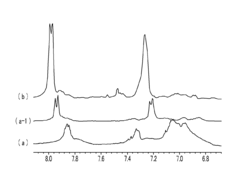
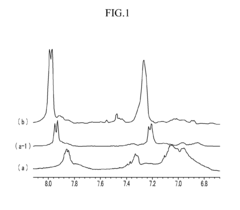
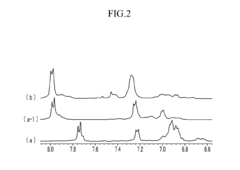
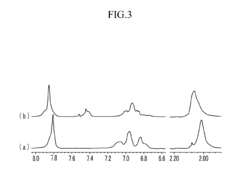
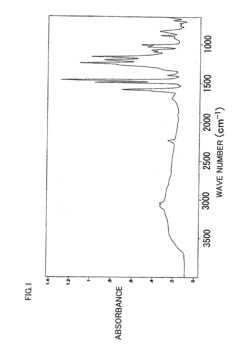
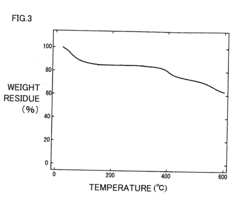
Poly(aryl ether) based membranes with improved conductivity
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PAE Membrane Technology Background and Objectives
Poly(aryl ether) (PAE) based membranes represent a significant advancement in polymer membrane technology, with applications spanning from fuel cells to water purification systems. The evolution of these membranes can be traced back to the 1960s when researchers first began exploring aromatic polymers for their exceptional thermal and chemical stability. Over subsequent decades, PAE membranes have undergone substantial refinement, particularly in addressing conductivity limitations that initially restricted their widespread adoption.
The technological trajectory of PAE membranes has been characterized by progressive improvements in their chemical structure, morphology, and functional properties. Early iterations focused primarily on mechanical durability, while recent developments have shifted toward enhancing ionic conductivity without compromising structural integrity. This evolution reflects broader trends in membrane technology, where performance optimization increasingly requires multidisciplinary approaches combining polymer chemistry, materials science, and electrochemistry.
Current research objectives in PAE membrane technology center on achieving significantly improved ionic conductivity while maintaining the inherent advantages of these materials, including excellent mechanical properties, chemical resistance, and thermal stability. Specifically, researchers aim to surpass the conductivity threshold of 100 mS/cm under operating conditions, which would position PAE membranes as viable alternatives to perfluorosulfonic acid membranes in various electrochemical applications.
The strategic importance of this research extends beyond academic interest, addressing critical challenges in renewable energy technologies, particularly fuel cells and flow batteries. Enhanced conductivity in PAE membranes could substantially improve energy conversion efficiency, reduce system costs, and extend operational lifetimes of these devices. Additionally, advances in this field align with global sustainability goals by enabling more efficient energy storage and conversion systems.
From a materials science perspective, the technical objectives include developing novel sulfonation methods, exploring hybrid organic-inorganic composites, and implementing innovative cross-linking strategies to create more efficient ion transport channels within the membrane structure. Researchers are also investigating the relationship between polymer architecture and conductivity, seeking to establish design principles that can guide future membrane development.
The anticipated technological trajectory suggests that PAE membranes with improved conductivity could reach commercial viability within the next 5-7 years, potentially disrupting established markets currently dominated by more expensive alternatives. This timeline assumes continued progress in addressing key challenges, particularly the conductivity-swelling trade-off that has historically limited the practical application of these materials.
The technological trajectory of PAE membranes has been characterized by progressive improvements in their chemical structure, morphology, and functional properties. Early iterations focused primarily on mechanical durability, while recent developments have shifted toward enhancing ionic conductivity without compromising structural integrity. This evolution reflects broader trends in membrane technology, where performance optimization increasingly requires multidisciplinary approaches combining polymer chemistry, materials science, and electrochemistry.
