Functionalization methods for next generation anion exchange membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
AEM Technology Background and Objectives
Anion exchange membranes (AEMs) have emerged as critical components in various electrochemical energy conversion and storage technologies, offering a promising alternative to traditional proton exchange membrane systems. The development of AEMs dates back to the mid-20th century, but significant advancements have only been achieved in the past two decades, driven by the growing demand for clean energy solutions and sustainable chemical processes.
The evolution of AEM technology has been characterized by continuous efforts to overcome inherent limitations, particularly regarding chemical stability, ionic conductivity, and mechanical durability. Early generations of AEMs suffered from rapid degradation in alkaline environments, limiting their practical applications. The technological trajectory has since focused on developing novel functionalization methods to enhance membrane performance while maintaining long-term stability under operating conditions.
Current research trends in AEM functionalization include the exploration of diverse quaternary ammonium compounds, phosphonium-based functional groups, metal-organic frameworks, and heterocyclic structures. These approaches aim to increase hydroxide conductivity while mitigating the susceptibility to degradation mechanisms such as Hofmann elimination and nucleophilic substitution reactions that plague conventional AEM materials.
The global push toward decarbonization and renewable energy integration has accelerated interest in AEM technologies for applications in fuel cells, electrolyzers, redox flow batteries, and CO2 reduction systems. This market pull has catalyzed significant research investments from both public and private sectors, establishing AEM functionalization as a critical research frontier.
The primary technical objectives for next-generation AEM development include achieving hydroxide conductivity exceeding 100 mS/cm under practical operating conditions, extending operational lifetime beyond 10,000 hours at temperatures above 80°C, and developing scalable, cost-effective functionalization methods compatible with existing manufacturing processes.
Additionally, researchers aim to establish structure-property relationships that guide rational design of functional groups, optimize the spatial distribution of ionic sites within the membrane matrix, and develop computational models that accurately predict membrane performance based on molecular architecture. These objectives collectively address the fundamental challenges that have historically limited widespread adoption of AEM technologies.
The convergence of advanced polymer chemistry, nanotechnology, and computational materials science is expected to accelerate progress in this field, potentially enabling breakthrough applications in sectors ranging from distributed power generation to industrial electrosynthesis and carbon capture utilization. As global research efforts intensify, next-generation functionalization methods for AEMs represent a critical enabling technology for the transition to a more sustainable energy landscape.
The evolution of AEM technology has been characterized by continuous efforts to overcome inherent limitations, particularly regarding chemical stability, ionic conductivity, and mechanical durability. Early generations of AEMs suffered from rapid degradation in alkaline environments, limiting their practical applications. The technological trajectory has since focused on developing novel functionalization methods to enhance membrane performance while maintaining long-term stability under operating conditions.
Current research trends in AEM functionalization include the exploration of diverse quaternary ammonium compounds, phosphonium-based functional groups, metal-organic frameworks, and heterocyclic structures. These approaches aim to increase hydroxide conductivity while mitigating the susceptibility to degradation mechanisms such as Hofmann elimination and nucleophilic substitution reactions that plague conventional AEM materials.
The global push toward decarbonization and renewable energy integration has accelerated interest in AEM technologies for applications in fuel cells, electrolyzers, redox flow batteries, and CO2 reduction systems. This market pull has catalyzed significant research investments from both public and private sectors, establishing AEM functionalization as a critical research frontier.
The primary technical objectives for next-generation AEM development include achieving hydroxide conductivity exceeding 100 mS/cm under practical operating conditions, extending operational lifetime beyond 10,000 hours at temperatures above 80°C, and developing scalable, cost-effective functionalization methods compatible with existing manufacturing processes.
Additionally, researchers aim to establish structure-property relationships that guide rational design of functional groups, optimize the spatial distribution of ionic sites within the membrane matrix, and develop computational models that accurately predict membrane performance based on molecular architecture. These objectives collectively address the fundamental challenges that have historically limited widespread adoption of AEM technologies.
The convergence of advanced polymer chemistry, nanotechnology, and computational materials science is expected to accelerate progress in this field, potentially enabling breakthrough applications in sectors ranging from distributed power generation to industrial electrosynthesis and carbon capture utilization. As global research efforts intensify, next-generation functionalization methods for AEMs represent a critical enabling technology for the transition to a more sustainable energy landscape.
