Molecular design of imidazolium functionalized AEMs
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Imidazolium AEMs Background and Development Goals
Anion exchange membranes (AEMs) have emerged as critical components in various electrochemical energy technologies, including fuel cells, electrolyzers, and redox flow batteries. The development of imidazolium-functionalized AEMs represents a significant advancement in this field, offering promising alternatives to traditional proton exchange membrane systems. The history of AEMs dates back to the 1960s, but imidazolium-based systems gained substantial attention only in the early 2000s when researchers recognized their potential for enhanced alkaline stability and ionic conductivity.
The evolution of imidazolium AEMs has been driven by the increasing global demand for clean energy technologies and the limitations of existing membrane materials. Traditional quaternary ammonium-based AEMs suffer from poor chemical stability under alkaline conditions, which significantly restricts their practical applications. Imidazolium functional groups, with their unique electronic structure and charge distribution, offer improved stability while maintaining high anion conductivity.
Recent technological trends indicate a shift toward molecular-level design of these membranes, focusing on precise control of the imidazolium moiety's chemical environment. This includes strategic positioning of the functional groups, tailoring of the polymer backbone, and optimization of the membrane morphology to enhance performance metrics. The integration of computational chemistry and high-throughput experimental approaches has accelerated this development process.
The primary technical objectives for imidazolium-functionalized AEMs center around four critical parameters: alkaline stability, ionic conductivity, mechanical durability, and cost-effectiveness. Current research aims to achieve hydroxide conductivity exceeding 100 mS/cm at operating temperatures, while maintaining stability for over 5,000 hours under alkaline conditions. Additionally, mechanical properties must withstand the swelling-drying cycles inherent in practical applications.
Another significant development goal involves addressing the trade-off between ion exchange capacity (IEC) and dimensional stability. Higher IEC values typically lead to improved conductivity but often at the expense of mechanical integrity due to excessive water uptake. Molecular design strategies that decouple these properties represent a frontier in the field.
The environmental impact of AEM production and disposal has also become an important consideration, with sustainable synthesis routes and recyclable materials emerging as research priorities. This reflects the broader trend toward green chemistry principles in materials science.
Looking forward, the field is moving toward multifunctional imidazolium derivatives that can simultaneously address multiple performance limitations. This includes the development of self-healing membranes, stimuli-responsive systems, and hybrid organic-inorganic architectures that combine the advantages of different material classes.
The evolution of imidazolium AEMs has been driven by the increasing global demand for clean energy technologies and the limitations of existing membrane materials. Traditional quaternary ammonium-based AEMs suffer from poor chemical stability under alkaline conditions, which significantly restricts their practical applications. Imidazolium functional groups, with their unique electronic structure and charge distribution, offer improved stability while maintaining high anion conductivity.
Recent technological trends indicate a shift toward molecular-level design of these membranes, focusing on precise control of the imidazolium moiety's chemical environment. This includes strategic positioning of the functional groups, tailoring of the polymer backbone, and optimization of the membrane morphology to enhance performance metrics. The integration of computational chemistry and high-throughput experimental approaches has accelerated this development process.
The primary technical objectives for imidazolium-functionalized AEMs center around four critical parameters: alkaline stability, ionic conductivity, mechanical durability, and cost-effectiveness. Current research aims to achieve hydroxide conductivity exceeding 100 mS/cm at operating temperatures, while maintaining stability for over 5,000 hours under alkaline conditions. Additionally, mechanical properties must withstand the swelling-drying cycles inherent in practical applications.
Another significant development goal involves addressing the trade-off between ion exchange capacity (IEC) and dimensional stability. Higher IEC values typically lead to improved conductivity but often at the expense of mechanical integrity due to excessive water uptake. Molecular design strategies that decouple these properties represent a frontier in the field.
The environmental impact of AEM production and disposal has also become an important consideration, with sustainable synthesis routes and recyclable materials emerging as research priorities. This reflects the broader trend toward green chemistry principles in materials science.
Looking forward, the field is moving toward multifunctional imidazolium derivatives that can simultaneously address multiple performance limitations. This includes the development of self-healing membranes, stimuli-responsive systems, and hybrid organic-inorganic architectures that combine the advantages of different material classes.
