Hydration dependent conductivity in anion exchange membranes
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
AEM Hydration Conductivity Background and Objectives
Anion exchange membranes (AEMs) have emerged as critical components in various electrochemical energy conversion and storage technologies, including fuel cells, electrolyzers, and redox flow batteries. The historical development of AEMs can be traced back to the 1950s, but significant advancements have only been achieved in the past two decades. The relationship between hydration and ionic conductivity represents one of the most fundamental yet complex aspects of AEM technology that continues to challenge researchers worldwide.
Water management in AEMs presents a delicate balance - sufficient hydration is essential for facilitating hydroxide ion transport, yet excessive water uptake can lead to dimensional instability and mechanical failure. This hydration-conductivity relationship follows distinctly different mechanisms compared to proton exchange membranes (PEMs), primarily due to the larger size of hydroxide ions and their different transport mechanisms through the polymer matrix.
Recent technological trends indicate a shift toward developing AEMs with precisely controlled hydration channels and optimized ionic clusters. The evolution of synthetic approaches has moved from simple quaternization of existing polymers toward sophisticated architectures with segregated hydrophilic and hydrophobic domains. This structural evolution aims to create efficient ion-conducting pathways while maintaining dimensional stability across varying hydration states.
The global research landscape shows accelerating interest in understanding the fundamental science behind hydration-dependent conductivity. Publications in this field have grown exponentially since 2010, with particular emphasis on in-situ characterization techniques that allow real-time observation of water-ion interactions within membrane structures.
The primary technical objectives of current research include: developing AEMs with high hydroxide conductivity (>100 mS/cm) at moderate relative humidity levels (40-80%); achieving stable conductivity across varying hydration conditions; understanding the molecular-level interactions between water, hydroxide ions, and polymer backbones; and establishing predictive models that can accurately correlate membrane chemistry and morphology with hydration-dependent conductivity.
Additionally, researchers aim to elucidate the complex interplay between hydration, alkaline stability, and mechanical properties - three interdependent factors that often require careful optimization trade-offs. The ultimate goal is to develop AEMs that maintain high conductivity with minimal hydration fluctuations across operating conditions, thereby enabling more efficient and stable electrochemical devices for clean energy applications.
Water management in AEMs presents a delicate balance - sufficient hydration is essential for facilitating hydroxide ion transport, yet excessive water uptake can lead to dimensional instability and mechanical failure. This hydration-conductivity relationship follows distinctly different mechanisms compared to proton exchange membranes (PEMs), primarily due to the larger size of hydroxide ions and their different transport mechanisms through the polymer matrix.
Recent technological trends indicate a shift toward developing AEMs with precisely controlled hydration channels and optimized ionic clusters. The evolution of synthetic approaches has moved from simple quaternization of existing polymers toward sophisticated architectures with segregated hydrophilic and hydrophobic domains. This structural evolution aims to create efficient ion-conducting pathways while maintaining dimensional stability across varying hydration states.
The global research landscape shows accelerating interest in understanding the fundamental science behind hydration-dependent conductivity. Publications in this field have grown exponentially since 2010, with particular emphasis on in-situ characterization techniques that allow real-time observation of water-ion interactions within membrane structures.
The primary technical objectives of current research include: developing AEMs with high hydroxide conductivity (>100 mS/cm) at moderate relative humidity levels (40-80%); achieving stable conductivity across varying hydration conditions; understanding the molecular-level interactions between water, hydroxide ions, and polymer backbones; and establishing predictive models that can accurately correlate membrane chemistry and morphology with hydration-dependent conductivity.
Additionally, researchers aim to elucidate the complex interplay between hydration, alkaline stability, and mechanical properties - three interdependent factors that often require careful optimization trade-offs. The ultimate goal is to develop AEMs that maintain high conductivity with minimal hydration fluctuations across operating conditions, thereby enabling more efficient and stable electrochemical devices for clean energy applications.
Market Analysis for Hydration-Enhanced AEM Applications
The global market for anion exchange membranes (AEMs) with hydration-dependent conductivity is experiencing significant growth, driven primarily by increasing demand for clean energy technologies. The market size for AEMs was valued at approximately $320 million in 2022 and is projected to reach $650 million by 2028, representing a compound annual growth rate (CAGR) of 12.5%. This growth trajectory is substantially higher than the broader membrane market, which grows at around 7-8% annually.
