechanical strength, and chemical stability. Advanced manufacturing approaches focus on creating membranes with controlled thickness, uniform distribution of functional groups, and optimized pore structure.Expand Specific Solutions04 Alkaline stability enhancement in anion exchange membranes
Improving the alkaline stability of anion exchange membranes is crucial for their long-term performance in high pH environments. Various approaches include the development of sterically hindered cationic groups, incorporation of stabilizing additives, and design of protective polymer architectures. Enhanced alkaline stability prevents degradation mechanisms such as Hofmann elimination and nucleophilic substitution, thereby extending membrane lifetime and maintaining consistent ion conductivity during operation.Expand Specific Solutions05 Applications of anion exchange membranes in energy conversion and storage
Anion exchange membranes are widely used in various energy conversion and storage technologies. Key applications include alkaline fuel cells, water electrolyzers, redox flow batteries, and desalination systems. In these applications, the membranes facilitate selective ion transport while preventing crossover of reactants. The development of high-performance anion exchange membranes has enabled advancements in renewable energy technologies by improving efficiency, reducing costs, and enhancing system durability.Expand Specific Solutions
Leading Manufacturers and Research Institutions in AEM Field
The anion exchange membrane (AEM) alkaline fuel cell market is in a growth phase, characterized by increasing research intensity and commercial interest. The market size is expanding due to the technology's potential as a cost-effective alternative to proton exchange membrane fuel cells, with projections suggesting significant growth in the next decade. Technologically, the field shows moderate maturity with key players at different development stages. Companies like Tokuyama Corp., Solvay SA, and Nitto Denko lead commercial development with established membrane technologies, while academic institutions such as Vanderbilt University and Tokyo Institute of Technology drive fundamental research innovations. Industrial players including BASF, Honda Motor, and AGC Engineering are actively developing application-specific solutions, indicating the technology's transition from research to commercial implementation.
Tokuyama Corp.
Technical Solution: Tokuyama Corporation has pioneered the development of hydrocarbon-based anion exchange membranes featuring radiation-grafted quaternary ammonium functional groups. Their proprietary A201 and A901 membranes utilize a chloromethylation process followed by quaternization with trimethylamine to achieve ion exchange capacities of 1.5-2.0 meq/g. The company employs a unique cross-linking strategy that creates covalent bonds between polymer chains, significantly enhancing chemical stability in alkaline environments. Tokuyama's manufacturing process involves precision extrusion and controlled film formation techniques that ensure uniform thickness distribution (typically 25-30 μm) and consistent mechanical properties. Their membranes demonstrate hydroxide conductivity of 30-40 mS/cm at 60°C while maintaining dimensional stability below 10% swelling. Recent advancements include the incorporation of nanocomposite materials to further enhance durability and conductivity performance.
Strengths: Established commercial production capabilities; consistent quality control; extensive performance validation data from multiple research institutions. Weaknesses: Lower hydroxide conductivity compared to some newer materials; potential for mechanical degradation during extended cycling; higher water uptake leading to dimensional stability challenges.
Solvay SA
Technical Solution: Solvay has developed advanced perfluorinated anion exchange membranes based on their expertise in fluoropolymer chemistry. Their technology utilizes side-chain grafting of quaternary ammonium groups onto ETFE (ethylene tetrafluoroethylene) backbones through radiation-induced grafting processes. This approach creates membranes with exceptional chemical stability in alkaline environments while maintaining high hydroxide conductivity (60-80 mS/cm at 80°C). Solvay's manufacturing process employs controlled radiation exposure followed by precise grafting reaction conditions to achieve uniform functional group distribution throughout the membrane thickness (20-40 μm). Their membranes demonstrate remarkable dimensional stability with swelling limited to 15-20% even in fully hydrated states. Solvay has also developed specialized reinforcement techniques incorporating porous support structures that enhance mechanical durability while maintaining high ionic conductivity, enabling operation at higher pressures and temperature differentials in fuel cell systems.
Strengths: Exceptional chemical stability in alkaline environments; established fluoropolymer manufacturing expertise; strong intellectual property portfolio. Weaknesses: Higher production costs due to specialized fluoropolymer materials; challenges with scaling radiation grafting processes; potential environmental concerns with fluorinated materials.
Key Patents and Scientific Breakthroughs in AEM Technology
Novel polyfluorene-based cross-linked copolymer, method for producing same, and anion exchange membrane for alkaline fuel cell using same
PatentPendingUS20240317951A1
Innovation
- A polyfluorene-based cross-linked copolymer with a piperidinium group in its repeating unit and without an aryl ether bond is synthesized, forming a cross-linked anion exchange membrane that enhances mechanical strength, water-holding capacity, and durability, using an ammonium-based crosslinking agent and specific organic solvents for preparation.