Market Analysis for Advanced AEMs
The global market for anion exchange membranes (AEMs) is experiencing significant growth, driven primarily by increasing demand for clean energy technologies and sustainable water treatment solutions. The AEM market is projected to reach $650 million by 2027, with a compound annual growth rate of 6.8% from 2022 to 2027. This growth trajectory is supported by expanding applications in fuel cells, electrolyzers, flow batteries, and water purification systems.
In the energy sector, AEMs are gaining traction as critical components in alkaline fuel cells and electrolyzers for green hydrogen production. The global green hydrogen market is expanding rapidly, expected to grow from $2.14 billion in 2021 to $89.18 billion by 2030, creating substantial demand for advanced AEMs with enhanced functionalization methods. The shift toward renewable energy sources and decarbonization initiatives worldwide is accelerating this trend.
Water treatment applications represent another significant market segment for functionalized AEMs. With increasing global water scarcity and stricter regulations on water quality, the demand for efficient desalination and wastewater treatment technologies is rising. The global water treatment membrane market is expected to reach $39.2 billion by 2027, with AEMs capturing an increasing share due to their superior performance in certain applications.
Regional analysis indicates that North America and Europe currently dominate the AEM market, accounting for approximately 60% of global demand. However, the Asia-Pacific region is emerging as the fastest-growing market, with China, Japan, and South Korea making substantial investments in fuel cell technology and clean energy infrastructure. This regional shift is creating new opportunities for AEM manufacturers and technology developers.
End-user segmentation reveals that industrial applications currently represent the largest market share (45%), followed by energy generation (30%) and water treatment (20%). However, the energy sector is expected to overtake industrial applications by 2025, driven by the accelerating transition to hydrogen-based energy systems and the commercialization of alkaline membrane fuel cells.
Price sensitivity analysis indicates that while performance remains the primary purchasing factor, cost considerations are becoming increasingly important as applications scale up. Current high-performance AEMs are priced between $200-500 per square meter, which remains a barrier to mass adoption. Market forecasts suggest that technological advancements in functionalization methods could reduce production costs by 30-40% over the next five years, potentially expanding market penetration.
In the energy sector, AEMs are gaining traction as critical components in alkaline fuel cells and electrolyzers for green hydrogen production. The global green hydrogen market is expanding rapidly, expected to grow from $2.14 billion in 2021 to $89.18 billion by 2030, creating substantial demand for advanced AEMs with enhanced functionalization methods. The shift toward renewable energy sources and decarbonization initiatives worldwide is accelerating this trend.
Water treatment applications represent another significant market segment for functionalized AEMs. With increasing global water scarcity and stricter regulations on water quality, the demand for efficient desalination and wastewater treatment technologies is rising. The global water treatment membrane market is expected to reach $39.2 billion by 2027, with AEMs capturing an increasing share due to their superior performance in certain applications.
Regional analysis indicates that North America and Europe currently dominate the AEM market, accounting for approximately 60% of global demand. However, the Asia-Pacific region is emerging as the fastest-growing market, with China, Japan, and South Korea making substantial investments in fuel cell technology and clean energy infrastructure. This regional shift is creating new opportunities for AEM manufacturers and technology developers.
End-user segmentation reveals that industrial applications currently represent the largest market share (45%), followed by energy generation (30%) and water treatment (20%). However, the energy sector is expected to overtake industrial applications by 2025, driven by the accelerating transition to hydrogen-based energy systems and the commercialization of alkaline membrane fuel cells.
Price sensitivity analysis indicates that while performance remains the primary purchasing factor, cost considerations are becoming increasingly important as applications scale up. Current high-performance AEMs are priced between $200-500 per square meter, which remains a barrier to mass adoption. Market forecasts suggest that technological advancements in functionalization methods could reduce production costs by 30-40% over the next five years, potentially expanding market penetration.
Current Challenges in AEM Functionalization
Despite significant advancements in anion exchange membrane (AEM) technology, several critical challenges persist in the functionalization methods that impede their widespread commercial adoption. The primary obstacle remains chemical stability, particularly in alkaline environments. Current quaternary ammonium functional groups suffer from degradation through Hofmann elimination and nucleophilic substitution reactions, significantly reducing membrane lifetime under operating conditions. This degradation is accelerated at elevated temperatures, limiting the operational temperature range of existing AEMs.