Fuel cell applications currently dominate the hydration-enhanced AEM market, accounting for roughly 45% of total demand. The automotive sector represents the largest end-user segment, with major manufacturers increasingly incorporating alkaline membrane fuel cells into their hydrogen vehicle prototypes. Toyota, Hyundai, and Honda have all announced significant investments in this technology over the past three years, indicating strong commercial interest.
Water electrolysis for green hydrogen production constitutes the second-largest application segment at 30% market share, with projected growth rates exceeding 15% annually through 2030. This acceleration is largely attributed to global decarbonization initiatives and substantial government funding for hydrogen infrastructure development across North America, Europe, and East Asia.
Regional analysis reveals that North America currently leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the fastest growth is occurring in the Asia-Pacific region, particularly in China, Japan, and South Korea, where government policies strongly support clean energy technologies and manufacturing capabilities are rapidly expanding.
Customer segmentation shows three primary buyer categories: energy technology companies (55%), research institutions (25%), and industrial manufacturers (20%). The energy technology segment is growing most rapidly as commercialization of AEM-based systems accelerates.
Key market drivers include increasing renewable energy integration requiring efficient energy storage solutions, stringent emission regulations worldwide, and declining manufacturing costs as production scales up. The price sensitivity varies significantly by application, with research applications being less price-sensitive than mass-market industrial applications.
Market challenges include competition from proton exchange membrane (PEM) technologies, which currently dominate many applications due to their maturity and established supply chains. Additionally, durability concerns under variable hydration conditions remain a significant barrier to wider commercial adoption, particularly in applications requiring thousands of operating hours.
Fuel cell applications currently dominate the hydration-enhanced AEM market, accounting for roughly 45% of total demand. The automotive sector represents the largest end-user segment, with major manufacturers increasingly incorporating alkaline membrane fuel cells into their hydrogen vehicle prototypes. Toyota, Hyundai, and Honda have all announced significant investments in this technology over the past three years, indicating strong commercial interest.
Water electrolysis for green hydrogen production constitutes the second-largest application segment at 30% market share, with projected growth rates exceeding 15% annually through 2030. This acceleration is largely attributed to global decarbonization initiatives and substantial government funding for hydrogen infrastructure development across North America, Europe, and East Asia.
Regional analysis reveals that North America currently leads the market with 38% share, followed by Europe (32%) and Asia-Pacific (25%). However, the fastest growth is occurring in the Asia-Pacific region, particularly in China, Japan, and South Korea, where government policies strongly support clean energy technologies and manufacturing capabilities are rapidly expanding.
Customer segmentation shows three primary buyer categories: energy technology companies (55%), research institutions (25%), and industrial manufacturers (20%). The energy technology segment is growing most rapidly as commercialization of AEM-based systems accelerates.
Key market drivers include increasing renewable energy integration requiring efficient energy storage solutions, stringent emission regulations worldwide, and declining manufacturing costs as production scales up. The price sensitivity varies significantly by application, with research applications being less price-sensitive than mass-market industrial applications.
Market challenges include competition from proton exchange membrane (PEM) technologies, which currently dominate many applications due to their maturity and established supply chains. Additionally, durability concerns under variable hydration conditions remain a significant barrier to wider commercial adoption, particularly in applications requiring thousands of operating hours.
Current Challenges in Anion Exchange Membrane Conductivity
Despite significant advancements in anion exchange membrane (AEM) technology, several critical challenges persist regarding conductivity performance, particularly in relation to hydration dependency. The fundamental issue lies in the delicate balance between membrane hydration and ion conductivity. Unlike proton exchange membranes, AEMs exhibit more complex hydration-conductivity relationships due to the larger size of hydroxide ions and their distinct transport mechanisms.
A primary technical obstacle is the trade-off between ion exchange capacity (IEC) and dimensional stability. Higher IEC values generally improve conductivity but simultaneously increase water uptake, leading to excessive swelling and mechanical degradation. This dimensional instability compromises the membrane's long-term operational reliability, especially during hydration-dehydration cycles commonly experienced in real-world applications.
The hydroxide ion mobility within AEMs presents another significant challenge. These ions demonstrate approximately half the inherent mobility of protons in aqueous environments, creating an intrinsic limitation to achievable conductivity values. Additionally, hydroxide transport mechanisms are highly dependent on water content and configuration within the membrane microstructure, making conductivity particularly sensitive to hydration fluctuations.
