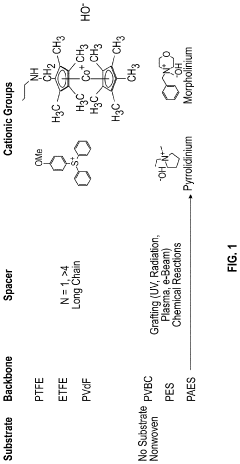
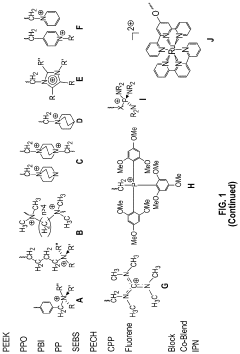
Degradation pathways of cationic groups in AEMs
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AEM Cationic Degradation Background and Objectives
Anion Exchange Membranes (AEMs) have emerged as critical components in various electrochemical energy conversion and storage technologies, including fuel cells, electrolyzers, and flow batteries. The development of AEMs has gained significant momentum over the past two decades as researchers seek alternatives to proton exchange membrane systems. The primary advantage of AEMs lies in their ability to operate under alkaline conditions, enabling the use of non-precious metal catalysts and potentially reducing system costs.
The cationic functional groups in AEMs play a pivotal role in facilitating hydroxide ion transport. Historically, quaternary ammonium (QA) groups have been the most widely employed cationic moieties due to their relatively straightforward synthesis and acceptable initial conductivity. However, the degradation of these cationic groups under operating conditions has emerged as one of the most significant barriers to widespread AEM technology adoption.
The evolution of AEM technology can be traced back to the early 2000s when researchers began exploring alternatives to perfluorosulfonic acid membranes. Initial efforts focused on simple polystyrene-based materials with pendant QA groups, which demonstrated proof of concept but suffered from rapid degradation. By the 2010s, more chemically stable polymer backbones were introduced, yet cationic group stability remained problematic.
Current technical objectives in this field center on understanding the fundamental degradation mechanisms of cationic groups under alkaline conditions and elevated temperatures. Researchers aim to elucidate the specific chemical pathways through which degradation occurs, including nucleophilic substitution, Hofmann elimination, and radical-induced degradation. This mechanistic understanding is essential for designing more stable cationic functionalities.
The field has progressed from merely observing degradation to actively investigating its molecular basis through advanced characterization techniques such as NMR spectroscopy, mass spectrometry, and computational modeling. These tools have revealed that degradation pathways are complex and often interdependent, necessitating a multifaceted approach to enhance stability.
The ultimate technical goal is to develop cationic groups that maintain high hydroxide conductivity while resisting degradation under operating conditions of 60-80°C in alkaline environments for thousands of hours. This requires innovative molecular designs that can mitigate known degradation mechanisms while maintaining the essential ion transport properties required for efficient electrochemical device operation.
Recent research has begun exploring alternative cationic structures beyond traditional QA groups, including guanidinium, phosphonium, and metal-coordinated complexes, each offering potential advantages in terms of chemical stability or conductivity. The field is now at a critical juncture where fundamental understanding of degradation pathways must translate into practical, durable materials for commercial applications.
The cationic functional groups in AEMs play a pivotal role in facilitating hydroxide ion transport. Historically, quaternary ammonium (QA) groups have been the most widely employed cationic moieties due to their relatively straightforward synthesis and acceptable initial conductivity. However, the degradation of these cationic groups under operating conditions has emerged as one of the most significant barriers to widespread AEM technology adoption.
The evolution of AEM technology can be traced back to the early 2000s when researchers began exploring alternatives to perfluorosulfonic acid membranes. Initial efforts focused on simple polystyrene-based materials with pendant QA groups, which demonstrated proof of concept but suffered from rapid degradation. By the 2010s, more chemically stable polymer backbones were introduced, yet cationic group stability remained problematic.