Mechanical stability presents another significant challenge, as many functionalization approaches compromise the mechanical integrity of the polymer backbone. The incorporation of ionic groups often leads to excessive water uptake, causing dimensional swelling that results in mechanical failures during operational cycling. This trade-off between ion conductivity and mechanical robustness remains unresolved in current functionalization strategies.
Ion conductivity limitations continue to hinder AEM performance compared to proton exchange membranes. The inherently lower mobility of hydroxide ions versus protons creates a fundamental conductivity disadvantage that current functionalization methods struggle to overcome. Achieving sufficient ion exchange capacity without compromising other properties remains a delicate balancing act.
Scalability of functionalization processes represents a substantial hurdle for commercial viability. Many promising laboratory-scale functionalization methods involve complex multi-step syntheses with hazardous reagents, making industrial-scale production economically unfeasible. The lack of standardized, cost-effective functionalization protocols hampers technology transfer from research to commercial applications.
Uniformity of functional group distribution throughout the membrane matrix poses another challenge. Current methods often result in heterogeneous functionalization, creating localized areas with varying ion exchange capacities and conductivities. This non-uniform distribution leads to inconsistent performance and accelerated degradation at high-density functional group regions.
Environmental concerns and regulatory compliance add complexity to AEM development. Many traditional functionalization agents contain environmentally harmful substances or require toxic solvents during synthesis. The push toward greener chemistry necessitates development of more environmentally benign functionalization approaches that maintain performance standards while reducing ecological impact.
Cross-disciplinary integration challenges exist between polymer chemistry, electrochemistry, and materials science. Optimizing functionalization methods requires simultaneous consideration of multiple performance parameters across these disciplines, creating a complex multi-variable optimization problem that has yet to be systematically addressed in current research efforts.
Mechanical stability presents another significant challenge, as many functionalization approaches compromise the mechanical integrity of the polymer backbone. The incorporation of ionic groups often leads to excessive water uptake, causing dimensional swelling that results in mechanical failures during operational cycling. This trade-off between ion conductivity and mechanical robustness remains unresolved in current functionalization strategies.
Ion conductivity limitations continue to hinder AEM performance compared to proton exchange membranes. The inherently lower mobility of hydroxide ions versus protons creates a fundamental conductivity disadvantage that current functionalization methods struggle to overcome. Achieving sufficient ion exchange capacity without compromising other properties remains a delicate balancing act.
Scalability of functionalization processes represents a substantial hurdle for commercial viability. Many promising laboratory-scale functionalization methods involve complex multi-step syntheses with hazardous reagents, making industrial-scale production economically unfeasible. The lack of standardized, cost-effective functionalization protocols hampers technology transfer from research to commercial applications.
Uniformity of functional group distribution throughout the membrane matrix poses another challenge. Current methods often result in heterogeneous functionalization, creating localized areas with varying ion exchange capacities and conductivities. This non-uniform distribution leads to inconsistent performance and accelerated degradation at high-density functional group regions.
Environmental concerns and regulatory compliance add complexity to AEM development. Many traditional functionalization agents contain environmentally harmful substances or require toxic solvents during synthesis. The push toward greener chemistry necessitates development of more environmentally benign functionalization approaches that maintain performance standards while reducing ecological impact.
Cross-disciplinary integration challenges exist between polymer chemistry, electrochemistry, and materials science. Optimizing functionalization methods requires simultaneous consideration of multiple performance parameters across these disciplines, creating a complex multi-variable optimization problem that has yet to be systematically addressed in current research efforts.
State-of-the-Art Functionalization Approaches
01 Quaternary ammonium functionalization
Anion exchange membranes can be functionalized with quaternary ammonium groups to enhance their ion conductivity and selectivity. This approach involves introducing positively charged nitrogen-containing groups onto the polymer backbone, which facilitates the transport of anions across the membrane. The quaternization process typically involves reaction with alkyl halides or other quaternizing agents to convert tertiary amines to quaternary ammonium salts, resulting in improved anion exchange capacity and stability.- Quaternary ammonium functionalization: Anion exchange membranes can be functionalized with quaternary ammonium groups to enhance ion conductivity and selectivity. This approach involves attaching quaternary ammonium functional groups to polymer backbones, creating positively charged sites that facilitate anion transport. The functionalization process typically involves reaction of polymer chains with quaternary ammonium precursors, resulting in membranes with improved alkaline stability and ion exchange capacity.