Water management within AEMs remains problematic across varying operating conditions. At low hydration levels, insufficient water molecules are available to facilitate efficient ion hopping mechanisms, severely restricting conductivity. Conversely, excessive hydration dilutes the fixed charge concentration, potentially reducing conductivity despite increased water content. This non-linear relationship creates significant engineering challenges for maintaining optimal performance across diverse environmental conditions.
The chemical microstructure of AEMs further complicates conductivity optimization. Current membrane architectures often feature inadequate phase separation between hydrophilic and hydrophobic domains, resulting in inefficient ion transport pathways. The formation of isolated water clusters rather than continuous hydrated channels impedes hydroxide mobility, particularly at intermediate hydration levels where percolation networks may be incomplete.
Temperature dependency adds another layer of complexity. Unlike proton conductivity, which generally follows Arrhenius behavior, anion conductivity in AEMs often exhibits more complex temperature relationships mediated by hydration effects. At elevated temperatures, water retention becomes increasingly difficult, potentially negating the expected conductivity improvements from enhanced ion mobility.
Interfacial resistance between membrane layers and electrode surfaces represents a growing concern, particularly in multi-layer systems. These interfaces can experience localized dehydration or irregular hydration profiles, creating conductivity bottlenecks that limit overall system performance regardless of bulk membrane properties.
A primary technical obstacle is the trade-off between ion exchange capacity (IEC) and dimensional stability. Higher IEC values generally improve conductivity but simultaneously increase water uptake, leading to excessive swelling and mechanical degradation. This dimensional instability compromises the membrane's long-term operational reliability, especially during hydration-dehydration cycles commonly experienced in real-world applications.
The hydroxide ion mobility within AEMs presents another significant challenge. These ions demonstrate approximately half the inherent mobility of protons in aqueous environments, creating an intrinsic limitation to achievable conductivity values. Additionally, hydroxide transport mechanisms are highly dependent on water content and configuration within the membrane microstructure, making conductivity particularly sensitive to hydration fluctuations.
Water management within AEMs remains problematic across varying operating conditions. At low hydration levels, insufficient water molecules are available to facilitate efficient ion hopping mechanisms, severely restricting conductivity. Conversely, excessive hydration dilutes the fixed charge concentration, potentially reducing conductivity despite increased water content. This non-linear relationship creates significant engineering challenges for maintaining optimal performance across diverse environmental conditions.
The chemical microstructure of AEMs further complicates conductivity optimization. Current membrane architectures often feature inadequate phase separation between hydrophilic and hydrophobic domains, resulting in inefficient ion transport pathways. The formation of isolated water clusters rather than continuous hydrated channels impedes hydroxide mobility, particularly at intermediate hydration levels where percolation networks may be incomplete.
Temperature dependency adds another layer of complexity. Unlike proton conductivity, which generally follows Arrhenius behavior, anion conductivity in AEMs often exhibits more complex temperature relationships mediated by hydration effects. At elevated temperatures, water retention becomes increasingly difficult, potentially negating the expected conductivity improvements from enhanced ion mobility.
Interfacial resistance between membrane layers and electrode surfaces represents a growing concern, particularly in multi-layer systems. These interfaces can experience localized dehydration or irregular hydration profiles, creating conductivity bottlenecks that limit overall system performance regardless of bulk membrane properties.
State-of-the-Art Solutions for Hydration-Dependent Ionic Conductivity
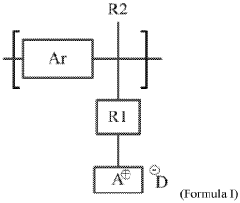
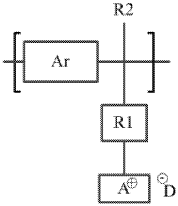
01 Polymer modification for enhanced conductivity
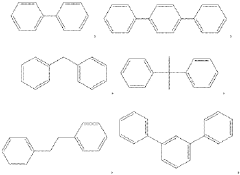
Various polymer modifications can significantly enhance the ionic conductivity of anion exchange membranes. These modifications include the incorporation of functional groups, cross-linking strategies, and the development of block copolymers. By optimizing the polymer structure, researchers can achieve higher hydroxide ion conductivity while maintaining mechanical stability and chemical durability, which are essential for applications in fuel cells and electrolysis systems.- Polymer modification for enhanced conductivity: Various polymer modifications can significantly enhance the ionic conductivity of anion exchange membranes. These modifications include the incorporation of functional groups, crosslinking strategies, and the development of block copolymers. By optimizing the polymer structure, researchers can achieve higher hydroxide ion conductivity while maintaining mechanical stability and chemical durability, which are essential for applications in fuel cells and electrolysis systems.
