Data Integrity in HPLC Systems: Validation Protocols
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HPLC Data Integrity Background and Objectives
High-performance liquid chromatography (HPLC) has evolved significantly since its introduction in the 1960s, becoming a cornerstone analytical technique in pharmaceutical, environmental, food safety, and clinical laboratories. The evolution of HPLC systems has paralleled advancements in computing technology, transitioning from simple chart recorders to sophisticated digital data acquisition systems capable of processing vast amounts of analytical information.
Data integrity in HPLC systems refers to the completeness, consistency, and accuracy of data throughout its lifecycle. This concept has gained paramount importance as regulatory bodies worldwide have intensified their focus on ensuring the reliability of analytical results. The FDA, EMA, MHRA, and other regulatory agencies have published guidance documents specifically addressing data integrity concerns in analytical laboratories, highlighting its critical role in ensuring product quality and patient safety.
The historical context of data integrity issues in analytical chemistry reveals a pattern of challenges, from paper-based record falsification to more complex digital data manipulation. Notable cases of data integrity violations in pharmaceutical companies have resulted in warning letters, consent decrees, and significant financial penalties, underscoring the serious consequences of non-compliance.
Current technological trends in HPLC systems include increased automation, integration with laboratory information management systems (LIMS), cloud storage capabilities, and advanced data analytics. These developments, while enhancing efficiency and analytical capabilities, also introduce new complexities in maintaining data integrity throughout the analytical workflow.
The primary objective of validation protocols for HPLC data integrity is to establish systematic approaches that ensure data reliability from sample preparation through analysis and reporting. These protocols aim to address vulnerabilities in the data lifecycle, implement appropriate technical and procedural controls, and create an environment where data integrity is embedded in laboratory culture rather than treated as a mere compliance exercise.
Secondary objectives include harmonizing validation approaches across different regulatory jurisdictions, developing scalable solutions suitable for laboratories of varying sizes and resources, and creating frameworks that can adapt to evolving technologies without compromising fundamental data integrity principles.
The anticipated trajectory for HPLC data integrity includes greater integration of artificial intelligence for anomaly detection, blockchain technology for immutable audit trails, and biometric authentication systems to enhance security. These technological advancements must be balanced with practical implementation considerations to ensure that data integrity solutions remain accessible and effective across the diverse landscape of analytical laboratories worldwide.
Data integrity in HPLC systems refers to the completeness, consistency, and accuracy of data throughout its lifecycle. This concept has gained paramount importance as regulatory bodies worldwide have intensified their focus on ensuring the reliability of analytical results. The FDA, EMA, MHRA, and other regulatory agencies have published guidance documents specifically addressing data integrity concerns in analytical laboratories, highlighting its critical role in ensuring product quality and patient safety.
The historical context of data integrity issues in analytical chemistry reveals a pattern of challenges, from paper-based record falsification to more complex digital data manipulation. Notable cases of data integrity violations in pharmaceutical companies have resulted in warning letters, consent decrees, and significant financial penalties, underscoring the serious consequences of non-compliance.
Current technological trends in HPLC systems include increased automation, integration with laboratory information management systems (LIMS), cloud storage capabilities, and advanced data analytics. These developments, while enhancing efficiency and analytical capabilities, also introduce new complexities in maintaining data integrity throughout the analytical workflow.
The primary objective of validation protocols for HPLC data integrity is to establish systematic approaches that ensure data reliability from sample preparation through analysis and reporting. These protocols aim to address vulnerabilities in the data lifecycle, implement appropriate technical and procedural controls, and create an environment where data integrity is embedded in laboratory culture rather than treated as a mere compliance exercise.
Secondary objectives include harmonizing validation approaches across different regulatory jurisdictions, developing scalable solutions suitable for laboratories of varying sizes and resources, and creating frameworks that can adapt to evolving technologies without compromising fundamental data integrity principles.
The anticipated trajectory for HPLC data integrity includes greater integration of artificial intelligence for anomaly detection, blockchain technology for immutable audit trails, and biometric authentication systems to enhance security. These technological advancements must be balanced with practical implementation considerations to ensure that data integrity solutions remain accessible and effective across the diverse landscape of analytical laboratories worldwide.