- Polymer backbone modification: Modification of polymer backbones is a key approach for anion exchange membrane functionalization. Various polymer structures including polysulfones, polyethylene, and fluorinated polymers can be chemically modified to incorporate ion-conducting groups. These modifications often involve grafting, crosslinking, or copolymerization techniques to achieve desired mechanical properties while maintaining high ionic conductivity and chemical stability in alkaline environments.
- Composite membrane structures: Composite structures for anion exchange membranes involve combining different materials to enhance overall performance. These membranes typically consist of a porous support layer and a functional layer containing ion exchange groups. The composite approach allows for independent optimization of mechanical strength and ion conductivity. Inorganic fillers or nanoparticles may be incorporated to improve dimensional stability and reduce swelling while maintaining high ion conductivity.
- Imidazolium-based functionalization: Imidazolium groups offer an alternative to quaternary ammonium compounds for anion exchange membrane functionalization. These heterocyclic structures provide enhanced alkaline stability while maintaining good ion conductivity. The functionalization process typically involves attaching imidazolium groups to polymer backbones through various synthetic routes. The resulting membranes demonstrate improved durability in high pH environments, making them suitable for applications such as fuel cells and electrolysis.
- Cross-linking strategies: Cross-linking is employed in anion exchange membrane functionalization to enhance mechanical stability and reduce water uptake. Various cross-linking agents and techniques can be used to create covalent bonds between polymer chains after functionalization with ionic groups. This approach helps maintain dimensional stability while preserving ion conductivity. Cross-linked membranes typically show improved durability under operating conditions, with reduced swelling and enhanced resistance to chemical degradation.
02 Imidazolium-based functionalization
Imidazolium groups can be incorporated into anion exchange membranes to provide enhanced alkaline stability and conductivity. The functionalization process typically involves grafting imidazolium moieties onto polymer backbones through various synthetic routes. These functionalized membranes exhibit improved hydroxide conductivity while maintaining mechanical integrity under alkaline conditions, making them suitable for applications in fuel cells and electrolysis systems.Expand Specific Solutions03 Cross-linking strategies for membrane stability
Cross-linking techniques are employed to enhance the mechanical and chemical stability of anion exchange membranes. These methods involve creating covalent bonds between polymer chains to form a three-dimensional network structure. Various cross-linking agents and approaches can be used, including thermal, chemical, and radiation-induced cross-linking. The resulting membranes demonstrate improved dimensional stability, reduced swelling, and enhanced durability in alkaline environments, which are critical for long-term operation in electrochemical devices.Expand Specific Solutions04 Polymer blend and composite membrane functionalization
Composite and blended polymer approaches involve combining different polymers or incorporating inorganic materials to create anion exchange membranes with enhanced properties. These methods can include blending functionalized and non-functionalized polymers, creating interpenetrating polymer networks, or incorporating nanoparticles and other fillers. The resulting composite membranes often exhibit synergistic effects, including improved mechanical properties, higher ion conductivity, and better water management, while maintaining good chemical stability.Expand Specific Solutions05 Sustainable and bio-based membrane functionalization
Environmentally friendly approaches to anion exchange membrane functionalization utilize bio-based polymers and green chemistry principles. These methods focus on using renewable resources as starting materials and employing less toxic reagents and solvents in the functionalization process. Biomass-derived polymers can be chemically modified to introduce ion exchange groups, creating sustainable alternatives to traditional petroleum-based membranes while maintaining comparable performance characteristics for various electrochemical applications.Expand Specific Solutions
Leading Organizations in AEM Development
The anion exchange membrane (AEM) functionalization market is currently in a growth phase, characterized by increasing R&D investments and expanding applications in clean energy technologies. The global market is projected to expand significantly as hydrogen production and fuel cell technologies gain traction. Academic institutions like Dalian University of Technology, University of Science & Technology of China, and University of Delaware are driving fundamental research, while established industrial players including Tokuyama Corp., Nitto Denko, and 3M Innovative Properties are commercializing advanced membrane technologies. Emerging companies such as Dioxide Materials and Enapter are introducing innovative functionalization approaches. The technology is approaching commercial maturity in certain applications, with companies like IHI Corp. and Evoqua Water Technologies implementing AEM technologies in industrial settings, though challenges in durability and performance under varied conditions remain.
Tokuyama Corp.