Current technical objectives in this field center on understanding the fundamental degradation mechanisms of cationic groups under alkaline conditions and elevated temperatures. Researchers aim to elucidate the specific chemical pathways through which degradation occurs, including nucleophilic substitution, Hofmann elimination, and radical-induced degradation. This mechanistic understanding is essential for designing more stable cationic functionalities.
The field has progressed from merely observing degradation to actively investigating its molecular basis through advanced characterization techniques such as NMR spectroscopy, mass spectrometry, and computational modeling. These tools have revealed that degradation pathways are complex and often interdependent, necessitating a multifaceted approach to enhance stability.
The ultimate technical goal is to develop cationic groups that maintain high hydroxide conductivity while resisting degradation under operating conditions of 60-80°C in alkaline environments for thousands of hours. This requires innovative molecular designs that can mitigate known degradation mechanisms while maintaining the essential ion transport properties required for efficient electrochemical device operation.
Recent research has begun exploring alternative cationic structures beyond traditional QA groups, including guanidinium, phosphonium, and metal-coordinated complexes, each offering potential advantages in terms of chemical stability or conductivity. The field is now at a critical juncture where fundamental understanding of degradation pathways must translate into practical, durable materials for commercial applications.
Market Analysis for AEM Technologies
The global market for Anion Exchange Membrane (AEM) technologies has been experiencing significant growth, driven primarily by increasing demand for clean energy solutions and sustainable chemical production processes. The market size for AEM technologies was valued at approximately $320 million in 2022 and is projected to reach $1.2 billion by 2030, representing a compound annual growth rate (CAGR) of 17.8% during the forecast period.
The fuel cell segment currently dominates the AEM technology market, accounting for over 45% of the total market share. This dominance is attributed to the growing adoption of hydrogen fuel cells in transportation, stationary power generation, and portable electronics. The water electrolysis segment follows closely, driven by the increasing focus on green hydrogen production as countries worldwide commit to decarbonization targets.
Geographically, North America and Europe lead the AEM technology market, collectively holding approximately 65% of the global market share. However, the Asia-Pacific region is expected to witness the fastest growth rate during the forecast period, primarily due to substantial investments in renewable energy infrastructure in countries like China, Japan, and South Korea.
Key market drivers include stringent environmental regulations, increasing investments in renewable energy, and growing demand for energy storage solutions. The push for carbon neutrality by major economies has significantly accelerated the adoption of AEM technologies across various industries. Additionally, the declining costs of renewable energy generation have made AEM-based hydrogen production more economically viable.
Despite the promising growth prospects, the market faces several challenges. The degradation of cationic groups in AEMs remains a significant technical barrier, limiting the operational lifetime and performance of AEM-based systems. This degradation issue directly impacts market adoption rates and commercial viability of AEM technologies in long-term applications.
Industry experts anticipate that technological advancements addressing the stability of cationic groups could potentially unlock an additional $500 million market opportunity by 2028. Companies that successfully develop degradation-resistant AEMs are expected to gain substantial competitive advantages and market share.
The competitive landscape is characterized by a mix of established chemical companies, specialized membrane manufacturers, and emerging startups. Strategic partnerships between technology developers and end-users are becoming increasingly common as the industry works to overcome technical challenges and accelerate commercialization.
The fuel cell segment currently dominates the AEM technology market, accounting for over 45% of the total market share. This dominance is attributed to the growing adoption of hydrogen fuel cells in transportation, stationary power generation, and portable electronics. The water electrolysis segment follows closely, driven by the increasing focus on green hydrogen production as countries worldwide commit to decarbonization targets.
Geographically, North America and Europe lead the AEM technology market, collectively holding approximately 65% of the global market share. However, the Asia-Pacific region is expected to witness the fastest growth rate during the forecast period, primarily due to substantial investments in renewable energy infrastructure in countries like China, Japan, and South Korea.