Regulatory Demand Analysis for HPLC Validation
The regulatory landscape for High-Performance Liquid Chromatography (HPLC) validation has evolved significantly over the past decade, driven by increasing concerns about data integrity in pharmaceutical and biopharmaceutical industries. Regulatory bodies worldwide have intensified their focus on ensuring the reliability and accuracy of analytical data generated by HPLC systems, recognizing their critical role in quality control and product release decisions.
The FDA, EMA, and other global regulatory authorities have established comprehensive guidelines specifically addressing data integrity in analytical testing. The FDA's guidance on "Data Integrity and Compliance with cGMP" emphasizes the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available), which have become fundamental requirements for HPLC validation protocols.
Market research indicates that regulatory citations related to data integrity violations in HPLC systems have increased by 30% between 2018 and 2022, highlighting the growing regulatory scrutiny in this area. The most common citations involve inadequate audit trail review, unauthorized data manipulation, and insufficient system access controls.
The implementation of 21 CFR Part 11 (Electronic Records; Electronic Signatures) by the FDA has significantly shaped validation requirements for computerized HPLC systems. Similarly, the EU GMP Annex 11 on Computerized Systems has established parallel standards for European markets, creating a harmonized approach to data integrity validation.
Recent regulatory trends show increasing demands for risk-based validation approaches that focus on critical aspects of data integrity. This includes comprehensive assessment of potential vulnerabilities in data acquisition, processing, and storage within HPLC systems. Regulatory inspections now routinely examine the technical controls implemented to prevent data manipulation and ensure complete data retention.
The Japanese PMDA and China's NMPA have also strengthened their regulatory frameworks regarding analytical method validation and data integrity, aligning with international standards while maintaining region-specific requirements. This global harmonization trend is driving pharmaceutical companies to develop standardized validation protocols that can satisfy multiple regulatory jurisdictions simultaneously.
Industry surveys reveal that approximately 65% of pharmaceutical companies have had to significantly upgrade their HPLC validation protocols in the past three years to meet evolving regulatory expectations. The financial impact of these compliance efforts is substantial, with medium to large pharmaceutical companies investing significantly in technical infrastructure and procedural improvements.
Regulatory authorities are increasingly focusing on the human factors in data integrity, requiring companies to demonstrate robust training programs and clear accountability structures for personnel involved in HPLC testing. This holistic approach combines technical validation with procedural and organizational controls to ensure comprehensive data integrity protection.
The FDA, EMA, and other global regulatory authorities have established comprehensive guidelines specifically addressing data integrity in analytical testing. The FDA's guidance on "Data Integrity and Compliance with cGMP" emphasizes the ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, plus Complete, Consistent, Enduring, and Available), which have become fundamental requirements for HPLC validation protocols.
Market research indicates that regulatory citations related to data integrity violations in HPLC systems have increased by 30% between 2018 and 2022, highlighting the growing regulatory scrutiny in this area. The most common citations involve inadequate audit trail review, unauthorized data manipulation, and insufficient system access controls.
The implementation of 21 CFR Part 11 (Electronic Records; Electronic Signatures) by the FDA has significantly shaped validation requirements for computerized HPLC systems. Similarly, the EU GMP Annex 11 on Computerized Systems has established parallel standards for European markets, creating a harmonized approach to data integrity validation.
Recent regulatory trends show increasing demands for risk-based validation approaches that focus on critical aspects of data integrity. This includes comprehensive assessment of potential vulnerabilities in data acquisition, processing, and storage within HPLC systems. Regulatory inspections now routinely examine the technical controls implemented to prevent data manipulation and ensure complete data retention.
The Japanese PMDA and China's NMPA have also strengthened their regulatory frameworks regarding analytical method validation and data integrity, aligning with international standards while maintaining region-specific requirements. This global harmonization trend is driving pharmaceutical companies to develop standardized validation protocols that can satisfy multiple regulatory jurisdictions simultaneously.