Current research objectives in PAE membrane technology center on achieving significantly improved ionic conductivity while maintaining the inherent advantages of these materials, including excellent mechanical properties, chemical resistance, and thermal stability. Specifically, researchers aim to surpass the conductivity threshold of 100 mS/cm under operating conditions, which would position PAE membranes as viable alternatives to perfluorosulfonic acid membranes in various electrochemical applications.
The strategic importance of this research extends beyond academic interest, addressing critical challenges in renewable energy technologies, particularly fuel cells and flow batteries. Enhanced conductivity in PAE membranes could substantially improve energy conversion efficiency, reduce system costs, and extend operational lifetimes of these devices. Additionally, advances in this field align with global sustainability goals by enabling more efficient energy storage and conversion systems.
From a materials science perspective, the technical objectives include developing novel sulfonation methods, exploring hybrid organic-inorganic composites, and implementing innovative cross-linking strategies to create more efficient ion transport channels within the membrane structure. Researchers are also investigating the relationship between polymer architecture and conductivity, seeking to establish design principles that can guide future membrane development.
The anticipated technological trajectory suggests that PAE membranes with improved conductivity could reach commercial viability within the next 5-7 years, potentially disrupting established markets currently dominated by more expensive alternatives. This timeline assumes continued progress in addressing key challenges, particularly the conductivity-swelling trade-off that has historically limited the practical application of these materials.
Market Analysis for High-Conductivity Polymer Membranes
The global market for high-conductivity polymer membranes has experienced significant growth in recent years, driven primarily by increasing demand in energy storage, fuel cells, and water treatment applications. The poly(aryl ether) based membrane segment specifically has gained substantial traction due to its superior thermal stability, mechanical strength, and chemical resistance compared to conventional alternatives.
The energy storage sector represents the largest market share for these advanced membranes, with lithium-ion batteries leading consumption. The automotive industry's shift toward electric vehicles has created unprecedented demand, with the market for battery separators expected to grow at a compound annual growth rate of 12% through 2028. This transition is further accelerated by governmental policies promoting clean energy technologies across North America, Europe, and Asia-Pacific regions.
Fuel cell applications constitute another rapidly expanding market segment, particularly proton exchange membrane fuel cells (PEMFCs) and anion exchange membrane fuel cells (AEMFCs). The hydrogen economy initiatives in Japan, South Korea, and Germany have established strong demand foundations, while China's aggressive investment in fuel cell technology has created a robust growth environment for high-conductivity membranes.
Water treatment and purification systems represent the third major application area, with reverse osmosis and electrodialysis processes requiring increasingly efficient ion-conductive membranes. The global water crisis and stricter environmental regulations have intensified market demand, particularly in water-scarce regions across the Middle East, North Africa, and parts of Asia.
Geographically, Asia-Pacific dominates the market consumption, accounting for approximately 45% of global demand, followed by North America and Europe. China has emerged as both the largest producer and consumer, driven by its massive manufacturing base for electronics and aggressive clean energy policies.
The market structure remains relatively concentrated, with major chemical corporations and specialized membrane manufacturers controlling significant market share. However, the landscape is becoming increasingly competitive as new entrants from emerging economies introduce cost-effective alternatives, particularly for less demanding applications.
Price sensitivity varies significantly across application segments, with high-end applications like aerospace and medical devices willing to pay premium prices for superior performance, while mass-market applications like consumer electronics exert considerable downward price pressure. This dynamic has created distinct market tiers with different value propositions and competitive dynamics.
Future market growth is expected to be driven by technological advancements enabling higher conductivity at lower costs, expansion of renewable energy infrastructure, and increasing adoption of electric vehicles. The development of poly(aryl ether) membranes with improved conductivity represents a critical innovation pathway that could significantly reshape competitive positioning within this high-growth market.