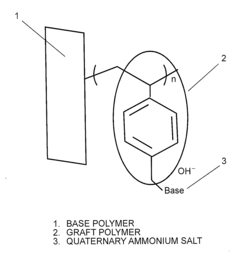
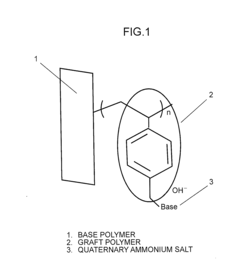
- Quaternary ammonium functionalization: Quaternary ammonium groups are widely used as ion-conducting sites in anion exchange membranes. The density, distribution, and chemical structure of these quaternary ammonium groups significantly impact the membrane's conductivity. Research focuses on optimizing these parameters to achieve high anion conductivity while addressing challenges related to alkaline stability. Various synthetic approaches are employed to incorporate these functional groups into polymer backbones.
- Composite and hybrid membrane structures: Composite and hybrid membrane structures combine different materials to enhance conductivity and stability. These structures often incorporate inorganic fillers, such as metal oxides or nanoparticles, into polymer matrices. The resulting membranes benefit from the synergistic properties of both components, leading to improved ion conductivity, mechanical strength, and chemical stability under operating conditions.
- Temperature and humidity effects on conductivity: The conductivity of anion exchange membranes is significantly influenced by operating temperature and humidity conditions. Research focuses on developing membranes that maintain high conductivity across a wide range of temperatures and humidity levels. This includes strategies to improve water retention capabilities and to ensure stable performance under varying environmental conditions, which is crucial for practical applications in energy conversion devices.
- Novel ion-conducting groups and mechanisms: Beyond traditional quaternary ammonium groups, researchers are exploring novel ion-conducting functionalities to enhance anion exchange membrane conductivity. These include phosphonium, imidazolium, and metal-organic frameworks as ion-conducting sites. Additionally, innovative ion transport mechanisms are being investigated to facilitate faster ion movement through the membrane structure, potentially leading to breakthrough improvements in conductivity performance.
02 Composite membranes with inorganic fillers
The integration of inorganic fillers into anion exchange membranes creates composite structures with improved conductivity properties. Materials such as metal oxides, nanoparticles, and layered materials can create additional ion transport pathways and enhance water retention. These composite membranes demonstrate superior ionic conductivity under various operating conditions while reducing swelling and improving dimensional stability.Expand Specific Solutions03 Quaternary ammonium functionalization techniques
Quaternary ammonium groups are widely used as ion-conducting moieties in anion exchange membranes. Advanced functionalization techniques focus on optimizing the distribution, density, and stability of these groups to enhance conductivity. Methods include controlled grafting, post-polymerization modification, and the development of novel quaternization reagents that improve alkaline stability while maintaining high ionic conductivity.Expand Specific Solutions04 Temperature and humidity effects on conductivity
The conductivity of anion exchange membranes is significantly influenced by operating temperature and humidity conditions. Research focuses on developing membranes with stable conductivity across a wide range of temperatures and humidity levels. Strategies include incorporating hydrophilic domains, designing specific microstructures, and developing self-humidifying membranes that maintain high conductivity even under low humidity conditions.Expand Specific Solutions05 Novel membrane architectures and fabrication methods
Innovative membrane architectures and fabrication techniques are being developed to enhance anion conductivity. These include pore-filling membranes, reinforced structures, electrospun nanofiber supports, and 3D-printed designs. Advanced fabrication methods allow for precise control over membrane morphology, thickness, and uniformity, resulting in optimized ion transport pathways and improved overall conductivity performance for various electrochemical applications.Expand Specific Solutions
Leading Research Groups and Industrial Players in AEM Technology
The anion exchange membrane (AEM) hydration-dependent conductivity market is in a growth phase, with increasing research focus due to its potential in clean energy applications. The market is expanding as demand for fuel cells and electrolyzers rises, though precise size estimates remain limited. Technologically, the field shows moderate maturity with significant ongoing development. Leading players include established chemical corporations like Tokuyama Corp., Asahi Kasei, and Hanwha Solutions, alongside research powerhouses such as MIT, Caltech, and CNRS. University-industry collaborations are accelerating innovation, particularly between institutions like Dalian University of Technology and commercial entities. Recent advancements in membrane materials and hydration management techniques suggest the technology is approaching commercial viability for broader energy applications.