Key market drivers include stringent environmental regulations, increasing investments in renewable energy, and growing demand for energy storage solutions. The push for carbon neutrality by major economies has significantly accelerated the adoption of AEM technologies across various industries. Additionally, the declining costs of renewable energy generation have made AEM-based hydrogen production more economically viable.
Despite the promising growth prospects, the market faces several challenges. The degradation of cationic groups in AEMs remains a significant technical barrier, limiting the operational lifetime and performance of AEM-based systems. This degradation issue directly impacts market adoption rates and commercial viability of AEM technologies in long-term applications.
Industry experts anticipate that technological advancements addressing the stability of cationic groups could potentially unlock an additional $500 million market opportunity by 2028. Companies that successfully develop degradation-resistant AEMs are expected to gain substantial competitive advantages and market share.
The competitive landscape is characterized by a mix of established chemical companies, specialized membrane manufacturers, and emerging startups. Strategic partnerships between technology developers and end-users are becoming increasingly common as the industry works to overcome technical challenges and accelerate commercialization.
Current Challenges in AEM Cationic Stability
Despite significant advancements in anion exchange membrane (AEM) technology, the stability of cationic groups remains a critical challenge that limits widespread commercial adoption. The degradation of these functional groups occurs through several well-documented pathways, with nucleophilic substitution and Hofmann elimination being the most prevalent mechanisms. Under alkaline conditions, hydroxide ions attack quaternary ammonium groups, leading to progressive degradation and ultimately compromising membrane performance.
The nucleophilic substitution pathway involves direct attack of hydroxide ions on the α-carbon adjacent to the quaternary nitrogen, resulting in demethylation and formation of tertiary amines. These degraded species exhibit lower ionic conductivity and reduced mechanical stability. Research has shown that this pathway is particularly problematic at operating temperatures above 60°C, creating a significant barrier for high-temperature applications.
Hofmann elimination represents another major degradation route, wherein hydroxide ions abstract β-hydrogen atoms, leading to the formation of alkenes and tertiary amines. This mechanism is especially pronounced in cations with β-hydrogens aligned perpendicular to the nitrogen p-orbital, facilitating E2 elimination reactions. The resulting chemical changes not only reduce ion exchange capacity but also create hydrophobic regions within the membrane that impair water management.
Recent studies have identified additional degradation mechanisms including ylide formation, where carbanions form at the α-carbon position and subsequently undergo rearrangement or further reactions. This pathway becomes particularly relevant in sterically hindered cationic structures where traditional degradation routes are partially suppressed.
The rate of degradation is heavily influenced by environmental factors including temperature, humidity levels, and mechanical stress. Accelerated testing protocols have revealed that degradation rates increase exponentially with temperature, following Arrhenius behavior. This temperature sensitivity creates a challenging trade-off between operational efficiency and membrane longevity.
Current commercial AEMs typically demonstrate stability for only 2,000-5,000 hours under realistic operating conditions, falling significantly short of the 40,000+ hours required for many practical applications. This stability gap represents perhaps the most significant barrier to widespread AEM adoption in fuel cells, electrolyzers, and other electrochemical devices.
The molecular architecture surrounding the cationic group substantially impacts degradation resistance. Bulky substituents that shield the cation center have shown promise in enhancing stability, but often at the expense of ionic conductivity. This fundamental trade-off between stability and performance continues to challenge researchers developing next-generation materials.
The nucleophilic substitution pathway involves direct attack of hydroxide ions on the α-carbon adjacent to the quaternary nitrogen, resulting in demethylation and formation of tertiary amines. These degraded species exhibit lower ionic conductivity and reduced mechanical stability. Research has shown that this pathway is particularly problematic at operating temperatures above 60°C, creating a significant barrier for high-temperature applications.