Industry surveys reveal that approximately 65% of pharmaceutical companies have had to significantly upgrade their HPLC validation protocols in the past three years to meet evolving regulatory expectations. The financial impact of these compliance efforts is substantial, with medium to large pharmaceutical companies investing significantly in technical infrastructure and procedural improvements.
Regulatory authorities are increasingly focusing on the human factors in data integrity, requiring companies to demonstrate robust training programs and clear accountability structures for personnel involved in HPLC testing. This holistic approach combines technical validation with procedural and organizational controls to ensure comprehensive data integrity protection.
Current Challenges in HPLC Data Integrity
Despite significant advancements in HPLC technology, data integrity remains a critical challenge for analytical laboratories. The complexity of modern HPLC systems, which integrate sophisticated hardware with complex software platforms, creates multiple vulnerability points where data integrity can be compromised. One primary challenge is the inconsistent implementation of audit trail functionality across different vendor platforms, making standardized validation approaches difficult to establish and maintain.
Electronic data management presents particular difficulties, with many laboratories struggling to transition from paper-based systems to fully electronic workflows. This transition often creates hybrid systems where data integrity gaps emerge between electronic acquisition and paper documentation, introducing opportunities for transcription errors and data manipulation. The absence of comprehensive data lifecycle management strategies further exacerbates these issues.
System security vulnerabilities constitute another significant challenge. Many HPLC systems operate on outdated operating systems with inadequate security protocols, creating potential entry points for unauthorized access. Password management practices frequently fall short of regulatory expectations, with shared logins and insufficient privilege controls being commonplace in busy laboratory environments.
Data archiving and retrieval processes present ongoing difficulties, particularly for organizations managing long-term stability studies or maintaining data for extended regulatory compliance periods. The rapid evolution of storage technologies and file formats raises concerns about future data accessibility, with many laboratories lacking robust strategies for ensuring data remains readable throughout its required retention period.
Regulatory expectations regarding data integrity continue to evolve, creating a moving compliance target for laboratories. The interpretation gap between regulatory guidance documents and practical implementation creates uncertainty, particularly for global organizations navigating different regional requirements. Recent FDA and EMA inspections have increasingly focused on data governance frameworks rather than just technical controls, requiring laboratories to develop more comprehensive approaches.
Personnel training deficiencies represent a persistent challenge, with many laboratory staff lacking sufficient understanding of data integrity principles beyond basic procedural compliance. The technical complexity of modern HPLC systems often exceeds the training provided, creating situations where users may inadvertently compromise data integrity through improper system use or troubleshooting attempts.
Integration challenges between HPLC systems and laboratory information management systems (LIMS) or electronic laboratory notebooks (ELN) create additional data integrity risks at transfer points. These interface vulnerabilities often lack adequate validation, creating potential for data corruption or loss during routine workflows.
Electronic data management presents particular difficulties, with many laboratories struggling to transition from paper-based systems to fully electronic workflows. This transition often creates hybrid systems where data integrity gaps emerge between electronic acquisition and paper documentation, introducing opportunities for transcription errors and data manipulation. The absence of comprehensive data lifecycle management strategies further exacerbates these issues.
System security vulnerabilities constitute another significant challenge. Many HPLC systems operate on outdated operating systems with inadequate security protocols, creating potential entry points for unauthorized access. Password management practices frequently fall short of regulatory expectations, with shared logins and insufficient privilege controls being commonplace in busy laboratory environments.
Data archiving and retrieval processes present ongoing difficulties, particularly for organizations managing long-term stability studies or maintaining data for extended regulatory compliance periods. The rapid evolution of storage technologies and file formats raises concerns about future data accessibility, with many laboratories lacking robust strategies for ensuring data remains readable throughout its required retention period.
Regulatory expectations regarding data integrity continue to evolve, creating a moving compliance target for laboratories. The interpretation gap between regulatory guidance documents and practical implementation creates uncertainty, particularly for global organizations navigating different regional requirements. Recent FDA and EMA inspections have increasingly focused on data governance frameworks rather than just technical controls, requiring laboratories to develop more comprehensive approaches.
Personnel training deficiencies represent a persistent challenge, with many laboratory staff lacking sufficient understanding of data integrity principles beyond basic procedural compliance. The technical complexity of modern HPLC systems often exceeds the training provided, creating situations where users may inadvertently compromise data integrity through improper system use or troubleshooting attempts.