Massachusetts Institute of Technology
Technical Solution: MIT has developed advanced anion exchange membranes (AEMs) with precisely engineered hydrophilic-hydrophobic microphase separation to optimize hydration-dependent conductivity. Their approach involves synthesizing block copolymers with controlled architecture where hydrophilic blocks contain quaternary ammonium groups for anion conduction, while hydrophobic blocks provide mechanical stability. MIT researchers have demonstrated that by controlling the spatial distribution of ionic groups and engineering interconnected hydrophilic channels, they can achieve high hydroxide conductivity (>100 mS/cm) under optimal hydration conditions while minimizing excessive membrane swelling[1]. Their technology incorporates specially designed side chains that maintain sufficient water content even at lower relative humidity, enabling more consistent conductivity across varying hydration levels[2]. Recent innovations include the development of AEMs with self-healing properties that can recover conductivity after dehydration cycles.
Strengths: Superior ionic conductivity at various hydration levels through precise microphase separation engineering; excellent mechanical stability even at high water uptake; innovative polymer architecture that balances water retention and mechanical properties. Weaknesses: Complex synthesis procedures may increase manufacturing costs; potential long-term durability issues under repeated hydration/dehydration cycles; chemical stability in highly alkaline environments remains challenging.
Asahi Kasei Corp.
Technical Solution: Asahi Kasei has pioneered commercial anion exchange membranes with optimized hydration-dependent conductivity through their proprietary "controlled hydration network" technology. Their approach involves synthesizing hydrocarbon-based polymer backbones with precisely distributed quaternary ammonium functional groups and engineered hydrophilic domains. The company's latest generation AEMs feature cross-linked structures with specific hydrophilic-hydrophobic balance that maintains optimal water content for ion conduction while preventing excessive swelling[3]. Asahi Kasei's membranes incorporate specially designed side chains that create interconnected ionic channels, allowing hydroxide ions to move efficiently even at moderate hydration levels. Their manufacturing process includes a proprietary treatment that enhances the membrane's dimensional stability under varying humidity conditions, addressing a key challenge in AEM technology[4]. The company has successfully scaled production of these membranes for commercial applications in alkaline fuel cells and electrolyzers.
Strengths: Established mass production capabilities with consistent quality control; excellent balance between conductivity and mechanical properties; proven long-term stability in commercial applications. Weaknesses: Higher cost compared to some competing materials; performance degradation at extremely low humidity conditions; requires specific operating parameters to maintain optimal hydration levels.
Critical Patents and Research on Water-Mediated Anion Transport
Anion conducting electrolyte membrane and manufacturing method thereof
PatentInactiveUS20130280626A1
Innovation
- A polymeric anion conducting electrolyte membrane is developed using a fluorine polymer or olefinic/aromatic polymer with graft-polymerized halogenated alkyl groups, quaternized with strong organic bases to form stable weak acid-weak base quaternary ammonium salts, balancing conductivity and water uptake.
Phosphate anion-quaternary ammonium ion pair coordinated polymer membranes
PatentWO2020112721A1
Innovation
- Development of phosphate anion-quaternary ammonium ion pair coordinated polymer membranes, where phosphoric acid molecules cluster around quaternary ammonium groups to facilitate proton transport, reducing dependence on water and enhancing conductivity at low humidity.
Durability and Stability Considerations for Hydrated AEMs
The durability and stability of anion exchange membranes (AEMs) under hydrated conditions represent critical challenges that must be addressed for their successful implementation in fuel cells and other electrochemical applications. The hydration-dependent conductivity of AEMs is intrinsically linked to their long-term performance, as water content significantly influences both ionic conductivity and mechanical integrity.