Hofmann elimination represents another major degradation route, wherein hydroxide ions abstract β-hydrogen atoms, leading to the formation of alkenes and tertiary amines. This mechanism is especially pronounced in cations with β-hydrogens aligned perpendicular to the nitrogen p-orbital, facilitating E2 elimination reactions. The resulting chemical changes not only reduce ion exchange capacity but also create hydrophobic regions within the membrane that impair water management.
Recent studies have identified additional degradation mechanisms including ylide formation, where carbanions form at the α-carbon position and subsequently undergo rearrangement or further reactions. This pathway becomes particularly relevant in sterically hindered cationic structures where traditional degradation routes are partially suppressed.
The rate of degradation is heavily influenced by environmental factors including temperature, humidity levels, and mechanical stress. Accelerated testing protocols have revealed that degradation rates increase exponentially with temperature, following Arrhenius behavior. This temperature sensitivity creates a challenging trade-off between operational efficiency and membrane longevity.
Current commercial AEMs typically demonstrate stability for only 2,000-5,000 hours under realistic operating conditions, falling significantly short of the 40,000+ hours required for many practical applications. This stability gap represents perhaps the most significant barrier to widespread AEM adoption in fuel cells, electrolyzers, and other electrochemical devices.
The molecular architecture surrounding the cationic group substantially impacts degradation resistance. Bulky substituents that shield the cation center have shown promise in enhancing stability, but often at the expense of ionic conductivity. This fundamental trade-off between stability and performance continues to challenge researchers developing next-generation materials.
Established Approaches to Mitigate Cationic Degradation
01 Quaternary ammonium degradation mechanisms
Quaternary ammonium (QA) cationic groups in anion exchange membranes are susceptible to degradation through several pathways, primarily nucleophilic substitution and Hofmann elimination reactions. These degradation mechanisms are accelerated in alkaline environments, where hydroxide ions attack the β-hydrogen atoms adjacent to the quaternary nitrogen, leading to the formation of tertiary amines and alkenes. This degradation significantly reduces membrane conductivity and mechanical stability over time, limiting the operational lifetime of AEMs in fuel cell applications.- Quaternary ammonium degradation mechanisms: Quaternary ammonium (QA) groups are commonly used cationic groups in AEMs but are susceptible to degradation through several pathways. The main degradation mechanisms include nucleophilic substitution (SN2) reactions, Hofmann elimination, and direct hydroxide attack. These reactions are accelerated at elevated temperatures and high pH environments, leading to decreased ion conductivity and mechanical stability of the membrane. Understanding these degradation pathways is crucial for developing more stable AEM materials.
- Stabilized cationic group structures: Various structural modifications have been developed to enhance the stability of cationic groups in AEMs. These include sterically hindered quaternary ammonium groups, resonance-stabilized structures, and multi-cationic centers. Incorporating bulky substituents around the cationic center can prevent nucleophilic attack, while resonance stabilization distributes the positive charge, reducing susceptibility to degradation. These structural improvements significantly extend the operational lifetime of AEMs under alkaline conditions.
- Alternative cationic functional groups: Beyond traditional quaternary ammonium groups, alternative cationic functionalities have been explored to improve AEM stability. These include phosphonium, imidazolium, pyridinium, guanidinium, and metal-organic complexes. Each offers distinct advantages in terms of chemical stability, with some showing significantly improved resistance to degradation under alkaline conditions. The selection of appropriate cationic groups depends on the specific application requirements and operating conditions of the membrane.
- Polymer backbone effects on cation stability: The polymer backbone structure significantly influences the stability of attached cationic groups in AEMs. Aromatic backbones generally provide better stability than aliphatic ones, while fluorinated polymers can enhance resistance to hydroxide attack. The distance between the cationic group and the backbone, as well as the flexibility of the connecting spacer, affects degradation rates. Optimizing these structural parameters can mitigate degradation pathways and improve overall membrane durability.