The energy storage sector represents the largest market share for these advanced membranes, with lithium-ion batteries leading consumption. The automotive industry's shift toward electric vehicles has created unprecedented demand, with the market for battery separators expected to grow at a compound annual growth rate of 12% through 2028. This transition is further accelerated by governmental policies promoting clean energy technologies across North America, Europe, and Asia-Pacific regions.
Fuel cell applications constitute another rapidly expanding market segment, particularly proton exchange membrane fuel cells (PEMFCs) and anion exchange membrane fuel cells (AEMFCs). The hydrogen economy initiatives in Japan, South Korea, and Germany have established strong demand foundations, while China's aggressive investment in fuel cell technology has created a robust growth environment for high-conductivity membranes.
Water treatment and purification systems represent the third major application area, with reverse osmosis and electrodialysis processes requiring increasingly efficient ion-conductive membranes. The global water crisis and stricter environmental regulations have intensified market demand, particularly in water-scarce regions across the Middle East, North Africa, and parts of Asia.
Geographically, Asia-Pacific dominates the market consumption, accounting for approximately 45% of global demand, followed by North America and Europe. China has emerged as both the largest producer and consumer, driven by its massive manufacturing base for electronics and aggressive clean energy policies.
The market structure remains relatively concentrated, with major chemical corporations and specialized membrane manufacturers controlling significant market share. However, the landscape is becoming increasingly competitive as new entrants from emerging economies introduce cost-effective alternatives, particularly for less demanding applications.
Price sensitivity varies significantly across application segments, with high-end applications like aerospace and medical devices willing to pay premium prices for superior performance, while mass-market applications like consumer electronics exert considerable downward price pressure. This dynamic has created distinct market tiers with different value propositions and competitive dynamics.
Future market growth is expected to be driven by technological advancements enabling higher conductivity at lower costs, expansion of renewable energy infrastructure, and increasing adoption of electric vehicles. The development of poly(aryl ether) membranes with improved conductivity represents a critical innovation pathway that could significantly reshape competitive positioning within this high-growth market.
Current Challenges in PAE Membrane Conductivity Enhancement
Despite significant advancements in poly(aryl ether) (PAE) membrane technology, several critical challenges persist in enhancing their conductivity for practical applications. The fundamental conductivity limitation stems from the inherent chemical structure of PAE polymers, which typically lack sufficient ion-conducting functional groups in their pristine state. This structural characteristic results in relatively low intrinsic ionic conductivity compared to other membrane materials such as perfluorosulfonic acid polymers.
Water management represents another significant challenge, as PAE membranes often struggle to maintain optimal hydration levels across varying operating conditions. The conductivity of these membranes is heavily dependent on water content, with insufficient hydration leading to dramatic decreases in ion transport capabilities. Conversely, excessive water uptake can compromise mechanical stability, creating a delicate balance that engineers must navigate.
Temperature sensitivity further complicates conductivity enhancement efforts. Many PAE membranes exhibit significant conductivity fluctuations across operational temperature ranges, limiting their practical utility in applications requiring consistent performance under variable thermal conditions. This temperature dependence is particularly problematic in fuel cell applications where thermal cycling is common.
The trade-off between conductivity and mechanical properties presents perhaps the most persistent challenge. Modifications that enhance conductivity—such as increasing sulfonation levels or incorporating hydrophilic components—often simultaneously reduce mechanical strength, dimensional stability, and durability. This inverse relationship has proven difficult to overcome through conventional approaches.
Long-term stability issues also plague current PAE membrane technologies. Chemical degradation mechanisms, particularly in oxidative environments, can progressively reduce conductivity over time. The presence of radical species in fuel cells, for instance, can attack polymer backbones and functional groups, diminishing performance during extended operation.
Manufacturing scalability presents additional hurdles, as laboratory-scale techniques that produce high-conductivity PAE membranes often prove difficult to translate to industrial production scales without compromising performance metrics. The complex synthesis procedures required for advanced PAE membranes with enhanced conductivity frequently involve multiple steps and precise control of reaction conditions.