Integration challenges between HPLC systems and laboratory information management systems (LIMS) or electronic laboratory notebooks (ELN) create additional data integrity risks at transfer points. These interface vulnerabilities often lack adequate validation, creating potential for data corruption or loss during routine workflows.
Current Validation Protocol Solutions
01 Data integrity protection mechanisms in HPLC systems
Various mechanisms are implemented in HPLC systems to ensure data integrity, including encryption, checksums, and digital signatures. These technologies help prevent unauthorized modifications to chromatography data and ensure that analytical results remain trustworthy throughout their lifecycle. Such protection mechanisms are crucial for maintaining regulatory compliance in pharmaceutical and other regulated industries where HPLC is commonly used.- Data integrity protection mechanisms in HPLC systems: Various mechanisms are implemented in HPLC systems to ensure data integrity, including encryption, checksums, and digital signatures. These mechanisms help prevent unauthorized modifications to chromatography data and ensure that the data remains accurate and reliable throughout its lifecycle. Advanced protection systems can detect tampering attempts and maintain audit trails of all data access and modifications.
- Audit trail and electronic record management: HPLC systems incorporate comprehensive audit trail capabilities that track all user actions, data modifications, and system events. These audit trails are designed to be tamper-evident and provide a complete history of data from creation through processing and reporting. Electronic record management systems ensure that chromatography data is stored securely with appropriate metadata and can be retrieved in its original form, supporting regulatory compliance requirements.
- System recovery and error handling for data preservation: HPLC systems implement robust error handling and system recovery mechanisms to preserve data integrity during system failures or unexpected shutdowns. These mechanisms include transaction logging, redundant storage, and automated backup procedures that ensure chromatography data is not lost or corrupted. Recovery protocols can restore the system to a known good state while maintaining the integrity of previously collected data.
- User access control and authentication: Access control systems in HPLC platforms ensure that only authorized personnel can access, modify, or delete chromatography data. These systems implement role-based permissions, multi-factor authentication, and secure login procedures. User management features allow administrators to define granular access rights based on job responsibilities, ensuring appropriate data handling while maintaining traceability of all user actions.
- Data validation and integrity checking algorithms: HPLC systems employ sophisticated validation algorithms to verify data integrity throughout the analytical workflow. These algorithms check for data consistency, detect anomalies, and ensure compliance with predefined acceptance criteria. Automated data verification processes can identify potential issues such as integration errors, baseline problems, or instrument malfunctions that might compromise data quality, allowing for corrective actions before results are reported.
02 Audit trail and electronic records management
HPLC systems incorporate comprehensive audit trail capabilities that track all user actions, data modifications, and system events. These features enable traceability of all operations performed on the system, including who made changes, what changes were made, and when they occurred. Electronic records management ensures proper storage, retrieval, and archiving of chromatography data in compliance with regulatory requirements such as 21 CFR Part 11.Expand Specific Solutions03 System validation and error detection
HPLC systems employ validation protocols and error detection algorithms to ensure data reliability. These include system suitability tests, automatic error checking, and data verification processes that identify anomalies or inconsistencies in analytical results. Continuous monitoring of system performance parameters helps detect potential issues that could compromise data integrity before they affect analytical results.Expand Specific Solutions04 User access control and authentication
Robust user access control mechanisms are implemented in HPLC systems to prevent unauthorized data access or manipulation. These include role-based access controls, multi-factor authentication, and user permission management that restrict system functionality based on user credentials. Such measures ensure that only authorized personnel can perform specific operations on the system, thereby maintaining data integrity throughout the analytical workflow.Expand Specific Solutions05 Data backup and recovery solutions
HPLC systems incorporate automated backup procedures and recovery mechanisms to protect against data loss. These solutions include redundant storage, automated backup scheduling, and disaster recovery protocols that ensure data can be restored in case of system failures. By maintaining multiple copies of critical data across different storage locations, these systems minimize the risk of permanent data loss and ensure business continuity.Expand Specific Solutions
Key Stakeholders in HPLC System Validation
The HPLC data integrity validation protocols market is currently in a growth phase, with increasing regulatory scrutiny driving adoption across pharmaceutical and biotechnology sectors. The market size is expanding steadily, projected to reach significant value as compliance requirements intensify globally. From a technological maturity perspective, the landscape shows varied sophistication levels among key players. Companies like Intel, Hitachi, and Siemens demonstrate advanced capabilities in data integrity solutions, while pharmaceutical technology providers such as EMC and Dell EMC offer specialized validation protocols. Emerging players including Huawei and Alibaba are rapidly developing competitive offerings, particularly in cloud-based compliance systems. The integration of automation and AI-driven validation tools represents the cutting edge, with Synopsys and Cadence Design Systems pioneering algorithmic approaches to data integrity verification in analytical instrumentation.