- Environmental factors affecting degradation: Several environmental factors accelerate the degradation of cationic groups in AEMs. Temperature has a significant impact, with degradation rates increasing exponentially at elevated temperatures. Humidity levels, mechanical stress, and the presence of contaminants also influence degradation pathways. Understanding these factors helps in developing testing protocols that accurately predict membrane lifetime and in designing operational strategies that minimize degradation under real-world conditions.
02 Stabilized cationic group structures
Novel cationic group structures have been developed to enhance the alkaline stability of AEMs. These include sterically hindered quaternary ammonium groups, where bulky substituents protect the cationic center from nucleophilic attack, and resonance-stabilized structures such as imidazolium, pyridinium, and guanidinium groups. The incorporation of electron-withdrawing or electron-donating groups strategically positioned around the cationic center can also modify electron density distribution, thereby reducing susceptibility to degradation pathways and extending membrane lifetime under alkaline conditions.Expand Specific Solutions03 Polymer backbone effects on cation stability
The chemical structure of the polymer backbone significantly influences the stability of attached cationic groups in AEMs. Aromatic backbones generally provide better stability than aliphatic ones due to their rigidity and ability to disperse charge. The distance between the polymer backbone and the cationic group (spacer length) affects degradation rates, with optimal spacer lengths balancing flexibility and stability. Cross-linking strategies can also enhance mechanical stability and reduce water uptake, which indirectly improves cation stability by limiting exposure to hydroxide ions and reducing membrane swelling.Expand Specific Solutions04 Environmental factors affecting degradation
Environmental conditions significantly impact the degradation rates of cationic groups in AEMs. Higher temperatures accelerate all degradation mechanisms, particularly Hofmann elimination. Humidity levels affect water uptake and ion mobility, influencing the concentration of hydroxide ions near cationic sites. The presence of metal impurities can catalyze degradation reactions, while mechanical stress from swelling/deswelling cycles can create microcracks that expose more cationic sites to degradative species. Understanding these environmental factors is crucial for predicting membrane lifetime and developing appropriate mitigation strategies.Expand Specific Solutions05 Novel cationic chemistries for enhanced stability
Emerging research focuses on developing novel cationic chemistries with inherently higher alkaline stability. Metal-organic frameworks incorporating cationic sites show promising stability characteristics. Phosphonium and sulfonium-based cations offer alternative degradation profiles compared to ammonium groups. Hybrid structures combining different types of cationic groups can provide synergistic stability effects. Reversible cation formation mechanisms are being explored to enable self-healing capabilities in membranes, where degraded cationic sites can regenerate under certain conditions, significantly extending the operational lifetime of AEMs in electrochemical devices.Expand Specific Solutions
Leading Organizations in AEM Development
The degradation of cationic groups in Anion Exchange Membranes (AEMs) represents a critical challenge in the emerging clean energy sector. The market is currently in an early growth phase, with increasing investments as hydrogen technologies gain traction. Academic institutions (Yale, MIT, University of California) are driving fundamental research, while commercial players like Ionomr Innovations, 1s1 Energy, and Nitto Denko are advancing practical applications. Large corporations including Siemens, 3M, and China Petroleum & Chemical Corp are strategically positioning themselves in this space. The technology remains in development stage, with most solutions at TRL 4-6, as researchers work to overcome stability limitations that currently restrict widespread commercialization of AEM-based systems for energy applications.
Yale University
Technical Solution: Yale University researchers have made significant advances in understanding and mitigating degradation pathways of cationic groups in AEMs. Their approach focuses on developing novel cationic chemistries beyond traditional quaternary ammonium compounds, including phosphonium, guanidinium, and imidazolium-based structures with enhanced alkaline stability. Yale's research has demonstrated that strategic placement of electron-withdrawing groups near cationic centers can significantly reduce susceptibility to hydroxide attack by decreasing electron density at vulnerable sites. Their technology incorporates cations directly attached to aromatic rings rather than through alkyl spacers, eliminating vulnerable β-hydrogen positions commonly associated with Hofmann elimination. Yale has also pioneered the development of sterically hindered cations where bulky substituents physically block nucleophilic attack pathways. Additionally, their research has revealed that controlling membrane hydration through precise tuning of hydrophilic/hydrophobic balance can significantly impact degradation rates by modulating local hydroxide concentration around cationic sites. Testing has shown membranes based on their most stable cationic structures retain over 85% conductivity after 1,000 hours at 80°C in 1M KOH solutions.