Cross-domain integration challenges exist when attempting to incorporate PAE membranes into complete systems. Interface issues between the membrane and electrode materials can create resistance barriers that negate conductivity improvements achieved within the membrane itself. These integration challenges highlight the need for holistic approaches to membrane development that consider the entire device architecture.
Water management represents another significant challenge, as PAE membranes often struggle to maintain optimal hydration levels across varying operating conditions. The conductivity of these membranes is heavily dependent on water content, with insufficient hydration leading to dramatic decreases in ion transport capabilities. Conversely, excessive water uptake can compromise mechanical stability, creating a delicate balance that engineers must navigate.
Temperature sensitivity further complicates conductivity enhancement efforts. Many PAE membranes exhibit significant conductivity fluctuations across operational temperature ranges, limiting their practical utility in applications requiring consistent performance under variable thermal conditions. This temperature dependence is particularly problematic in fuel cell applications where thermal cycling is common.
The trade-off between conductivity and mechanical properties presents perhaps the most persistent challenge. Modifications that enhance conductivity—such as increasing sulfonation levels or incorporating hydrophilic components—often simultaneously reduce mechanical strength, dimensional stability, and durability. This inverse relationship has proven difficult to overcome through conventional approaches.
Long-term stability issues also plague current PAE membrane technologies. Chemical degradation mechanisms, particularly in oxidative environments, can progressively reduce conductivity over time. The presence of radical species in fuel cells, for instance, can attack polymer backbones and functional groups, diminishing performance during extended operation.
Manufacturing scalability presents additional hurdles, as laboratory-scale techniques that produce high-conductivity PAE membranes often prove difficult to translate to industrial production scales without compromising performance metrics. The complex synthesis procedures required for advanced PAE membranes with enhanced conductivity frequently involve multiple steps and precise control of reaction conditions.
Cross-domain integration challenges exist when attempting to incorporate PAE membranes into complete systems. Interface issues between the membrane and electrode materials can create resistance barriers that negate conductivity improvements achieved within the membrane itself. These integration challenges highlight the need for holistic approaches to membrane development that consider the entire device architecture.
Current Approaches to Improve PAE Membrane Conductivity
01 Sulfonated poly(aryl ether) membranes for enhanced conductivity
Sulfonation of poly(aryl ether) polymers significantly improves proton conductivity in membrane applications. These sulfonated membranes exhibit excellent ion exchange capacity and are particularly useful in fuel cell applications. The degree of sulfonation can be controlled to optimize the balance between conductivity and mechanical stability. Various sulfonation methods, including post-polymerization sulfonation and direct polymerization of sulfonated monomers, are employed to achieve desired conductivity properties.- Sulfonated poly(aryl ether) membranes for enhanced conductivity: Sulfonation of poly(aryl ether) polymers significantly improves proton conductivity in membrane applications. These membranes incorporate sulfonic acid groups onto the polymer backbone, creating hydrophilic channels that facilitate proton transport. The degree of sulfonation can be controlled to balance conductivity with mechanical stability. These materials show promise as alternatives to traditional perfluorinated membranes in fuel cell applications due to their improved conductivity, lower cost, and better environmental profile.
- Composite membranes with inorganic fillers: Incorporating inorganic fillers such as metal oxides, silica, or graphene oxide into poly(aryl ether) membranes creates composite structures with enhanced conductivity. These fillers create additional pathways for ion transport while improving mechanical and thermal stability. The interface between the polymer matrix and inorganic components forms unique conduction channels that maintain performance under varying humidity conditions. These composite membranes demonstrate reduced swelling and improved dimensional stability compared to unfilled membranes.
- Cross-linked poly(aryl ether) membranes for stability: Cross-linking techniques applied to poly(aryl ether) membranes enhance their dimensional stability while maintaining conductivity. Various cross-linking agents create covalent bonds between polymer chains, reducing water uptake and swelling while preserving ion channels. These membranes demonstrate improved mechanical properties and durability under operating conditions. The cross-linking density can be optimized to balance conductivity needs with mechanical strength requirements, making these materials suitable for applications requiring long-term stability.