Hitachi Ltd.
Technical Solution: Hitachi's approach to data integrity in HPLC systems centers on their comprehensive Chromaster and LaChrom Elite HPLC platforms, which incorporate multi-layered data security protocols. Their systems implement 21 CFR Part 11 compliant software that provides complete audit trails, electronic signatures, and user authentication with multiple privilege levels. Hitachi has developed a unique "Data Guardian" technology that creates tamper-evident data records by applying cryptographic hashing algorithms to raw chromatography data, ensuring that any unauthorized modifications are immediately detectable. Their validation protocols include Automated System Suitability Testing (SST) that continuously monitors critical performance parameters and automatically flags deviations from predetermined acceptance criteria. Hitachi's systems also feature real-time data verification mechanisms that compare acquired data against predefined validation parameters throughout the analytical process.
Strengths: Hitachi's long history in both analytical instrumentation and information technology gives them unique capabilities in integrating robust data security with high-performance chromatography. Their systems offer exceptional audit trail functionality and user management. Weaknesses: Their validation software interfaces can be complex for new users, requiring substantial training, and their premium data integrity features come at a higher price point than some competitors.
EMC IP Holding Co. LLC
Technical Solution: EMC's approach to HPLC data integrity focuses on secure storage infrastructure and specialized data management solutions for analytical laboratories. Their SourceOne Archiving platform has been adapted specifically for chromatography data, providing immutable storage capabilities that prevent unauthorized modifications to raw HPLC data files. EMC's validation protocols for HPLC systems center on their Information Lifecycle Management (ILM) framework, which establishes policies for data retention, protection, and eventual disposition in compliance with regulatory requirements. Their system implements blockchain-inspired technologies to create tamper-evident audit trails for all HPLC data transactions, ensuring that the complete history of data acquisition, processing, and reporting is preserved. EMC's solution includes automated data verification routines that compare checksums before and after any data transfer or storage operation, alerting users to any potential data corruption issues.
Strengths: EMC's extensive experience in enterprise data storage provides exceptional data protection capabilities, with particular strengths in long-term archiving and disaster recovery for critical HPLC data. Their solutions scale effectively for organizations with large data volumes. Weaknesses: Their offerings focus primarily on data storage and management rather than the analytical workflow itself, requiring integration with third-party HPLC control software, which can create implementation challenges.
Critical Technologies for Data Integrity Assurance
Process of conducting high throughput testing high performance liquid chromatography
PatentActiveNZ731893A
Innovation
- Development of a simplified sample preparation technique for high throughput testing in HPLC that involves adding the entire sample vial to a wide mouth disposable bottle, which reduces sample handling and potential contamination.
- Implementation of overnight shaking followed by centrifugation as part of the sample preparation protocol, which potentially improves extraction efficiency and sample homogeneity.
- Adaptation of traditional HPLC methods into a high throughput testing framework specifically designed for pharmaceutical composition validation, increasing efficiency while maintaining analytical rigor.