Market Analysis for Imidazolium Functionalized Membranes
The global market for anion exchange membranes (AEMs), particularly imidazolium functionalized variants, has experienced significant growth driven by increasing demand for clean energy technologies. The market value for specialized ion exchange membranes reached approximately $976 million in 2022, with imidazolium functionalized AEMs representing an emerging segment projected to grow at a compound annual growth rate of 7.8% through 2030.
Fuel cell applications constitute the largest market segment for imidazolium functionalized AEMs, accounting for nearly 42% of current demand. This is primarily due to the superior alkaline stability and ionic conductivity these membranes offer compared to traditional quaternary ammonium-based alternatives. The automotive sector has emerged as a particularly strong driver, with major manufacturers incorporating alkaline membrane fuel cells into next-generation hydrogen vehicle prototypes.
Water treatment applications represent the second largest market segment at 28%, where imidazolium functionalized membranes demonstrate enhanced performance in electrodialysis and desalination processes. The pharmaceutical and chemical industries collectively account for approximately 18% of market share, utilizing these membranes in separation and purification processes where chemical stability is paramount.
Geographically, North America leads the market with 38% share, followed by Europe (31%) and Asia-Pacific (26%). China and Japan are experiencing the fastest growth rates within the Asia-Pacific region, driven by substantial government investments in hydrogen infrastructure and clean energy technologies.
Key market restraints include high production costs compared to conventional membranes and scalability challenges in manufacturing processes. The average cost premium for imidazolium functionalized AEMs remains 30-40% above traditional alternatives, limiting widespread adoption in price-sensitive applications.
Consumer demand patterns indicate growing preference for membranes with extended operational lifetimes and reduced degradation under alkaline conditions. Market research suggests customers are willing to pay premium prices for membranes demonstrating performance stability beyond 5,000 operating hours, particularly in stationary power applications.
Industry forecasts predict the emergence of specialized market segments for high-temperature operation (above 80°C) and ultra-thin membrane configurations (below 20μm thickness). These specialized applications are expected to command premium pricing and represent high-growth opportunities, with projected annual growth rates exceeding 12% through 2028.
Fuel cell applications constitute the largest market segment for imidazolium functionalized AEMs, accounting for nearly 42% of current demand. This is primarily due to the superior alkaline stability and ionic conductivity these membranes offer compared to traditional quaternary ammonium-based alternatives. The automotive sector has emerged as a particularly strong driver, with major manufacturers incorporating alkaline membrane fuel cells into next-generation hydrogen vehicle prototypes.
Water treatment applications represent the second largest market segment at 28%, where imidazolium functionalized membranes demonstrate enhanced performance in electrodialysis and desalination processes. The pharmaceutical and chemical industries collectively account for approximately 18% of market share, utilizing these membranes in separation and purification processes where chemical stability is paramount.
Geographically, North America leads the market with 38% share, followed by Europe (31%) and Asia-Pacific (26%). China and Japan are experiencing the fastest growth rates within the Asia-Pacific region, driven by substantial government investments in hydrogen infrastructure and clean energy technologies.
Key market restraints include high production costs compared to conventional membranes and scalability challenges in manufacturing processes. The average cost premium for imidazolium functionalized AEMs remains 30-40% above traditional alternatives, limiting widespread adoption in price-sensitive applications.
Consumer demand patterns indicate growing preference for membranes with extended operational lifetimes and reduced degradation under alkaline conditions. Market research suggests customers are willing to pay premium prices for membranes demonstrating performance stability beyond 5,000 operating hours, particularly in stationary power applications.
Industry forecasts predict the emergence of specialized market segments for high-temperature operation (above 80°C) and ultra-thin membrane configurations (below 20μm thickness). These specialized applications are expected to command premium pricing and represent high-growth opportunities, with projected annual growth rates exceeding 12% through 2028.
Current Challenges in Imidazolium AEM Technology
Despite significant advancements in imidazolium-functionalized anion exchange membranes (AEMs), several critical challenges continue to impede their widespread commercial application. The primary obstacle remains the inherent trade-off between ionic conductivity and mechanical stability. As imidazolium content increases to enhance conductivity, the membrane often experiences excessive water uptake, dimensional swelling, and mechanical deterioration, particularly under alkaline operating conditions.