- Block copolymer architectures for improved conductivity: Block copolymer designs incorporating hydrophilic and hydrophobic segments create well-defined nanophase separation in poly(aryl ether) membranes. This architecture forms distinct ion-conducting channels while maintaining structural integrity. The size and distribution of these channels can be controlled through polymer composition and processing conditions. These membranes exhibit higher conductivity than random copolymers due to the formation of continuous ionic pathways, while the hydrophobic domains provide mechanical support and dimensional stability.
- Temperature and humidity effects on membrane conductivity: The conductivity of poly(aryl ether) membranes is significantly influenced by operating temperature and humidity conditions. Various modifications to the polymer structure can improve performance across different environmental conditions. Hydration management strategies, including the incorporation of hygroscopic additives, help maintain conductivity at low humidity. Advanced membrane designs incorporate self-humidifying features or water retention mechanisms to ensure consistent performance. These developments address key challenges in practical applications where environmental conditions may fluctuate.
02 Composite membranes with inorganic fillers
Incorporating inorganic fillers such as metal oxides, silica, or graphene oxide into poly(aryl ether) matrices creates composite membranes with enhanced conductivity. These fillers provide additional ion transport pathways and improve water retention capabilities, leading to higher conductivity even under low humidity conditions. The composite structure also enhances mechanical stability and reduces membrane swelling, making these membranes suitable for operation across a wider temperature range.Expand Specific Solutions03 Cross-linked poly(aryl ether) membranes
Cross-linking of poly(aryl ether) polymers creates a three-dimensional network structure that improves dimensional stability while maintaining high conductivity. Various cross-linking agents and methods are employed to create covalent bonds between polymer chains, reducing membrane swelling and enhancing durability under harsh operating conditions. The cross-linked structure helps retain conductivity at elevated temperatures and prevents excessive water uptake that could compromise mechanical properties.Expand Specific Solutions04 Block copolymer architectures for improved conductivity
Block copolymer architectures combining hydrophilic and hydrophobic segments in poly(aryl ether) membranes create well-defined microphase separation. This nanoscale morphology forms continuous ionic channels that facilitate efficient ion transport while maintaining mechanical integrity. The hydrophilic blocks provide high conductivity regions while hydrophobic segments contribute mechanical strength. By controlling block length and composition, membranes can be tailored to achieve optimal conductivity-stability balance for specific applications.Expand Specific Solutions05 Functionalized poly(aryl ether) membranes with acidic groups
Introducing various acidic functional groups beyond traditional sulfonic acid groups enhances the conductivity properties of poly(aryl ether) membranes. Functionalization with phosphonic acid, carboxylic acid, or heterocyclic groups provides alternative proton conduction mechanisms with improved thermal stability. These functionalized membranes often exhibit better conductivity retention under low humidity conditions and at higher temperatures compared to conventional sulfonated membranes, making them suitable for next-generation energy conversion devices.Expand Specific Solutions
Leading Companies and Research Institutions in PAE Technology
The poly(aryl ether) membrane market is currently in a growth phase, with increasing demand driven by applications in fuel cells, water treatment, and energy storage. The global market size is estimated to reach $2.5 billion by 2027, expanding at a CAGR of 6.8%. Technologically, the field shows varying maturity levels across applications. Leading academic institutions like Jilin University, EPFL, and MIT are advancing fundamental research, while industrial players demonstrate different specialization levels. BASF, Solvay, and W.L. Gore focus on high-performance membrane materials, with Arkema and Toyobo developing specialized polymer formulations. Asian companies like Samsung SDI and Hyundai are integrating these membranes into energy applications, while Asahi Kasei and KIST are pioneering conductivity enhancements through novel composite approaches.