System for the Simultaneous Monitoring of Constituents of an Electroplating Bath
PatentPendingUS20240133074A1
Innovation
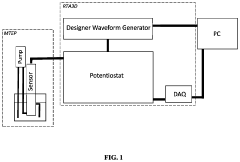
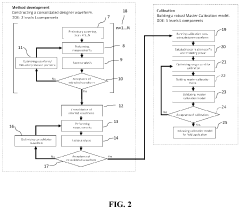
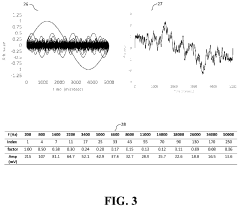
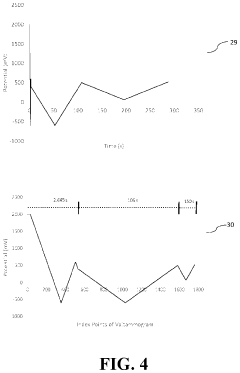
- The development of novel second-order, consolidated voltammetric waveforms combined with chemometric analysis and data compression techniques allows for the simultaneous measurement and analysis of all electroplating bath constituents without pretreatment, using a multi-frequency, variable amplitude waveform to generate a diagnostic voltammetric output that captures the synergistic interactions and maintains process control within the electroplating process.
Compliance Framework for Laboratory Information Systems
Laboratory Information Systems (LIS) must operate within a comprehensive compliance framework to ensure data integrity in HPLC systems. This framework encompasses regulatory requirements from multiple authorities including FDA 21 CFR Part 11, EU GMP Annex 11, GAMP 5, ISO 17025, and ICH Q10. These regulations collectively establish standards for electronic records, electronic signatures, data security, and system validation that directly impact HPLC data management.
The compliance framework for LIS requires implementation of technical controls that maintain data integrity throughout the entire data lifecycle. These controls include user access management with role-based permissions, comprehensive audit trails that capture all system activities, data encryption both at rest and in transit, and automated data backup procedures. Each of these controls must be validated according to predefined protocols to ensure consistent performance.
Risk assessment forms a critical component of the compliance framework, requiring organizations to identify potential vulnerabilities in their LIS that could compromise HPLC data integrity. This assessment should evaluate risks related to unauthorized access, data corruption, system failures, and procedural gaps. The framework mandates that risk mitigation strategies be documented and regularly reviewed as part of continuous improvement processes.
Standard Operating Procedures (SOPs) represent the procedural backbone of the compliance framework. These documents must clearly define all aspects of LIS operation, including system administration, data entry protocols, review processes, and exception handling. SOPs should specifically address HPLC data management, including chromatogram storage, integration parameters, and result calculations. Regular training on these procedures is essential for maintaining compliance.
Validation documentation within the framework must follow a structured approach, including requirements specifications, functional specifications, design specifications, risk assessments, test protocols, and summary reports. For HPLC systems specifically, the framework requires validation of data acquisition, processing algorithms, integration methods, and reporting functions. This documentation serves as evidence of system suitability during regulatory inspections.
Periodic system reviews are mandated by the compliance framework to ensure ongoing adherence to regulatory requirements. These reviews should evaluate system performance, assess the effectiveness of controls, identify emerging compliance gaps, and implement corrective actions. The framework specifies that these reviews be conducted at predetermined intervals and after significant system changes that might affect HPLC data integrity.
The compliance framework for LIS requires implementation of technical controls that maintain data integrity throughout the entire data lifecycle. These controls include user access management with role-based permissions, comprehensive audit trails that capture all system activities, data encryption both at rest and in transit, and automated data backup procedures. Each of these controls must be validated according to predefined protocols to ensure consistent performance.
Risk assessment forms a critical component of the compliance framework, requiring organizations to identify potential vulnerabilities in their LIS that could compromise HPLC data integrity. This assessment should evaluate risks related to unauthorized access, data corruption, system failures, and procedural gaps. The framework mandates that risk mitigation strategies be documented and regularly reviewed as part of continuous improvement processes.
Standard Operating Procedures (SOPs) represent the procedural backbone of the compliance framework. These documents must clearly define all aspects of LIS operation, including system administration, data entry protocols, review processes, and exception handling. SOPs should specifically address HPLC data management, including chromatogram storage, integration parameters, and result calculations. Regular training on these procedures is essential for maintaining compliance.