Strengths: Cutting-edge fundamental research on novel cationic structures; excellent mechanistic understanding of degradation pathways; innovative approaches to molecular design. Weaknesses: Current focus more on fundamental understanding than commercial application; potential challenges in scaling laboratory syntheses; higher complexity in membrane fabrication compared to conventional systems.
Ionomr Innovations, Inc.
Technical Solution: Ionomr has developed advanced hydrocarbon-based anion exchange membranes (AEMs) with enhanced stability against degradation of cationic groups. Their proprietary Aemion+® technology incorporates sterically hindered cations that significantly reduce susceptibility to nucleophilic attack and β-hydrogen elimination, two primary degradation pathways in AEMs. The company's approach involves embedding quaternary ammonium groups within a rigid polymer backbone structure that limits conformational mobility, thereby preventing the E2 elimination mechanism. Additionally, Ionomr has implemented specialized fluorinated polymer matrices that provide hydrophobic environments around the cationic groups, reducing hydroxide ion concentration in their vicinity and consequently slowing degradation rates. Their membranes demonstrate stability at temperatures up to 80°C under alkaline conditions for over 4,000 hours, representing a significant improvement over conventional AEMs.
Strengths: Superior chemical stability in alkaline environments; reduced degradation rates at elevated temperatures; maintains high ionic conductivity despite structural modifications for stability. Weaknesses: Higher manufacturing costs compared to traditional membranes; potential trade-offs between mechanical strength and ion conductivity; limited long-term operational data beyond laboratory testing conditions.
Critical Patents and Literature on AEM Stability
Energy conversion devices including stable ionenes
PatentWO2017190234A1
Innovation
- A catalyst-coated membrane featuring a random copolymer with specific anion counterbalances and degree of methylation, combined with a catalyst coating, which enhances chemical stability, mechanical properties, and conductivity, allowing for scalable production and resistance to CO2 impurities.
Systems including ion exchange membranes and methods of making the same
PatentActiveUS20220384833A1
Innovation
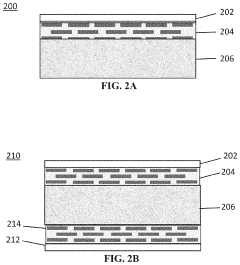
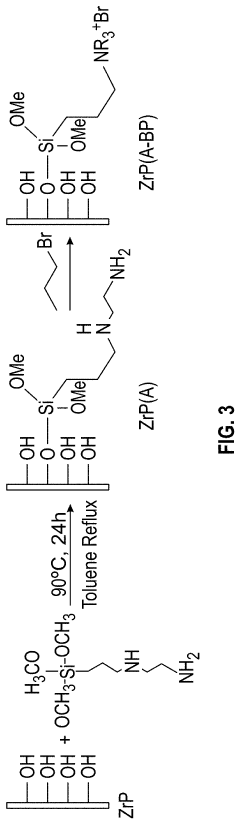
- A composite anion exchange membrane structure is developed, incorporating a layer of inorganic nanosheets, such as ZrP or LDH, on one or both sides of a polymeric AEM layer, along with a porous polymer binder layer, to enhance chemical stability and oxidation resistance.
Environmental Impact of AEM Materials
The environmental impact of Anion Exchange Membrane (AEM) materials is increasingly becoming a critical consideration in the development and deployment of these technologies. As cationic groups in AEMs undergo degradation through various pathways, they release potentially harmful compounds into the environment, raising concerns about their ecological footprint.