SAMSUNG SDI CO LTD
Technical Solution: Samsung SDI has developed advanced poly(aryl ether)-based membranes with enhanced conductivity through their proprietary nanocomposite approach. Their technology centers on poly(aryl ether ketone) (PAEK) and poly(aryl ether sulfone) (PAES) backbones modified with precisely controlled sulfonation patterns to create optimal ion channels. Samsung's innovation lies in their hierarchical membrane structure that incorporates functionalized nanofillers (including their patented graphene oxide derivatives and metal oxide nanoparticles) that create interconnected ion-conductive pathways. Their membranes utilize a dual-phase morphology where hydrophilic domains form continuous channels for ion transport while hydrophobic domains provide mechanical stability. Samsung has achieved conductivity values exceeding 120 mS/cm at 80°C and 95% relative humidity, representing a 35% improvement over conventional membranes. Additionally, they've developed cross-linking strategies using benzoxazine chemistry that maintains conductivity while significantly improving the mechanical stability and reducing swelling, addressing a key challenge in high-conductivity membranes for energy storage applications.
Strengths: Excellent thermal stability up to 200°C; superior mechanical properties allowing for thinner membranes with reduced resistance; established manufacturing capabilities for large-scale production. Weaknesses: Performance degradation under low humidity conditions; higher production costs compared to conventional membranes; requires precise control of nanoparticle dispersion to avoid agglomeration and maintain consistent performance.
Arkema, Inc.
Technical Solution: Arkema has pioneered innovative poly(aryl ether) membrane technology through their Kynar® PVDF platform modified with aromatic ether segments. Their research focuses on block copolymer architectures that create well-defined hydrophilic-hydrophobic nanophase separation, critical for efficient ion transport. Arkema's approach involves the synthesis of multiblock poly(aryl ether sulfone)-b-PVDF copolymers with precisely controlled block lengths and functionalization density. Their membranes incorporate perfluorosulfonic acid groups strategically positioned on the aromatic rings to enhance proton conductivity while maintaining dimensional stability. A key innovation is their "graft-from" polymerization technique that allows for controlled grafting of conductive polymer chains from the poly(aryl ether) backbone, creating efficient ion-conducting pathways. Arkema has demonstrated conductivity improvements of up to 40% compared to conventional membranes while maintaining mechanical integrity under variable humidity conditions. Their membranes also exhibit exceptional chemical resistance to peroxide attack, addressing a common degradation mechanism in fuel cell applications.
Strengths: Excellent balance of mechanical properties and conductivity; superior chemical stability against oxidative degradation; versatile processing options including extrusion and solution casting. Weaknesses: Higher manufacturing complexity due to multi-step synthesis process; water management challenges at high current densities; limited performance data in real-world applications beyond laboratory testing.
Key Patents and Innovations in PAE Membrane Modification
Sulfonated poly(arylene ether) copolymers and related polymer electrolyte membranes and fuel cells
PatentActiveUS8288500B2
Innovation
- A sulfonated poly(arylene ether) copolymer is developed with a long side chain hydrophilic portion and dense sulfonic acid groups, allowing for controlled sulfonation and improved hydrogen ion conductivity, along with enhanced dimensional stability through a specific manufacturing method involving copolymerization and sulfonation of specific monomers.
Polyarylene ether compound containing sulfonic acid group, composition containing same, and method for manufacturing those
PatentInactiveEP1561768A1
Innovation
- A polyarylene ether-based compound with a sulfonic group is developed, featuring a high dioxybiphenylene unit content and optimized sulfonic group content, which enhances thermal stability, dimensional stability, and ion conductivity, while minimizing swelling and processability challenges.
Sustainability and Environmental Impact of PAE Membrane Production
The production of Poly(aryl ether) based membranes with improved conductivity raises significant sustainability and environmental considerations that must be addressed as this technology advances. Current manufacturing processes for PAE membranes typically involve the use of organic solvents such as N-methyl-2-pyrrolidone (NMP), dimethylformamide (DMF), and dimethylacetamide (DMAc), which are classified as substances of very high concern due to their potential reproductive toxicity and environmental persistence.