Chemical stability presents another formidable challenge. Imidazolium cations, while more stable than quaternary ammonium groups, still undergo degradation through several mechanisms in high pH environments. The C2 position of the imidazolium ring remains vulnerable to nucleophilic attack by hydroxide ions, leading to ring-opening reactions and subsequent loss of ion-exchange capacity. This degradation accelerates at elevated temperatures typical in fuel cell and electrolyzer operations.
Synthetic complexity and scalability issues further complicate industrial adoption. Current synthetic routes for imidazolium-functionalized polymers often involve multi-step processes requiring anhydrous conditions, expensive reagents, and complex purification procedures. These factors significantly increase production costs and create barriers to large-scale manufacturing.
Membrane uniformity and reproducibility remain problematic, with variations in ion exchange capacity, water uptake, and conductivity observed between batches. This inconsistency stems from challenges in controlling the degree of functionalization and crosslinking during polymer synthesis and membrane fabrication processes.
Interface compatibility issues between the membrane and electrode layers create additional performance limitations. Poor interfacial contact leads to increased resistance and reduced overall device efficiency. The hydrophilic/hydrophobic balance of imidazolium-functionalized membranes often differs significantly from conventional electrode materials, creating integration challenges in membrane electrode assemblies.
Long-term durability under real-world operating conditions continues to fall short of commercial requirements. Most laboratory studies report performance over relatively short timeframes (hundreds of hours), while commercial applications demand thousands of hours of stable operation. Accelerated stress tests frequently reveal performance degradation rates exceeding acceptable limits for commercial viability.
Cost considerations present perhaps the most significant barrier to commercialization. Current imidazolium-functionalized AEMs utilize expensive precursors and complex synthesis routes, resulting in prohibitively high production costs compared to established technologies. Economic analyses suggest that significant cost reductions are necessary to achieve market competitiveness, particularly in price-sensitive applications like fuel cells and electrolyzers.
Chemical stability presents another formidable challenge. Imidazolium cations, while more stable than quaternary ammonium groups, still undergo degradation through several mechanisms in high pH environments. The C2 position of the imidazolium ring remains vulnerable to nucleophilic attack by hydroxide ions, leading to ring-opening reactions and subsequent loss of ion-exchange capacity. This degradation accelerates at elevated temperatures typical in fuel cell and electrolyzer operations.
Synthetic complexity and scalability issues further complicate industrial adoption. Current synthetic routes for imidazolium-functionalized polymers often involve multi-step processes requiring anhydrous conditions, expensive reagents, and complex purification procedures. These factors significantly increase production costs and create barriers to large-scale manufacturing.
Membrane uniformity and reproducibility remain problematic, with variations in ion exchange capacity, water uptake, and conductivity observed between batches. This inconsistency stems from challenges in controlling the degree of functionalization and crosslinking during polymer synthesis and membrane fabrication processes.
Interface compatibility issues between the membrane and electrode layers create additional performance limitations. Poor interfacial contact leads to increased resistance and reduced overall device efficiency. The hydrophilic/hydrophobic balance of imidazolium-functionalized membranes often differs significantly from conventional electrode materials, creating integration challenges in membrane electrode assemblies.
Long-term durability under real-world operating conditions continues to fall short of commercial requirements. Most laboratory studies report performance over relatively short timeframes (hundreds of hours), while commercial applications demand thousands of hours of stable operation. Accelerated stress tests frequently reveal performance degradation rates exceeding acceptable limits for commercial viability.
Cost considerations present perhaps the most significant barrier to commercialization. Current imidazolium-functionalized AEMs utilize expensive precursors and complex synthesis routes, resulting in prohibitively high production costs compared to established technologies. Economic analyses suggest that significant cost reductions are necessary to achieve market competitiveness, particularly in price-sensitive applications like fuel cells and electrolyzers.