The degradation products of quaternary ammonium groups, commonly used in AEMs, can include volatile organic compounds and nitrogen-containing substances that may contribute to water pollution when leached from membranes during operation or disposal. Studies have shown that these degradation byproducts can persist in aquatic environments, potentially affecting aquatic organisms and ecosystems through bioaccumulation processes.
Life cycle assessment (LCA) studies of AEM materials reveal that their environmental impact extends beyond the operational phase. The production of AEM materials involves energy-intensive processes and potentially hazardous chemicals, contributing to their overall environmental burden. Furthermore, the limited recyclability of degraded AEMs presents waste management challenges, as these materials often end up in landfills after their useful life.
Recent research has focused on developing more environmentally benign AEM materials with improved stability of cationic groups. Bio-based polymers and naturally derived cationic groups are being explored as sustainable alternatives to conventional petroleum-based materials. These bio-derived AEMs show promising degradation profiles with reduced environmental persistence and toxicity.
Regulatory frameworks worldwide are beginning to address the environmental implications of membrane materials, including AEMs. The European Union's REACH regulation and similar initiatives in other regions are imposing stricter requirements on chemical substances used in membrane manufacturing, driving the industry toward greener alternatives with reduced degradation-related environmental impacts.
The carbon footprint associated with AEM production and disposal is another environmental consideration. The energy requirements for synthesizing stable cationic groups and their subsequent degradation pathways influence the overall greenhouse gas emissions attributed to AEM technologies. Improving the stability of these functional groups not only enhances performance but also reduces the frequency of replacement, thereby lowering the cumulative environmental impact.
Water consumption during AEM manufacturing and the potential for water contamination during use represent additional environmental concerns. Degraded cationic fragments can alter water chemistry, potentially affecting downstream water treatment processes or natural water bodies if not properly managed.
The degradation products of quaternary ammonium groups, commonly used in AEMs, can include volatile organic compounds and nitrogen-containing substances that may contribute to water pollution when leached from membranes during operation or disposal. Studies have shown that these degradation byproducts can persist in aquatic environments, potentially affecting aquatic organisms and ecosystems through bioaccumulation processes.
Life cycle assessment (LCA) studies of AEM materials reveal that their environmental impact extends beyond the operational phase. The production of AEM materials involves energy-intensive processes and potentially hazardous chemicals, contributing to their overall environmental burden. Furthermore, the limited recyclability of degraded AEMs presents waste management challenges, as these materials often end up in landfills after their useful life.
Recent research has focused on developing more environmentally benign AEM materials with improved stability of cationic groups. Bio-based polymers and naturally derived cationic groups are being explored as sustainable alternatives to conventional petroleum-based materials. These bio-derived AEMs show promising degradation profiles with reduced environmental persistence and toxicity.
Regulatory frameworks worldwide are beginning to address the environmental implications of membrane materials, including AEMs. The European Union's REACH regulation and similar initiatives in other regions are imposing stricter requirements on chemical substances used in membrane manufacturing, driving the industry toward greener alternatives with reduced degradation-related environmental impacts.
The carbon footprint associated with AEM production and disposal is another environmental consideration. The energy requirements for synthesizing stable cationic groups and their subsequent degradation pathways influence the overall greenhouse gas emissions attributed to AEM technologies. Improving the stability of these functional groups not only enhances performance but also reduces the frequency of replacement, thereby lowering the cumulative environmental impact.
Water consumption during AEM manufacturing and the potential for water contamination during use represent additional environmental concerns. Degraded cationic fragments can alter water chemistry, potentially affecting downstream water treatment processes or natural water bodies if not properly managed.
Durability Testing Protocols for AEMs
Durability testing protocols for anion exchange membranes (AEMs) are critical for understanding and mitigating the degradation pathways of cationic groups, which represent the primary failure mechanism in these materials. Standardized testing methods have evolved significantly over the past decade, with researchers developing increasingly sophisticated approaches to simulate real-world operating conditions.