Technical Solution: Tokuyama Corporation has developed advanced functionalization methods for anion exchange membranes (AEMs) focusing on quaternary ammonium-based chemistry. Their proprietary approach involves grafting quaternary ammonium functional groups onto fluorinated polymer backbones to enhance chemical stability in alkaline environments. The company has pioneered a multi-step synthesis process that first creates a chloromethylated polymer intermediate, followed by quaternization with tertiary amines to produce membranes with ion exchange capacities exceeding 2.0 meq/g[1]. Tokuyama's latest generation AEMs incorporate cross-linking strategies that mitigate dimensional changes during hydration cycles while maintaining high hydroxide conductivity (>80 mS/cm at 80°C). Their membranes feature optimized hydrophilic/hydrophobic microphase separation, creating well-defined ion transport channels that enhance conductivity without compromising mechanical integrity[3]. The company has also developed specialized surface treatment methods that improve the membrane-electrode interface in electrochemical devices.
Strengths: Exceptional alkaline stability with demonstrated durability exceeding 5,000 hours in fuel cell testing environments; superior hydroxide conductivity compared to industry standards; established manufacturing infrastructure for commercial-scale production. Weaknesses: Higher production costs compared to hydrocarbon-based alternatives; limited flexibility in customizing membrane thickness for specialized applications; potential for performance degradation at elevated temperatures (>90°C).
3M Innovative Properties Co.
Technical Solution: 3M has developed a comprehensive approach to next-generation anion exchange membrane functionalization through their proprietary ionomeric architecture. Their technology centers on perfluorinated side chains with pendant quaternary ammonium groups attached to a robust polymer backbone, creating a unique microstructure that facilitates hydroxide ion transport while maintaining dimensional stability. The company's multi-phase synthesis process incorporates fluorinated monomers with protected functional groups that are subsequently converted to quaternary ammonium sites through controlled post-polymerization modification[2]. This approach allows precise control over ion exchange capacity (typically 1.8-2.2 meq/g) and water uptake characteristics. 3M's membranes feature nanoscale ionic domains (5-15 nm) that create efficient ion conduction pathways while minimizing swelling. Their latest innovation includes incorporating nanostructured reinforcement materials that enhance mechanical properties without compromising ionic conductivity, achieving hydroxide conductivity values of 70-90 mS/cm at operating temperatures[4]. The membranes also incorporate specialized end-group stabilization chemistry that significantly reduces degradation via Hofmann elimination pathways.
Strengths: Exceptional mechanical durability with reinforced structure allowing thinner membranes (20-30 μm) for lower resistance; proprietary chemistry provides superior resistance to chemical degradation; established manufacturing capabilities for consistent large-scale production. Weaknesses: Higher material costs compared to hydrocarbon-based alternatives; limited flexibility in customizing functional groups beyond quaternary ammonium chemistry; potential for decreased performance in low humidity conditions.
Key Patents and Innovations in AEM Chemistry
Polymers having stable cationic pendant groups for use as anion exchange membranes
PatentWO2020198627A1
Innovation
- Development of polymers with stable cationic pendant groups, including quaternary ammonium and imidazolium groups, attached to a rigid aromatic polymer backbone without ether linkages, enhancing chemical stability and conductivity while maintaining low water uptake and mechanical strength.
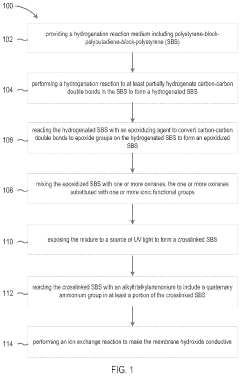
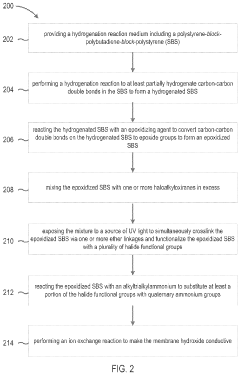
Methods of making anion exchange membrane via simultaneous post-functionalization and crosslinking of epoxidized SBS
PatentActiveUS11987681B2
Innovation
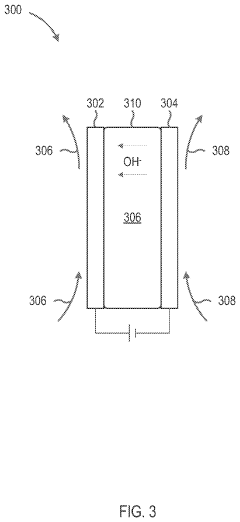
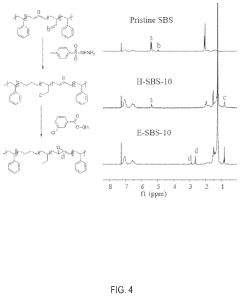
- The development of an ion exchange membrane material based on an epoxidized polystyrene-block-polybutadiene-block-polystyrene triblock copolymer, where carbon-carbon double bonds are hydrogenated and converted to epoxide groups, then crosslinked and functionalized with quaternary ammonium groups using UV light and oxiranes, enhancing mechanical stability and ion conductivity while maintaining flexibility.