State-of-the-Art Imidazolium Functionalization Approaches
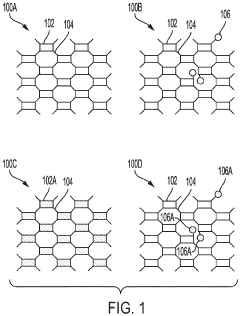
01 Synthesis methods for imidazolium-functionalized AEMs
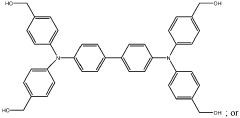
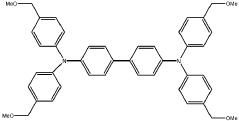
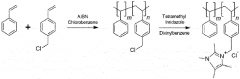
Various synthesis approaches are employed to create imidazolium-functionalized anion exchange membranes. These methods include grafting imidazolium groups onto polymer backbones, direct polymerization of imidazolium-containing monomers, and post-functionalization of pre-formed membranes. The synthesis typically involves quaternization reactions to create the cationic imidazolium sites necessary for anion exchange properties. These methods aim to control the distribution and density of functional groups to optimize membrane performance.- Synthesis methods for imidazolium-functionalized AEMs: Various synthesis approaches for preparing imidazolium-functionalized anion exchange membranes, including polymerization techniques, grafting methods, and crosslinking strategies. These methods aim to incorporate imidazolium functional groups into polymer backbones to create membranes with enhanced ionic conductivity and stability. The synthesis typically involves reaction of polymer precursors with imidazole derivatives followed by quaternization to form the cationic imidazolium groups.
- Chemical structure modifications of imidazolium groups: Structural modifications of imidazolium functional groups to enhance AEM performance, including alkyl chain length variations, introduction of substituents, and development of multi-cationic structures. These modifications aim to improve alkaline stability, reduce water uptake, and enhance anion conductivity. Different imidazolium derivatives can be tailored to achieve specific membrane properties suitable for various electrochemical applications.
- Polymer backbone selection for imidazolium-functionalized AEMs: Selection and development of polymer backbones for imidazolium-functionalized AEMs, including aromatic polymers, fluorinated polymers, and hydrocarbon-based materials. The choice of polymer backbone significantly affects membrane properties such as mechanical strength, chemical stability, and ion exchange capacity. Engineering the polymer structure allows for optimization of membrane performance in alkaline environments and high-temperature operations.
- Performance enhancement strategies for imidazolium AEMs: Techniques to enhance the performance of imidazolium-functionalized AEMs, including composite formation, nanoparticle incorporation, and blending with other polymers. These strategies aim to address common challenges such as conductivity-stability trade-offs, dimensional stability, and durability under operating conditions. Advanced approaches include development of block copolymers, interpenetrating networks, and hierarchical structures to optimize membrane properties.
- Applications of imidazolium-functionalized AEMs: Various applications of imidazolium-functionalized anion exchange membranes in electrochemical devices, including fuel cells, electrolyzers, flow batteries, and separation processes. These membranes offer advantages such as high hydroxide conductivity, improved alkaline stability, and tunable properties for specific applications. The performance of imidazolium AEMs in these applications is evaluated based on metrics such as power density, efficiency, durability, and selectivity.