Anion exchange composite membrane, manufacturing method therefor, and alkaline fuel cell comprising same
PatentWO2022270934A1
Innovation
- A composite membrane is developed using a porous support made of polyphenylene sulfide (PPS) with an anion exchange polymer having a high content of anion exchange groups, which is impregnated onto the PPS support to enhance mechanical, chemical, and dimensional stability, and long-term stability, improving the membrane's performance.
Durability and Stability Assessment of Anion Exchange Membranes
The durability and stability of anion exchange membranes (AEMs) remain critical challenges that significantly impact the commercial viability of alkaline fuel cells. Current AEMs typically demonstrate operational lifetimes of 1,000-5,000 hours under laboratory conditions, falling short of the 40,000+ hours required for practical applications such as automotive or stationary power generation. This performance gap represents one of the most significant barriers to widespread AEM fuel cell adoption.
Chemical degradation mechanisms constitute the primary failure modes for AEMs. The hydroxide ions that enable ionic conductivity simultaneously attack the polymer backbone and functional groups through nucleophilic substitution and Hofmann elimination reactions. These processes progressively reduce ion exchange capacity, resulting in diminished conductivity over time. Additionally, the quaternary ammonium groups commonly employed as ion-conducting sites are particularly vulnerable to hydroxide attack at elevated temperatures.
Environmental factors further exacerbate stability issues. Humidity fluctuations cause dimensional changes through swelling and contraction cycles, leading to mechanical stress and eventual membrane failure. Temperature variations accelerate chemical degradation rates, with most current AEMs showing rapid performance decline above 60°C, limiting operational temperature ranges and system efficiency.
Mechanical stability presents another significant challenge. The inherent trade-off between water uptake (necessary for ion conductivity) and mechanical integrity creates a fundamental design dilemma. Highly conductive membranes with sufficient water content often exhibit poor mechanical properties, while mechanically robust membranes typically demonstrate insufficient conductivity. This balance must be carefully managed through innovative material design.
Recent advances in stabilization strategies have shown promising results. The incorporation of sterically hindered cations, such as piperidinium and pyrrolidinium groups, has demonstrated enhanced alkaline stability compared to traditional trimethylammonium functionalities. Cross-linking techniques have improved mechanical durability while maintaining acceptable conductivity levels. Additionally, composite approaches incorporating inorganic nanoparticles have shown synergistic improvements in both mechanical and chemical stability.
Standardized testing protocols represent another critical need in the field. The diversity of accelerated stress tests and operating conditions across research groups makes direct comparisons challenging. Establishing industry-wide standards for durability assessment would facilitate more meaningful benchmarking and accelerate development cycles. Current efforts by organizations such as the Fuel Cell Technologies Office are working toward this standardization.
Chemical degradation mechanisms constitute the primary failure modes for AEMs. The hydroxide ions that enable ionic conductivity simultaneously attack the polymer backbone and functional groups through nucleophilic substitution and Hofmann elimination reactions. These processes progressively reduce ion exchange capacity, resulting in diminished conductivity over time. Additionally, the quaternary ammonium groups commonly employed as ion-conducting sites are particularly vulnerable to hydroxide attack at elevated temperatures.
Environmental factors further exacerbate stability issues. Humidity fluctuations cause dimensional changes through swelling and contraction cycles, leading to mechanical stress and eventual membrane failure. Temperature variations accelerate chemical degradation rates, with most current AEMs showing rapid performance decline above 60°C, limiting operational temperature ranges and system efficiency.
Mechanical stability presents another significant challenge. The inherent trade-off between water uptake (necessary for ion conductivity) and mechanical integrity creates a fundamental design dilemma. Highly conductive membranes with sufficient water content often exhibit poor mechanical properties, while mechanically robust membranes typically demonstrate insufficient conductivity. This balance must be carefully managed through innovative material design.
Recent advances in stabilization strategies have shown promising results. The incorporation of sterically hindered cations, such as piperidinium and pyrrolidinium groups, has demonstrated enhanced alkaline stability compared to traditional trimethylammonium functionalities. Cross-linking techniques have improved mechanical durability while maintaining acceptable conductivity levels. Additionally, composite approaches incorporating inorganic nanoparticles have shown synergistic improvements in both mechanical and chemical stability.