The accelerated stress tests (ASTs) have emerged as the predominant methodology, typically subjecting AEMs to elevated temperatures (60-80°C) in alkaline environments (1M KOH) for extended periods (500-1000 hours). These conditions deliberately intensify the degradation mechanisms to provide timely data on membrane stability. Recent protocols have incorporated additional stressors such as humidity cycling and mechanical strain to better replicate fuel cell operating environments.
In-situ monitoring techniques have become increasingly integrated into durability protocols, allowing for real-time observation of degradation phenomena. Electrochemical impedance spectroscopy (EIS) measurements during testing provide valuable insights into conductivity losses as cationic groups degrade. Similarly, periodic sampling for ion exchange capacity (IEC) determination has become standard practice in comprehensive testing protocols.
Chemical stability assessments specifically targeting cationic functional groups have been refined to isolate degradation mechanisms. These protocols typically involve ex-situ analysis using NMR spectroscopy to track changes in chemical structure, particularly focusing on quaternary ammonium groups that are susceptible to nucleophilic attack and Hofmann elimination reactions.
Mechanical durability testing has also been incorporated into comprehensive protocols, as the mechanical integrity of AEMs is often compromised as cationic groups degrade. Dimensional stability measurements during hydration/dehydration cycles provide critical data on how chemical degradation affects physical properties.
International standardization efforts have accelerated in recent years, with organizations such as the International Electrotechnical Commission (IEC) and the U.S. Department of Energy establishing benchmark protocols. These standards typically specify minimum testing durations, temperature ranges, and analytical techniques to ensure comparability across research institutions.
Round-robin testing involving multiple laboratories has become increasingly common to validate protocol reliability and reproducibility. These collaborative efforts have highlighted the importance of controlling testing parameters precisely, as minor variations in humidity, temperature cycling, or alkaline concentration can significantly impact degradation rates of cationic functional groups.
The accelerated stress tests (ASTs) have emerged as the predominant methodology, typically subjecting AEMs to elevated temperatures (60-80°C) in alkaline environments (1M KOH) for extended periods (500-1000 hours). These conditions deliberately intensify the degradation mechanisms to provide timely data on membrane stability. Recent protocols have incorporated additional stressors such as humidity cycling and mechanical strain to better replicate fuel cell operating environments.
In-situ monitoring techniques have become increasingly integrated into durability protocols, allowing for real-time observation of degradation phenomena. Electrochemical impedance spectroscopy (EIS) measurements during testing provide valuable insights into conductivity losses as cationic groups degrade. Similarly, periodic sampling for ion exchange capacity (IEC) determination has become standard practice in comprehensive testing protocols.
Chemical stability assessments specifically targeting cationic functional groups have been refined to isolate degradation mechanisms. These protocols typically involve ex-situ analysis using NMR spectroscopy to track changes in chemical structure, particularly focusing on quaternary ammonium groups that are susceptible to nucleophilic attack and Hofmann elimination reactions.
Mechanical durability testing has also been incorporated into comprehensive protocols, as the mechanical integrity of AEMs is often compromised as cationic groups degrade. Dimensional stability measurements during hydration/dehydration cycles provide critical data on how chemical degradation affects physical properties.
International standardization efforts have accelerated in recent years, with organizations such as the International Electrotechnical Commission (IEC) and the U.S. Department of Energy establishing benchmark protocols. These standards typically specify minimum testing durations, temperature ranges, and analytical techniques to ensure comparability across research institutions.
Round-robin testing involving multiple laboratories has become increasingly common to validate protocol reliability and reproducibility. These collaborative efforts have highlighted the importance of controlling testing parameters precisely, as minor variations in humidity, temperature cycling, or alkaline concentration can significantly impact degradation rates of cationic functional groups.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!