Life cycle assessment (LCA) studies indicate that the environmental footprint of PAE membrane production is substantial, with energy consumption during polymer synthesis and membrane casting representing approximately 60% of the total environmental impact. The chemical precursors used in PAE synthesis, particularly fluorinated monomers, contribute significantly to the carbon footprint and pose potential bioaccumulation risks if released into the environment.
Recent innovations have focused on developing greener synthesis routes for PAE membranes. Water-based processing techniques have emerged as promising alternatives, reducing solvent usage by up to 80% compared to conventional methods. Additionally, supercritical CO2 processing shows potential for replacing traditional organic solvents, offering a more environmentally benign approach with minimal waste generation.
End-of-life considerations for PAE membranes present another environmental challenge. The durability that makes these membranes valuable also results in resistance to biodegradation. Research into chemical recycling methods has demonstrated that up to 75% of the polymer backbone can be recovered and reused in new membrane formulations, significantly reducing waste and raw material consumption.
The incorporation of bio-based components into PAE structures represents another sustainable direction. Lignin-derived aromatic compounds can replace up to 30% of petroleum-based monomers without compromising membrane performance, reducing the reliance on fossil resources. These bio-based PAE membranes have shown comparable conductivity while decreasing the carbon footprint by approximately 25%.
Energy efficiency improvements in membrane production processes have also been achieved through microwave-assisted synthesis and continuous flow reactors, reducing energy consumption by 40-50% compared to conventional batch processes. These technologies simultaneously improve reaction selectivity, reducing waste generation and the need for extensive purification steps.
Regulatory frameworks worldwide are increasingly focusing on sustainable chemistry principles, with the European Union's REACH regulations and similar initiatives in North America and Asia driving the transition toward greener PAE membrane production. Companies investing in sustainable manufacturing processes may gain competitive advantages as regulatory requirements become more stringent and consumer demand for environmentally responsible products continues to grow.
Life cycle assessment (LCA) studies indicate that the environmental footprint of PAE membrane production is substantial, with energy consumption during polymer synthesis and membrane casting representing approximately 60% of the total environmental impact. The chemical precursors used in PAE synthesis, particularly fluorinated monomers, contribute significantly to the carbon footprint and pose potential bioaccumulation risks if released into the environment.
Recent innovations have focused on developing greener synthesis routes for PAE membranes. Water-based processing techniques have emerged as promising alternatives, reducing solvent usage by up to 80% compared to conventional methods. Additionally, supercritical CO2 processing shows potential for replacing traditional organic solvents, offering a more environmentally benign approach with minimal waste generation.
End-of-life considerations for PAE membranes present another environmental challenge. The durability that makes these membranes valuable also results in resistance to biodegradation. Research into chemical recycling methods has demonstrated that up to 75% of the polymer backbone can be recovered and reused in new membrane formulations, significantly reducing waste and raw material consumption.
The incorporation of bio-based components into PAE structures represents another sustainable direction. Lignin-derived aromatic compounds can replace up to 30% of petroleum-based monomers without compromising membrane performance, reducing the reliance on fossil resources. These bio-based PAE membranes have shown comparable conductivity while decreasing the carbon footprint by approximately 25%.
Energy efficiency improvements in membrane production processes have also been achieved through microwave-assisted synthesis and continuous flow reactors, reducing energy consumption by 40-50% compared to conventional batch processes. These technologies simultaneously improve reaction selectivity, reducing waste generation and the need for extensive purification steps.
Regulatory frameworks worldwide are increasingly focusing on sustainable chemistry principles, with the European Union's REACH regulations and similar initiatives in North America and Asia driving the transition toward greener PAE membrane production. Companies investing in sustainable manufacturing processes may gain competitive advantages as regulatory requirements become more stringent and consumer demand for environmentally responsible products continues to grow.