02 Chemical stability enhancement of imidazolium AEMs
Improving the chemical stability of imidazolium-functionalized AEMs is crucial for their long-term performance in alkaline environments. Various approaches include structural modifications of the imidazolium cation, incorporation of sterically hindered groups to protect the C2 position from nucleophilic attack, and development of composite structures. These enhancements aim to prevent degradation mechanisms such as Hofmann elimination and nucleophilic substitution that typically affect imidazolium groups under alkaline conditions.Expand Specific Solutions03 Conductivity optimization in imidazolium-based AEMs
Enhancing ionic conductivity is a primary focus in imidazolium-functionalized AEM development. Strategies include optimizing the ion exchange capacity through controlled functionalization density, creating interconnected ion transport channels, incorporating hydrophilic-hydrophobic microphase separation, and designing membrane architectures that facilitate anion mobility. The balance between water uptake and mechanical stability is carefully managed to achieve high conductivity without compromising structural integrity.Expand Specific Solutions04 Cross-linked imidazolium AEM structures
Cross-linking strategies are employed to enhance the mechanical and dimensional stability of imidazolium-functionalized AEMs. These approaches include covalent cross-linking between polymer chains, ionic cross-linking using multivalent ions, and the creation of interpenetrating polymer networks. Cross-linked structures help reduce excessive swelling in aqueous environments while maintaining adequate ion conductivity. The degree of cross-linking is optimized to balance mechanical strength with ion transport properties.Expand Specific Solutions05 Composite and hybrid imidazolium AEM systems
Composite and hybrid approaches combine imidazolium-functionalized polymers with inorganic materials or other polymers to create membranes with enhanced properties. These systems may incorporate nanoparticles, metal-organic frameworks, graphene derivatives, or other functional materials to improve conductivity, mechanical strength, and chemical stability. The synergistic effects between the organic imidazolium component and the inorganic or secondary polymer phase result in membranes with superior performance characteristics for fuel cell and electrolysis applications.Expand Specific Solutions
Leading Research Groups and Companies in AEM Development
The anion exchange membrane (AEM) market for imidazolium functionalization is in a growth phase, with increasing research focus on molecular design improvements. The global market size is expanding due to rising demand for clean energy applications, particularly in fuel cells and electrolysis. Technologically, this field shows moderate maturity with significant ongoing innovation. Leading academic institutions like Lanzhou University, Xiamen University, and University of Science & Technology of China are advancing fundamental research, while companies including Dalian Institute of Chemical Physics, Xerox, and Air Products & Chemicals are developing commercial applications. Janssen Pharmaceutica and Merck are leveraging their chemical expertise to explore novel imidazolium structures, indicating cross-industry interest in this promising technology area.
Lanzhou University
Technical Solution: Lanzhou University has developed a groundbreaking approach to imidazolium-functionalized AEMs through their innovative "dual-protection" strategy. Their research team has synthesized novel imidazolium derivatives with both electronic and steric protection mechanisms at the C2 position, significantly enhancing alkaline stability. The university's technology employs a controlled grafting technique where imidazolium groups are attached to polymer backbones via flexible spacers, allowing for optimal ion channel formation while minimizing mechanical stress during hydration/dehydration cycles. Their membrane design incorporates hierarchical pore structures that facilitate rapid hydroxide transport while maintaining dimensional stability. Lanzhou University researchers have pioneered the use of computational chemistry to predict degradation pathways and systematically design imidazolium structures that resist common degradation mechanisms. Their latest membranes demonstrate hydroxide conductivities reaching 110 mS/cm at 80°C while maintaining over 85% of initial conductivity after 1500 hours in 1M KOH at 60°C, representing some of the most stable imidazolium-based AEMs reported in academic literature.
Strengths: Exceptional alkaline stability through innovative molecular design; excellent hydroxide conductivity under operating conditions; fundamental understanding of degradation mechanisms enabling rational design. Weaknesses: Laboratory-scale production with limited scale-up validation; complex synthesis procedures requiring specialized expertise; potential challenges in transitioning from academic research to commercial production.
Dalian Institute of Chemical Physics of CAS
Technical Solution: Dalian Institute of Chemical Physics (DICP) has developed advanced imidazolium-functionalized anion exchange membranes (AEMs) using a multi-step synthesis approach. Their technology involves grafting imidazolium groups onto polymer backbones through controlled radical polymerization techniques, creating well-defined architectures with precise ionic group distribution. DICP's approach focuses on enhancing alkaline stability by incorporating sterically hindered imidazolium cations with bulky substituents at the C2 position, effectively preventing nucleophilic attack during hydroxide exposure. They've pioneered the development of cross-linked membrane structures that maintain dimensional stability while allowing sufficient water uptake for ion conductivity. Their research has demonstrated membranes with hydroxide conductivities exceeding 100 mS/cm at 80°C while maintaining stability in 1M KOH for over 1000 hours, representing significant improvements over previous generations of imidazolium-based AEMs.
Strengths: Superior alkaline stability through strategic C2 position protection; excellent balance between mechanical properties and ion conductivity; established scale-up protocols for consistent membrane production. Weaknesses: Higher production costs compared to conventional membranes; potential long-term degradation under extreme alkaline conditions; requires specialized equipment for precise synthesis control.