Chemical degradation mechanisms pose substantial threats to hydrated AEMs, particularly through nucleophilic attack by hydroxide ions on the cationic functional groups. This degradation is accelerated at elevated temperatures and higher hydration levels, leading to decreased ion exchange capacity and conductivity over time. The Hofmann elimination reaction, where quaternary ammonium groups decompose into tertiary amines and alkenes, becomes especially problematic under fully hydrated conditions.
Physical stability issues also emerge as AEMs undergo dimensional changes during hydration-dehydration cycles. The repeated swelling and contraction can lead to mechanical fatigue, resulting in microcrack formation and eventual membrane failure. Studies have shown that membranes with higher water uptake typically exhibit more pronounced dimensional instability, creating a challenging trade-off between conductivity and durability.
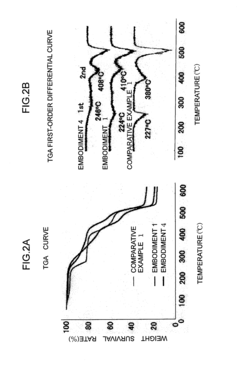
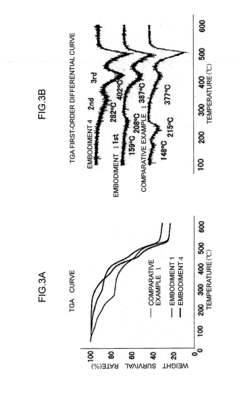
Temperature effects compound these stability concerns, as thermal cycling in hydrated environments accelerates both chemical and mechanical degradation pathways. Above 60°C, many conventional AEMs show dramatically reduced lifespans, with some quaternary ammonium-based membranes losing up to 50% of their conductivity within 500 hours of operation under hydrated conditions.
Recent advances in stabilizing strategies include the development of sterically hindered cations that resist nucleophilic attack and the incorporation of crosslinking agents that maintain dimensional stability while allowing sufficient water uptake for ion transport. Reinforced composite membranes with hydrophilic channels embedded in hydrophobic matrices have demonstrated promising results, maintaining over 80% of initial conductivity after 1000 hours at 80°C under fully hydrated conditions.
The relationship between hydration levels and membrane lifetime presents a complex optimization challenge. While higher hydration generally increases initial conductivity, it often compromises long-term stability. Controlled hydration protocols that maintain water content within an optimal range (typically 20-30% water uptake by weight) have been shown to extend membrane lifetimes significantly while preserving adequate conductivity for practical applications.
Chemical degradation mechanisms pose substantial threats to hydrated AEMs, particularly through nucleophilic attack by hydroxide ions on the cationic functional groups. This degradation is accelerated at elevated temperatures and higher hydration levels, leading to decreased ion exchange capacity and conductivity over time. The Hofmann elimination reaction, where quaternary ammonium groups decompose into tertiary amines and alkenes, becomes especially problematic under fully hydrated conditions.
Physical stability issues also emerge as AEMs undergo dimensional changes during hydration-dehydration cycles. The repeated swelling and contraction can lead to mechanical fatigue, resulting in microcrack formation and eventual membrane failure. Studies have shown that membranes with higher water uptake typically exhibit more pronounced dimensional instability, creating a challenging trade-off between conductivity and durability.
Temperature effects compound these stability concerns, as thermal cycling in hydrated environments accelerates both chemical and mechanical degradation pathways. Above 60°C, many conventional AEMs show dramatically reduced lifespans, with some quaternary ammonium-based membranes losing up to 50% of their conductivity within 500 hours of operation under hydrated conditions.
Recent advances in stabilizing strategies include the development of sterically hindered cations that resist nucleophilic attack and the incorporation of crosslinking agents that maintain dimensional stability while allowing sufficient water uptake for ion transport. Reinforced composite membranes with hydrophilic channels embedded in hydrophobic matrices have demonstrated promising results, maintaining over 80% of initial conductivity after 1000 hours at 80°C under fully hydrated conditions.
The relationship between hydration levels and membrane lifetime presents a complex optimization challenge. While higher hydration generally increases initial conductivity, it often compromises long-term stability. Controlled hydration protocols that maintain water content within an optimal range (typically 20-30% water uptake by weight) have been shown to extend membrane lifetimes significantly while preserving adequate conductivity for practical applications.
Environmental Impact and Sustainability of AEM Technologies
The environmental impact of anion exchange membrane (AEM) technologies represents a critical consideration in their development and deployment. AEM-based systems offer significant sustainability advantages compared to traditional technologies, particularly in energy generation and water treatment applications. The hydration-dependent conductivity of AEMs directly influences their efficiency and longevity, which in turn affects their overall environmental footprint.