Validation documentation within the framework must follow a structured approach, including requirements specifications, functional specifications, design specifications, risk assessments, test protocols, and summary reports. For HPLC systems specifically, the framework requires validation of data acquisition, processing algorithms, integration methods, and reporting functions. This documentation serves as evidence of system suitability during regulatory inspections.
Periodic system reviews are mandated by the compliance framework to ensure ongoing adherence to regulatory requirements. These reviews should evaluate system performance, assess the effectiveness of controls, identify emerging compliance gaps, and implement corrective actions. The framework specifies that these reviews be conducted at predetermined intervals and after significant system changes that might affect HPLC data integrity.
Risk Assessment Strategies for HPLC Data Management
Risk assessment in HPLC data management requires a systematic approach to identify, evaluate, and mitigate potential threats to data integrity. Organizations must first establish a comprehensive risk identification framework that encompasses both technical and procedural vulnerabilities within their HPLC systems. This includes evaluating hardware components, software configurations, network infrastructure, and human interaction points that could compromise data quality.
The assessment process should follow a structured methodology such as FMEA (Failure Mode and Effects Analysis) or HACCP (Hazard Analysis Critical Control Points) adapted for laboratory environments. These methodologies enable organizations to assign risk priority numbers based on severity, occurrence probability, and detection difficulty for each identified risk factor. Critical areas requiring particular attention include data acquisition interfaces, storage systems, backup procedures, and audit trail mechanisms.
Risk categorization should differentiate between systemic risks (affecting the entire data management infrastructure) and specific risks (limited to particular instruments or processes). High-priority risks typically include unauthorized data manipulation, system configuration changes, inadequate audit trails, and improper data transfer protocols. Medium-priority risks often encompass calibration drift, method parameter inconsistencies, and routine maintenance lapses.
Mitigation strategies must be tailored to the specific risk profile of the organization's HPLC operations. Technical controls such as electronic signatures, access restrictions, and automated data verification algorithms form the first line of defense. These should be complemented by procedural controls including comprehensive SOPs, regular training programs, and independent verification protocols.
Continuous monitoring represents a critical component of effective risk management. Organizations should implement automated monitoring systems that track key performance indicators related to data integrity, including audit trail completeness, system suitability test results, and deviation patterns. Statistical process control techniques can help identify anomalies that may indicate emerging risks before they impact data quality.
Regular risk reassessment is essential as laboratory technologies evolve and regulatory expectations change. A formal review cycle should be established, typically quarterly for high-risk systems and annually for lower-risk environments. This process should incorporate lessons learned from internal audits, regulatory inspections, and industry best practices to ensure the risk assessment framework remains current and effective.
The assessment process should follow a structured methodology such as FMEA (Failure Mode and Effects Analysis) or HACCP (Hazard Analysis Critical Control Points) adapted for laboratory environments. These methodologies enable organizations to assign risk priority numbers based on severity, occurrence probability, and detection difficulty for each identified risk factor. Critical areas requiring particular attention include data acquisition interfaces, storage systems, backup procedures, and audit trail mechanisms.
Risk categorization should differentiate between systemic risks (affecting the entire data management infrastructure) and specific risks (limited to particular instruments or processes). High-priority risks typically include unauthorized data manipulation, system configuration changes, inadequate audit trails, and improper data transfer protocols. Medium-priority risks often encompass calibration drift, method parameter inconsistencies, and routine maintenance lapses.
Mitigation strategies must be tailored to the specific risk profile of the organization's HPLC operations. Technical controls such as electronic signatures, access restrictions, and automated data verification algorithms form the first line of defense. These should be complemented by procedural controls including comprehensive SOPs, regular training programs, and independent verification protocols.
Continuous monitoring represents a critical component of effective risk management. Organizations should implement automated monitoring systems that track key performance indicators related to data integrity, including audit trail completeness, system suitability test results, and deviation patterns. Statistical process control techniques can help identify anomalies that may indicate emerging risks before they impact data quality.
Regular risk reassessment is essential as laboratory technologies evolve and regulatory expectations change. A formal review cycle should be established, typically quarterly for high-risk systems and annually for lower-risk environments. This process should incorporate lessons learned from internal audits, regulatory inspections, and industry best practices to ensure the risk assessment framework remains current and effective.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!