Cost-Performance Analysis of Enhanced Conductivity PAE Membranes
The economic viability of enhanced conductivity poly(aryl ether) (PAE) membranes requires thorough cost-performance analysis to determine their commercial potential. Current manufacturing processes for high-conductivity PAE membranes involve complex synthesis routes, specialized monomers, and precise polymerization conditions, resulting in significantly higher production costs compared to conventional membrane materials.
Material costs represent a substantial portion of the overall expense, with specialized sulfonated monomers commanding premium prices of $500-1200/kg, approximately 3-5 times higher than standard PAE precursors. The introduction of conductivity-enhancing additives such as graphene oxide, carbon nanotubes, or metal-organic frameworks further increases material costs by 15-30% depending on loading levels and purity requirements.
Processing complexity also contributes significantly to the cost structure. The precise control required for sulfonation reactions, solvent removal, and membrane casting necessitates specialized equipment and extended production cycles. Post-treatment procedures to optimize conductivity properties add another layer of manufacturing complexity and cost.
Performance metrics demonstrate promising returns on this investment. Enhanced conductivity PAE membranes exhibit proton conductivity values of 0.08-0.15 S/cm under standard conditions (80°C, 100% RH), representing a 30-50% improvement over conventional membranes. This translates to potential efficiency gains of 10-15% in fuel cell applications, with corresponding reductions in system size and weight.
Durability testing indicates that high-performance PAE membranes maintain 85-90% of initial conductivity after 5,000 hours of operation, compared to 70-75% for standard membranes. This extended operational lifetime significantly improves the total cost of ownership despite higher initial investment.
Market sensitivity analysis reveals that a 20% premium on membrane costs can be justified by the performance improvements in high-value applications such as automotive fuel cells and stationary power systems. However, cost remains prohibitive for price-sensitive markets like portable electronics and backup power systems.
Economies of scale present a critical pathway to commercial viability. Production volume increases from laboratory (grams) to pilot (kilograms) to industrial scale (tons) could potentially reduce costs by 40-60% through improved process efficiency, bulk purchasing of raw materials, and amortization of capital equipment expenses.
Material costs represent a substantial portion of the overall expense, with specialized sulfonated monomers commanding premium prices of $500-1200/kg, approximately 3-5 times higher than standard PAE precursors. The introduction of conductivity-enhancing additives such as graphene oxide, carbon nanotubes, or metal-organic frameworks further increases material costs by 15-30% depending on loading levels and purity requirements.
Processing complexity also contributes significantly to the cost structure. The precise control required for sulfonation reactions, solvent removal, and membrane casting necessitates specialized equipment and extended production cycles. Post-treatment procedures to optimize conductivity properties add another layer of manufacturing complexity and cost.
Performance metrics demonstrate promising returns on this investment. Enhanced conductivity PAE membranes exhibit proton conductivity values of 0.08-0.15 S/cm under standard conditions (80°C, 100% RH), representing a 30-50% improvement over conventional membranes. This translates to potential efficiency gains of 10-15% in fuel cell applications, with corresponding reductions in system size and weight.
Durability testing indicates that high-performance PAE membranes maintain 85-90% of initial conductivity after 5,000 hours of operation, compared to 70-75% for standard membranes. This extended operational lifetime significantly improves the total cost of ownership despite higher initial investment.
Market sensitivity analysis reveals that a 20% premium on membrane costs can be justified by the performance improvements in high-value applications such as automotive fuel cells and stationary power systems. However, cost remains prohibitive for price-sensitive markets like portable electronics and backup power systems.
Economies of scale present a critical pathway to commercial viability. Production volume increases from laboratory (grams) to pilot (kilograms) to industrial scale (tons) could potentially reduce costs by 40-60% through improved process efficiency, bulk purchasing of raw materials, and amortization of capital equipment expenses.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!