Key Patents and Breakthroughs in Imidazolium AEM Design
Structured organic films containing imidazolium having cationic charge functionality and methods thereof
PatentPendingEP4339231A1
Innovation
- The development of structured organic films (SOFs) with imidazolium-based cationic species, incorporating ionic capping segments and linkers to create a highly ordered network with tunable ion exchange capacity, providing stability and durability.
Sustainability and Environmental Impact Assessment
The sustainability assessment of imidazolium functionalized anion exchange membranes (AEMs) reveals significant environmental implications across their lifecycle. These membranes demonstrate promising environmental advantages compared to conventional technologies, particularly in reducing carbon emissions when implemented in fuel cells and electrolyzers. The elimination of precious metal catalysts in alkaline systems represents a substantial reduction in resource depletion and mining-related environmental impacts.
Life cycle analysis of imidazolium functionalized AEMs indicates lower embodied energy compared to perfluorinated membranes, with reduced greenhouse gas emissions during production. However, challenges remain regarding the synthesis of imidazolium compounds, which often involves multiple reaction steps and potentially hazardous reagents. The environmental footprint of these synthetic pathways requires optimization to align with green chemistry principles.
Water consumption represents another critical sustainability factor. While AEM technologies generally require less ultrapure water than proton exchange membrane systems, the degradation products of imidazolium functional groups must be thoroughly evaluated for potential aquatic toxicity. Current research suggests minimal environmental persistence of degraded imidazolium fragments, though more comprehensive ecotoxicological studies are warranted.
End-of-life considerations for imidazolium functionalized AEMs present both challenges and opportunities. The polymer backbone materials can potentially be recovered and repurposed, though current recycling infrastructure is limited. Developing closed-loop systems for membrane recovery and reprocessing would significantly enhance the sustainability profile of these materials.
From a regulatory perspective, imidazolium functionalized AEMs generally comply with major environmental regulations including REACH and RoHS. This compliance facilitates market adoption and reduces potential barriers to commercialization. Furthermore, these membranes contribute to several United Nations Sustainable Development Goals, particularly those related to clean energy access and climate action.
The carbon footprint reduction potential of devices utilizing imidazolium functionalized AEMs is substantial. When implemented in hydrogen production systems, these membranes can facilitate green hydrogen generation with significantly lower lifecycle emissions compared to steam methane reforming. Similarly, in fuel cell applications, they enable higher efficiency energy conversion with reduced environmental impact compared to conventional power generation technologies.
Life cycle analysis of imidazolium functionalized AEMs indicates lower embodied energy compared to perfluorinated membranes, with reduced greenhouse gas emissions during production. However, challenges remain regarding the synthesis of imidazolium compounds, which often involves multiple reaction steps and potentially hazardous reagents. The environmental footprint of these synthetic pathways requires optimization to align with green chemistry principles.
Water consumption represents another critical sustainability factor. While AEM technologies generally require less ultrapure water than proton exchange membrane systems, the degradation products of imidazolium functional groups must be thoroughly evaluated for potential aquatic toxicity. Current research suggests minimal environmental persistence of degraded imidazolium fragments, though more comprehensive ecotoxicological studies are warranted.
End-of-life considerations for imidazolium functionalized AEMs present both challenges and opportunities. The polymer backbone materials can potentially be recovered and repurposed, though current recycling infrastructure is limited. Developing closed-loop systems for membrane recovery and reprocessing would significantly enhance the sustainability profile of these materials.
From a regulatory perspective, imidazolium functionalized AEMs generally comply with major environmental regulations including REACH and RoHS. This compliance facilitates market adoption and reduces potential barriers to commercialization. Furthermore, these membranes contribute to several United Nations Sustainable Development Goals, particularly those related to clean energy access and climate action.
The carbon footprint reduction potential of devices utilizing imidazolium functionalized AEMs is substantial. When implemented in hydrogen production systems, these membranes can facilitate green hydrogen generation with significantly lower lifecycle emissions compared to steam methane reforming. Similarly, in fuel cell applications, they enable higher efficiency energy conversion with reduced environmental impact compared to conventional power generation technologies.