AEM fuel cells and electrolyzers demonstrate substantially lower environmental impacts than conventional systems due to their platinum-free catalysts. By eliminating the need for precious metals, AEM technologies reduce resource depletion and minimize the environmental damage associated with mining operations. The hydration management in these systems also enables more efficient operation at varying humidity levels, reducing water consumption in regions facing scarcity challenges.
Life cycle assessments of AEM technologies reveal favorable carbon footprints compared to traditional ion exchange systems. The manufacturing processes for AEMs typically require less energy and generate fewer emissions than perfluorinated membranes used in proton exchange membrane systems. Additionally, the biodegradability of many non-fluorinated AEM materials presents opportunities for reduced end-of-life environmental impacts, though this remains an area requiring further research.
Water consumption represents another crucial sustainability factor for AEM technologies. The hydration-dependent conductivity mechanism necessitates careful water management, but optimized systems can achieve significant water savings compared to conventional alternatives. Recent innovations in AEM materials that maintain conductivity at lower hydration levels have further improved water efficiency, making these technologies particularly valuable in water-stressed regions.
The chemical stability of AEMs also contributes to their sustainability profile. Membranes with enhanced durability require less frequent replacement, reducing waste generation and resource consumption over system lifetimes. Research into hydration-stable AEMs has yielded materials that maintain performance while experiencing fewer degradation issues, thereby extending operational lifespans and improving lifecycle sustainability metrics.
Energy efficiency gains from advanced AEM technologies translate directly into reduced greenhouse gas emissions when deployed at scale. The relationship between hydration levels and ionic conductivity has been optimized in newer materials to achieve higher efficiencies across varying operating conditions, maximizing environmental benefits. These improvements make AEM technologies increasingly viable alternatives to fossil fuel-dependent systems in multiple industrial applications.
Regulatory frameworks worldwide are beginning to recognize the environmental advantages of AEM technologies, with several jurisdictions offering incentives for their adoption. The continued development of hydration-optimized membranes will likely accelerate this trend, positioning AEM systems as key components in sustainable industrial transformation strategies and clean energy transitions.
AEM fuel cells and electrolyzers demonstrate substantially lower environmental impacts than conventional systems due to their platinum-free catalysts. By eliminating the need for precious metals, AEM technologies reduce resource depletion and minimize the environmental damage associated with mining operations. The hydration management in these systems also enables more efficient operation at varying humidity levels, reducing water consumption in regions facing scarcity challenges.
Life cycle assessments of AEM technologies reveal favorable carbon footprints compared to traditional ion exchange systems. The manufacturing processes for AEMs typically require less energy and generate fewer emissions than perfluorinated membranes used in proton exchange membrane systems. Additionally, the biodegradability of many non-fluorinated AEM materials presents opportunities for reduced end-of-life environmental impacts, though this remains an area requiring further research.
Water consumption represents another crucial sustainability factor for AEM technologies. The hydration-dependent conductivity mechanism necessitates careful water management, but optimized systems can achieve significant water savings compared to conventional alternatives. Recent innovations in AEM materials that maintain conductivity at lower hydration levels have further improved water efficiency, making these technologies particularly valuable in water-stressed regions.
The chemical stability of AEMs also contributes to their sustainability profile. Membranes with enhanced durability require less frequent replacement, reducing waste generation and resource consumption over system lifetimes. Research into hydration-stable AEMs has yielded materials that maintain performance while experiencing fewer degradation issues, thereby extending operational lifespans and improving lifecycle sustainability metrics.
Energy efficiency gains from advanced AEM technologies translate directly into reduced greenhouse gas emissions when deployed at scale. The relationship between hydration levels and ionic conductivity has been optimized in newer materials to achieve higher efficiencies across varying operating conditions, maximizing environmental benefits. These improvements make AEM technologies increasingly viable alternatives to fossil fuel-dependent systems in multiple industrial applications.
Regulatory frameworks worldwide are beginning to recognize the environmental advantages of AEM technologies, with several jurisdictions offering incentives for their adoption. The continued development of hydration-optimized membranes will likely accelerate this trend, positioning AEM systems as key components in sustainable industrial transformation strategies and clean energy transitions.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!