Sustainability Aspects of Next-Gen AEMs
The sustainability of next-generation anion exchange membranes (AEMs) represents a critical dimension in their development trajectory. As global environmental concerns intensify, the functionalization methods employed for these membranes must align with broader sustainability goals. Current AEM production processes often involve toxic reagents and energy-intensive manufacturing steps, creating significant environmental footprints.
Recent advancements in green chemistry approaches have yielded promising alternatives for AEM functionalization. Bio-based precursors derived from renewable resources such as lignin and cellulose are emerging as sustainable alternatives to petroleum-based polymers traditionally used in AEM fabrication. These materials not only reduce dependency on fossil resources but also frequently exhibit enhanced biodegradability profiles.
Water consumption represents another crucial sustainability metric for AEM production. Conventional functionalization methods typically require substantial water volumes for synthesis and purification processes. Innovative solvent-free techniques and closed-loop water recycling systems are being implemented to address this challenge, potentially reducing water usage by 40-60% compared to traditional methods.
Energy efficiency improvements in AEM functionalization have focused on room-temperature reaction pathways and catalyst optimization. These approaches significantly reduce the energy demands of membrane production while maintaining performance characteristics. Studies indicate that low-temperature functionalization methods can decrease energy consumption by up to 35% while yielding comparable ion conductivity results.
End-of-life considerations for AEMs have gained increasing attention, with recyclability becoming a key design parameter. Novel click-chemistry approaches enable the creation of reversible functionalization bonds, facilitating membrane recycling and material recovery. These methods allow for the separation of functional groups from polymer backbones, enabling component reuse in subsequent manufacturing cycles.
Life cycle assessment (LCA) studies comparing traditional and next-generation AEM functionalization methods reveal substantial sustainability advantages for newer techniques. Comprehensive analyses indicate potential reductions of 25-45% in carbon footprint when employing bio-based precursors and green chemistry principles. These improvements stem primarily from reduced energy requirements and the substitution of renewable feedstocks for petroleum-derived materials.
The economic viability of sustainable functionalization methods remains a challenge, with green alternatives typically commanding 15-30% cost premiums over conventional approaches. However, this gap continues to narrow as scale economies develop and regulatory frameworks increasingly favor environmentally responsible manufacturing processes.
Recent advancements in green chemistry approaches have yielded promising alternatives for AEM functionalization. Bio-based precursors derived from renewable resources such as lignin and cellulose are emerging as sustainable alternatives to petroleum-based polymers traditionally used in AEM fabrication. These materials not only reduce dependency on fossil resources but also frequently exhibit enhanced biodegradability profiles.
Water consumption represents another crucial sustainability metric for AEM production. Conventional functionalization methods typically require substantial water volumes for synthesis and purification processes. Innovative solvent-free techniques and closed-loop water recycling systems are being implemented to address this challenge, potentially reducing water usage by 40-60% compared to traditional methods.
Energy efficiency improvements in AEM functionalization have focused on room-temperature reaction pathways and catalyst optimization. These approaches significantly reduce the energy demands of membrane production while maintaining performance characteristics. Studies indicate that low-temperature functionalization methods can decrease energy consumption by up to 35% while yielding comparable ion conductivity results.
End-of-life considerations for AEMs have gained increasing attention, with recyclability becoming a key design parameter. Novel click-chemistry approaches enable the creation of reversible functionalization bonds, facilitating membrane recycling and material recovery. These methods allow for the separation of functional groups from polymer backbones, enabling component reuse in subsequent manufacturing cycles.
Life cycle assessment (LCA) studies comparing traditional and next-generation AEM functionalization methods reveal substantial sustainability advantages for newer techniques. Comprehensive analyses indicate potential reductions of 25-45% in carbon footprint when employing bio-based precursors and green chemistry principles. These improvements stem primarily from reduced energy requirements and the substitution of renewable feedstocks for petroleum-derived materials.