Standardized testing protocols represent another critical need in the field. The diversity of accelerated stress tests and operating conditions across research groups makes direct comparisons challenging. Establishing industry-wide standards for durability assessment would facilitate more meaningful benchmarking and accelerate development cycles. Current efforts by organizations such as the Fuel Cell Technologies Office are working toward this standardization.
Environmental Impact and Sustainability of AEM Fuel Cell Systems
The environmental impact of Anion Exchange Membrane (AEM) fuel cell systems represents a critical consideration in their development and deployment. AEM fuel cells offer significant sustainability advantages over conventional energy technologies and even other fuel cell types. Their operation produces only water and heat as byproducts, eliminating harmful emissions such as nitrogen oxides, sulfur oxides, and particulate matter that characterize fossil fuel combustion.
When examining the full lifecycle environmental footprint, AEM fuel cells demonstrate promising sustainability metrics. The membrane materials, typically composed of polymers with quaternary ammonium functional groups, can be synthesized using increasingly green chemistry approaches. Recent advances in membrane fabrication have reduced dependence on toxic solvents and energy-intensive processing steps, further enhancing their environmental credentials.
The hydrogen production pathway significantly influences the overall sustainability of AEM fuel cell systems. While traditional hydrogen production through steam methane reforming carries a substantial carbon footprint, green hydrogen generated via water electrolysis powered by renewable energy represents a truly sustainable fuel pathway. The compatibility of AEM fuel cells with non-platinum catalysts further reduces environmental impact by decreasing reliance on scarce noble metals.
End-of-life considerations for AEM fuel cell systems have received increasing attention. Research into recyclable membrane materials and recovery processes for valuable components has advanced significantly. Some polymer backbones used in AEMs can be chemically recycled, and catalysts can be recovered through established processes, minimizing waste and resource depletion.
From a broader sustainability perspective, AEM fuel cells contribute to energy system resilience and decarbonization goals. Their potential integration with intermittent renewable energy sources provides a pathway for energy storage and grid stabilization without the environmental drawbacks of battery technologies, particularly regarding resource extraction and disposal challenges.
Water management in AEM fuel cells presents both challenges and opportunities for sustainability. While these systems require water for operation, they generate clean water as a byproduct, potentially offering integrated water recovery in certain applications. This aspect is particularly valuable in water-stressed regions where combined energy and water solutions are increasingly sought.
The manufacturing scalability of AEM fuel cells also influences their sustainability profile. Current research focuses on developing production methods that minimize energy consumption and waste generation while maintaining performance standards. Advances in roll-to-roll processing and additive manufacturing techniques show promise for reducing the environmental footprint of mass production.
When examining the full lifecycle environmental footprint, AEM fuel cells demonstrate promising sustainability metrics. The membrane materials, typically composed of polymers with quaternary ammonium functional groups, can be synthesized using increasingly green chemistry approaches. Recent advances in membrane fabrication have reduced dependence on toxic solvents and energy-intensive processing steps, further enhancing their environmental credentials.
The hydrogen production pathway significantly influences the overall sustainability of AEM fuel cell systems. While traditional hydrogen production through steam methane reforming carries a substantial carbon footprint, green hydrogen generated via water electrolysis powered by renewable energy represents a truly sustainable fuel pathway. The compatibility of AEM fuel cells with non-platinum catalysts further reduces environmental impact by decreasing reliance on scarce noble metals.
End-of-life considerations for AEM fuel cell systems have received increasing attention. Research into recyclable membrane materials and recovery processes for valuable components has advanced significantly. Some polymer backbones used in AEMs can be chemically recycled, and catalysts can be recovered through established processes, minimizing waste and resource depletion.
From a broader sustainability perspective, AEM fuel cells contribute to energy system resilience and decarbonization goals. Their potential integration with intermittent renewable energy sources provides a pathway for energy storage and grid stabilization without the environmental drawbacks of battery technologies, particularly regarding resource extraction and disposal challenges.
Water management in AEM fuel cells presents both challenges and opportunities for sustainability. While these systems require water for operation, they generate clean water as a byproduct, potentially offering integrated water recovery in certain applications. This aspect is particularly valuable in water-stressed regions where combined energy and water solutions are increasingly sought.
The manufacturing scalability of AEM fuel cells also influences their sustainability profile. Current research focuses on developing production methods that minimize energy consumption and waste generation while maintaining performance standards. Advances in roll-to-roll processing and additive manufacturing techniques show promise for reducing the environmental footprint of mass production.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!