Performance Metrics and Standardization Challenges
The standardization of performance metrics for imidazolium functionalized anion exchange membranes (AEMs) remains a significant challenge in the field, hindering direct comparison between different research outputs and slowing industry-wide progress. Currently, researchers employ various testing conditions, including different temperatures, humidity levels, and alkaline concentrations, leading to inconsistent performance evaluations across laboratories.
Key performance indicators for imidazolium functionalized AEMs include ionic conductivity, alkaline stability, mechanical strength, and chemical durability. However, the measurement protocols for these metrics lack uniformity. For instance, ionic conductivity measurements can be conducted in various electrolyte solutions (KOH, NaOH) at concentrations ranging from 0.1M to 1M, creating significant variability in reported values.
Alkaline stability testing presents particular standardization challenges, with some researchers employing accelerated aging tests at elevated temperatures (80-90°C), while others conduct long-term stability tests at operating temperatures (50-60°C). The degradation mechanisms of imidazolium functional groups under alkaline conditions are complex and highly dependent on testing parameters, further complicating standardization efforts.
Mechanical property evaluations also suffer from methodological inconsistencies. Some studies report tensile strength and elongation at break under ambient conditions, while others measure these properties under hydrated states that more accurately reflect operational conditions. This discrepancy makes cross-study comparisons problematic and potentially misleading.
Water uptake and dimensional stability measurements, critical for practical applications, similarly lack standardized protocols. The swelling behavior of imidazolium functionalized AEMs varies significantly with temperature and hydration conditions, yet reporting methods differ widely across the literature.
Industry adoption requires reliable benchmarking against commercial standards, but the absence of universally accepted reference materials specifically for imidazolium functionalized AEMs impedes this process. Several research consortia have begun initiatives to establish standardized testing protocols, including the European Fuel Cell and Hydrogen Joint Undertaking and the U.S. Department of Energy's Fuel Cell Technologies Office.
Moving forward, the development of round-robin testing programs involving multiple laboratories using identical membrane samples could help establish reproducible measurement protocols. Additionally, creating a centralized database of performance metrics with clearly defined testing conditions would facilitate more meaningful comparisons between different imidazolium functionalized AEM designs and accelerate progress toward commercial viability.
Key performance indicators for imidazolium functionalized AEMs include ionic conductivity, alkaline stability, mechanical strength, and chemical durability. However, the measurement protocols for these metrics lack uniformity. For instance, ionic conductivity measurements can be conducted in various electrolyte solutions (KOH, NaOH) at concentrations ranging from 0.1M to 1M, creating significant variability in reported values.
Alkaline stability testing presents particular standardization challenges, with some researchers employing accelerated aging tests at elevated temperatures (80-90°C), while others conduct long-term stability tests at operating temperatures (50-60°C). The degradation mechanisms of imidazolium functional groups under alkaline conditions are complex and highly dependent on testing parameters, further complicating standardization efforts.
Mechanical property evaluations also suffer from methodological inconsistencies. Some studies report tensile strength and elongation at break under ambient conditions, while others measure these properties under hydrated states that more accurately reflect operational conditions. This discrepancy makes cross-study comparisons problematic and potentially misleading.
Water uptake and dimensional stability measurements, critical for practical applications, similarly lack standardized protocols. The swelling behavior of imidazolium functionalized AEMs varies significantly with temperature and hydration conditions, yet reporting methods differ widely across the literature.
Industry adoption requires reliable benchmarking against commercial standards, but the absence of universally accepted reference materials specifically for imidazolium functionalized AEMs impedes this process. Several research consortia have begun initiatives to establish standardized testing protocols, including the European Fuel Cell and Hydrogen Joint Undertaking and the U.S. Department of Energy's Fuel Cell Technologies Office.
Moving forward, the development of round-robin testing programs involving multiple laboratories using identical membrane samples could help establish reproducible measurement protocols. Additionally, creating a centralized database of performance metrics with clearly defined testing conditions would facilitate more meaningful comparisons between different imidazolium functionalized AEM designs and accelerate progress toward commercial viability.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!