The economic viability of sustainable functionalization methods remains a challenge, with green alternatives typically commanding 15-30% cost premiums over conventional approaches. However, this gap continues to narrow as scale economies develop and regulatory frameworks increasingly favor environmentally responsible manufacturing processes.
Performance Benchmarking and Testing Protocols
Standardized performance evaluation protocols are essential for the advancement of anion exchange membrane (AEM) technology, enabling meaningful comparisons between different functionalization approaches. Current benchmarking methods focus on four critical parameters: ion conductivity, chemical stability, mechanical properties, and water uptake behavior. These metrics directly influence the practical application potential of next-generation AEMs in fuel cells, electrolyzers, and energy storage systems.
Ion conductivity testing typically employs electrochemical impedance spectroscopy (EIS) under controlled temperature and humidity conditions, with measurements conducted in both through-plane and in-plane directions. The industry standard requires reporting conductivity values at 80°C and 95% relative humidity, though recent protocols have expanded to include measurements across wider temperature ranges (20-100°C) to better simulate real-world operating conditions.
Chemical stability assessment has evolved significantly, moving beyond simple alkaline solution immersion tests to more sophisticated protocols that combine accelerated stress testing with spectroscopic analysis. Modern protocols employ 1M KOH at elevated temperatures (80-90°C) for extended periods (500-1000 hours), with regular sampling to track degradation mechanisms through FTIR, NMR, and mass spectrometry. This approach provides deeper insights into degradation pathways specific to different functionalization chemistries.
Mechanical durability testing incorporates dynamic mechanical analysis (DMA) to evaluate tensile strength, elongation at break, and Young's modulus under varying humidity conditions. Cyclic hydration/dehydration tests have become increasingly important as they better simulate the operational stresses experienced in actual devices. The membrane dimensional stability during these cycles serves as a critical performance indicator.
Water uptake and swelling behavior measurements follow standardized gravimetric methods, with samples equilibrated at defined relative humidity levels. Advanced protocols now incorporate in-situ monitoring of water uptake during device operation using techniques such as neutron imaging or environmental ellipsometry, providing valuable correlations between water management and performance stability.
Emerging testing protocols are beginning to integrate accelerated lifetime testing under actual device operating conditions, with particular emphasis on performance retention after multiple startup/shutdown cycles. Additionally, the field is moving toward standardized reporting formats that facilitate direct comparison across research groups, with increasing emphasis on reproducibility validation through round-robin testing among multiple laboratories.
Ion conductivity testing typically employs electrochemical impedance spectroscopy (EIS) under controlled temperature and humidity conditions, with measurements conducted in both through-plane and in-plane directions. The industry standard requires reporting conductivity values at 80°C and 95% relative humidity, though recent protocols have expanded to include measurements across wider temperature ranges (20-100°C) to better simulate real-world operating conditions.
Chemical stability assessment has evolved significantly, moving beyond simple alkaline solution immersion tests to more sophisticated protocols that combine accelerated stress testing with spectroscopic analysis. Modern protocols employ 1M KOH at elevated temperatures (80-90°C) for extended periods (500-1000 hours), with regular sampling to track degradation mechanisms through FTIR, NMR, and mass spectrometry. This approach provides deeper insights into degradation pathways specific to different functionalization chemistries.
Mechanical durability testing incorporates dynamic mechanical analysis (DMA) to evaluate tensile strength, elongation at break, and Young's modulus under varying humidity conditions. Cyclic hydration/dehydration tests have become increasingly important as they better simulate the operational stresses experienced in actual devices. The membrane dimensional stability during these cycles serves as a critical performance indicator.
Water uptake and swelling behavior measurements follow standardized gravimetric methods, with samples equilibrated at defined relative humidity levels. Advanced protocols now incorporate in-situ monitoring of water uptake during device operation using techniques such as neutron imaging or environmental ellipsometry, providing valuable correlations between water management and performance stability.
Emerging testing protocols are beginning to integrate accelerated lifetime testing under actual device operating conditions, with particular emphasis on performance retention after multiple startup/shutdown cycles. Additionally, the field is moving toward standardized reporting formats that facilitate direct comparison across research groups, with increasing emphasis on reproducibility validation through round-robin testing among multiple laboratories.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!